Intracellular Glutathione Depletion by Oridonin Leads to Apoptosis in Hepatic Stellate Cells
Abstract
:1. Introduction
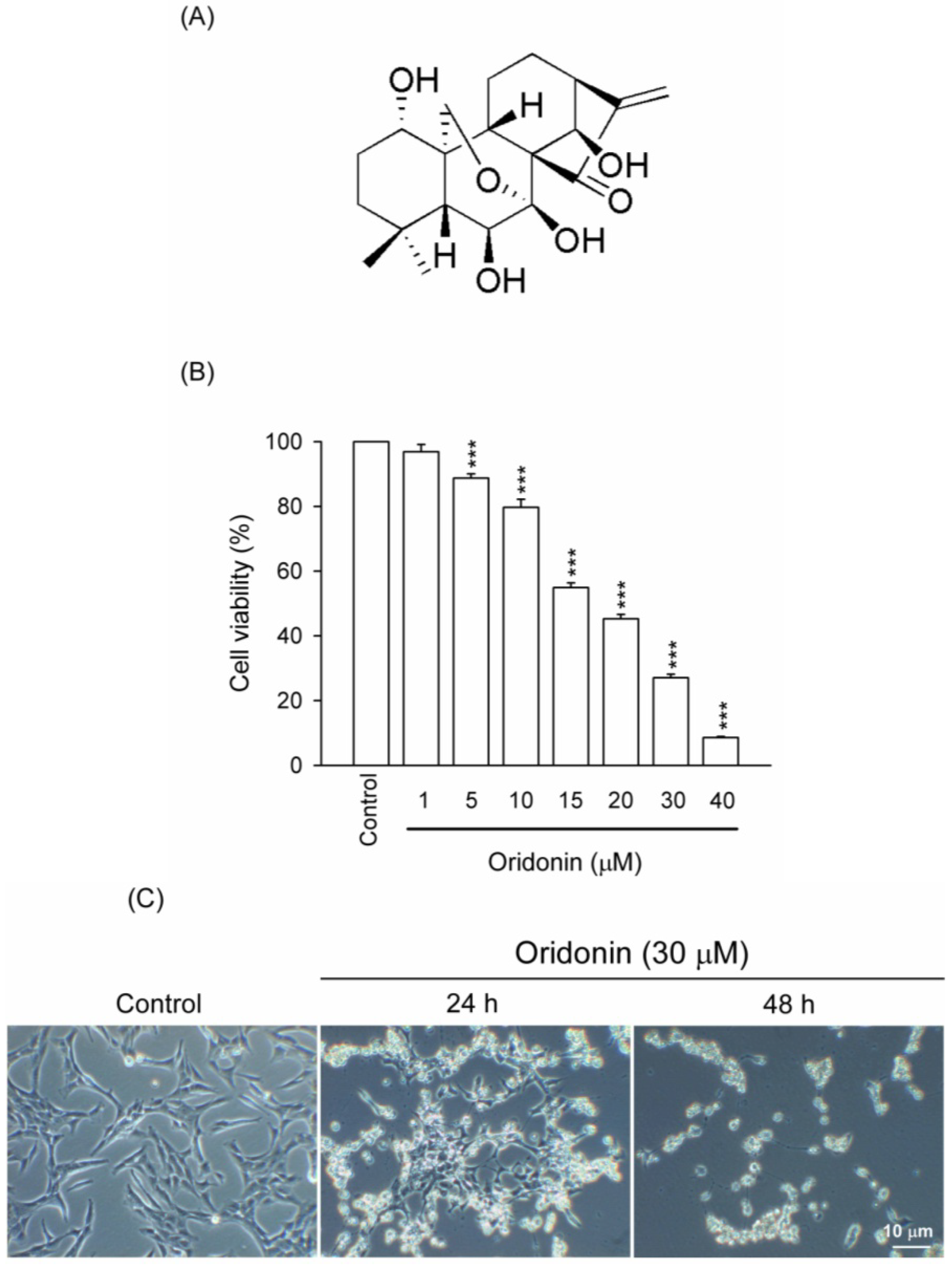
2. Results and Discussion
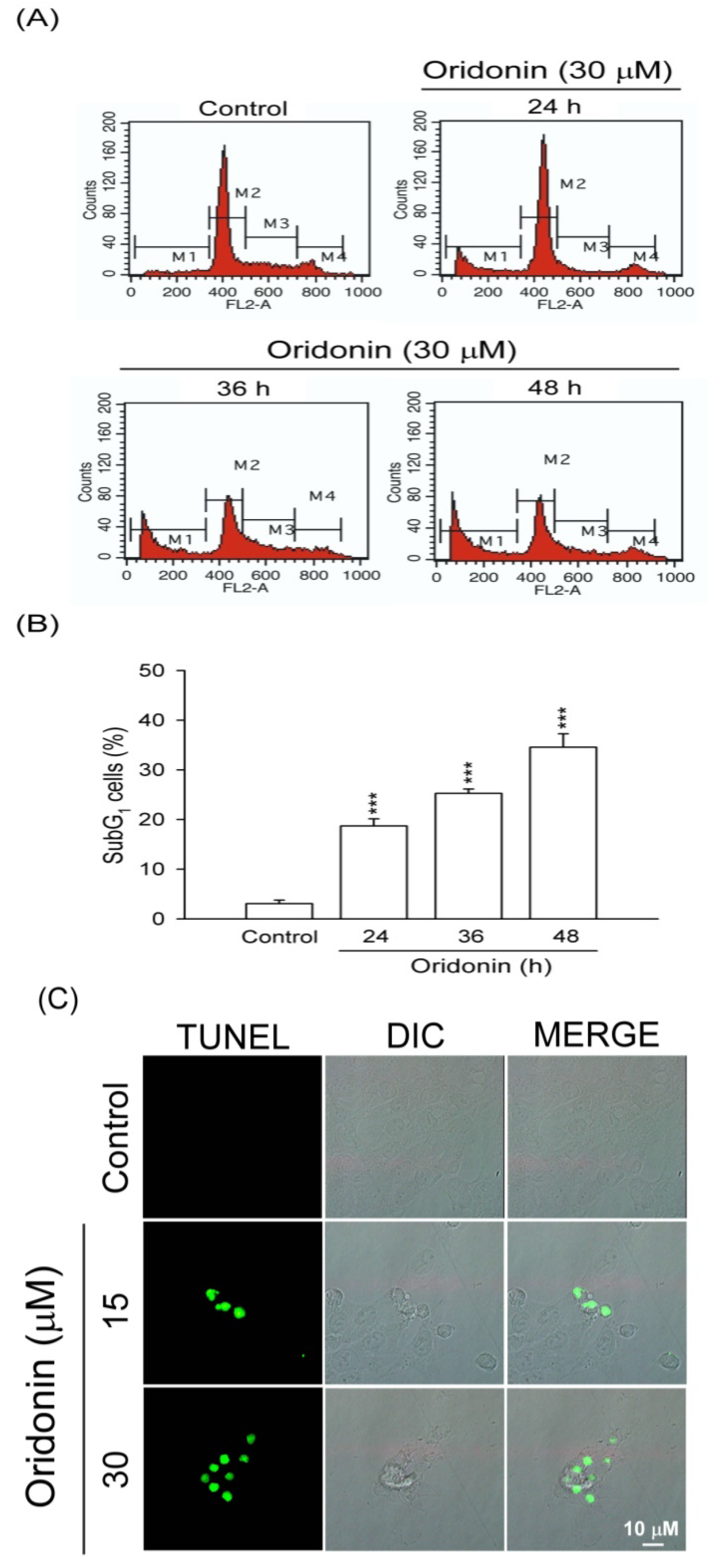
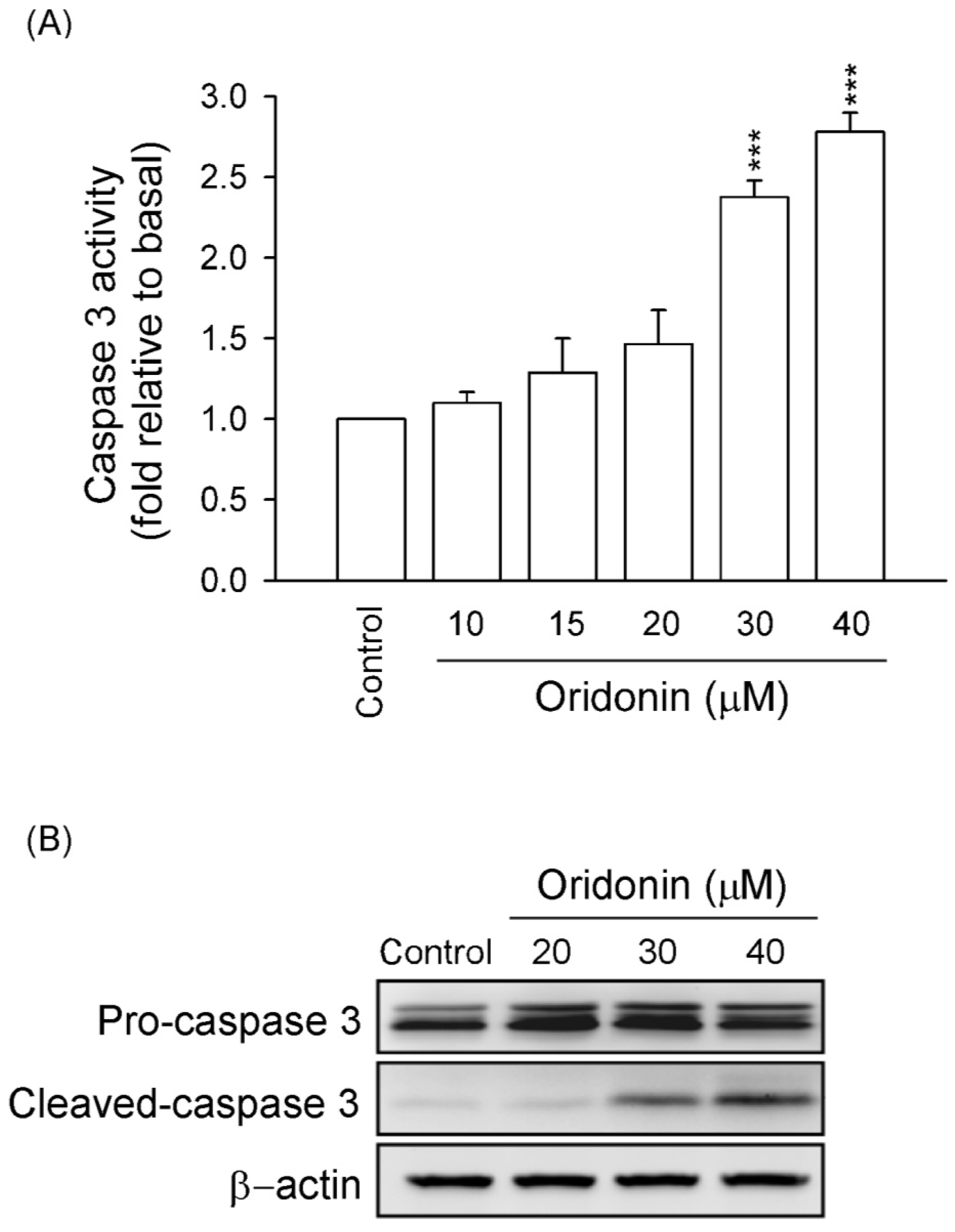
2.1. Oridonin Inhibited Cell Viability and Induced Apoptosis in HSC-T6 Cells
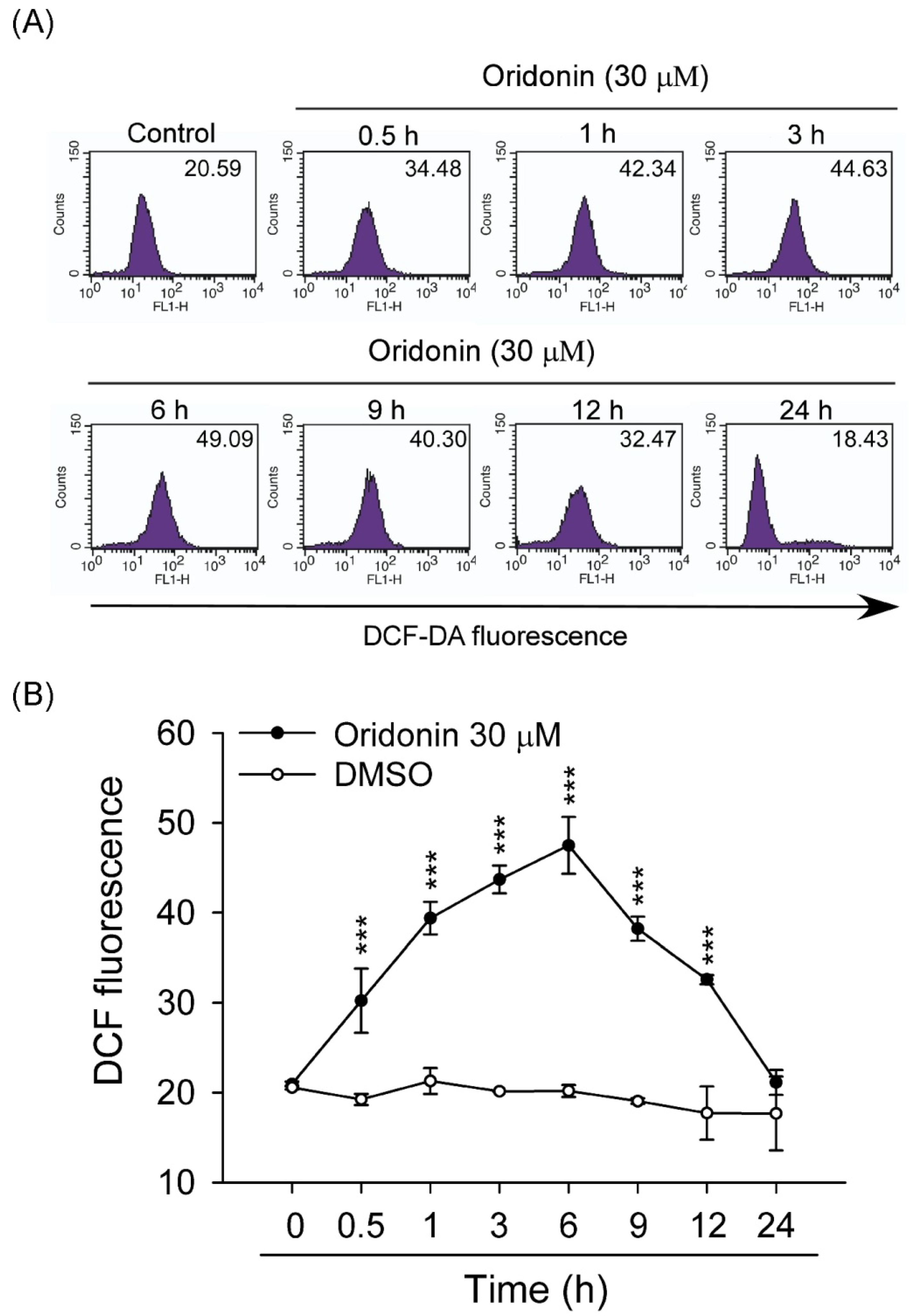
2.2. Oridonin Induced Intracellular ROS Generation
2.3. Oridonin Reduced Intracellular GSH Level

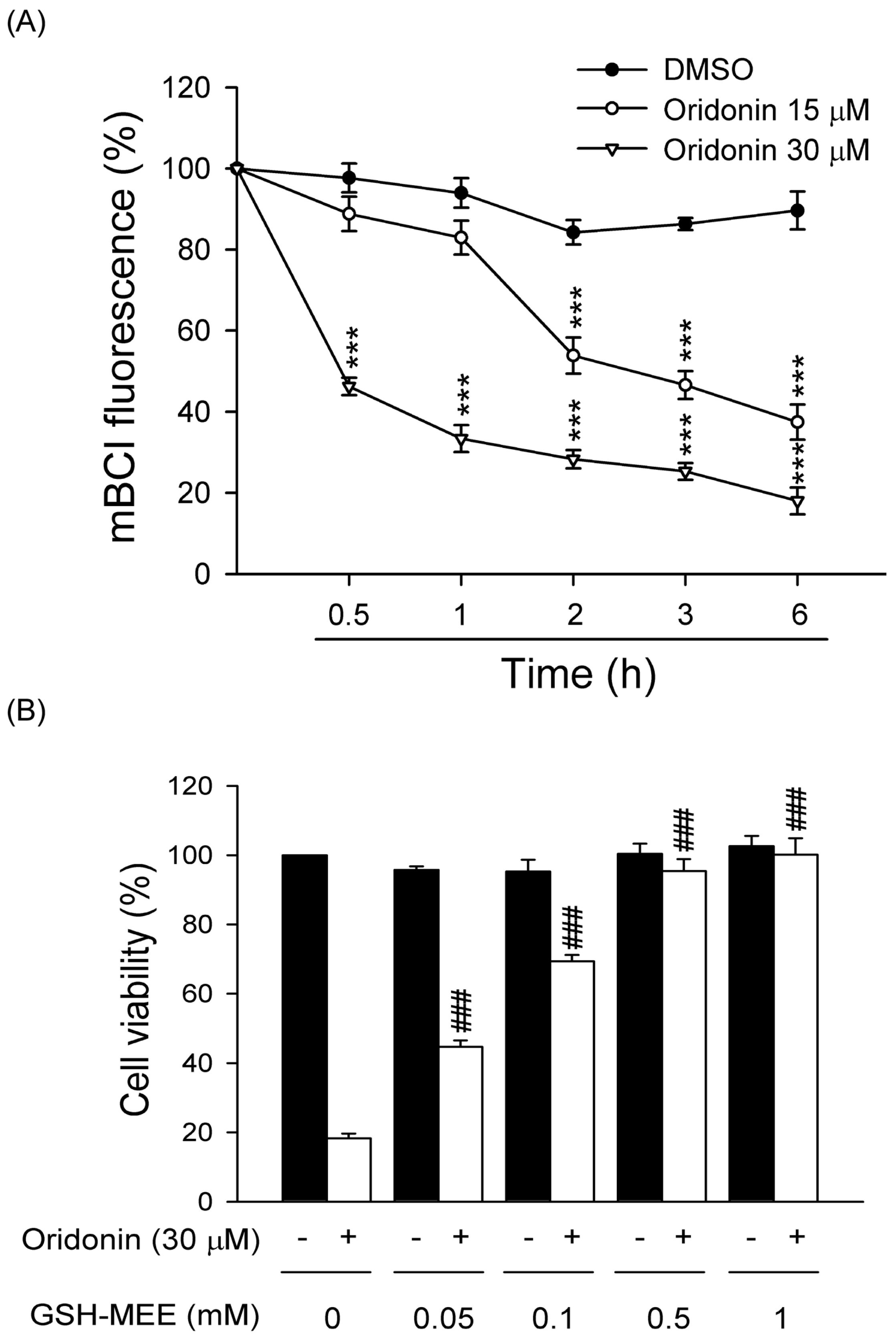
2.4. Effect of Oridonin on Phosphorylation of MAPKs

2.5. NAC Inhibited the Changes of Mitochondrial Membrane Potential, Caspase 3 Activity, and Subg1 Population in Oridonin-Treated HSC-T6 Cells
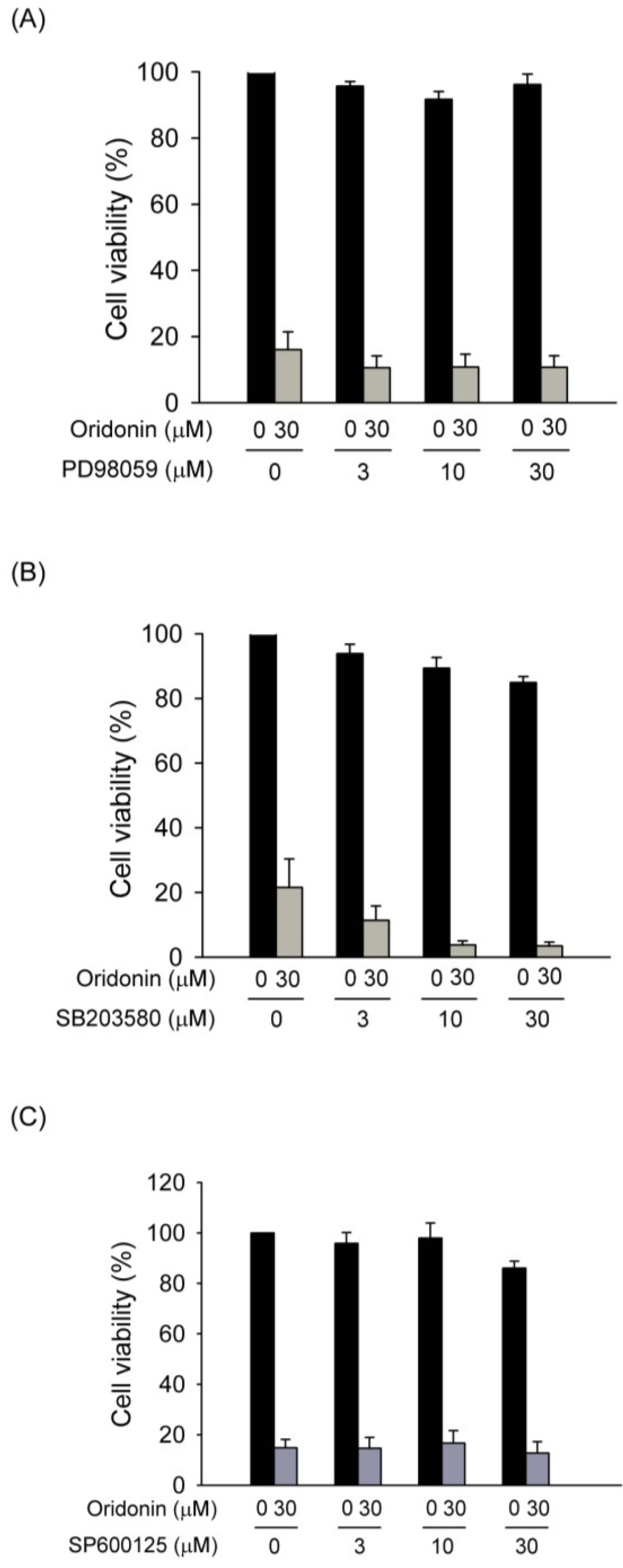
2.6. Discussion

3. Experimental
3.1. Reagents
3.2. Cell Culture
3.3. Cell Viability Assay
3.4. Analysis of Cell Cycle
3.5. Measurement of Intracellular ROS Generation
3.6. Measurement of Intracellular GSH Levels
3.7. Caspase 3 Activity Assay
3.8. Western Blotting
3.9. Assay of Mitochondrial Membrane Potential
3.10. Apoptosis Staining
3.11. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflictts of Interest
References
- Friedman, S.L. Evolving challenges in hepatic fibrosis. Nat. Rev. Gastroenterol. Hepatol. 2010, 7, 425–436. [Google Scholar] [CrossRef]
- Yin, C.; Evason, K.J.; Asahina, K.; Stainier, D.Y. Hepatic stellate cells in liver development, regeneration, and cancer. J. Clin. Invest. 2013, 123, 1902–1910. [Google Scholar] [CrossRef]
- Hernandez-Gea, V.; Friedman, S.L. Pathogenesis of liver fibrosis. Annu. Rev. Pathol. 2011, 6, 425–456. [Google Scholar] [CrossRef]
- Fallowfield, J.A. Therapeutic targets in liver fibrosis. Am. J. Physiol. Gastrointest. Liver Physiol. 2011, 300, G709–G715. [Google Scholar] [CrossRef]
- Wang, X.B.; Feng, Y.; Wang, N.; Cheung, F.; Wong, C.W. Recent progress on anti-liver fibrosis candidates in patents of herbal medicinal products. Recent Pat. Food Nutr. Agric. 2012, 4, 91–106. [Google Scholar] [CrossRef]
- Tian, W.; Chen, S.Y. Recent advances in the molecular basis of anti-neoplastic mechanisms of oridonin. Chin. J. Integr. Med. 2013, 19, 315–320. [Google Scholar] [CrossRef]
- Wong, A.M.; Zhang, Y.; Kesler, K.; Deng, M.; Burhenn, L.; Wang, D.; Moro, A.; Li, Z.; Heber, D. Genomic and in vivo evidence of synergy of a herbal extract compared to its most active ingredient: Rabdosia rubescens vs. oridonin. Exp. Ther. Med. 2010, 1, 1013–1017. [Google Scholar]
- Li, D.; Wu, L.J.; Tashiro, S.; Onodera, S.; Ikejima, T. Oridonin induces human epidermoid carcinoma A431 cell apoptosis through tyrosine kinase and mitochondrial pathway. J. Asian. Nat. Prod. Res. 2008, 10, 77–87. [Google Scholar] [CrossRef]
- Bai, N.; He, K.; Zhou, Z.; Tsai, M.L.; Zhang, L.; Quan, Z.; Shao, X.; Pan, M.H.; Ho, C.T. Ent-kaurane diterpenoids from Rabdosia rubescens and their cytotoxic effects on human cancer cell lines. Planta Med. 2010, 76, 140–145. [Google Scholar] [CrossRef]
- Xu, Y.; Xue, Y.; Wang, Y.; Feng, D.; Lin, S.; Xu, L. Multiple-modulation effects of Oridonin on the production of proinflammatory cytokines and neurotrophic factors in LPS-activated microglia. Int. Immunopharmacol. 2009, 9, 360–365. [Google Scholar] [CrossRef]
- Osawa, K.; Yasuda, H.; Maruyama, T.; Morita, H.; Takeya, K.; Itokawa, H.; Okuda, K. An investigation of diterpenes from the leaves of Rabdosia trichocarpa and their antibacterial activity against oral microorganisms. Chem. Pharm. Bull. (Tokyo) 1994, 42, 922–925. [Google Scholar] [CrossRef]
- Jin, H.; Tan, X.; Liu, X.; Ding, Y. Downregulation of AP-1 gene expression is an initial event in the oridonin-mediated inhibition of colorectal cancer: Studies in vitro and in vivo. J. Gastroenterol. Hepatol. 2011, 26, 706–715. [Google Scholar] [CrossRef]
- Wang, H.; Ye, Y.; Pan, S.Y.; Zhu, G.Y.; Li, Y.W.; Fong, D.W.; Yu, Z.L. Proteomic identification of proteins involved in the anticancer activities of oridonin in HepG2 cells. Phytomedicine 2011, 18, 163–169. [Google Scholar] [CrossRef]
- Kwan, H.Y.; Yang, Z.; Fong, W.F.; Hu, Y.M.; Yu, Z.L.; Hsiao, W.L. The anticancer effect of oridonin is mediated by fatty acid synthase suppression in human colorectal cancer cells. J. Gastroenterol. 2013, 48, 182–192. [Google Scholar] [CrossRef]
- Zhang, D.Y.; Friedman, S.L. Fibrosis-dependent mechanisms of hepatocarcinogenesis. Hepatology 2012, 56, 769–775. [Google Scholar] [CrossRef]
- Kocabayoglu, P.; Friedman, S.L. Cellular basis of hepatic fibrosis and its role in inflammation and cancer. Front. Biosci. (Schol Ed) 2013, 5, 217–230. [Google Scholar]
- Brunati, A.M.; Pagano, M.A.; Bindoli, A.; Rigobello, M.P. Thiol redox systems and protein kinases in hepatic stellate cell regulatory processes. Free Radic. Res. 2010, 44, 363–378. [Google Scholar] [CrossRef]
- Circu, M.L.; Aw, T.Y. Glutathione and apoptosis. Free Radic. Res. 2008, 42, 689–706. [Google Scholar] [CrossRef]
- Dunning, S.; Ur Rehman, A.; Tiebosch, M.H.; Hannivoort, R.A.; Haijer, F.W.; Woudenberg, J.; van den Heuvel, F.A.; Buist-Homan, M.; Faber, K.N.; Moshage, H. Glutathione and antioxidant enzymes serve complementary roles in protecting activated hepatic stellate cells against hydrogen peroxide-induced cell death. Biochim. Biophys. Acta 2013, 1832, 2027–2034. [Google Scholar] [CrossRef]
- Huang, J.; Wu, L.; Tashiro, S.; Onodera, S.; Ikejima, T. Reactive oxygen species mediate oridonin-induced HepG2 apoptosis through p53, MAPK, and mitochondrial signaling pathways. J. Pharmacol. Sci. 2008, 107, 370–379. [Google Scholar] [CrossRef]
- Franco, R.; Cidlowski, J.A. Glutathione efflux and cell death. Antioxid. Redox. Signal. 2012, 17, 1694–1713. [Google Scholar] [CrossRef]
- Issa, R.; Williams, E.; Trim, N.; Kendall, T.; Arthur, M.J.; Reichen, J.; Benyon, R.C.; Iredale, J.P. Apoptosis of hepatic stellate cells: Involvement in resolution of biliary fibrosis and regulation by soluble growth factors. Gut 2001, 48, 548–557. [Google Scholar] [CrossRef]
- Mallat, A.; Lotersztajn, S. Cellular mechanisms of tissue fibrosis. 5. Novel insights into liver fibrosis. Am. J. Physiol. Cell Physiol. 2013, 305, C789–C799. [Google Scholar]
- Schuppan, D.; Kim, Y.O. Evolving therapies for liver fibrosis. J. Clin. Invest. 2013, 123, 1887–1901. [Google Scholar] [CrossRef]
- Lou, H.; Gao, L.; Wei, X.; Zhang, Z.; Zheng, D.; Zhang, D.; Zhang, X.; Li, Y.; Zhang, Q. Oridonin nanosuspension enhances anti-tumor efficacy in SMMC-7721 cells and H22 tumor bearing mice. Colloids Surf. B. Biointerfaces 2011, 87, 319–325. [Google Scholar] [CrossRef]
- Duan, C.; Gao, J.; Zhang, D.; Jia, L.; Liu, Y.; Zheng, D.; Liu, G.; Tian, X.; Wang, F.; Zhang, Q. Galactose-decorated pH-responsive nanogels for hepatoma-targeted delivery of oridonin. Biomacromolecules 2011, 12, 4335–4343. [Google Scholar] [CrossRef]
- Ray, K. Liver: Hepatic stellate cells hold the key to liver fibrosis. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 74. [Google Scholar] [CrossRef]
- Puche, J.E.; Saiman, Y.; Friedman, S.L. Hepatic stellate cells and liver fibrosis. Comp. Physiol. 2013, 3, 1473–1492. [Google Scholar]
- Mederacke, I.; Hsu, C.C.; Troeger, J.S.; Huebener, P.; Mu, X.; Dapito, D.H.; Pradere, J.P.; Schwabe, R.F. Fate tracing reveals hepatic stellate cells as dominant contributors to liver fibrosis independent of its aetiology. Nat. Commun. 2013, 4, 2823. [Google Scholar]
- Bohanon, F.J.; Wang, X.; Ding, C.; Radhakrishnan, G.L.; Rastellini, C.; Zhou, J.; Radhakrishnan, R.S. Oridonin Induces apoptosis and cell cycle arrest in activated Hepatic stellate cells and reduces proliferation and extracellular Matrix protein production. J. Surg. Res. 2014, 186, 547. [Google Scholar]
- Dunning, S.; Hannivoort, R.A.; de Boer, J.F.; Buist-Homan, M.; Faber, K.N.; Moshage, H. Superoxide anions and hydrogen peroxide inhibit proliferation of activated rat stellate cells and induce different modes of cell death. Liver Int. 2009, 29, 922–932. [Google Scholar] [CrossRef]
- Bae, S.; Lee, E.J.; Lee, J.H.; Park, I.C.; Lee, S.J.; Hahn, H.J.; Ahn, K.J.; An, S.; An, I.S.; Cha, H.J. Oridonin protects HaCaT keratinocytes against hydrogen peroxide-induced oxidative stress by altering microRNA expression. Int. J. Mol. Med. 2014, 33, 185–193. [Google Scholar]
- Yu, Y.; Fan, S.M.; Song, J.K.; Tashiro, S.; Onodera, S.; Ikejima, T. Hydroxyl radical .OH played a pivotal role in oridonin-induced apoptosis and autophagy in human epidermoid carcinoma A431 cells. Biol. Pharm. Bull 2012, 35, 2148–2159. [Google Scholar] [CrossRef]
- Britton, R.S.; Bacon, B.R. Role of free radicals in liver diseases and hepatic fibrosis. Hepatogastroenterology 1994, 41, 343–348. [Google Scholar]
- Tsukamoto, H.; Rippe, R.; Niemela, O.; Lin, M. Roles of oxidative stress in activation of Kupffer and Ito cells in liver fibrogenesis. J. Gastroenterol. Hepatol. 1995, 10 (Suppl. 1), S50–S53. [Google Scholar] [CrossRef]
- Kim, J.H.; Jeong, Y.J.; Hong, J.M.; Kim, H.R.; Kang, J.S.; Lee, W.J.; Hwang, Y.I. Chronic vitamin C insufficiency aggravated thioacetamide-induced liver fibrosis in gulo-knockout mice. Free Radic. Biol. Med. 2013, 67, 81–90. [Google Scholar]
- Zhu, M.; Hong, D.; Bao, Y.; Wang, C.; Pan, W. Oridonin induces the apoptosis of metastatic hepatocellular carcinoma cells via a mitochondrial pathway. Oncol. Lett. 2013, 6, 1502–1506. [Google Scholar]
- Dodson, M.; Darley-Usmar, V.; Zhang, J. Cellular metabolic and autophagic pathways: Traffic control by redox signaling. Free Radic. Biol. Med. 2013, 63, 207–221. [Google Scholar] [CrossRef]
- Runchel, C.; Matsuzawa, A.; Ichijo, H. Mitogen-activated protein kinases in mammalian oxidative stress responses. Antioxid. Redox Signal. 2011, 15, 205–218. [Google Scholar] [CrossRef]
- Chowdhury, A.A.; Chaudhuri, J.; Biswas, N.; Manna, A.; Chatterjee, S.; Mahato, S.K.; Chaudhuri, U.; Jaisankar, P.; Bandyopadhyay, S. Synergistic apoptosis of CML cells by buthionine sulfoximine and hydroxychavicol correlates with activation of AIF and GSH-ROS-JNK-ERK-iNOS pathway. PLoS One 2013, 8, e73672. [Google Scholar] [CrossRef]
- Szuster-Ciesielska, A.; Mizerska-Dudka, M.; Daniluk, J.; Kandefer-Szerszen, M. Butein inhibits ethanol-induced activation of liver stellate cells through TGF-beta, NFkappaB, p38, and JNK signaling pathways and inhibition of oxidative stress. J. Gastroenterol. 2013, 48, 222–237. [Google Scholar] [CrossRef]
- Yu, J.Y.; Lee, S.Y.; Son, Y.O.; Shi, X.; Park, S.S.; Lee, J.C. Continuous presence of H2O2 induces mitochondrial-mediated, MAPK- and caspase-independent growth inhibition and cytotoxicity in human gingival fibroblasts. Toxicol. In Vitro 2012, 26, 561–570. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compound are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kuo, L.-M.; Kuo, C.-Y.; Lin, C.-Y.; Hung, M.-F.; Shen, J.-J.; Hwang, T.-L. Intracellular Glutathione Depletion by Oridonin Leads to Apoptosis in Hepatic Stellate Cells. Molecules 2014, 19, 3327-3344. https://doi.org/10.3390/molecules19033327
Kuo L-M, Kuo C-Y, Lin C-Y, Hung M-F, Shen J-J, Hwang T-L. Intracellular Glutathione Depletion by Oridonin Leads to Apoptosis in Hepatic Stellate Cells. Molecules. 2014; 19(3):3327-3344. https://doi.org/10.3390/molecules19033327
Chicago/Turabian StyleKuo, Liang-Mou, Chan-Yen Kuo, Chen-Yu Lin, Min-Fa Hung, Jiann-Jong Shen, and Tsong-Long Hwang. 2014. "Intracellular Glutathione Depletion by Oridonin Leads to Apoptosis in Hepatic Stellate Cells" Molecules 19, no. 3: 3327-3344. https://doi.org/10.3390/molecules19033327
APA StyleKuo, L.-M., Kuo, C.-Y., Lin, C.-Y., Hung, M.-F., Shen, J.-J., & Hwang, T.-L. (2014). Intracellular Glutathione Depletion by Oridonin Leads to Apoptosis in Hepatic Stellate Cells. Molecules, 19(3), 3327-3344. https://doi.org/10.3390/molecules19033327





