Pharmacokinetics of BMEDA after Intravenous Administration in Beagle Dogs
Abstract
:1. Introduction
2. Results
Method Validation
| Nominal concentration (ng/mL) | Observed concentration (ng/mL) | Precision (RSD %) | Accuracy (% Bias) |
|---|---|---|---|
| Intra-assay (n = 6) | |||
| 10 | 8.7 ± 0.6 | 6.9 | −12.9 |
| 30 | 29.4 ± 1.6 | 5.5 | −2.1 |
| 240 | 269.5 ± 14.0 | 5.2 | 12.3 |
| 2400 | 2303.4 ± 199.6 | 8.7 | −4.0 |
| Inter-assay (n = 18) | |||
| 10 | 9.5 ± 1.0 | 10.7 | −4.8 |
| 30 | 30.2 ± 1.6 | 5.4 | 0.7 |
| 240 | 257.0 ± 16.5 | 6.4 | 7.1 |
| 2400 | 2229.9 ± 142 | 6.4 | −7.1 |
| Conc. (ng/mL) | Matrix effect (%) | Recovery (%) |
|---|---|---|
| 30 | 104.3 | 98.9 |
| 240 | 110.0 | 86.3 |
| 2400 | 107.4 | 89.3 |
| Average | 107.2 ± 2.9 | 91.5 ± 6.6 |
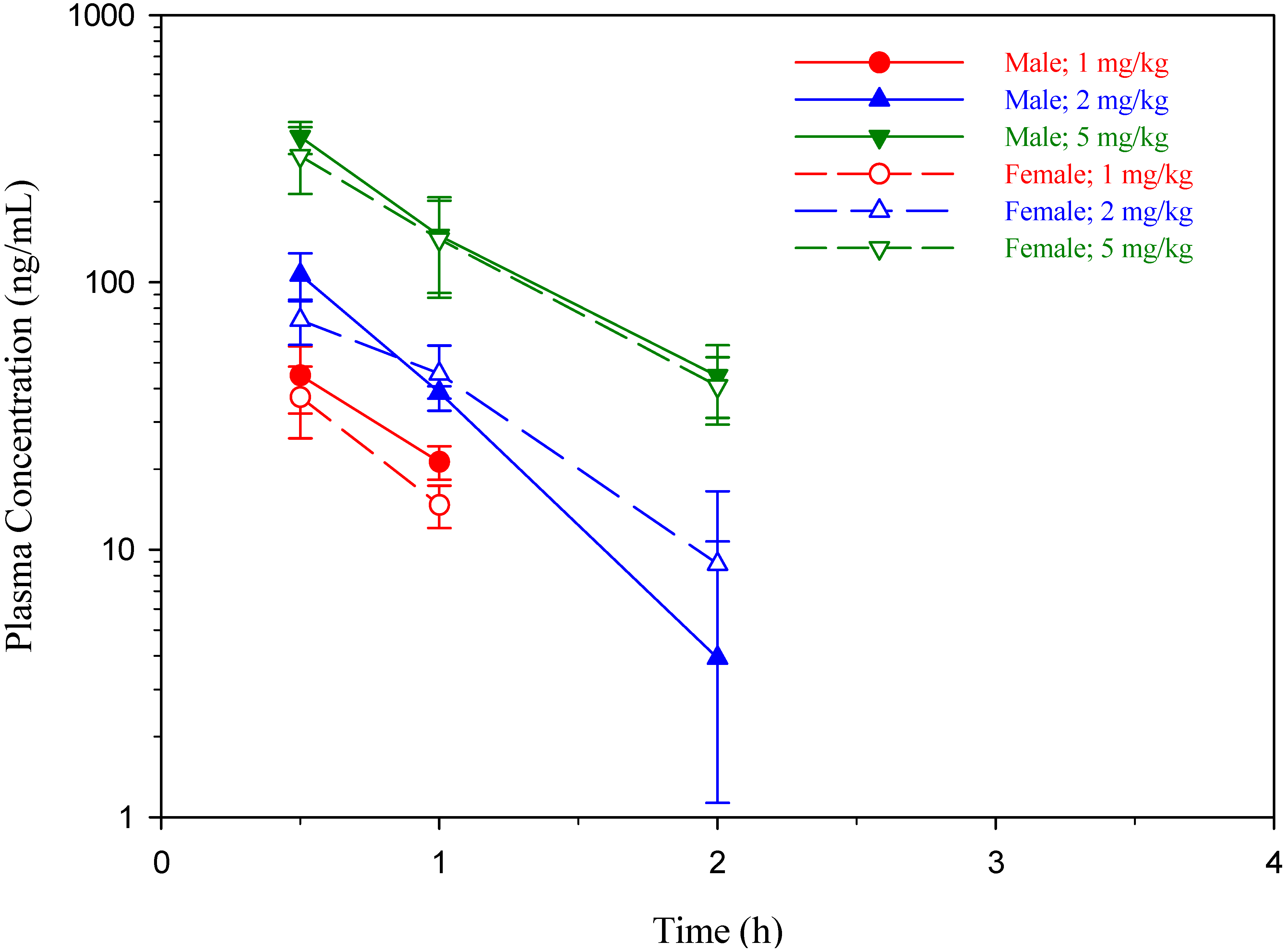
| Parameter | Dose (mg/kg) | Male (N = 3) | Female (N = 3) | Combined (N = 6) | |||
|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | ||
| Cmax (ng/mL) | 1 | 44.9 | 12.7 | 37.2 | 11.1 | 41.1 | 11.5 |
| 2 | 107 | 21.6 | 73.2 | 12.4 | 89.9 | 24.2 | |
| 5 | 350 | 48.3 | 297 | 83.4 | 324 | 67.4 | |
| AUC0–t (h·ng/mL) | 1 | 26.6 | 8.04 | 22.3 | 6.14 | 24.5 | 6.83 |
| 2 | 71.7 | 18.4 | 67.7 | 15.6 | 69.7 | 15.4 | |
| 5 | 310 | 62.2 | 278 | 87.4 | 294 | 70.0 | |
| AUC0–∞ (h·ng/mL) | 1 | ND | ND | ND | ND | ND | ND |
| 2 | 96.8 a | NC a | 83.1 a | NC a | 90.0 b | 9.69 b | |
| 5 | 343 | 75.2 | 310 | 95.0 | 326 | 78.8 | |
| Vss (mL/kg) | 1 | ND | ND | ND | ND | ND | ND |
| 2 | 18,887 a | NC a | 25,688 a | NC a | 22,288 b | 4809 b | |
| 5 | 14,473 | 2284 | 17,243 | 5281 | 15,858 | 3942 | |
| CL (mL/h/kg) | 1 | ND | ND | ND | ND | ND | ND |
| 2 | 20,663 a | NC a | 24,077 a | NC a | 22,370 b | 2414 b | |
| 5 | 15,074 | 3475 | 17,126 | 4777 | 16,100 | 3901 | |
| T1/2 (h) | 1 | ND | ND | ND | ND | ND | ND |
| 2 | 0.5 a | NC a | 0.6 a | NC a | 0.6 b | 0.1 b | |
| 5 | 0.5 | 0.1 | 0.5 | 0.1 | 0.5 | 0.1 | |
| MRT0–∞ (h) | 1 | ND | ND | ND | ND | ND | ND |
| 2 | 0.9 a | NC a | 1.1 a | NC a | 1.0 b | 0.1 b | |
| 5 | 1.0 | 0.1 | 1.0 | 0.0 | 1.0 | 0.1 | |
3. Discussion
| Parameter | Dose (mg/kg) | Male (N = 3) | Female (N = 3) | Combined (N = 6) | |||
|---|---|---|---|---|---|---|---|
| Mean | Ratio | Mean | Ratio | Mean | Ratio | ||
| Cmax (ng/mL) | 1 | 44.9 | − | 37.2 | − | 41.1 | − |
| 2 | 107 | 2.38 | 73.2 | 1.97 | 89.9 | 2.19 | |
| 5 | 350 | 7.80 | 297 | 7.98 | 324 | 7.88 | |
| AUC0-t (h·ng/mL) | 1 | 26.6 | − | 22.3 | − | 24.5 | − |
| 2 | 71.7 | 2.70 | 67.7 | 3.04 | 69.7 | 2.84 | |
| 5 | 310 | 11.7 | 278 | 12.5 | 294 | 12.0 | |
| AUC0-∞ (h·ng/mL) | 1 | ND | − | ND | − | ND | − |
| 2 | 96.8 a | − | 83.1 a | − | 90.0 b | − | |
| 5 | 343 | 3.54 c | 310 | 3.73 c | 326 | 3.62 c | |
4. Experimental
4.1. Reagents
4.2. Experimental Animals
4.3. Liquid Chromatography-Mass Spectrometry/Mass Spectrometry
4.4. Method Validation
4.5. Pharmacokinetic Analyses
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Parkin, D.M.; Bray, F.; Ferlay, J.; Pisani, P. Estimating the world cancer burden: Globocan 2000. Int. J. Cancer 2001, 94, 153–156. [Google Scholar] [CrossRef]
- Larsson, S.C.; Wolk, A. Meat consumption and risk of colorectal cancer: A meta-analysis of prospective studies. Int. J. Cancer 2006, 119, 2657–2664. [Google Scholar] [CrossRef]
- Ting, G.; Chang, C.H.; Wang, H.E. Cancer nanotargeted radiopharmaceuticals for tumor imaging and therapy. Anticancer Res. 2009, 29, 4107–4118. [Google Scholar]
- Lombardi, L.; Morelli, F.; Cinieri, S.; Santini, D.; Silvestris, N.; Fazio, N.; Orlando, L.; Tonini, G.; Colucci, G.; Maiello, E. Adjuvant colon cancer chemotherapy: Where we are and where we’ll go. Cancer Treat. Rev. 2010, 36, S34–S41. [Google Scholar] [CrossRef]
- Peer, D.; Karp, J.M.; Hong, S.; Farokhzad, O.C.; Margalit, R.; Langer, R. Nanocarriers as an emerging platform for cancer therapy. Nat. Nanotechnol. 2007, 2, 751–760. [Google Scholar] [CrossRef]
- Torchilin, V.P. Recent advances with liposomes as pharmaceutical carriers. Nat. Rev. Drug Discov. 2005, 4, 145–160. [Google Scholar] [CrossRef]
- Emfietzoglou, D.; Kostarelos, K.; Sgouros, G. An analytic dosimetry study for the use of radionuclide-liposome conjugates in internal radiotherapy. J. Nucl. Med. 2001, 42, 499–504. [Google Scholar]
- Mitra, A.; Nan, A.; Line, B.R.; Ghandehari, H. Nanocarriers for nuclear imaging and radiotherapy of cancer. Curr. Pharm. Des. 2006, 12, 4729–4749. [Google Scholar] [CrossRef]
- Chang, C.H.; Stabin, M.G.; Chang, Y.J.; Chen, L.C.; Chen, M.H.; Chang, T.J.; Lee, T.W.; Ting, G. Comparative dosimetric evaluation of nanotargeted 188Re-(DXR)-liposome for internal radiotherapy. Cancer Biother. Radiopharm. 2008, 23, 749–758. [Google Scholar] [CrossRef]
- Hamoudeh, M.; Kamleh, M.A.; Diab, R.; Fessi, H. Radionuclides delivery systems for nuclear imaging and radiotherapy of cancer. Adv. Drug Deliv. Rev. 2008, 60, 1329–1346. [Google Scholar] [CrossRef]
- Knapp, F.F., Jr.; Beets, A.L.; Guhlke, S.; Zamora, P.O.; Bender, H.; Palmedo, H.; Biersack, H.J. Availability of rhenium-188 from the alumina-based tungsten-188/rhenium-188 generator for preparation of rhenium-188-labeled radiopharmaceuticals for cancer treatment. Anticancer Res. 1997, 17, 1783–1795. [Google Scholar]
- Chen, L.C.; Chang, C.H.; Yu, C.Y.; Chang, Y.J.; Wu, Y.H.; Lee, W.C.; Yeh, C.H.; Lee, T.W.; Ting, G. Pharmacokinetics, Micro-SPECT/CT imaging and therapeutic efficacy of 188Re-DXR-liposome in C26 colon carcinoma ascites mice model. Nucl. Med. Biol. 2008, 35, 883–893. [Google Scholar] [CrossRef]
- Chen, L.C.; Wu, Y.H.; Liu, I.H.; Ho, C.L.; Lee, W.C.; Chang, C.H.; Lan, K.L.; Ting, G.; Lee, T.W.; Shien, J.H. Pharmacokinetics, dosimetry and comparative efficacy of 188Re-liposome and 5-fu in a CT26-luc lung-metastatic mice model. Nucl. Med. Biol. 2012, 39, 35–43. [Google Scholar] [CrossRef]
- Hsu, C.W.; Chang, Y.J.; Chang, C.H.; Chen, L.C.; Lan, K.L.; Ting, G.; Lee, T.W. Comparative therapeutic efficacy of rhenium-188 radiolabeled-liposome and 5-fluorouracil in LS-174T human colon carcinoma solid tumor xenografts. Cancer Biother. Radiopharm. 2012, 27, 481–489. [Google Scholar] [CrossRef]
- Chang, Y.J.; Hsu, C.W.; Chang, C.H.; Lan, K.L.; Ting, G.; Lee, T.W. Therapeutic efficacy of 188Re-liposome in a C26 murine colon carcinoma solid tumor model. Investig. New Drugs 2013, 31, 801–811. [Google Scholar] [CrossRef]
- Liu, C.M.; Tsai, C.C.; Yu, C.Y.; Lee, W.C.; Ho, C.L.; Chang, T.J.; Chang, C.H.; Lee, T.W. Extended acute toxicity study of 188Re-liposome in rats. J. Appl. Toxicol. 2013, 33, 886–893. [Google Scholar] [CrossRef]
- Bao, A.; Goins, B.; Klipper, R.; Negrete, G.; Mahindaratne, M.; Phillips, W.T. A novel liposome radiolabeling method using 99mTc-“SNS/S” complexes: In vitro and in vivo evaluation. J. Pharm. Sci. 2003, 92, 1893–1904. [Google Scholar] [CrossRef]
- Bao, A.; Goins, B.; Klipper, R.; Negrete, G.; Phillips, W.T. 186Re-liposome labeling using 186Re-SNS/S complexes: In vitro stability, imaging, and biodistribution in rats. J. Nucl. Med. 2003, 44, 1992–1999. [Google Scholar]
- Bao, A.; Goins, B.; Klipper, R.; Negrete, G.; Phillips, W.T. Direct 99mTc labeling of pegylated liposomal doxorubicin (doxil) for pharmacokinetic and non-invasive imaging studies. J. Pharmacol. Exp. Ther. 2004, 308, 419–425. [Google Scholar]
- Tsai, C.C.; Chang, C.H.; Chen, L.C.; Chang, Y.J.; Lan, K.L.; Wu, Y.H.; Hsu, C.W.; Liu, I.H.; Ho, C.L.; Lee, W.C.; et al. Biodistribution and pharmacokinetics of 188Re-liposomes and their comparative therapeutic efficacy with 5-fluorouracil in C26 colonic peritoneal carcinomatosis mice. Int. J. Nanomed. 2011, 6, 2607–2619. [Google Scholar]
- Chang, C.H.; Chiu, S.P.; Chiang, T.C.; Lee, T.W. Acute intravenous injection toxicity of BMEDA in mice. Drug Chem. Toxicol. 2011, 34, 20–24. [Google Scholar] [CrossRef]
- Liu, S.Y.; Chang, C.H.; Lee, T.W. Institute of Nuclear Energy Research: Taoyuan, Taiwan, Unpublished work. 2013.
- Lin, J.H. Dose-dependent pharmacokinetics: Experimental observations and theoretical considerations. Biopharm. Drug Dispos. 1994, 15, 1–31. [Google Scholar] [CrossRef]
- Shand, D.G.; Rangno, R.E. The disposition of propranolol. I. Elimination during oral absorption in man. Pharmacology 1972, 7, 159–168. [Google Scholar] [CrossRef]
- Walle, T.; Conradi, E.C.; Walle, U.K.; Fagan, T.C.; Gaffney, T.E. The predictable relationship between plasma levels and dose during chronic propranolol therapy. Clin. Pharmacol. Ther. 1978, 24, 668–677. [Google Scholar]
- Makar, A.B.; Mannering, G.J. Kinetics of ethanol metabolism in the intact rat and monkey. Biochem. Pharmacol. 1970, 19, 2017–2022. [Google Scholar] [CrossRef]
- Levy, G.; Yacobi, A. Letter: Effect of plasma protein binding on elimination of warfarin. J. Pharm. Sci. 1974, 63, 805–806. [Google Scholar] [CrossRef]
- Schary, W.L.; Rowland, M. Protein binding and hepatic clearance: Studies with tolbutamide, a drug of low intrinsic clearance, in the isolated perfused rat liver preparation. J. Pharmacokinet. Biopharm. 1983, 11, 225–243. [Google Scholar] [CrossRef]
- Jusko, W.J.; Gretch, M. Plasma and tissue protein binding of drugs in pharmacokinetics. Drug Metab. Rev. 1976, 5, 43–140. [Google Scholar] [CrossRef]
- Lin, J.H. Species differences in protein binding of diflunisal. Drug Metab. Dispos. 1989, 17, 221–223. [Google Scholar]
- Levy, G.; Tsuchiya, T.; Amsel, L.P. Limited capacity for salicyl phenolic glucuronide formation and its effect on the kinetics of salicylate elimination in man. Clin. Pharmacol. Ther. 1972, 13, 258–268. [Google Scholar]
- Perrier, D.; Gibaldi, M. General derivation of the equation for time to reach a certain fraction of steady state. J. Pharm. Sci. 1982, 71, 474–475. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chang, C.-H.; Liu, S.-Y.; Lee, T.-W. Pharmacokinetics of BMEDA after Intravenous Administration in Beagle Dogs. Molecules 2014, 19, 538-549. https://doi.org/10.3390/molecules19010538
Chang C-H, Liu S-Y, Lee T-W. Pharmacokinetics of BMEDA after Intravenous Administration in Beagle Dogs. Molecules. 2014; 19(1):538-549. https://doi.org/10.3390/molecules19010538
Chicago/Turabian StyleChang, Chih-Hsien, Si-Yen Liu, and Te-Wei Lee. 2014. "Pharmacokinetics of BMEDA after Intravenous Administration in Beagle Dogs" Molecules 19, no. 1: 538-549. https://doi.org/10.3390/molecules19010538




