4-Aminobenzoic Acid-Coated Maghemite Nanoparticles as Potential Anticancer Drug Magnetic Carriers: A Case Study on Highly Cytotoxic Cisplatin-Like Complexes Involving 7-Azaindoles
Abstract
:1. Introduction
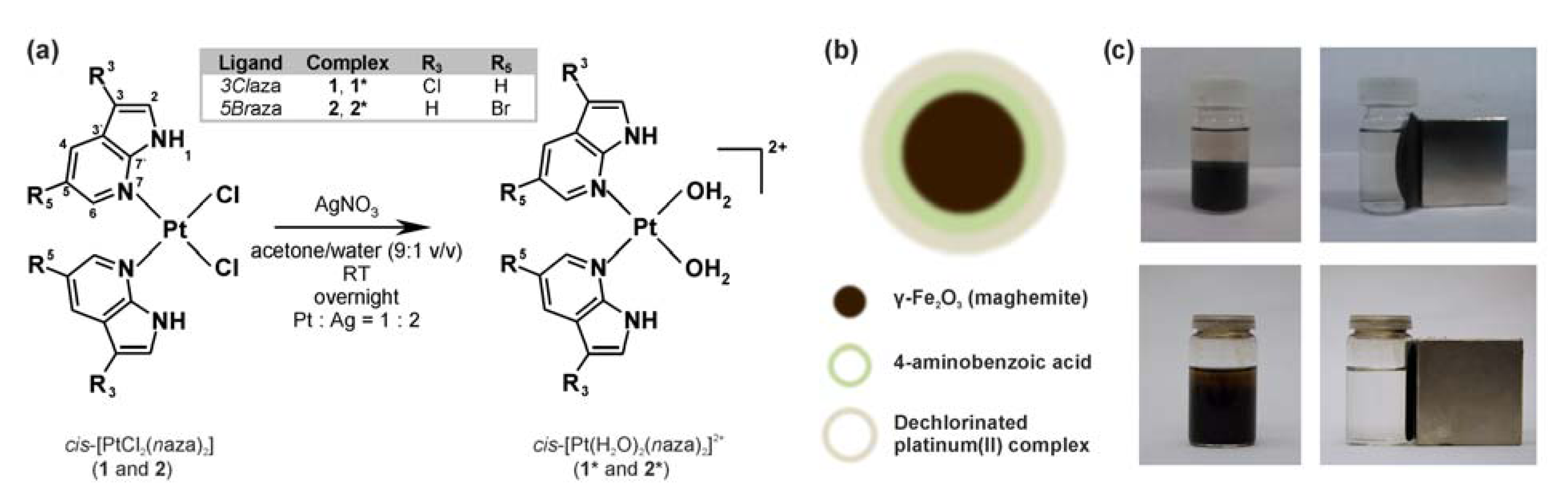
2. Results and Discussion
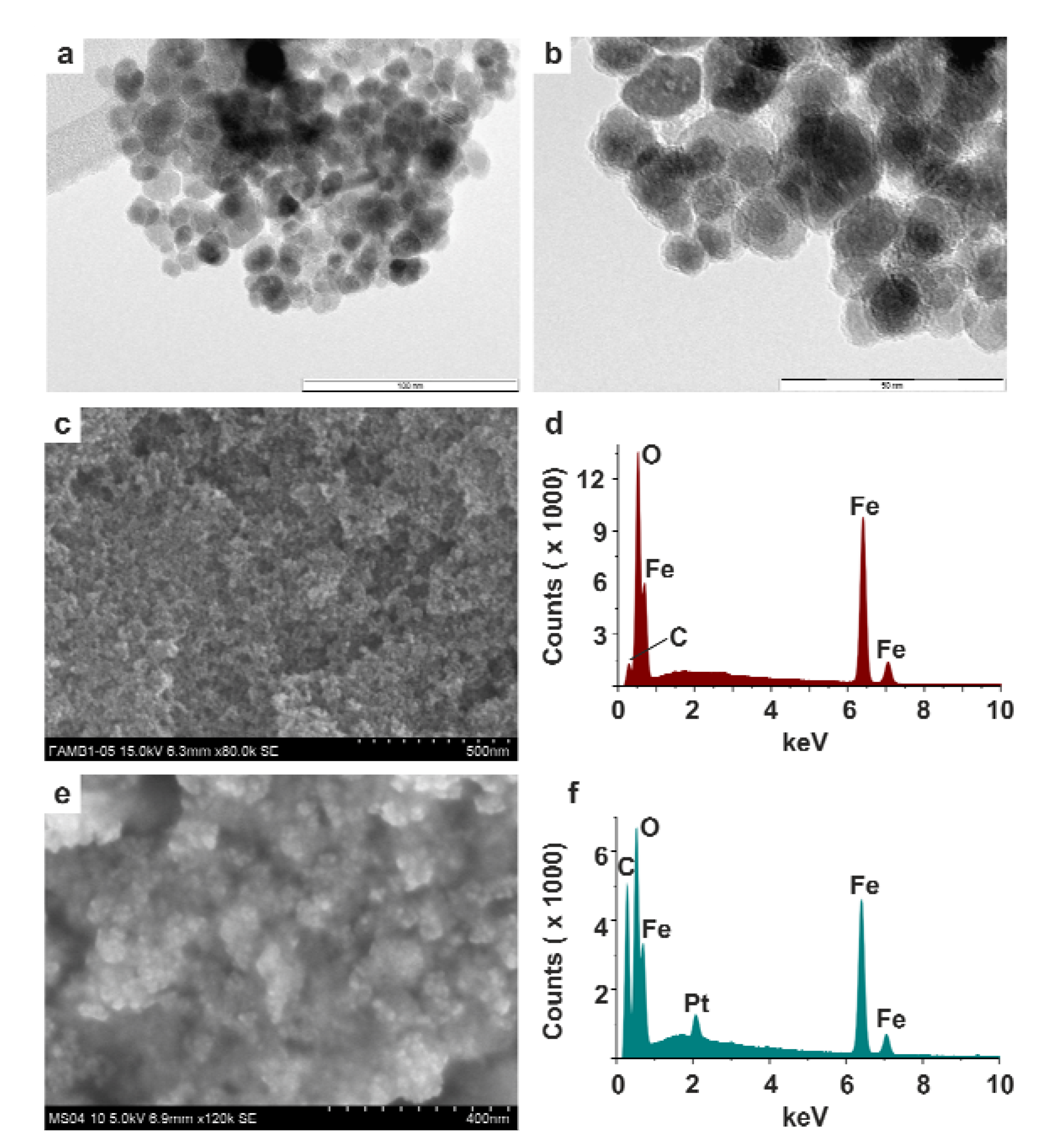
2.1. Preparation and Properties of PABA@FeNPs
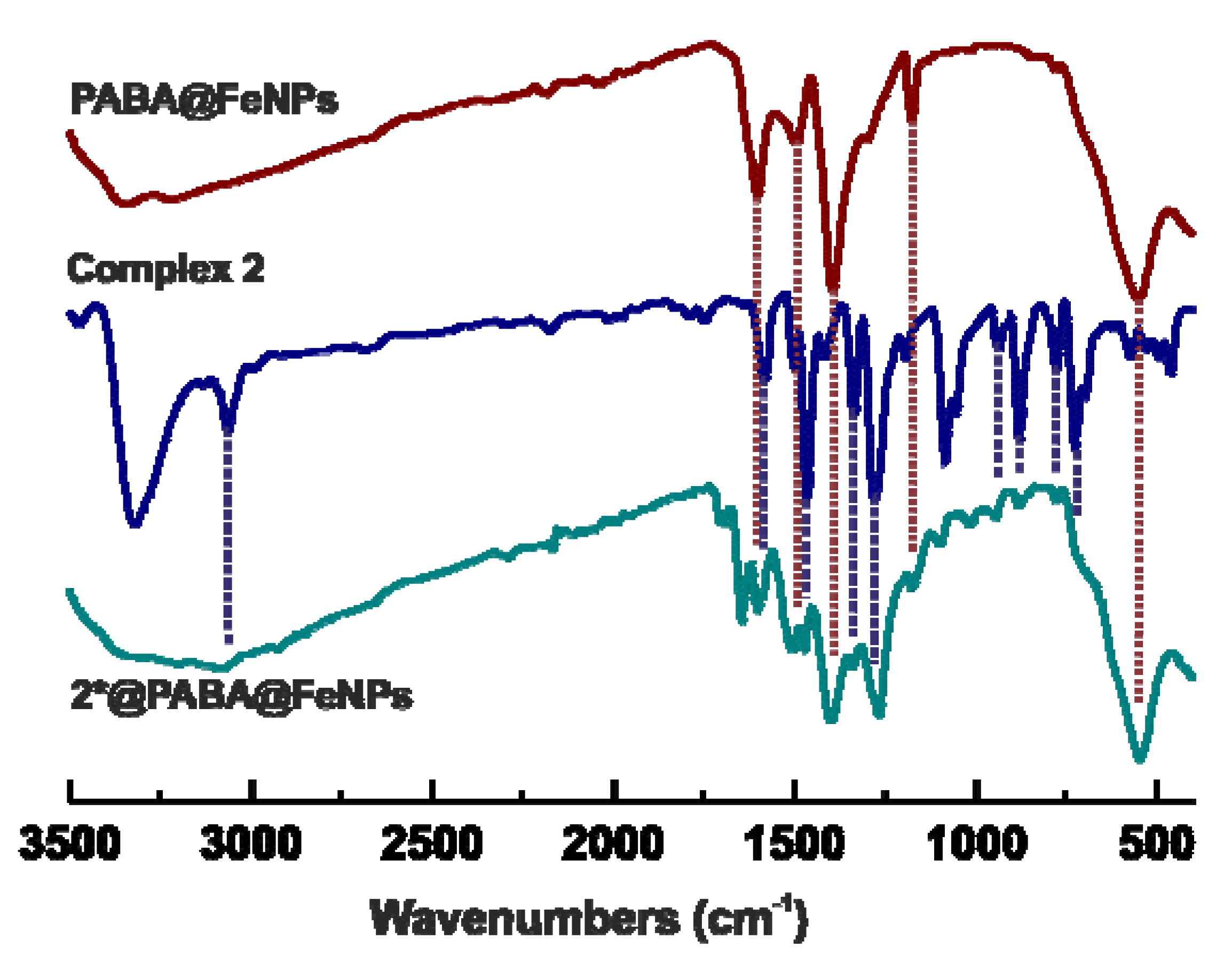
2.2. Functionalization of PABA@FeNPs by Platinum(II) Complexes
2.3. Mössbauer Spectroscopy
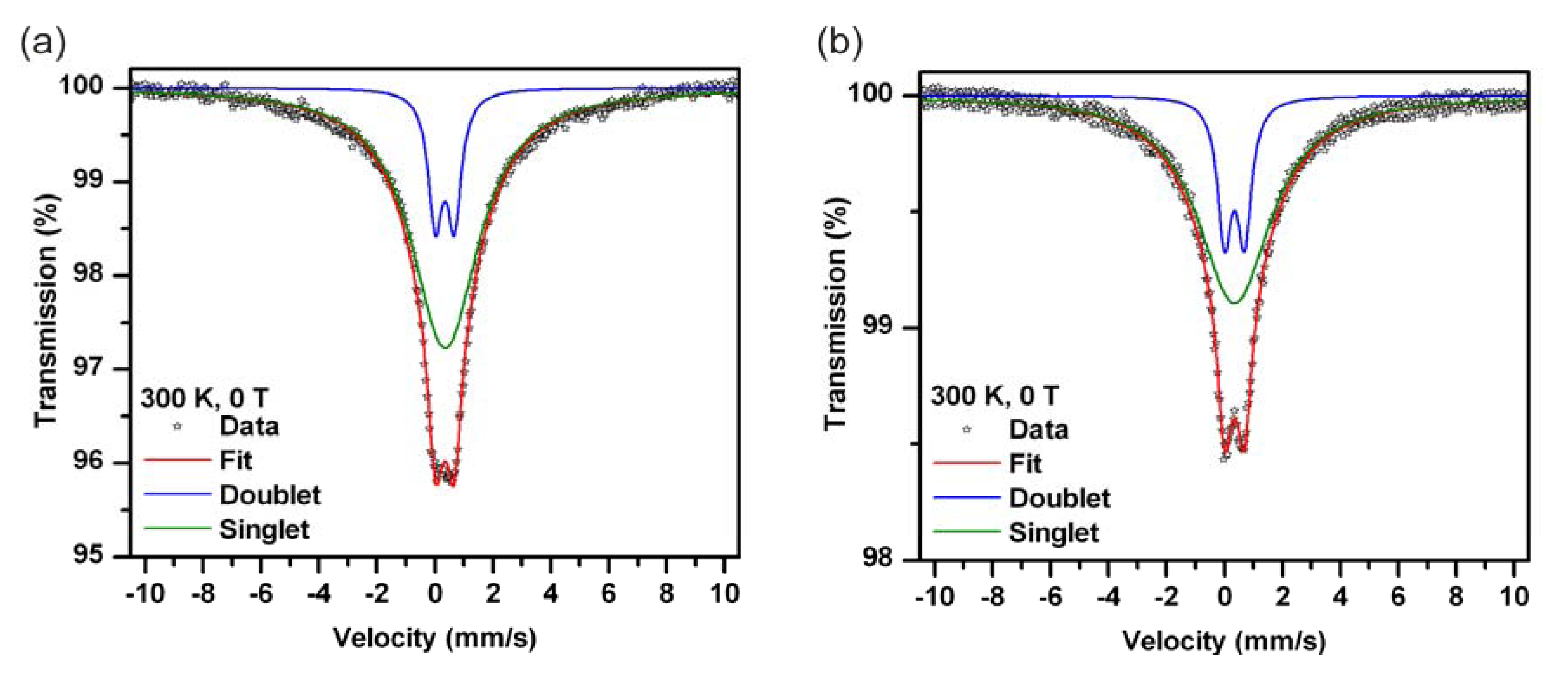
| Sample | Component | δ ± 0.01 (mm/s) | ΔEQ ± 0.01 (mm/s) | RA ± 1 (%) | Assignment |
|---|---|---|---|---|---|
| PABA@FeNPs | Doublet | 0.34 | 0.66 | 18 | Fe3+ relaxation component |
| Singlet | 0.35 | 82 | Fe3+ superparamagnetic component | ||
| 2*PABA@FeNPs | Doublet | 0.35 | 0.69 | 88 | Fe3+ relaxation component |
| Singlet | 0.35 | 78 | Fe3+ superparamagnetic component |
3. Experimental
3.1. Materials and Methods
3.2. PABA@FeNPs Nanoparticles
3.3. Synthesis of 1*@PABA@FeNPs and 2*@PABA@FeNPs
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kelland, L. The resurgence of platinum-based cancer chemotherapy. Nat. Rev. Cancer 2007, 7, 573–584. [Google Scholar] [CrossRef]
- Butler, J.S.; Sadler, P.J. Targeted delivery of platinum-based anticancer complexes. Curr. Opin. Chem. Biol. 2013, 17, 175–188. [Google Scholar] [CrossRef]
- Zhang, Y.; Chan, H.F.; Leong, K.W. Advanced materials and processing for drug delivery: The past and the future. Adv. Drug Deliv. Rev. 2013, 65, 104–120. [Google Scholar] [CrossRef]
- Goncalves, A.S.; Macedo, A.S.; Souto, E.B. Therapeutic nanosystems for oncology nanomedicine. Clin. Transl. Oncol. 2012, 14, 883–890. [Google Scholar] [CrossRef]
- Chomoucka, J.; Drbohlavova, J.; Huska, D.; Adam, V.; Kizek, R.; Hubalek, J. Magnetic nanoparticles and targeted drug delivering. Pharmacol. Res. 2010, 62, 144–149. [Google Scholar] [CrossRef]
- Kievit, F.M.; Zhang, M. Surface Engineering of iron oxide nanoparticles for targeted cancer therapy. Acc. Chem. Res. 2011, 44, 853–862. [Google Scholar] [CrossRef]
- Gupta, A.K.; Gupta, M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 2005, 26, 3995–4021. [Google Scholar] [CrossRef]
- Laurent, S.; Forge, D.; Port, M.; Roch, A.; Robic, C.; Elst, L.V.; Muller, R.N. Magnetic iron oxide nanoparticles: Synthesis, Stabilization, Vectorization, Physicochemical characterizations, and biological applications. Chem. Rev. 2008, 108, 2064–2110. [Google Scholar] [CrossRef]
- Kelkar, S.S.; Reineke, T.M. Theranostics: Combining Imaging and Therapy. Bioconjugate. Chem. 2011, 22, 1879–1903. [Google Scholar] [CrossRef]
- Wang, N.; Guan, Y.; Yang, L.; Jia, L.; Wei, X.; Liu, H.; Guo, G. Magnetic nanoparticles (MNPs) covalently coated by PEO–PPO–PEO block copolymer for drug delivery. J. Colloid Interface Sci. 2013, 395, 50–57. [Google Scholar] [CrossRef]
- Nowicka, A.M.; Kowalczyk, A.; Jarzebinska, A.; Donten, M.; Krysinski, P.; Stojek, Z. Progress in targeting tumor cells by using drug-magnetic nanoparticles conjugate. Biomacromolecules 2013, 14, 828–833. [Google Scholar] [CrossRef]
- Karsten, S.; Nan, A.; Turcu, R.; Liebscher, J. A new access to polypyrrole-based functionalized magnetic core-shell nanoparticles. J. Polym. Sci. Part. A Polym. Chem. 2012, 50, 3986–3995. [Google Scholar] [CrossRef]
- Fan, C.H.; Ting, C.Y.; Lin, H.J.; Wang, C.H.; Liu, H.L.; Yen, T.C.; Yeh, C.K. SPIO-conjugated, doxorubicin-loaded microbubbles for concurrent MRI and focused-ultrasound enhanced brain-tumor drug delivery. Biomaterials 2013, 34, 3706–3715. [Google Scholar] [CrossRef]
- Gautiera, J.; Munniera, E.; Paillard, A.; Hervéa, K.; Douziech-Eyrollesa, L.; Soucé, M.; Duboisa, P.; Chourpa, I. A pharmaceutical study of doxorubicin-loaded PEGylated nanoparticles for magnetic drug targeting. Int. J. Pharm. 2012, 423, 16–25. [Google Scholar] [CrossRef]
- Kim, J.E.; Shin, J.Y.; Cho, M.H. Magnetic nanoparticles: An update of application for drug delivery and possible toxic effects. Arch. Toxicol. 2012, 86, 685–700. [Google Scholar] [CrossRef]
- Qi, L.; Wu, L.; Zheng, S.; Wang, Y.; Fu, H.; Cui, D. Cell-Penetrating Magnetic Nanoparticles for Highly Efficient Delivery and Intracellular Imaging of siRNA. Biomacromolecules 2012, 13, 2723–2730. [Google Scholar] [CrossRef]
- Huang, P.; Li, Z.; Lin, J.; Yang, D.; Gao, G.; Xu, C.; Bao, L.; Zhang, C.; Wang, K.; Song, H.; et al. Photosensitizer-conjugated magnetic nanoparticles for in vivo simultaneous magnetofluorescent imaging and targeting therapy. Biomaterials 2011, 32, 3447–3458. [Google Scholar] [CrossRef]
- Pan, B.; Cui, D.; Sheng, Y.; Ozkan, C.; Gao, F.; He, R.; Li, Q.; Xu, P.; Huang, T. Dendrimer-modified magnetic nanoparticles enhance efficiency of gene delivery system. Cancer Res. 2007, 67, 8156–8163. [Google Scholar] [CrossRef]
- Knezevic, N.Z.; Ruiz-Hernandez, E.; Hennink, W.E.; Vallet-Regi, M. Magnetic mesoporous silica-based core/shellnanoparticlesfor biomedical applications. RSC Adv. 2013, 3, 9584–9593. [Google Scholar] [CrossRef]
- Wang, J.; Wang, X.; Song, Y.; Wang, J.; Zhang, C.; Chang, C.; Yan, J.; Qiu, L.; Wua, M.; Guo, Z. A platinum anticancer theranostic agent with magnetic targeting potential derived from maghemite nanoparticles. Chem. Sci. 2013, 4, 2605–2612. [Google Scholar] [CrossRef]
- Wang, J.; Wang, X.; Song, Y.; Zhu, C.; Wang, J.; Wang, K.; Guo, Z. Detecting and delivering platinum anticancer drugs using fluorescent maghemite nanoparticles. Chem. Commun. 2013, 49, 2786–2788. [Google Scholar] [CrossRef]
- Deng, H.; Lei, Z. Preparation and characterization of hollow Fe3O4/SiO2@PEG–PLA nanoparticles for drug delivery. Composites Part. B 2013, 54, 194–199. [Google Scholar] [CrossRef]
- Devi, S.V.; Prakash, T. Kinetics of cisplatin release by in-vitro using poly(d,l-Lactide) coated Fe3O4 nanocarriers. IEEE Trans. Nanobiosci. 2013, 12, 60–63. [Google Scholar] [CrossRef]
- Ashjari, M.; Khoee, S.; Mahdavian, A.R. Controlling the morphology and surface property of magnetic/cisplatin-loaded nanocapsules via W/O/W double emulsion method. Colloids Surf. A Physicochem. Eng. Asp. 2012, 408, 87–96. [Google Scholar] [CrossRef]
- Xie, M.; Xu, Y.; Liu, J.; Zhang, T.; Zhang, H. Preparation and characterization of Folate targeting magnetic nanomedicine loaded with cisplatin. J. Nanomater. 2012, 2012, 921034. [Google Scholar]
- Arias, J.L.; Reddy, L.H.; Othman, M.; Gillet, B.; Desmaele, D.; Zouhiri, F.; Dosio, F.; Gref, R.; Couvreur, P. Squalene based nanocomposites: A new platform for the design of multifunctional pharmaceutical theragnostics. ACS Nano. 2011, 5, 1513–1521. [Google Scholar] [CrossRef]
- Xing, R.; Wang, X.; Zhang, C.; Wang, J.; Zhang, Y.; Song, Y.; Guo, Z. Superparamagnetic magnetite nanocrystal clusters as potential magnetic carriers for the delivery of platinum anticancer drugs. J. Mater. Chem. 2011, 21, 11142–11149. [Google Scholar]
- Sonoda, A.; Nitta, N.; Nitta-Seko, A.; Ohta, S.; Takamatsu, S.; Ikehata, Y.; Nagano, I.; Jo, J.; Tabata, Y.; Takahashi, M.; et al. Complex comprised of dextran magnetite and conjugated cisplatin exhibiting selective hyperthermic and controlled-release potential. Int. J. Nanomed. 2010, 5, 499–504. [Google Scholar]
- Yang, J.; Lee, H.; Hyung, W.; Park, S.B.; Haam, S. Magnetic PECA nanoparticles as drug carriers for targeted delivery: Synthesis and release characteristics. J. Microencapsul. 2006, 23, 203–212. [Google Scholar] [CrossRef]
- Li, K.; Chen, B.; Xu, L.; Feng, J.; Xia, G.; Cheng, J.; Wang, J.; Gao, F.; Wang, X. Reversal of multidrug resistance by cisplatin-loaded magnetic Fe3O4 nanoparticles in A549/DDP lung cancer cells in vitro and in vivo. Int. J. Nanomed. 2013, 8, 1867–1877. [Google Scholar]
- Zhang, Z.; Chai, A. Core-shell magnetite-silica composite nanoparticles enhancing DNA damage induced by a photoactive platinum-diimine complex in red light. J. Inor. Biochem. 2012, 117, 71–76. [Google Scholar] [CrossRef]
- Štarha, P.; Marek, J.; Trávníček, Z. Cisplatin and oxaliplatin derivatives involving 7-azaindole: Structural characterisations. Polyhedron 2012, 33, 104–409. [Google Scholar]
- Štarha, P.; Trávníček, Z.; Popa, A.; Popa, I.; Muchová, T.; Brabec, V. How to modify 7-azaindole to form cytotoxic Pt(II) complexes: Highly in vitro anticancer effective cisplatin derivatives involving halogeno-substituted 7-azaindole. J. Inorg. Biochem. 2012, 105, 57–63. [Google Scholar]
- Muchová, T.; Prachařová, J.; Štarha, P.; Olivová, R.; Vrána, O.; Benešová, B.; Kašpárková, J.; Trávníček, Z.; Brabec, V. Insight into the toxic effects ofcis-dichloridoplatinum(II) complexes containing 7-azaindole halogeno derivatives in tumor cells. J. Biol. Inorg. Chem. 2013, 18, 579–589. [Google Scholar] [CrossRef]
- Maity, D.; Zoppellaro, G.; Sedenkova, V.; Tucek, J.; Safarova, K.; Polakova, K.; Tomankova, K.; Diwoky, C.; Stollberger, R.; Machala, L.; et al. Surface design of core–shell superparamagnetic iron oxide nanoparticles drives record relaxivity values in functional MRI contrast agents. Chem. Commun. 2012, 48, 11398–11400. [Google Scholar] [CrossRef]
- Hamoudeh, M.; Al Faraj, A.; Canet-Soulas, E.; Bessueille, F.; Leonard, D.; Fessi, H. Pharmaceutical nanotechnology elaboration of PLLA-based superparamagnetic nanoparticles: Characterization, magnetic behaviour study and in vitro relaxivity evaluation. Int. J. Pharm. 2007, 338, 248–257. [Google Scholar] [CrossRef]
- Dorniani, D.; Bin Hussein, M.Z.; Kura, A.U.; Fakurazi, S.; Shaari, A.H.; Ahmad, Z. Preparation of Fe3O4 magnetic nanoparticles coated with gallic acid for drug delivery. Int. J. Nanomed. 2012, 7, 5745–5756. [Google Scholar]
- Cafaggi, S.; Esposito, M.; Parodi, B.; Viale, M. A water-soluble 1,2-diaminocyclohexaneplatinum(II) complex containing procaine hydrochloride: Synthesis and antiproliferative activity in vitro. Pharmazie 1994, 49, 617–618. [Google Scholar]
- Kukushkin, Y.N.; Blyumental, T.O.; Konovalov, L.V. Complex compounds of platinum(II) with p- and m-aminobenzoic acids. Zh. Obshch. Khim. 1979, 49, 1376–1379. [Google Scholar]
- Rudyi, R.I.; Solomentseva, A.I.; Cherkashina, N.V.; Evstafeva, O.N.; Salyn, Y.V.; Moiseev, I.I. Complexes of platinum(II) and platinum(IV) with aromatic amines. Koord. Khim. 1976, 2, 499–506. [Google Scholar]
- Davies, G.R.; Hewertson, W.; Mais, R.H.B.; Owston, P.G.; Patel, C.G. π-Complexes of platinum (II) with unsaturated hydrocarbons. Part II. Crystal and molecular structure of trans-dichloro-(π-di-t-butylacetyl-ene)-p-toluidineplatinum (II). J. Chem. Soc. A 1970, 1873–1877. [Google Scholar]
- Rochon, F.D.; Bonnier, C. Study of Pt(II)-aromatic amines complexes of the types cis- and trans-Pt(amine)2I2, [Pt(amine)4]I2 and I(amine)Pt(μ-I)2Pt(amine)I. Inorg. Chim. Acta 2007, 360, 461–472. [Google Scholar]
- Karthikeyan, B. Density functional calculations on the structure, vibrational frequencies and normal modes of 7-Azaindole. Spectrochim. Acta 2006, A64, 1083–1087. [Google Scholar] [CrossRef]
- Ye, X.; Lin, D.; Jiao, Z.; Zhang, L. The thermal stability of nanocrystalline maghemite Fe2O3. J. Phys. D-Appl. Phys. 1998, 31, 2739–2744. [Google Scholar] [CrossRef]
- Cornell, R.M.; Schwertmann, U. The Iron Oxides: Structure, Properties, Reactions, Occurrence and Uses; Wiley-VCH Publishers: Weinheim, Germany, 2003. [Google Scholar]
- Prochazka, R.; Tucek, P.; Tucek, J.; Marek, J.; Mashlan, M.; Pechousek, J. Statistical analysis and digital processing of the Mössbauer spectra. Meas. Sci. Technol. 2010, 21, 025107. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1, 2, 1*, 2*, PABA@FeNPs, 1*PABA@FeNPs and 2*PABA@FeNPs are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Štarha, P.; Stavárek, M.; Tuček, J.; Trávníček, Z. 4-Aminobenzoic Acid-Coated Maghemite Nanoparticles as Potential Anticancer Drug Magnetic Carriers: A Case Study on Highly Cytotoxic Cisplatin-Like Complexes Involving 7-Azaindoles. Molecules 2014, 19, 1622-1634. https://doi.org/10.3390/molecules19021622
Štarha P, Stavárek M, Tuček J, Trávníček Z. 4-Aminobenzoic Acid-Coated Maghemite Nanoparticles as Potential Anticancer Drug Magnetic Carriers: A Case Study on Highly Cytotoxic Cisplatin-Like Complexes Involving 7-Azaindoles. Molecules. 2014; 19(2):1622-1634. https://doi.org/10.3390/molecules19021622
Chicago/Turabian StyleŠtarha, Pavel, Martin Stavárek, Jiří Tuček, and Zdeněk Trávníček. 2014. "4-Aminobenzoic Acid-Coated Maghemite Nanoparticles as Potential Anticancer Drug Magnetic Carriers: A Case Study on Highly Cytotoxic Cisplatin-Like Complexes Involving 7-Azaindoles" Molecules 19, no. 2: 1622-1634. https://doi.org/10.3390/molecules19021622
APA StyleŠtarha, P., Stavárek, M., Tuček, J., & Trávníček, Z. (2014). 4-Aminobenzoic Acid-Coated Maghemite Nanoparticles as Potential Anticancer Drug Magnetic Carriers: A Case Study on Highly Cytotoxic Cisplatin-Like Complexes Involving 7-Azaindoles. Molecules, 19(2), 1622-1634. https://doi.org/10.3390/molecules19021622