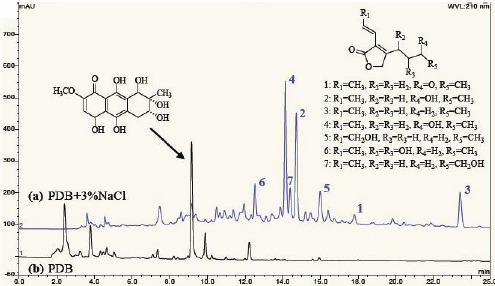
Pestalafuranones F–J, Five New Furanone Analogues from the Endophytic Fungus Nigrospora sp. BM-2
Abstract
:1. Introduction
2. Results and Discussion
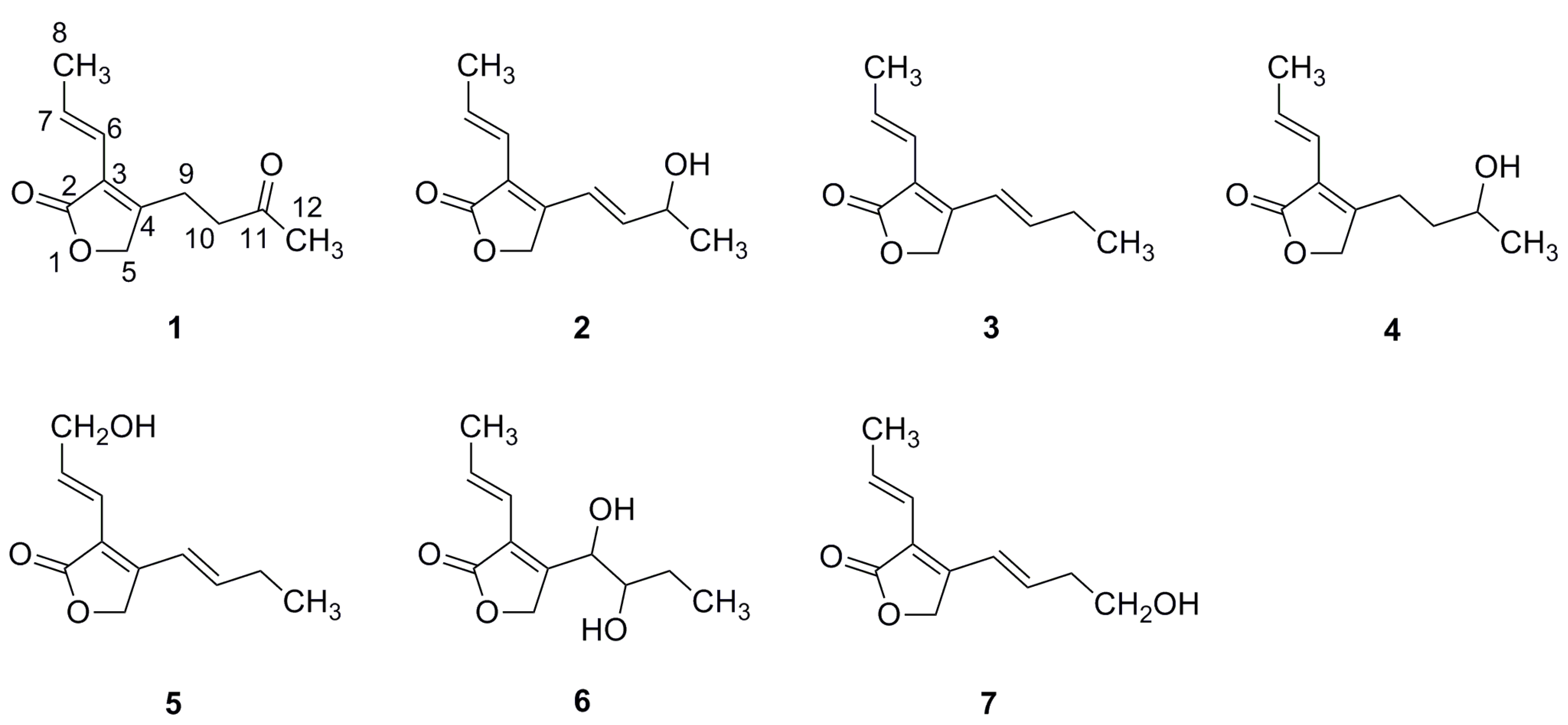
2.1. Structural Characterization
| No. | 3 | 4 | 5 | 6 | 7 |
|---|---|---|---|---|---|
| 5 | 4.85, s | 4.69, s | 4.89, s | 4.89, d (3.5) | 4.91, s |
| 6 | 6.23, dd (1.6, 16.0) | 6.12, dd (1.0, 15.8) | 6.51, dt (1.8, 16.0) | 6.08, dd (1.5, 15.7) | 6.28, dd (1.5, 16.0) |
| 7 | 6.92, m | 6.87, m | 7.08, m | 6.97, m | 6.99, m |
| 8 | 1.89, d (6.8) | 1.87, d (6.6) | 4.36, d (4.8) | 1.86, d (6.3) | 1.94, d (6.8) |
| 9 | 6.54, d (16.0) | 2.61, m | 6.59, d (16.0) | 4.87, d (4.0) | 6.70, d (16.0) |
| 10 | 6.07, dt (6.6, 16.0) | 1.64, m | 6.13, dt (6.6, 16.0) | 3.87, dt (4.0, 8.1) | 6.08, dt (7.2, 16.0) |
| 11 | 2.28, m | 3.84, dt (6.2, 12.3) | 2.30, m | 1.40, m | 2.57, q (6.1) |
| 12 | 1.10, t (7.4) | 1.26, dd (0.6, 6.8) | 1.10, t (7.4) | 1.00, t (7.4) | 3.84, t (6.1) |
| No. | 3 | 4 | 5 | 6 | 7 |
|---|---|---|---|---|---|
| 2 | 173.3, qC | 173.2, qC | 173.1, qC | 172.7, qC | 173.1, qC |
| 3 | 120.7, qC | 122.9, qC | 119.8, qC | 124.3, qC | 121.4, qC |
| 4 | 150.6, qC | 158.1, qC | 152.6, qC | 155.4, qC | 149.8, qC |
| 5 | 68.5, CH2 | 70.7, CH2 | 68.6, CH2 | 69.8, CH2 | 68.5, CH2 |
| 6 | 118.6, CH | 118.3, CH | 116.7, CH | 118.1, CH | 118.5, CH |
| 7 | 133.3, CH | 132.6, CH | 135.1, CH | 134.3, CH | 133.8, CH |
| 8 | 19.4, CH3 | 19.3, CH3 | 63.4, CH2 | 19.3, CH3 | 19.4, CH3 |
| 9 | 119.5, CH | 23.2, CH2 | 119.3, CH | 70.1, CH | 122.6, CH |
| 10 | 140.8, CH | 36.7, CH2 | 141.9, CH | 75.4, CH | 135.0, CH |
| 11 | 26.6, CH2 | 67.3, CH | 26.6, CH2 | 25.0, CH2 | 36.7, CH2 |
| 12 | 12.9, CH3 | 23.9, CH3 | 12.8, CH3 | 10.2, CH3 | 61.5, CH2 |
2.2. Cytotoxic Activities
3. Experimental
3.1. General
3.2. Fungal Material
3.3. Extraction and Isolation
3.4. Cytotoxicity Testing
4. Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Kogej, T.; Gostincar, C.; Volkmann, M.; Gorbushina, A.A.; Gunde-Cimerman, N. Mycosporines in extremophilic fungi-novel complementary osmolytes? Environ. Chem. 2006, 3, 105–110. [Google Scholar] [CrossRef]
- Tamburini, E.; Mastromei, G. Do bacterial cryptic genes really exist? Res. Microbiol. 2000, 151, 179–182. [Google Scholar] [CrossRef]
- Peng, X.P.; Wang, Y.; Liu, P.P.; Hong, K.; Chen, H.; Yin, X.; Zhu, W.M. Aromatic compounds from the Halotolerant Fungal strain of Wallemia. sebi PXP-89 in a hypersaline medium. Arch. Pharm. Res. 2011, 34, 907–912. [Google Scholar] [CrossRef]
- Zheng, J.K.; Xu, Z.H.; Wang, Y.; Hong, K.; Liu, P.P.; Zhu, W.M. Cyclic tripeptides from the Halotolerant Fungus Aspergillus sclerotiorum PT06–1. J. Nat. Prod. 2010, 73, 1133–1137. [Google Scholar] [CrossRef]
- Peng, X.P.; Wang, Y.; Sun, K.L.; Liu, P.P.; Yin, X.; Zhu, W.M. Cerebrosides and 2-pyridone alkaloids from the Halotolerant Fungus Penicillium. chrysogenum grown in a hypersaline medium. J. Nat. Prod. 2011, 74, 1298–1302. [Google Scholar] [CrossRef]
- Wu, Z.; Raven, P.H. Flora of China, Volume 22: Poaceae-Saccharum Linn.; Missouri Botanical Garden Press: St. Louis, MO, USA; Science Press: Beijing, China, 2006. [Google Scholar]
- Liu, H.T.; Liu, S.C.; Guo, L.D.; Zhang, Y.G.; Cui, L.J.; Ding, G. New furanones from the plant endophytic fungus Pestalotiopsis. besseyi. Molecules 2012, 17, 14015–14021. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are not available from the authors because bioactivity tests of those compounds are going on.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, H.; Deng, Z.; Guo, Z.; Tu, X.; Wang, J.; Zou, K. Pestalafuranones F–J, Five New Furanone Analogues from the Endophytic Fungus Nigrospora sp. BM-2. Molecules 2014, 19, 819-825. https://doi.org/10.3390/molecules19010819
Zhang H, Deng Z, Guo Z, Tu X, Wang J, Zou K. Pestalafuranones F–J, Five New Furanone Analogues from the Endophytic Fungus Nigrospora sp. BM-2. Molecules. 2014; 19(1):819-825. https://doi.org/10.3390/molecules19010819
Chicago/Turabian StyleZhang, Hongqi, Zhangshuang Deng, Zhiyong Guo, Xuan Tu, Junzhi Wang, and Kun Zou. 2014. "Pestalafuranones F–J, Five New Furanone Analogues from the Endophytic Fungus Nigrospora sp. BM-2" Molecules 19, no. 1: 819-825. https://doi.org/10.3390/molecules19010819
APA StyleZhang, H., Deng, Z., Guo, Z., Tu, X., Wang, J., & Zou, K. (2014). Pestalafuranones F–J, Five New Furanone Analogues from the Endophytic Fungus Nigrospora sp. BM-2. Molecules, 19(1), 819-825. https://doi.org/10.3390/molecules19010819