Saccharide Substituted Zinc Phthalocyanines: Optical Properties, Interaction with Bovine Serum Albumin and Near Infrared Fluorescence Imaging for Sentinel Lymph Nodes
Abstract
:1. Introduction
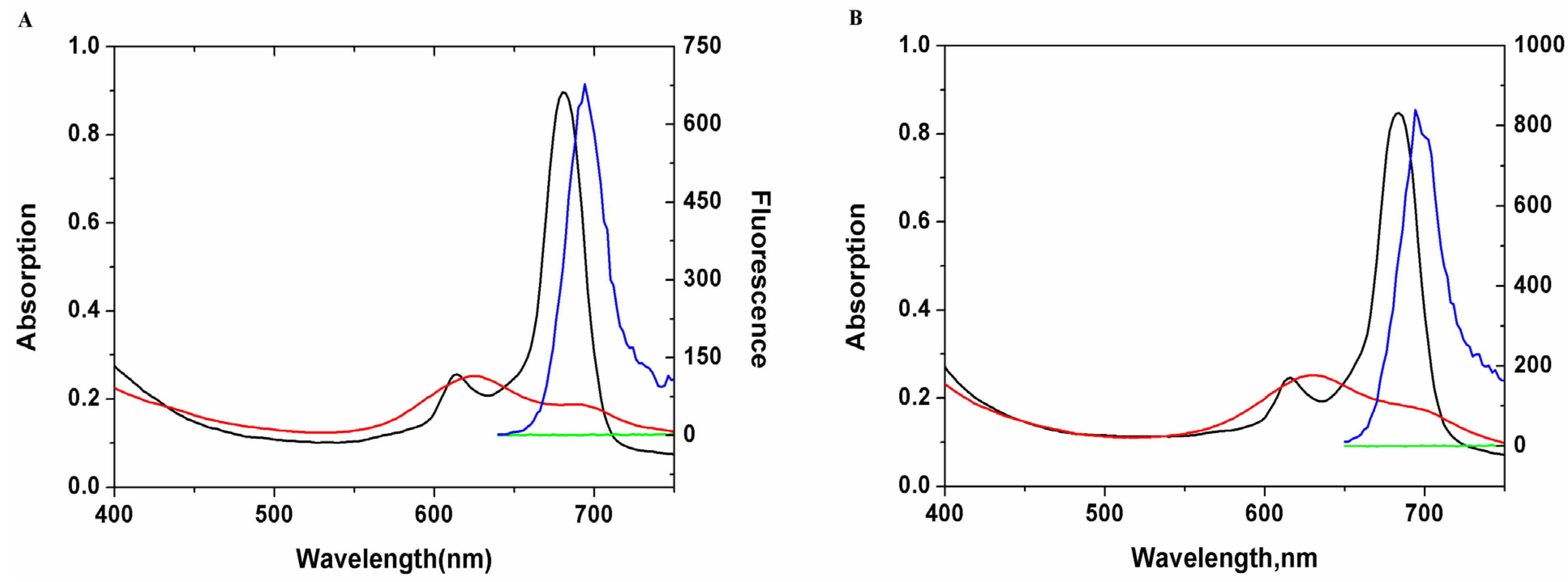
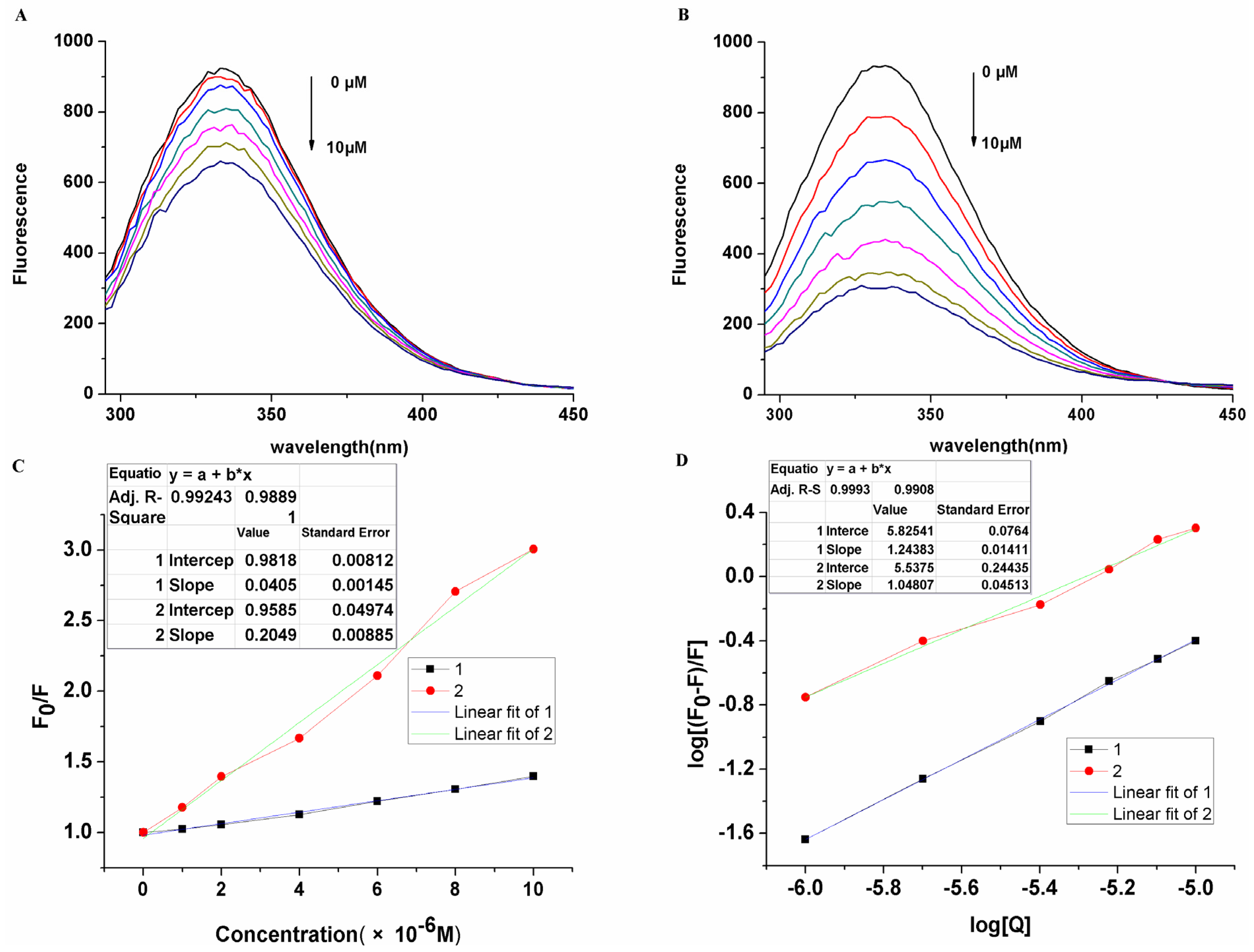
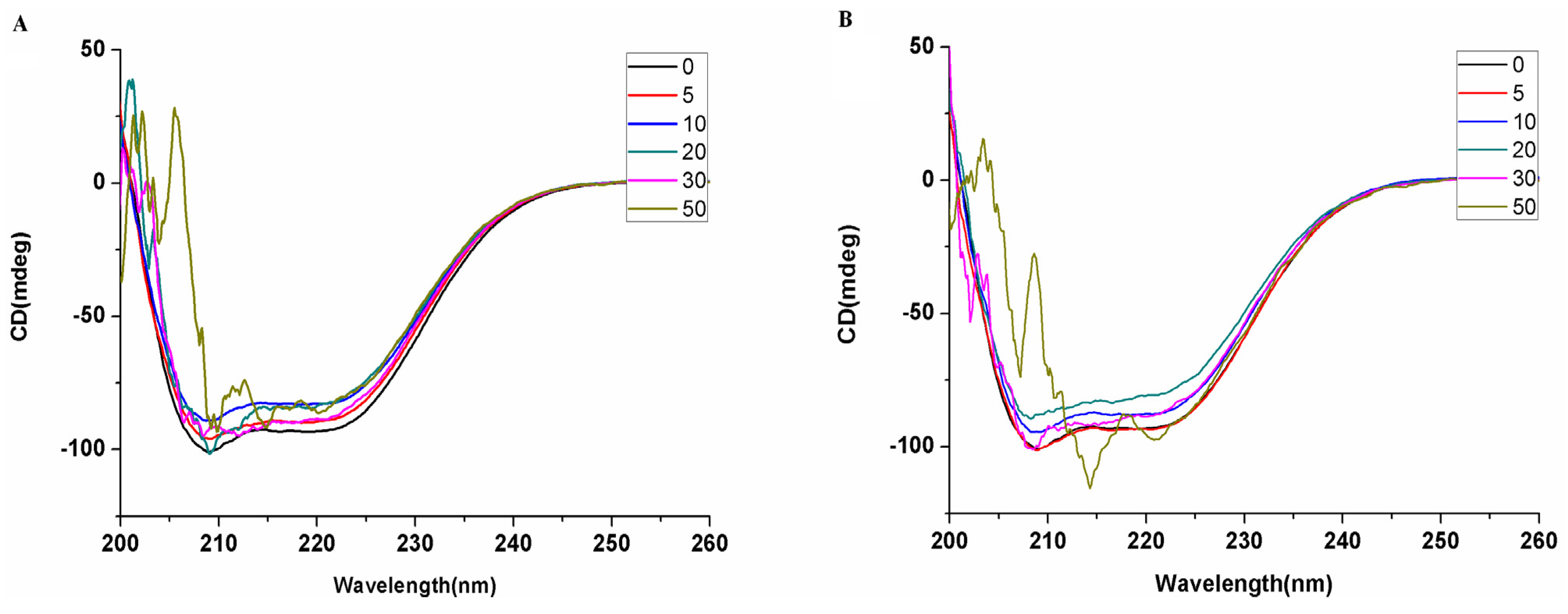
2. Results and Discussion
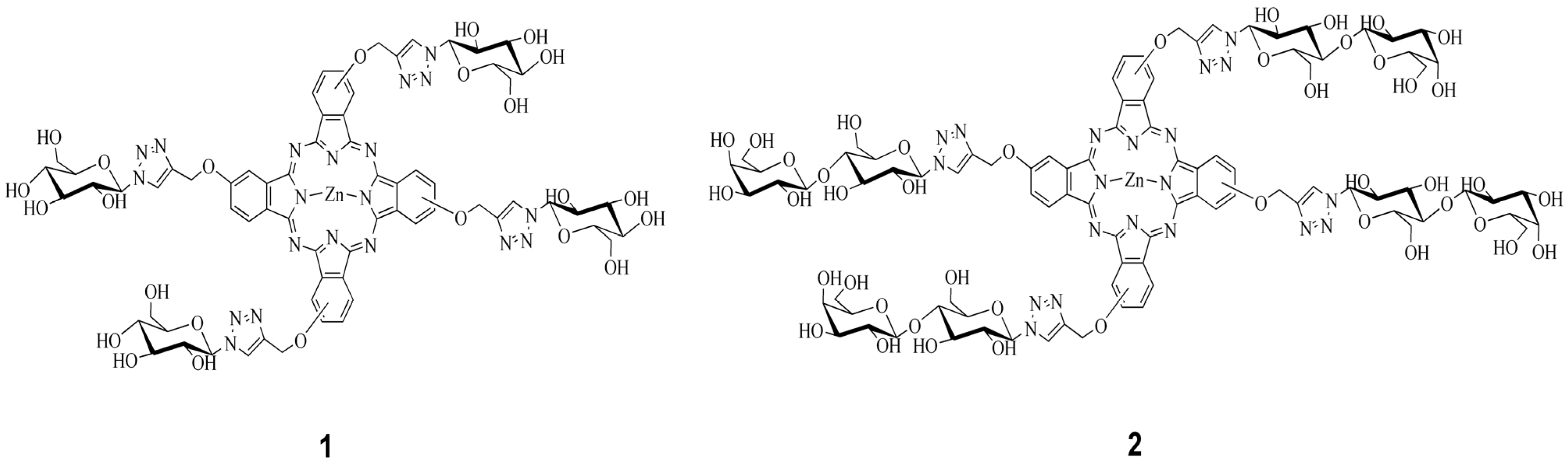


3. Experimental
3.1. Materials
3.2. Octanol/Water Partition Coefficients of Saccharide Substituted Zinc Phthalocyanines
3.3. UV-Vis and Fluorescence Spectra of Saccharide Substituted Zinc Phthalocyanines
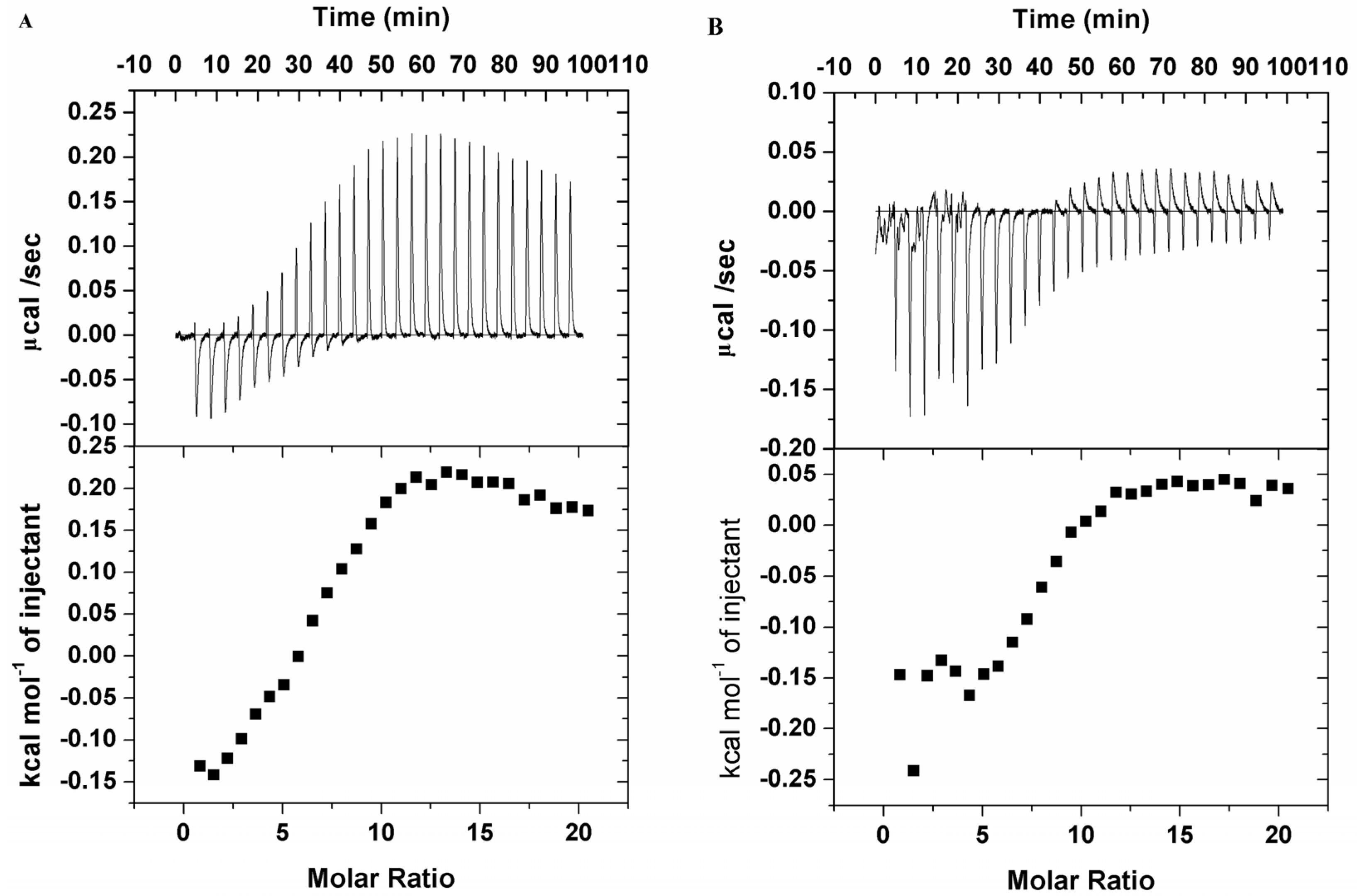
3.4. Interaction of Saccharide Substituted Zinc Phthalocyanines with BSA
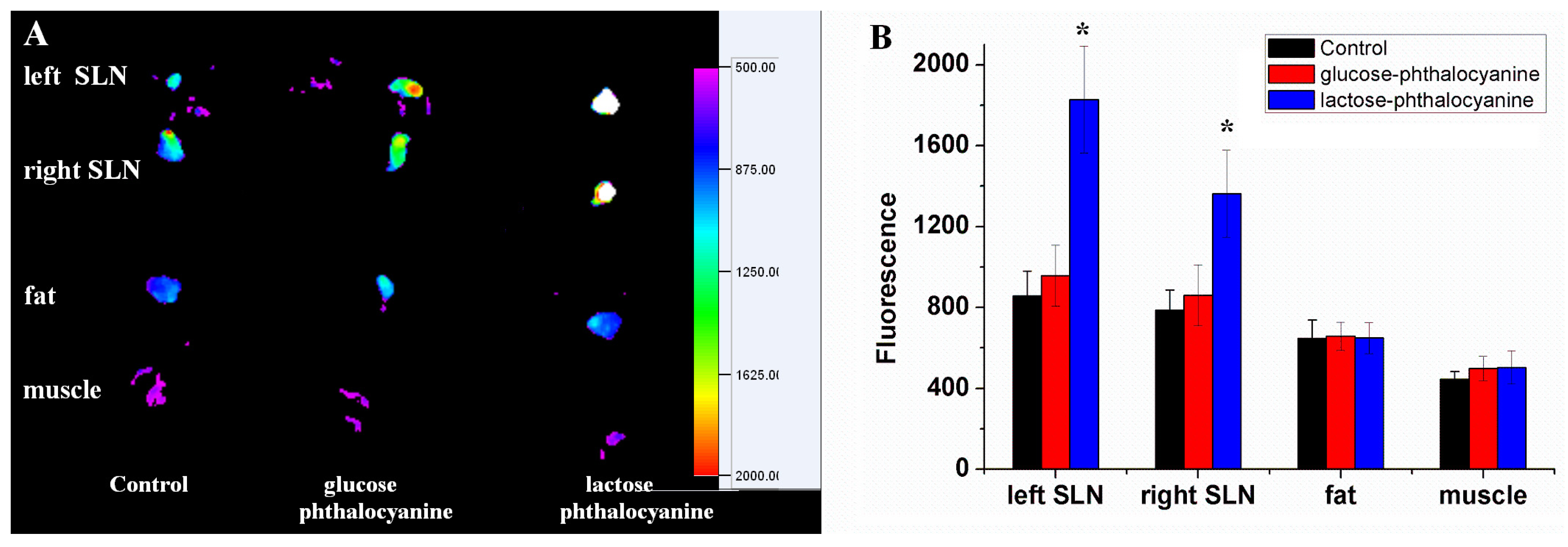
3.5. In Vivo Imaging and Distribution of Saccharide Substituted Zinc Phthalocyanines for SLN
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Weissleder, R.; Pittet, M.J. Imaging in the era of molecular oncology. Nature 2008, 452, 580–589. [Google Scholar] [CrossRef]
- Luker, G.D.; Luker, K.E. Optical imaging: Current applications and future directions. J. Nuclear Med. 2008, 49, 1–4. [Google Scholar] [CrossRef]
- Hilderbrand, S.A.; Weissleder, R. Near-infrared fluorescence: Application to in vivo molecular imaging. Curr. Opin. Chem. Biol. 2010, 14, 71–79. [Google Scholar] [CrossRef]
- Kobayashi, H.; Ogawa, M.; Alford, R.; Choyke, P.L.; Urano, Y. New strategies for fluorescent probe design in medical diagnostic imaging. Chem. Rev. 2010, 110, 2620–2640. [Google Scholar] [CrossRef]
- Rao, J.; Dragulescu-Andrasi, A.; Yao, H. Fluorescence imaging in vivo: Recent advances. Curr. Opin. Biotechnol. 2007, 18, 17–25. [Google Scholar] [CrossRef]
- Magalotti, S.; Gustafson, T.P.; Cao, Q.; Abendschein, D.R.; Pierce, R.A.; Berezin, M.Y.; Akers, W.J. Evaluation of inflammatory response to acute ischemia using near-infrared fluorescent reactive oxygen sensors. Mol. Imaging Biol. 2013, 15, 423–430. [Google Scholar] [CrossRef]
- Cheng, T.C.; Roffler, S.R.; Tzou, S.C.; Chuang, K.H.; Su, Y.C.; Chuang, C.H.; Leu, Y.L. An activity-based near-infrared glucuronide trapping probe for imaging β-glucuronidase expression in deep tissues. J. Am. Chem. Soc. 2012, 134, 3103–3110. [Google Scholar]
- Keereweer, S.; Hutteman, M.; Kerrebijn, J.D.F.; van de Velde, C.L.J.H.; Vahrmeijer, A.; Lowik, C.W.G.M. Translational optical imaging in diagnosis and treatment of cancer. Curr. Pharm. Biotechnol. 2012, 13, 498–503. [Google Scholar] [CrossRef]
- Crane, L.M.A.; Themelis, G.; Arts, H.J.G.; Buddingh, K.T.; Brouwers, A.H.; Ntziachristos, V.; van der Zee, A.G.J. Intraoperative near-infrared fluorescence imaging for sentinel lymph node detection in vulvar cancer: First clinical results. Gynecol. Oncol. 2011, 120, 291–295. [Google Scholar] [CrossRef]
- Sevick-Muraca, E.M. Translation of near-infrared fluorescence imaging technologies: Emerging clinical applications. Annu. Rev. Med. 2012, 63, 217–231. [Google Scholar] [CrossRef]
- Troyan, S.L.; Frangioni, J.V. The FLARE™ intraoperative near-infrared fluorescence imaging system: A first-in-human clinical trial in breast cancer sentinel lymph node mapping. Ann. Surg. Oncol. 2009, 16, 2943–2952. [Google Scholar] [CrossRef]
- Te Velde, E.A.; Veerman, T.; Subramaniam, V.; Ruers, T. The use of fluorescent dyes and probes in surgical oncology. Eur. J. Surg. Oncol. 2010, 36, 6–15. [Google Scholar] [CrossRef]
- Kachala, S.S.; Servais, E.L.; Park, B.J.; Rusch, V.W.; Adusumilli, P.S. Therapeutic sentinel lymph node imaging. Semin. Thorac. Cardiovasc. Surg. 2010, 21, 327–338. [Google Scholar]
- Aswathy, R.G.; Yoshida, Y.; Maekawa, T.; Kumar, D.S. Near-infrared quantum dots for deep tissue imaging. Anal. Bioanal. Chem. 2010, 397, 1417–1435. [Google Scholar] [CrossRef]
- Çamur, M.; Bulut, M.; Kandaz, M.; Güney, O. Synthesis, characterization and fluorescence behavior of new fluorescent probe phthalocyanines bearing coumarin substituents. Polyhedron 2009, 28, 233–238. [Google Scholar] [CrossRef]
- Nesterova, I.V.; Erdem, S.S.; Pakhomov, S.; Hammer, R.P.; Soper, S.A. Phthalocyanine dimerization-based molecular beacons using near-IR fluorescence. J. Am. Chem. Soc. 2009, 131, 2432–2433. [Google Scholar]
- Choi, C.F.; Tsang, P.T.; Huang, J.D.; Chan, E.Y.; Ko, W.H.; Fong, W.P.; Ng, D.K. Synthesis and in vitro photodynamic activity of new hexadeca-carboxy phthalocyanines. Chem. Commun. 2004, 19, 2236–2237. [Google Scholar]
- Soares, A.R.; Tomé, J.P.; Neves, M.G.; Tomé, A.C.; Cavaleiro, J.A.; Torres, T. Synthesis of water-soluble phthalocyanines bearing four or eight D-galactose units. Carbohydr. Res. 2009, 344, 507–510. [Google Scholar] [CrossRef]
- Aggarwal, A.; Singh, S.; Zhang, Y.; Anthes, M.; Samaroo, D.; Gao, R.; Drain, C.M. Synthesis and photophysics of an octathioglycosylated zinc (II) phthalocyanine. Tetrahedron Lett. 2011, 52, 5456–5459. [Google Scholar] [CrossRef]
- Choi, C.F.; Huang, J.D.; Lo, P.C.; Fong, W.P.; Ng, D.K. Glycosylated zinc (II) phthalocyanines as efficient photosensitisers for photodynamic therapy. Synthesis, photophysical properties and in vitro photodynamic activity. Org. Biomol. Chem. 2008, 6, 2173–2181. [Google Scholar] [CrossRef]
- Kimani, S.G.; Shmigol, T.A.; Hammond, S.; Phillips, J.B.; Bruce, J.I.; MacRobert, A.J.; Golding, J.P. Fully protected glycosylated Zinc (II) phthalocyanine shows high uptake and photodynamic cytotoxicity in MCF-7 cancer cells. Photochem. Photobiol. 2013, 89, 139–149. [Google Scholar] [CrossRef]
- Lv, F.; Li, Y.; Cao, B.; Liu, T. Galactose substituted zinc phthalocyanines as near infrared fluorescence probes for liver cancer imaging. J. Mater. Sci. Mater. Med. 2013, 24, 811–819. [Google Scholar] [CrossRef]
- Lv, F.; He, X.; Lu, L.; Wu, L.; Liu, T. Synthesis, properties and near-infrared imaging evaluation of glucose conjugated zinc phthalocyanines via Click reaction. J. Por. Phthal. 2012, 16, 77–84. [Google Scholar] [CrossRef]
- Lv, F.; He, X.; Wu, L.; Liu, T. Lactose substituted zinc phthalocyanine: A near infrared fluorescence imaging probe for liver cancer targeting. Bioorg. Med. Chem. Lett. 2013, 23, 1878–1882. [Google Scholar] [CrossRef]
- Vuignier, K.; Schappler, J.; Veuthey, J.L.; Carrupt, P.A.; Martel, S. Drug-protein binding: A critical review of analytical tools. Anal. Bioanal. Chem. 2010, 398, 53–66. [Google Scholar] [CrossRef]
- Sułkowska, A. Interaction of drugs with bovine and human serum albumin. J. Mol. Struct. 2002, 614, 227–232. [Google Scholar] [CrossRef]
- Peters, T., Jr. All about Albumin: Biochemistry, Genetics, and Medical Applications; Academic Press: Salt Lake City, UT, USA, 1995. [Google Scholar]
- Alarcón, E.; Edwards, A.M.; Garcia, A.M.; Muñoz, M.; Aspée, A.; Borsarelli, C.D.; Lissi, E.A. Photophysics and photochemistry of zinc phthalocyanine/bovine serum albumin adducts. Photochem. Photobiol. Sci. 2009, 8, 255–263. [Google Scholar] [CrossRef]
- Cheatum, C.M. Drug-protein interactions: Mechanisms of potency. Nat. Chem. 2013, 5, 152–153. [Google Scholar] [CrossRef]
- Bi, S.; Sun, Y.; Qiao, C.; Zhang, H.; Liu, C. Binding of several anti-tumor drugs to bovine serum albumin: Fluorescence study. J. Lumin. 2009, 129, 541–547. [Google Scholar] [CrossRef]
- Hein, C.D.; Liu, X.M.; Wang, D. Click chemistry, a powerful tool for pharmaceutical. Pharm. Res. 2008, 25, 2216–2230. [Google Scholar] [CrossRef]
- Tron, G.C.; Pirali, T.; Billington, R.A.; Canonico, P.L.; Sorba, G.; Genazzani, AA. Click chemistry reactions in medicinal chemistry: Applications of the 1,3-dipolar cycloaddition between azides and alkynes. Med. Res. Rev. 2008, 28, 278–308. [Google Scholar] [CrossRef]
- Lv, F.; Cao, B.; Cui, Y.; Liu, T. Zinc phthalocyanine labelled polyethylene glycol: Preparation, characterization, interaction with bovine serum albumin and near infrared fluorescence imaging in vivo. Molecules 2012, 17, 6348–6361. [Google Scholar] [CrossRef]
- Joshi, P.; Chakraborty, S.; Dey, S.; Shanker, V.; Ansari, Z.A.; Singh, S.P.; Chakrabarti, P. Binding of chloroquine-conjugated gold nanoparticles with bovine serum albumin. J. Colloid Interface Sci. 2011, 355, 402–409. [Google Scholar] [CrossRef]
- Ross, P.D.; Subramanian, S. Thermodynamics of protein association reactions: Forces contributing to stability. Biochemistry 1981, 20, 3096–3102. [Google Scholar] [CrossRef]
- Citrin, D.; Lee, A.K.; Scott, T.; Sproull, M.; Ménard, C.; Tofilon, P.J.; Kevin Camphausen, K. In vivo tumor imaging in mice with near-infrared labeled endostatin. Mol. Cancer Ther. 2004, 3, 481–488. [Google Scholar]
- Meier, R.; Boddington, S.; Krug, C.; Acosta, F.L.; Thullier, D.; Henning, T.D.; Sutton, E.J.; Tavri, S.; Lotz, J.C.; Daldrup-Link, H.E. Detection of postoperative granulation tissue with an ICG-enhanced integrated OI-/X-ray System. J. Transl. Med. 2008, 6, 73–82. [Google Scholar] [CrossRef]
- Herbert, B.J.; Dorsey, J.G. n-Octanol-water partition coefficient estimation by micellar electrokinetic capillary chromatography. Anal. Chem. 1995, 67, 744–749. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1 and 2 are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lu, L.; Lv, F.; Cao, B.; He, X.; Liu, T. Saccharide Substituted Zinc Phthalocyanines: Optical Properties, Interaction with Bovine Serum Albumin and Near Infrared Fluorescence Imaging for Sentinel Lymph Nodes. Molecules 2014, 19, 525-537. https://doi.org/10.3390/molecules19010525
Lu L, Lv F, Cao B, He X, Liu T. Saccharide Substituted Zinc Phthalocyanines: Optical Properties, Interaction with Bovine Serum Albumin and Near Infrared Fluorescence Imaging for Sentinel Lymph Nodes. Molecules. 2014; 19(1):525-537. https://doi.org/10.3390/molecules19010525
Chicago/Turabian StyleLu, Li, Feng Lv, Bo Cao, Xujun He, and Tianjun Liu. 2014. "Saccharide Substituted Zinc Phthalocyanines: Optical Properties, Interaction with Bovine Serum Albumin and Near Infrared Fluorescence Imaging for Sentinel Lymph Nodes" Molecules 19, no. 1: 525-537. https://doi.org/10.3390/molecules19010525



