Synthesis and Photophysical Property Studies of the 2,6,8-Triaryl-4-(phenylethynyl)quinazolines
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis of the 2-Aryl-6,8-dibromoquinazolin-4(3H)-Ones
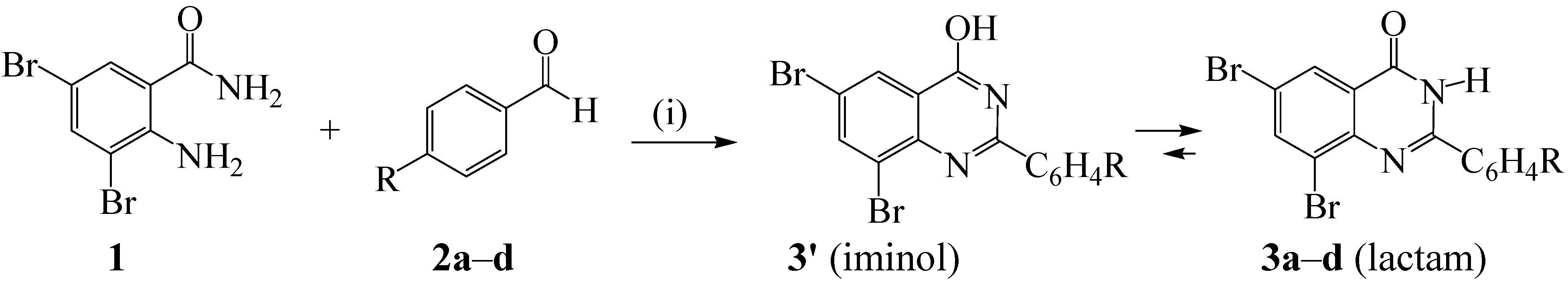
| R | %Yield | |
|---|---|---|
| 3a | 4-H | 80 |
| 3b | 4-F | 89 |
| 3c | 4-Cl | 94 |
| 3d | 4-OMe | 91 |
2.2. Oxidative Aromatization of 2-Aryl-6,8-dibromoquinazolin-4(3H)-Ones
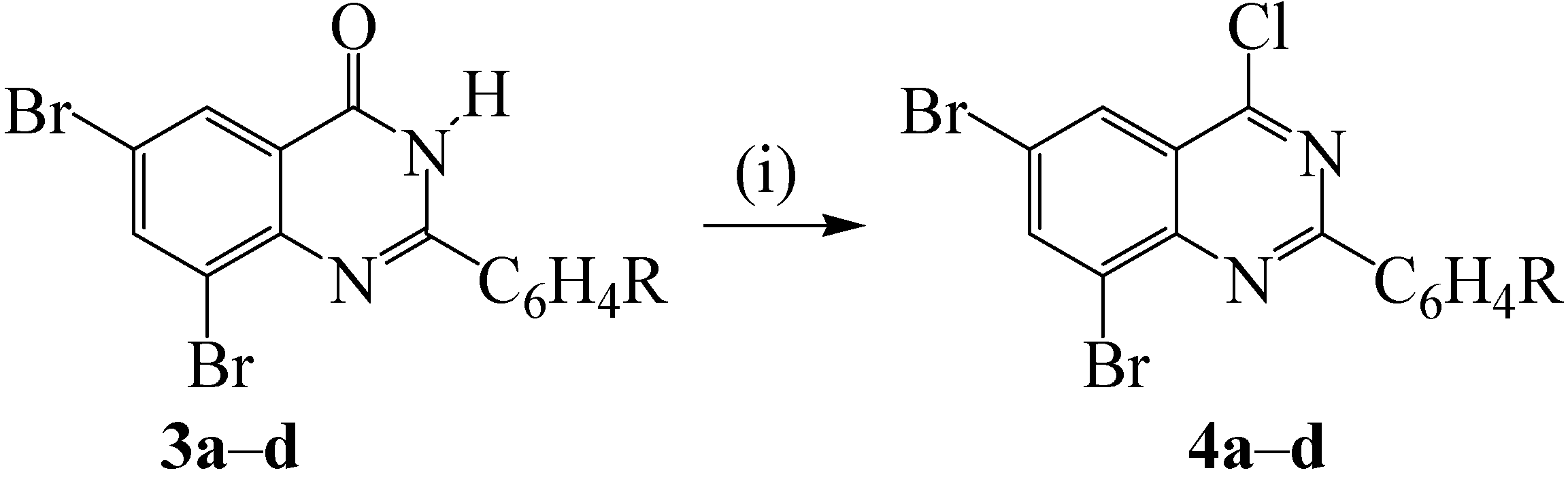
| R | % Yield 4 | |
|---|---|---|
| 4a | 4-H | 77 |
| 4b | 4-F | 81 |
| 4c | 4-Cl | 77 |
| 4d | 4-OMe | 91 |
2.3. Sonogashira Cross-Coupling of the 2-Aryl-6,8-dibromo-4-chloroquinazolines

| R | R' | % Yield 5 | |
|---|---|---|---|
| 5a | 4-H | -C6H5 | 60 |
| 5b | 4-F | -C6H5 | 72 |
| 5c | 4-Cl | -C6H5 | 69 |
| 5d | 4-OMe | -C6H5 | 72 |
| 5e | 4-H | 2-pyridyl- | 65 |
| 5f | 4-F | 2-pyridyl- | 67 |
| 5g | 4-OMe | 2-pyridyl- | 53 |
| 5h | 4-H | -CH(OH)CH3 | 56 |
2.4. Suzuki-Miyaura Cross-Coupling of the 2-Aryl-6,8-dibromo-4-(phenylethynyl)quinazolines
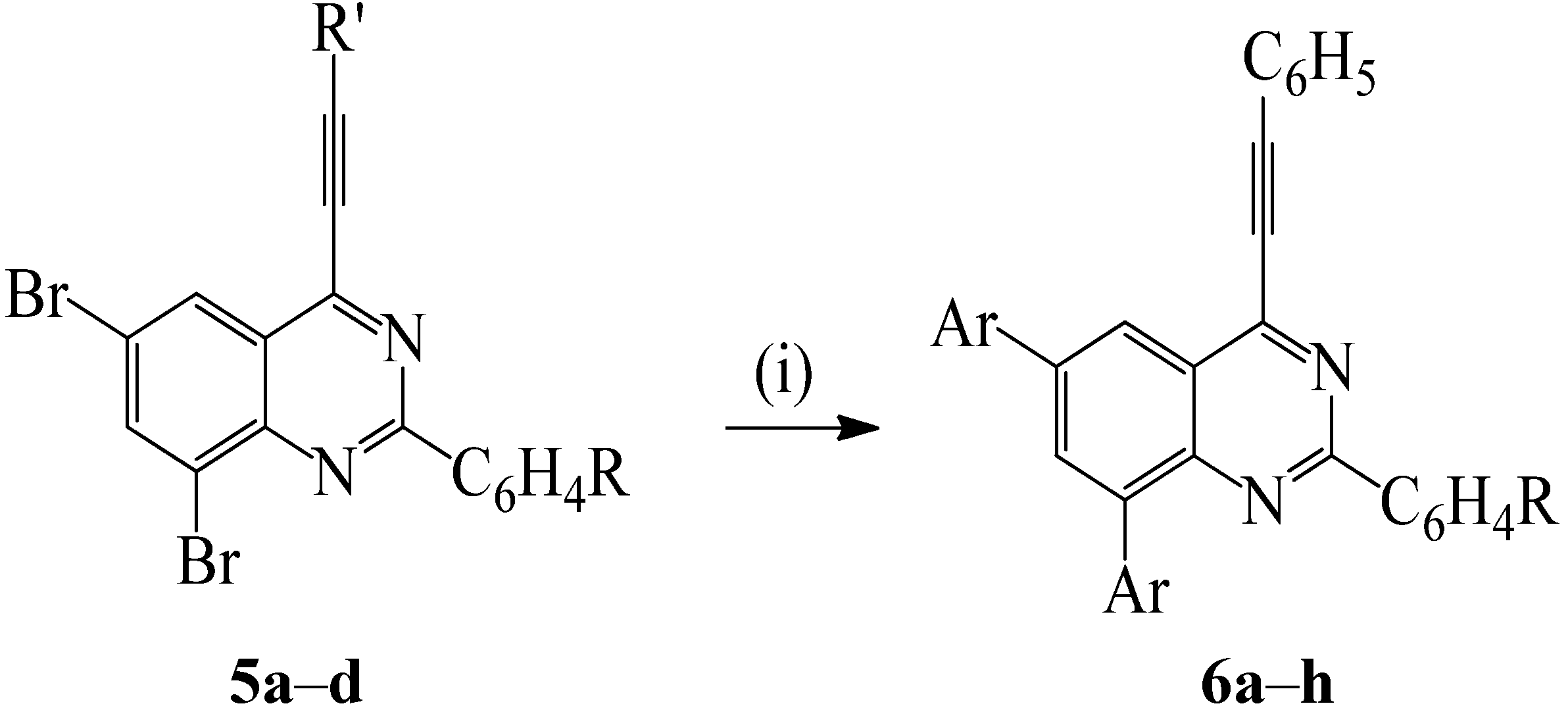
| R | Ar | % Yield 6 | |
|---|---|---|---|
| 6a | 4-H | -C6H5 | 61 |
| 6b | 4-F | -C6H5 | 69 |
| 6c | 4-Cl | -C6H5 | 88 |
| 6d | 4-OMe | -C6H5 | 68 |
| 6e | 4-H | 4-FC6H4- | 64 |
| 6f | 4-F | 4-FC6H4- | 76 |
| 6g | 4-Cl | 4-FC6H4- | 77 |
| 6h | 4-OMe | 4-FC6H4- | 58 |
| 6i | 4-H | 4-MeOC6H4- | 70 |
| 6j | 4-F | 4-MeOC6H4- | 62 |
| 6k | 4-Cl | 4-MeOC6H4- | 76 |
| 6l | 4-OMe | 4-MeOC6H4- | 52 |
2.5. Photophysical Property Studies of Compounds 6
2.5.1. UV-Vis Absorption Properties of the 2,6,8-Triaryl-4-(phenylethynyl)quinazolines 6a–l
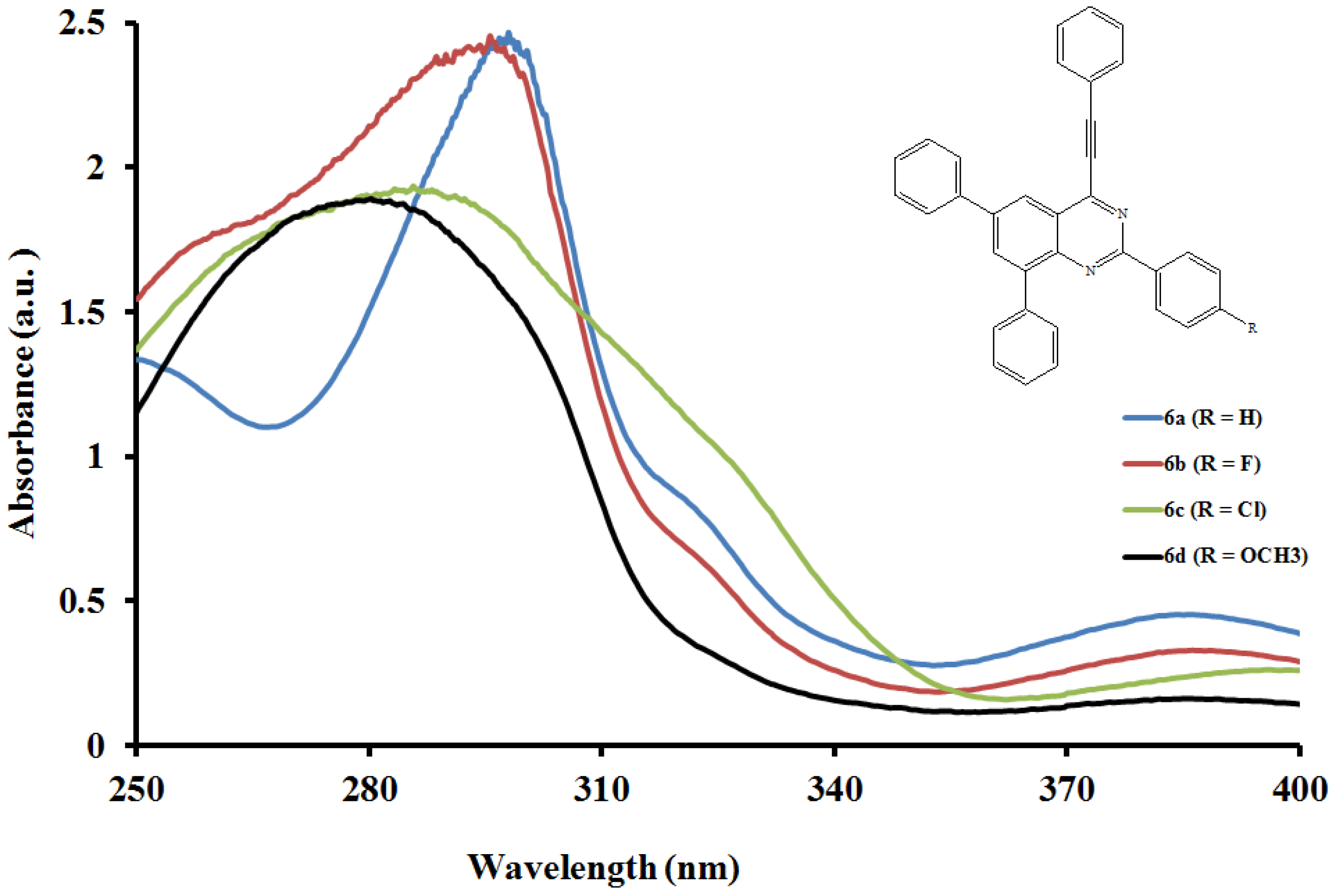
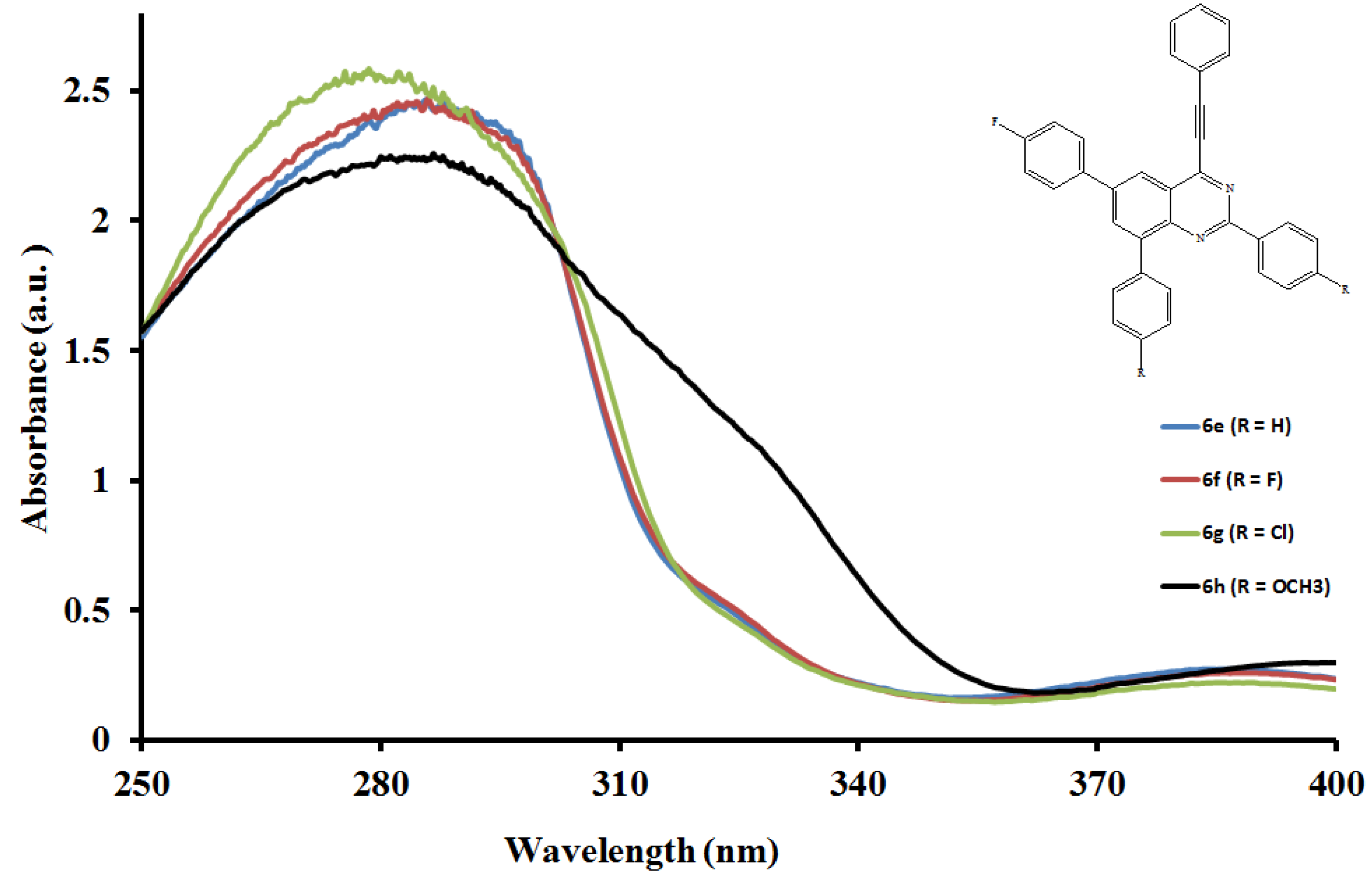

2.5.2. Emission properties of the 2,6,8-triaryl-4-(phenylethynyl)quinazolines 6a–l



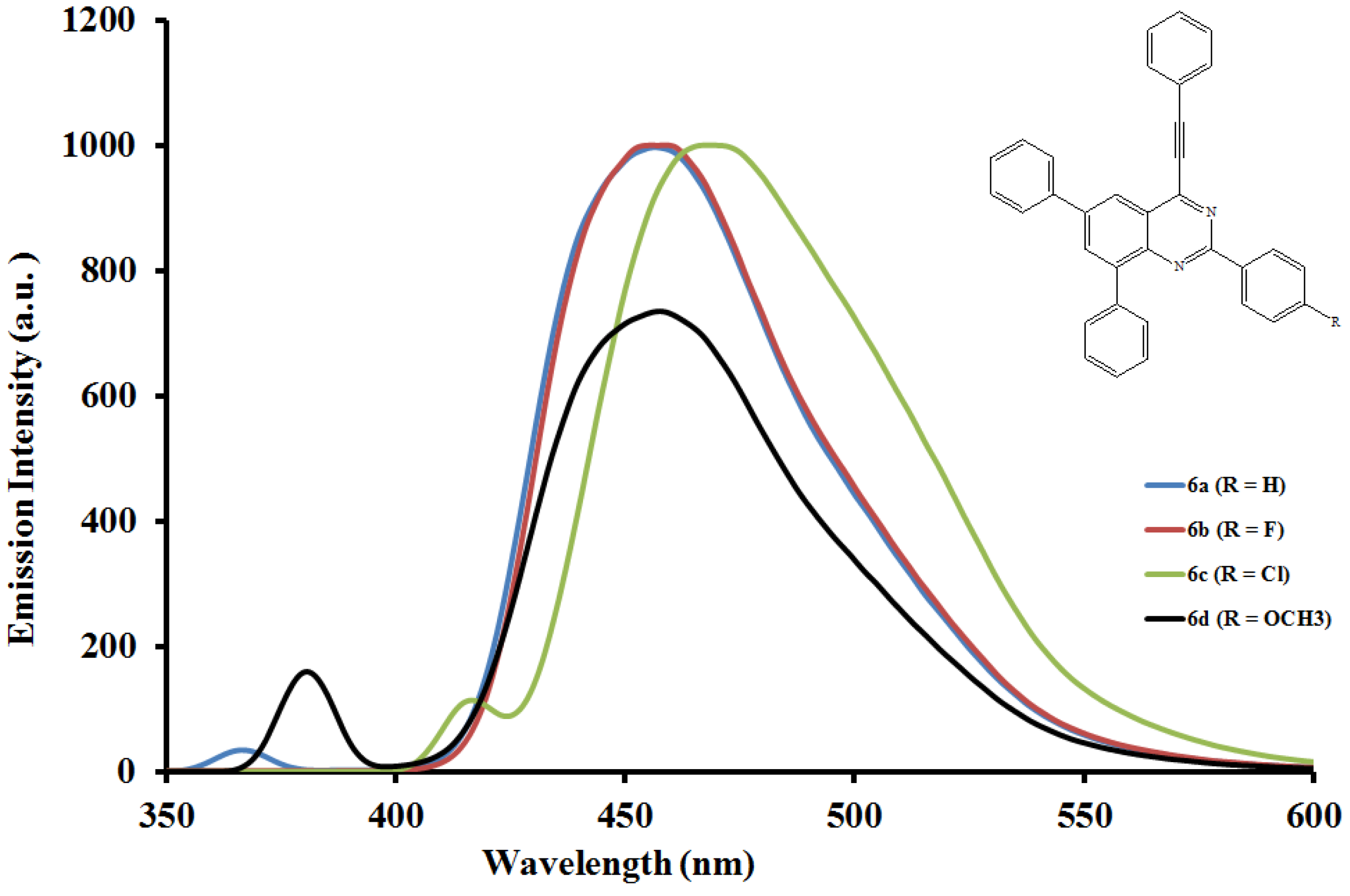

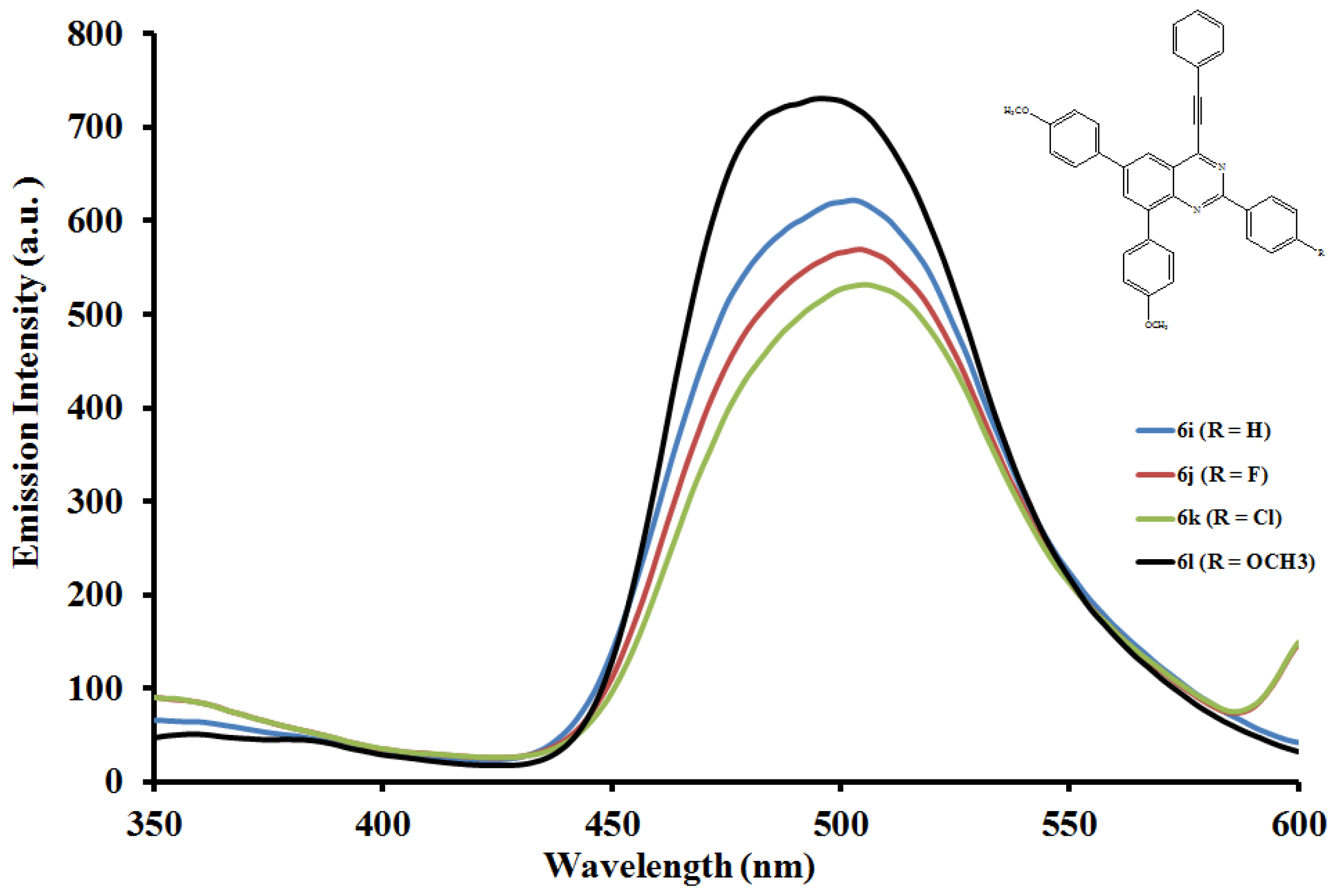
| Compounds | λmax (nm) CHCl3 | (ε) × 103 Mol−1cm−1 | λem (nm) CHCl3 | λem (nm) DMF | (a) Quantum yields (Φ) | Stokes shift CHCl3 |
|---|---|---|---|---|---|---|
| 6a | 298.0 | 11.216 | 454.5 | 456.5 | 0.071 | 156.5 |
| 6b | 295.6 | 11.163 | 455.0 | 459.0 | 0.078 | 159.4 |
| 6c | 285.7 | 8.798 | 462.5 | 467.0 | 0.102 | 176.8 |
| 6d | 280.3 | 8.601 | 455.0 | 458.0 | 0.105 | 174.7 |
| 6e | 285.7 | 11.216 | 454.5 | 457.5 | 0.071 | 168.8 |
| 6f | 284.5 | 11.181 | 456.0 | 457.0 | 0.081 | 171.5 |
| 6g | 278.5 | 11.754 | 455.0 | 458.5 | 0.076 | 176.5 |
| 6h | 286.6 | 10.271 | 463.0 | 466.5 | 0.088 | 176.4 |
| 6i | 292.9 | 9.725 | 478.5 | 503.0 | 0.088 | 185.6 |
| 6j | 276.4 | 12.063 | 479.0 | 504.5 | 0.072 | 202.6 |
| 6k | 276.7 | 12.268 | 480.0 | 505.5 | 0.070 | 203.3 |
| 6l | 273.4 | 10.422 | 479.5 | 496.0 | 0.081 | 206.1 |
2.5.3. Quantum Chemical Calculations

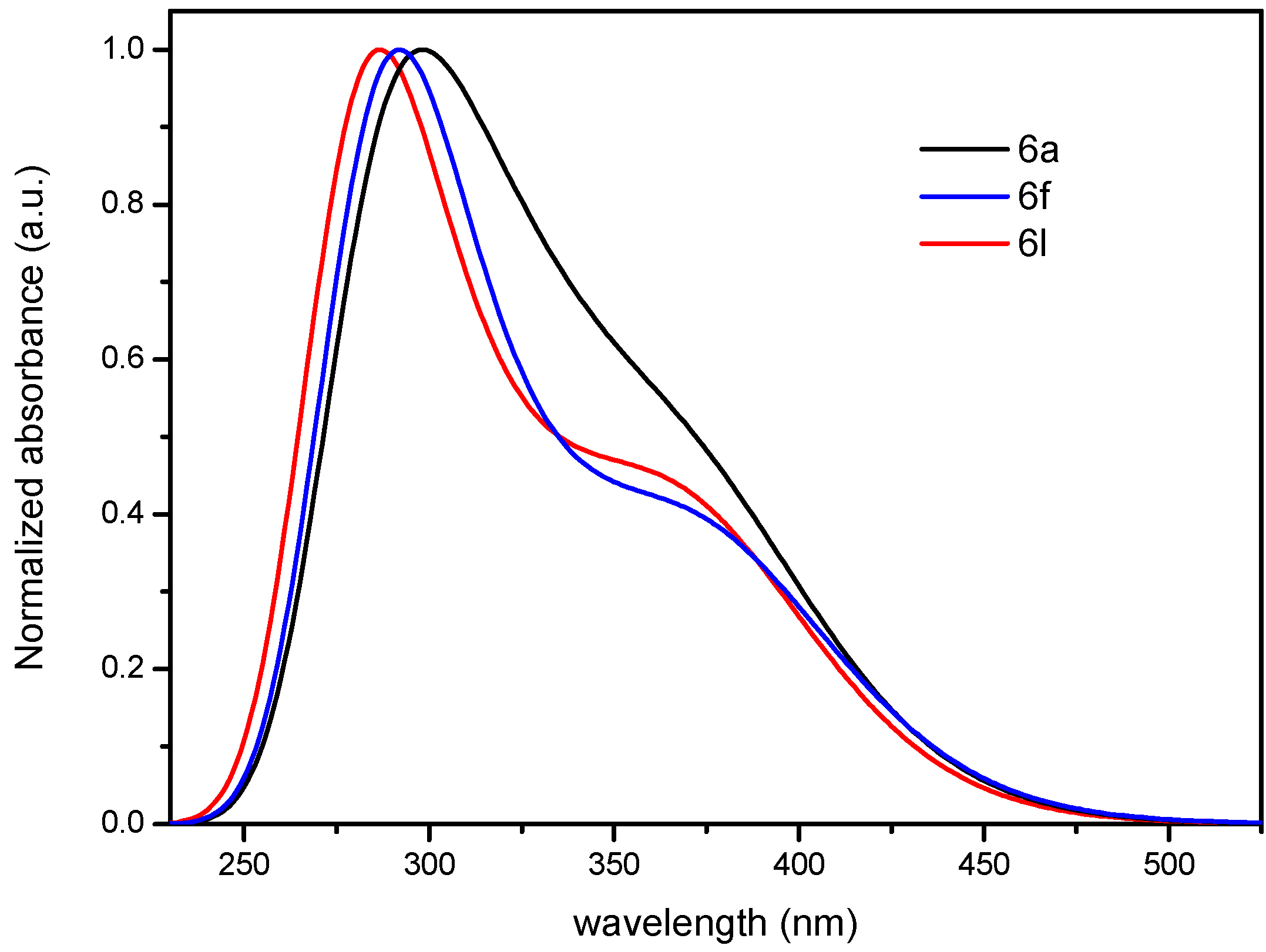
3. Experimental
3.1. General
3.2. I2–Promoted Cyclocondensation of 3,5-Dibromoanthranilamide and Arylaldehydes
Typical Procedure
3.3. Oxidative Aromatization of 3a–d with SOCl2-DMF Mixture
Typical Procedure
3.4. Sonogashira Cross-Coupling of 4a–d with Terminal Acetylynes
Typical Procedure
3.5. Typical Procedure for the Suzuki-Miyaura Cross-Coupling of 5a–d with Arylboronic Acids
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Grushin, V.V.; Alper, H. Transformations of chloroarenes, catalyzed by transition-metal complexes. Chem. Rev. 1994, 94, 1047–1062. [Google Scholar] [CrossRef]
- Hassan, J.; Sévignon, M.; Gozzi, C.; Schulz, E.; Lemaire, M. Aryl-aryl bond formation one century after the discovery of the Ullmann reaction. Chem. Rev. 2002, 102, 1359–1469. [Google Scholar] [CrossRef]
- Garcia, Y.; Schoenebeck, F.; Legault, C.Y.; Merlic, C.A.; Houk, K.N. Theoretical bond dissociation energies of halo-heterocycles: Trends and relationships to regioselectivity in palladium-catalyzed cross-coupling reactions. J. Am. Chem. Soc. 2009, 131, 6632–6639. [Google Scholar] [CrossRef]
- Mangalagiu, I.; Benneche, T.; Undheim, K. Trialkylalanes in palladium-catalyzed chemo- and regioselective alkylations. Tetrahedron Lett. 1996, 37, 1309–1312. [Google Scholar] [CrossRef]
- Mangalagiu, I.; Benneche, T.; Undheim, K. Ethenylation and alkynylation in palladium-catalyzed carbosubstitution in heteroazines. Acta Chem. Scand. 1996, 50, 914–917. [Google Scholar] [CrossRef]
- Charpiot, B.; Brun, J.; Donze, I.; Naef, R.; Stefani, M.; Mueller, T. Quinazolines: Combined type 3- and 4-phosphodiesterase inhibitors. Bioorg. Med. Chem. Lett. 1998, 8, 2891–2896. [Google Scholar] [CrossRef]
- Wipf, P.; George, K.M. Regioselective palladium-catalyzed cross-coupling reactions of 2,4,7-trichloroquinazoline. SynLett. 2010, 644–648. [Google Scholar] [CrossRef]
- Mphahlele, M.J. Regioselective alkynylation of 2-aryl-4-chloro-3-iodoquinolines and aubsequent arylation or amination of the 2-aryl-3-(alkynyl)-4-chloroquinolines. Tetrahedron 2010, 66, 8261–8266. [Google Scholar] [CrossRef]
- Khoza, T.A.; Maluleka, M.M.; Mama, N.; Mphahlele, M.J. Synthesis and photophysical properties of the 2-aryl-6,8-bis(arylethenyl)-4-methoxyquinolines. Molecules 2012, 17, 14186–14204. [Google Scholar] [CrossRef]
- Hisano, T.; Ichikawa, M.; Nakagawa, A.; Tsuji, M. Organosulfur compounds. XII. Syntheses and pharmacological activities of 2-heterocyclic-substituted 4(3H)-quinazolinones. Chem. Pharm. Bull. 1975, 23, 1910–1916. [Google Scholar] [CrossRef]
- Abdel-Jalil, R.J.; Aldoqum, H.M.; Ayoub, M.T.; Voelter, W. Synthesis and antitumor activity of 2-aryl-7-fluoro-6-(4-methyl-1-piperazinyl)-4(3H)-quinazolinones. Heterocycles 2005, 65, 2061–2070. [Google Scholar] [CrossRef]
- Mitobe, Y.; Ito, S.; Mizutani, T.; Nagase, T.; Sato, N.; Tokita, S. Development of a selective and potent radioactive ligand for histamine H3 receptors: A compound potentially useful for receptor occupancy studies. Bioorg. Med. Chem. Lett. 2009, 19, 4075–4078. [Google Scholar] [CrossRef]
- Balakumar, C.; Lamaba, P.; Kishore, D.P.; Narayana, B.L.; Rao, K.V.; Rajwinder, K.; Rao, A.R.; Shireesha, B.; Narsaiah, B. Synthesis, Anti-inflammatory evaluation and docking studies of some new fluorinated fused quinazolines. Eur. J. Med. Chem. 2010, 45, 4904–4913. [Google Scholar] [CrossRef]
- López, S.E.; Rosales, M.E.; Urdaneta, N.; Godoy, M.V.; Charris, J.E. The synthesis of substituted 2-aryl-4(3H)-quinazolinones using NaHSO3/DMA. Steric effect upon the cyclization-dehydrogenation step. J. Chem. Res. 2000, 258–259. [Google Scholar]
- Naleway, J.J.; Fox, C.J.M.; Robinhold, D.; Terpetsching, E.; Olsen, N.A.; Haugland, R.P. Synthesis and use of new fluorogenic precipitating substrates. Tetrahedron Lett. 1994, 35, 8569–8572. [Google Scholar]
- Abdel-Jalil, R.J.; Voelterb, W.; Saeed, M. A novel method for the synthesis of 4(3H)-quinazolinones. Tetrahedron Lett. 2004, 45, 3475–3476. [Google Scholar] [CrossRef]
- Wang, G.-W.; Miao, C.-B.; Kang, H. Benign and efficient synthesis of 2-substituted 4(3H)-quinazolinones mediated by iron(III) chloride hexahydrate in refluxing water. Bull. Chem. Soc. Jpn. 2006, 79, 1426–1430. [Google Scholar] [CrossRef]
- Bakavoli, M.; Rahimizadeh, M.; Shiri, A.; Ebrahimpour, Z. Molecular iodine oxidative cyclocondesation of ortho-aminobenzamide with aryl aldehydes: A new and efficient access to quinazolin-4(3H)-ones. Iran. J. Org. Chem. 2009, 1, 39–42. [Google Scholar]
- Barlaam, B.; Harris, C.S.; Lecog, J.; Nguyen, H.T.H. Preparation of 6-aminoquinazolin-4(3H)-ones via direct SNAr on the quinazoline ring. Tetrahedron 2012, 68, 534–543. [Google Scholar] [CrossRef]
- Jiang, J.B.; Hesson, D.P.; Dusak, B.A.; Dexter, D.L.; Kang, G.J.; Hamel, E. Synthesis and biological evaluation of 2-styrylquinazolin-4(3H)-ones, a new class of antimitotic anticancer agents which inhibit tubulin polymerization. J. Med. Chem. 1990, 33, 1721–1728. [Google Scholar] [CrossRef]
- Arnott, E.A.; Chan, L.C.; Cox, B.G.; Meyrick, B.; Phillips, A. POCl3 chlorination of 4-quinazolinones. J. Org. Chem. 2011, 76, 1653–1661. [Google Scholar] [CrossRef]
- El-Badry, Y.A.-M. Synthesis and reactions of 2-carboxyvinyl-4-chloro-6,8-dibromoquinazoline and some new fused triazolo-quinazoline derivatives. Acta Chim. Slov. 2010, 57, 836–841. [Google Scholar]
- Kitano, Y.; Suzuki, T.; Kawahara, E.; Yamasaki, T. Synthesis and inhibitory activity of 4-alkynyl and 4-alkenylquinazolines: Identification of new scaffolds for potent EGFR tyrosine kinase inhibitors. Bioorg. Med. Chem. Lett. 2007, 17, 5863–5867. [Google Scholar] [CrossRef]
- Akrawi, O.A.; Mohammed, H.H.; Langer, P. Synthesis and Suzuki-Miyaura reactions of 3,6,8-tribromoquinoline: A structural revision. SynLett. 2013, 1121–1124. [Google Scholar]
- Ho-Joon, L.; Hao, X.; Seong-Min, P.; Seog-IL, P.; Taek, A.; Dong-Kyu, P.; Samson, A.J.; Tae-Woo, K. Synthesis and properties of diarylamino-substituted linear and dendritic oligoquinolines for organic light-emitting diodes. Bull. Korean Chem. Soc. 2012, 33, 1627–1637. [Google Scholar] [CrossRef]
- Diaz, A.N.J. Absorption and emission spectroscopy and photochemistry of 1,10-anthraquinone derivatives: A review. Photochem. Photobiol. A 1990, 53, 141–167. [Google Scholar] [CrossRef]
- Kalinowski, J. Excimers and exciplexes in organic electroluminescence. Mat. Sci.-Poland 2009, 27, 735–756. [Google Scholar]
- Chen, G.S.; Talekar, R.S.; Wong, K.-T.; Chi, L.-C.; Chern, J.-W. Physical properties of 8-substituted 5,7-dichloro-2-styrylquinolines as potential light emitting materials. J. Chin. Chem. Soc. 2007, 54, 1387–1394. [Google Scholar]
- Yanai, T.; Tew, D.; Handy, N. A new hybrid exchange-correlation functional using the Coulomb-attenuating method (CAM-B3LYP). Chem. Phys. Lett. 2004, 393, 51–57. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09; Revision B.01; Gaussian, Inc.: Wallingford, CT, USA, 2010. [Google Scholar]
- Zerner, M.C. Semiempirical Molecular Orbital Methods. In Reviews in Computational Chemistry; Lipkowitz, K.B., Boyd, D.B., Eds.; VCH Publishing: New York, NY, USA, 1991; Volume 2, pp. 313–366. [Google Scholar]
- Sunahara, H.; Urano, Y.; Kojima, H.; Nagano, T. Design and synthesis of a library of BODIPY-based environmental polarity sensors utilizing photoinduced electron-transfer-controlled fluorescence on/off switching. J. Am. Chem. Soc. 2007, 129, 5597–5604. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 3a–d, 4a–d, 5a–h and 6a–l are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mphahlele, M.J.; Paumo, H.K.; El-Nahas, A.M.; El-Hendawy, M.M. Synthesis and Photophysical Property Studies of the 2,6,8-Triaryl-4-(phenylethynyl)quinazolines. Molecules 2014, 19, 795-818. https://doi.org/10.3390/molecules19010795
Mphahlele MJ, Paumo HK, El-Nahas AM, El-Hendawy MM. Synthesis and Photophysical Property Studies of the 2,6,8-Triaryl-4-(phenylethynyl)quinazolines. Molecules. 2014; 19(1):795-818. https://doi.org/10.3390/molecules19010795
Chicago/Turabian StyleMphahlele, Malose Jack, Hugues Kadem Paumo, Ahmed M. El-Nahas, and Morad M. El-Hendawy. 2014. "Synthesis and Photophysical Property Studies of the 2,6,8-Triaryl-4-(phenylethynyl)quinazolines" Molecules 19, no. 1: 795-818. https://doi.org/10.3390/molecules19010795






