Laccase Immobilized on a PAN/Adsorbents Composite Nanofibrous Membrane for Catechol Treatment by a Biocatalysis/Adsorption Process
Abstract
:1. Introduction
2. Results and Discussion
2.1. Morphologies of MMT, GO, MMT/GO Composites, and PAN/MMT/GO Composite Nanofibers

2.2. Relationship between Enzyme Structure and Activity

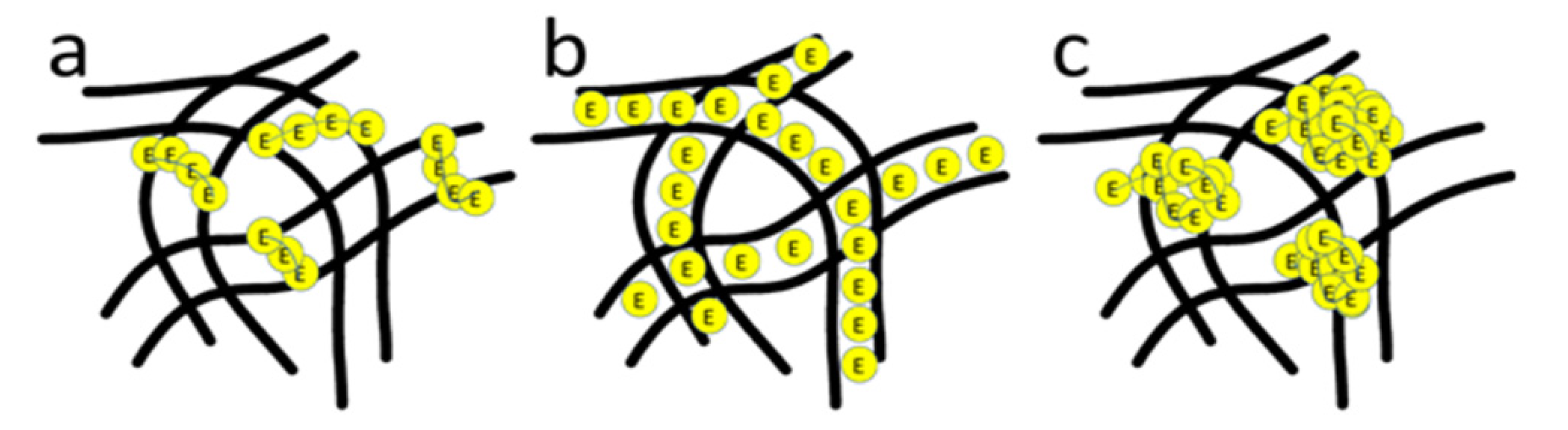
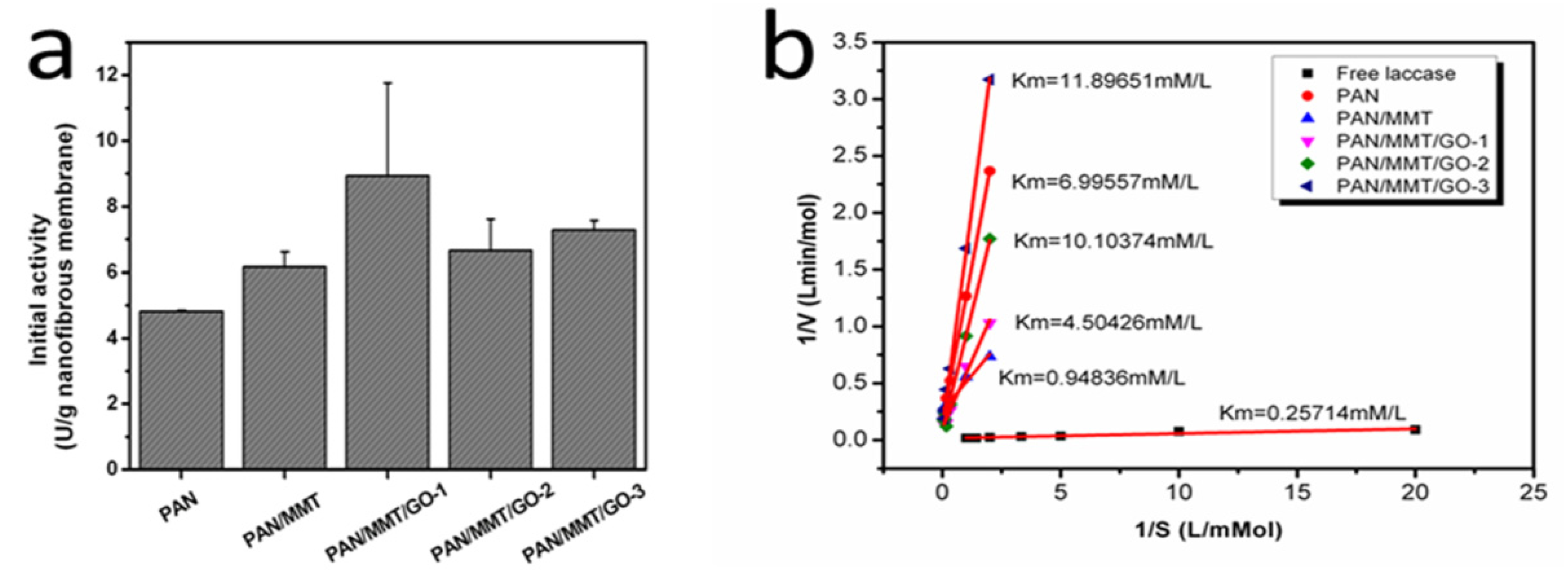
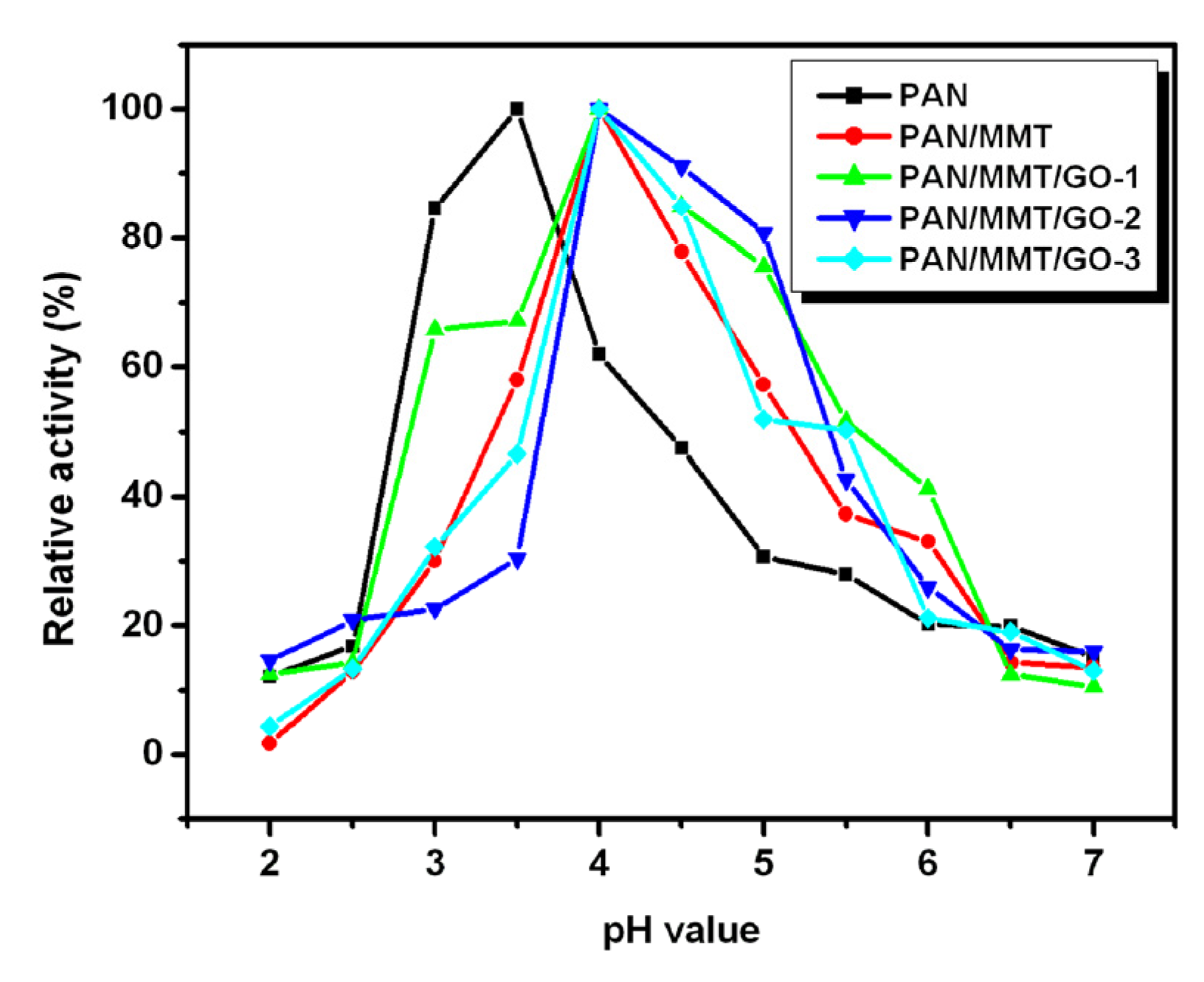
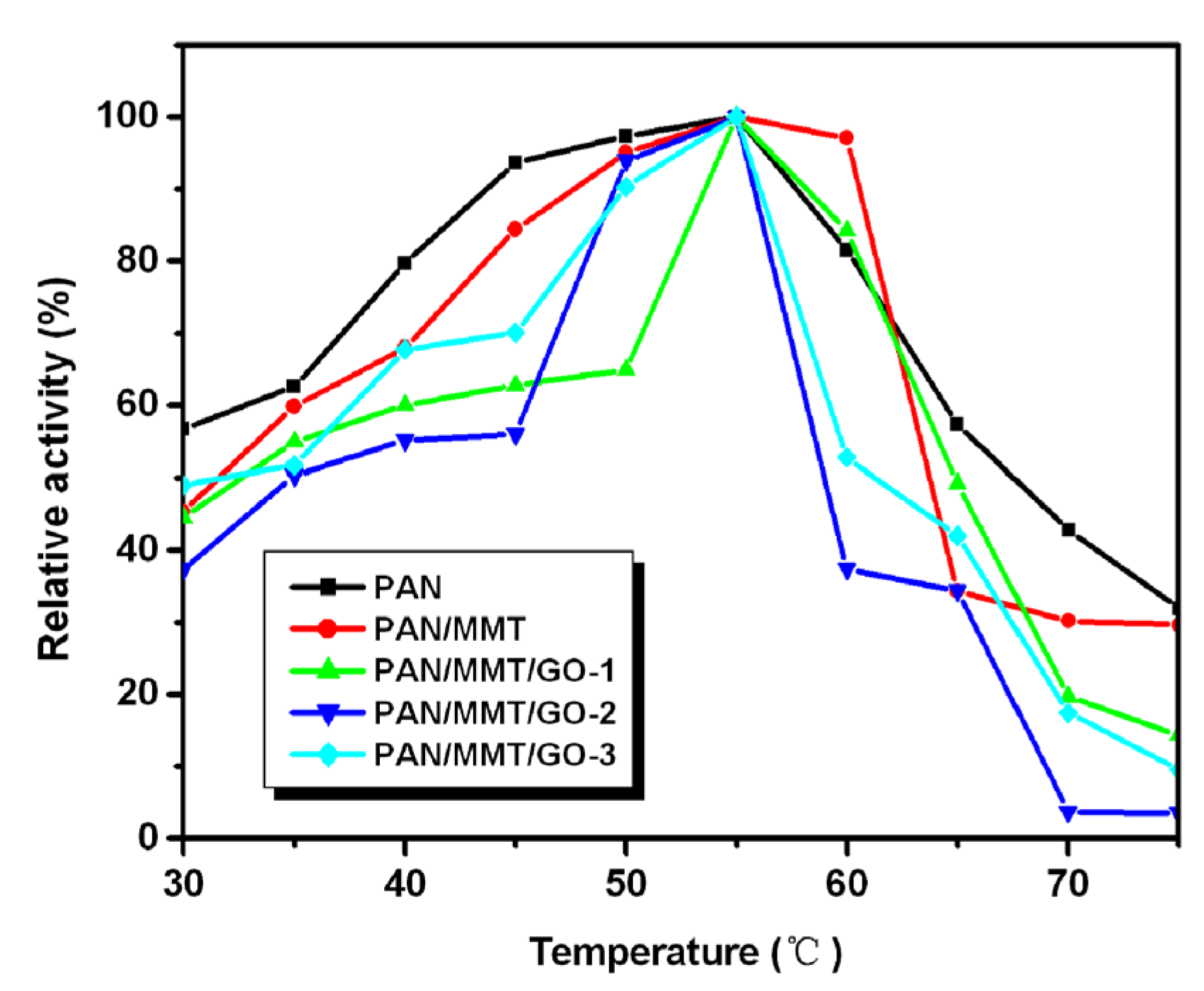
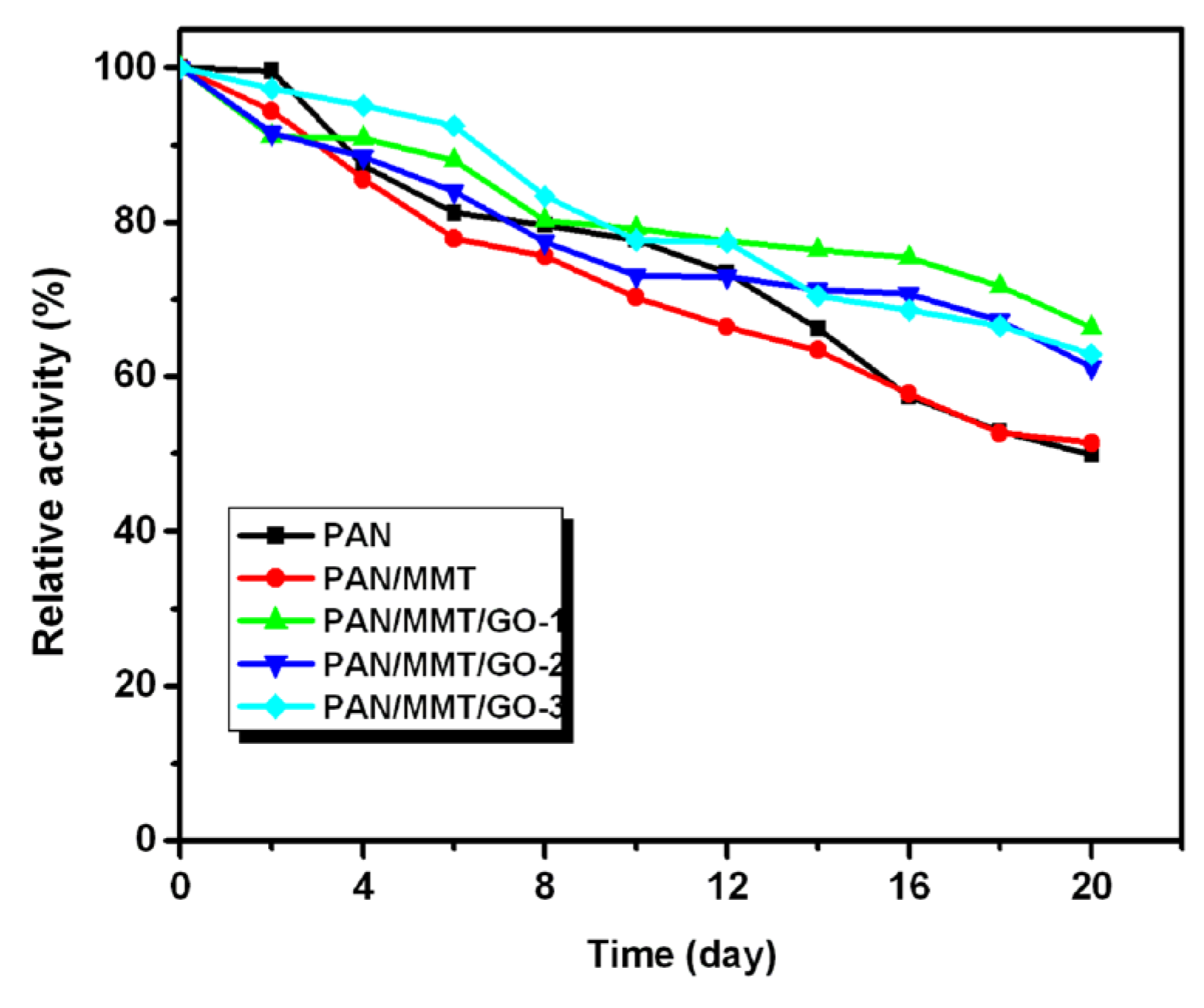
2.3. Immobilized Enzyme Properties (pH, Temperature, Storage, Reusability)

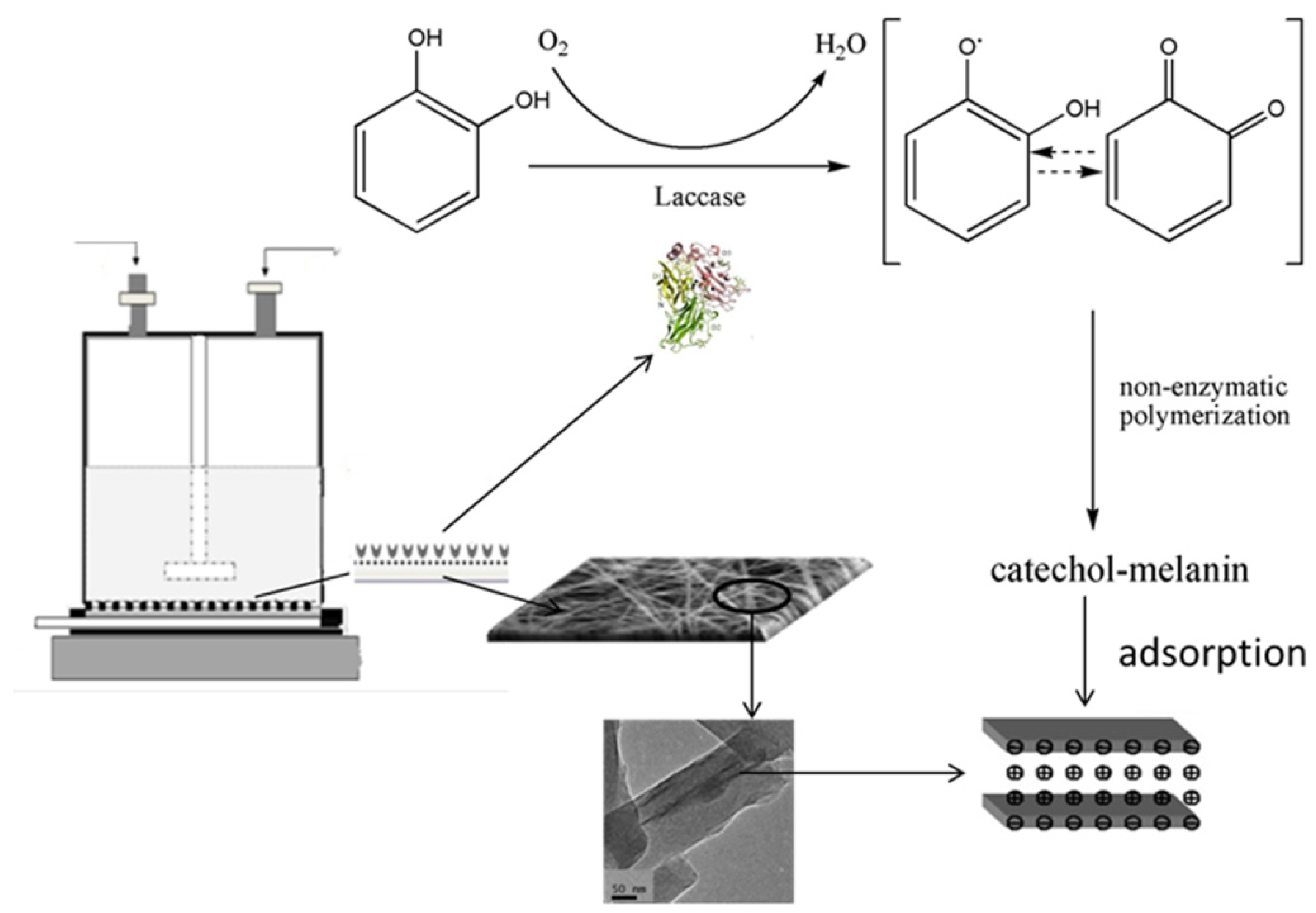
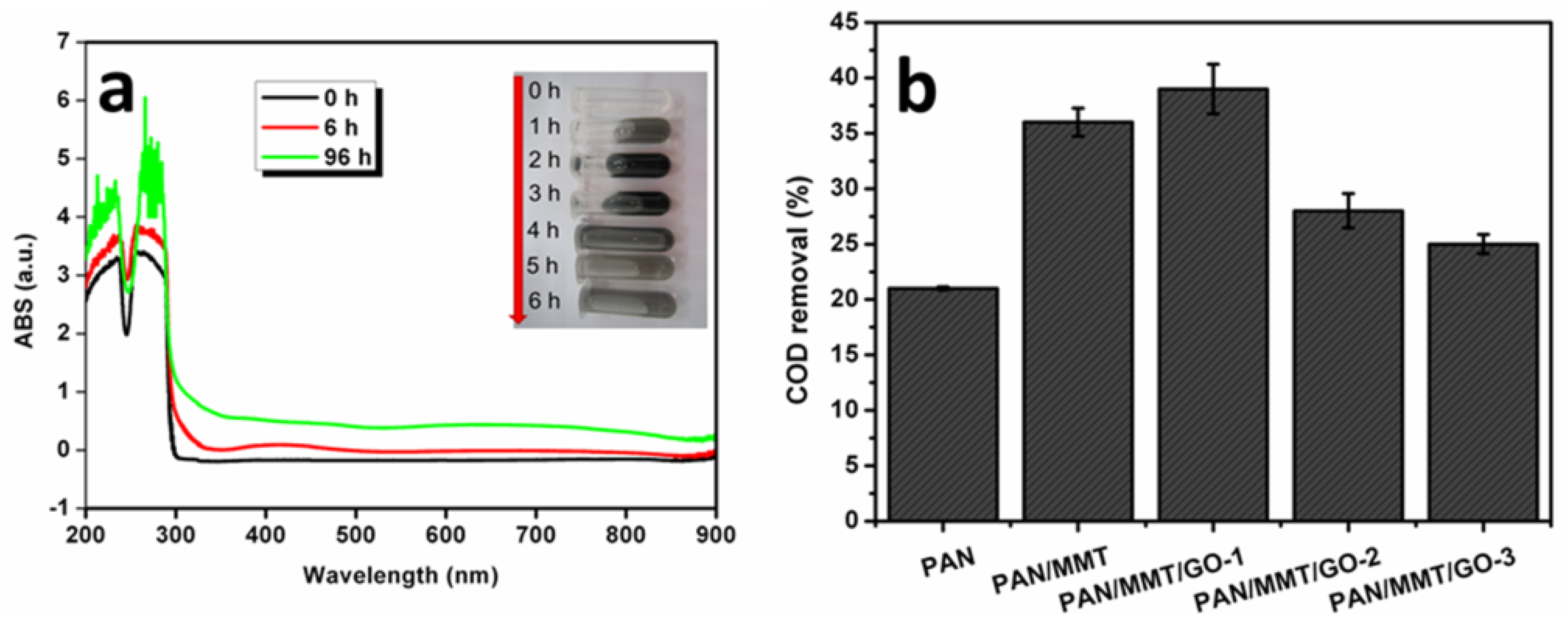
2.4. Catechol Treatment by a Homemade Membrane Reactor Treatment
3. Experimental
3.1. Chemicals
3.2. Preparation of Electrospun PAN, PAN/MMT and PAN/MMT/GO Composite Nanofibers
3.3. Immobilization of Laccase
3.4. Determination of Immobilized Laccase Activity
3.5. Catecholl Degradation
3.6. Characterizations
4. Conclusions
Acknowledgments
Author Contributions
Conflictts of Interest
References
- Flory, A.R.; Requesens, D.V.; Devaiah, S.P.; Teoh, K.T.; Mansfield, S.D.; Hood, E.E. Development of a green binder system for paper products. BMC Biotechnol. 2013, 13, 1–14. [Google Scholar] [CrossRef]
- Fackler, K.; Kuncinger, T.; Ters, T.; Srebotnik, E. Laccase-catalyzed functionalization with 4-hydroxy-3-methoxybenzylurea significantly improves internal bond of particle boards. Holzforschung 2008, 62, 223–229. [Google Scholar]
- Ashrafi, S.D.; Rezaei, S.; Forootanfar, H.; Mahvi, A.H.; Faramarzi, M.A. The enzymatic decolorization and detoxification of synthetic dyes by the laccase from a soil-isolated ascomycete, Paraconiothyrium variabile. Int. Biodeter. Biodegr. 2013, 85, 173–181. [Google Scholar] [CrossRef]
- Zhang, Y.; Zeng, Z.T.; Zeng, G.M.; Liu, X.M.; Liu, Z.F.; Chen, M.; Liu, L.; Li, J.; Xie, G. Effect of Triton X-100 on the removal of aqueous phenol by laccase analyzed with a combined approach of experiments and molecular docking. Colloid Surf. B 2012, 97, 7–12. [Google Scholar] [CrossRef]
- Dhillon, G.S.; Kaur, S.; Brar, S.K.; Verma, M. Flocculation and Haze Removal from Crude Beer Using In-House Produced Laccase from Trametes versicolor Cultured on Brewer’s Spent Grain. J. Agric. Food Chem. 2012, 60, 7895–7904. [Google Scholar] [CrossRef]
- Basto, C.; Tzanov, T.; Cavaco-Paulo, A. Combined ultrasound-laccase assisted bleaching of cotton. Ultrason. Sonochem. 2007, 14, 350–354. [Google Scholar] [CrossRef]
- Fernandez-Fernandez, M.; Sanroman, M.A.; Moldes, D. Recent developments and applications of immobilized laccase. Biotechnol. Adv. 2013, 31, 1808–1825. [Google Scholar] [CrossRef]
- Sheldon, R.A.; van Pelt, S. Enzyme immobilisation in biocatalysis: Why, what and how. Chem. Soc. Rev. 2013, 42, 6223–6235. [Google Scholar]
- Bayramoglu, G.; Arica, M.Y. Immobilization of laccase onto poly(glycidylmethacrylate) brush grafted poly(hydroxyethylmethacrylate) films: Enzymatic oxidation of phenolic compounds. Mat. Sci. Eng. C-Mater. 2009, 29, 1990–1997. [Google Scholar] [CrossRef]
- Xu, X.H.; Lu, P.; Zhou, Y.M.; Zhao, Z.Z.; Guo, M.Q. Laccase immobilized on methylene blue modified mesoporous silica mcm-41/pva. Mat. Sci. Eng. C-Mater. 2009, 29, 2160–2164. [Google Scholar]
- Qiu, H.J.; Xu, C.X.; Huang, X.R.; Ding, Y.; Qu, Y.B.; Gao, P.J. Immobilization of laccase on nanoporous gold: Comparative studies on the immobilization strategies and the particle size effects. J. Phys. Chem. C 2009, 113, 2521–2525. [Google Scholar] [CrossRef]
- Garcia-Galan, C.; Berenguer-Murcia, A.; Fernandez-Lafuente, R.; Rodrigues, R.C. Potential of different enzyme immobilization strategies to improve enzyme performance. Adv. Synth. Catal. 2011, 353, 2885–2904. [Google Scholar] [CrossRef]
- Wang, Z.G.; Wan, L.S.; Liu, Z.M.; Huang, X.J.; Xu, Z.K. Enzyme immobilization on electrospun polymer nanofibers: An overview. J. Mol. Catal. B.-Enzym. 2009, 56, 189–195. [Google Scholar] [CrossRef]
- Ye, P.; Xu, Z.K.; Wu, J.; Innocent, C.; Seta, P. Nanofibrous membranes containing reactive groups: Electrospinning from poly(acrylonitrile-co-maleic acid) for lipase immobilization. Macromolecules 2006, 39, 1041–1045. [Google Scholar] [CrossRef]
- Li, S.F.; Chen, J.P.; Wu, W.T. Electrospun polyacrylonitrile nanofibrous membranes for lipase immobilization. J. Mol. Catal. B.-Enzym. 2007, 47, 117–124. [Google Scholar] [CrossRef]
- Xu, R.; Chi, C.L.; Li, F.T.; Zhang, B.R. Laccase-Polyacrylonitrile Nanofibrous membrane: Highly immobilized, Stable, Reusable, and efficacious for 2,4,6-trichlorophenol removal. Acs. Appl. Mater. Inter. 2013, 5, 12554–12560. [Google Scholar] [CrossRef]
- Feng, Q.; Wang, Q.Q.; Tang, B.; Wei, A.F.; Wang, X.Q.; Wei, Q.F.; Huang, F.L.; Cai, Y.B.; Hou, D.Y.; Bi, S.M. Immobilization of catalases on amidoxime polyacrylonitrile nanofibrous membranes. Polym. Int. 2013, 62, 251–256. [Google Scholar] [CrossRef]
- Gupta, A.; Dhakate, S.R.; Pahwa, M.; Sinha, S.; Chand, S.; Mathur, R.B. Geranyl acetate synthesis catalyzed by Thermomyces lanuginosus lipase immobilized on electrospun polyacrylonitrile nanofiber membrane. Process. Biochem. 2013, 48, 124–132. [Google Scholar] [CrossRef]
- Huang, X.J.; Chen, P.C.; Huang, F.; Ou, Y.; Chen, M.R.; Xu, Z.K. Immobilization of Candida rugosa lipase on electrospun cellulose nanofiber membrane. J. Mol. Catal. B.-Enzym. 2011, 70, 95–100. [Google Scholar] [CrossRef]
- Zhu, J.; Sun, G. Lipase immobilization on glutaraldehyde-activated nanofibrous membranes for improved enzyme stabilities and activities. React. Funct. Polym. 2012, 72, 839–845. [Google Scholar] [CrossRef]
- Chattopadhyay, S.; Sen, R. A comparative performance evaluation of jute and eggshell matrices to immobilize pancreatic lipase. Process Biochem. 2012, 47, 749–757. [Google Scholar] [CrossRef]
- Cetinus, S.A.; Sahin, E.; Saraydin, D. Preparation of Cu(II) adsorbed chitosan beads for catalase immobilization. Food Chem. 2009, 114, 962–969. [Google Scholar] [CrossRef]
- Wang, L.; Wang, A.Q. Adsorption properties of Congo Red from aqueous solution onto surfactant-modified montmorillonite. J. Hazard. Mater. 2008, 160, 173–180. [Google Scholar] [CrossRef]
- Ma, H.L.; Kermasha, S.; Gao, J.M.; Borges, R.M.; Yu, X.Z. Laccase-catalyzed oxidation of phenolic compounds in organic media. J. Mol. Catal. B.-Enzym. 2009, 57, 89–95. [Google Scholar] [CrossRef]
- Shakir, K.; Ghoneimy, H.F.; Elkafrawy, A.F.; Beheir, S.G.; Refaat, M. Removal of catechol from aqueous solutions by adsorption onto organophilic-bentonite. J. Hazard. Mater. 2008, 150, 765–773. [Google Scholar]
- Alm, M.Y.; Martinez, C.E.; Archibald, D.D.; Zimmerman, A.R.; Bollag, J.M.; Dec, J. Transformation of catechol in the presence of a laccase and birnessite. Soil Biol. Biochem. 2006, 38, 1015–1020. [Google Scholar] [CrossRef]
- Wang, Q.Q.; Peng, L.; Du, Y.Z.; Xu, J.; Cai, Y.B.; Feng, Q.; Huang, F.L.; Wei, Q.F. Fabrication of hydrophilic nanoporous PMMA/O-MMT composite microfibrous membrane and its use in enzyme immobilization. J. Porous Mat. 2013, 20, 457–464. [Google Scholar] [CrossRef]
- Wang, Q.Q.; Peng, L.; Li, G.H.; Zhang, P.; Li, D.W.; Huang, F.L.; Wei, Q.F. Activity of Laccase Immobilized on TiO2-Montmorillonite Complexes. Int. J. Mol. Sci. 2013, 14, 12520–12532. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds including PAN, PAN/MMT, PAN/MMT-GO, PAN/MMT-GO-Lac composite nanofibrous membrane are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, Q.; Cui, J.; Li, G.; Zhang, J.; Li, D.; Huang, F.; Wei, Q. Laccase Immobilized on a PAN/Adsorbents Composite Nanofibrous Membrane for Catechol Treatment by a Biocatalysis/Adsorption Process. Molecules 2014, 19, 3376-3388. https://doi.org/10.3390/molecules19033376
Wang Q, Cui J, Li G, Zhang J, Li D, Huang F, Wei Q. Laccase Immobilized on a PAN/Adsorbents Composite Nanofibrous Membrane for Catechol Treatment by a Biocatalysis/Adsorption Process. Molecules. 2014; 19(3):3376-3388. https://doi.org/10.3390/molecules19033376
Chicago/Turabian StyleWang, Qingqing, Jing Cui, Guohui Li, Jinning Zhang, Dawei Li, Fenglin Huang, and Qufu Wei. 2014. "Laccase Immobilized on a PAN/Adsorbents Composite Nanofibrous Membrane for Catechol Treatment by a Biocatalysis/Adsorption Process" Molecules 19, no. 3: 3376-3388. https://doi.org/10.3390/molecules19033376
APA StyleWang, Q., Cui, J., Li, G., Zhang, J., Li, D., Huang, F., & Wei, Q. (2014). Laccase Immobilized on a PAN/Adsorbents Composite Nanofibrous Membrane for Catechol Treatment by a Biocatalysis/Adsorption Process. Molecules, 19(3), 3376-3388. https://doi.org/10.3390/molecules19033376



