Amazonian Plant Natural Products: Perspectives for Discovery of New Antimalarial Drug Leads
Abstract
:1. Introduction
2. Malaria Drug Resistance: Urging the Discovery and Development of New Drugs
3. Amazonian Plants: Sources of New Antimalarial Drugs Leads
4. Field Installed Capacity and Recent Findings from the Manaus Drug Discovery Group and Others
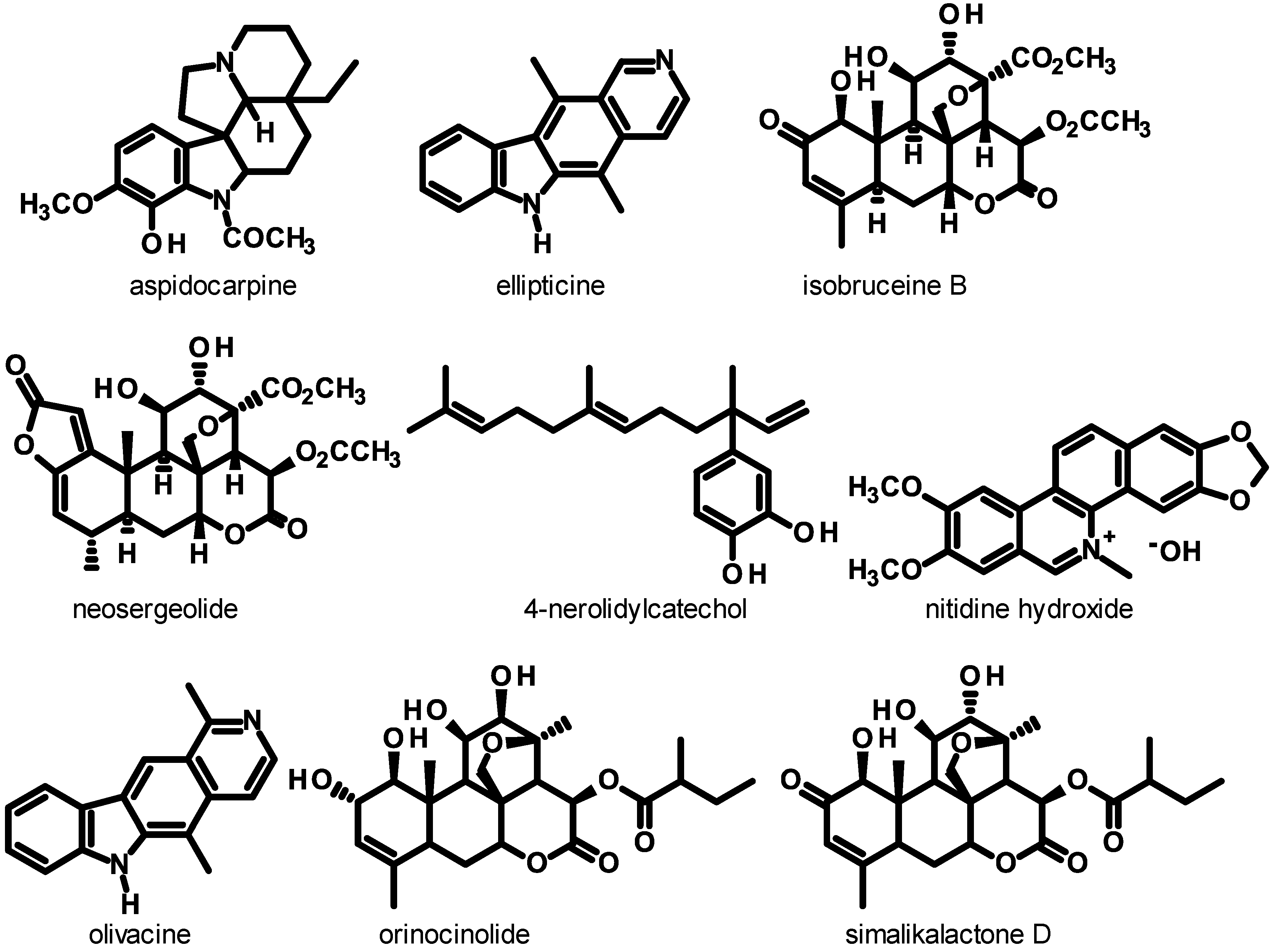
| Plant species | Family | Common name | Collection place | Part used | Active component | IC50 (μM) | Pf strain | Source |
|---|---|---|---|---|---|---|---|---|
| Aspidosperma desmanthum Benth. ex Müll. Arg. | Apocynaceae | araracanga | Brazil | bark | aspidocarpine | 0.019 | K1 | [29,32] |
| Aspidosperma ulei Markgr. | Apocynaceae | Brazil | root bark | uleine, olivacine | 1.2–3.7 | K1, 3D7, W2 | [33,34,35] | |
| Aspidosperma vargasii A. DC. | Apocynaceae | amarelão | Brazil | bark | ellipticine | 0.073–0.81 | K1, 3D7 | [29,33] |
| Carapa guianensis Aubl. | Meliaceae | andiroba | Peru | seed/flower oil | gedunins, an andirobin, mexicanolides, phragmalin-type limonoids (andirolides) | 2.5–15 | FCR-3 | [36] |
| Moronobea coccinea Aubl. | Clusiaceae | manniballi | French Guiana | latex | polyprenylated acylphloroglucinols, isogarcinol,cycloxanthochymol, garcinol | 2.1–37 | FcB1 | [37] |
| Picrolemma sprucei Hook. f. | Simaroubaceae | caferana | Brazil | root, stem | isobrucein B, neosergeolide | 0.002–0.008 | K1 | [29,38] |
| Piper peltatum L. | Piperaceae | caapeba | Brazil | root, leaf, flowering part | 4-nerolidylcatechol | 0.67 | K1 | [29] |
| Quassia amara L. | Simaroubaceae | quinquina de Cayenne | French Guiana | fresh leaf | simalikalactone D, simalikalactone E | 0.010–0.068 | FcB1, F32, W2 | [39,40] |
| Rheedia acuminata (Ruiz & Pavon) Planchon & Triana | Clusiaceae | cerillo, botoncillo, caraño | French Guiana | root bark | prenylated xanthones, polyprenylated acylphloroglucinols | 3.2–15 | FcB1 | [37] |
| Simaba orinocensis Kunth | Simaroubaceae | Peru | root bark | orinocinolide, simalikalactone D | 0.0063–0.018 | D6, W2 | [41] | |
| Tabebuia incana A.H. Gentry | Bignoniaceae | pau d´arco | Brazil | bark | 5 & 8-hydroxy hydroxyethyl naphtho[2,3-b]furan-4,9-diones | 0.67 | FcB2 | [42,43] |
| Tachia grandiflora Maguire & Weaver | Gentianaceae | caferana | Brazil | leaf | amplexine (djalonenol) | 35 | W2 | [44,45] |
| Tapirira guianensis Aubl. | Anacardiaceae | piojo | French Guiana | bark | cyclic alkyl polyols | 4.7–5.4 | F32, FcB1 | [46] |
| Zanthoxylum. rhoifolium Lam | Rutaceae | tachuelo | French Guiana | trunk bark | avicine hydroxide, nitidine hydroxide, fagaridine | <0.27–38 | FCB1 | [47] |
5. New Generation Antimalarials: Desirable and Essential Characteristics
6. Drug Discovery for P. falciparum: The Path to Appropriate Drug-Sensitive Assays
7. Plasmodium vivax Malaria: Neglected and Misinterpreted
8. Drug Discovery for P. vivax: The Next Step Forward
9. Hypnozoite Drug Tests: The Greatest Challenge
10. Perspectives
11. Concluding Remarks
Acknowledgments
Conflicts of Interest
References
- malERA Consultative Group on Drugs. A research agenda for malaria eradication: Drugs. PLoS Med. 2011, 8, e1000402. [CrossRef]
- White, N.J. Counter perspective: Artemisinin resistance: Facts, fears, and fables. Am. J. Trop. Med. Hyg. 2012, 87, 785. [Google Scholar] [CrossRef]
- Phyo, A.P.; Nkhoma, S.; Stepniewska, K.; Ashley, E.A.; Nair, S.; McGready, R.; ler Moo, C.; Al-Saai, S.; Dondorp, A.M.; Lwin, K.M.; et al. Emergence of artemisinin-resistant malaria on the western border of Thailand: a longitudinal study. Lancet 2012, 379, 1960–1966. [Google Scholar] [CrossRef]
- Milliken, W. Plants for Malaria, Plants for Fever: Medicinal Species in Latin America, A Bibliographic Survey; Royal Botanic Gardens, Kew: Richmond, VA, USA, 1997; p. 116. [Google Scholar]
- Ruiz, L.; Ruiz, L.; Maco, M.; Cobos, M.; Gutierrez-Choquevilca, A.L.; Roumy, V. Plants used by native Amazonian groups from the Nanay River (Peru) for the treatment of malaria. J. Ethnopharmacol. 2011, 133, 917–921. [Google Scholar] [CrossRef]
- Vigneron, M.; Deparis, X.; Deharo, E.; Bourdy, G. Antimalarial remedies in French Guiana: a knowledge attitudes and practices study. J. Ethnopharmacol. 2005, 98, 351–360. [Google Scholar] [CrossRef]
- Bero, J.; Quetin-Leclercq, J. Natural products published in 2009 from plants traditionally used to treat malaria. Planta Med. 2011, 77, 631–640. [Google Scholar] [CrossRef] [Green Version]
- Deharo, E.; Ginsburg, H. Analysis of additivity and synergism in the anti-plasmodial effect of purified compounds from plant extracts. Malar. J. 2011, 10 (Suppl 1), S5. [Google Scholar] [CrossRef]
- Andrade-Neto, V.F.; Brandao, M.G.; Nogueira, F.; Rosario, V.E.; Krettli, A.U. Ampelozyziphus amazonicus Ducke (Rhamnaceae), a medicinal plant used to prevent malaria in the Amazon Region, hampers the development of Plasmodium berghei sporozoites. Int. J. Parasitol. 2008, 38, 1505–1511. [Google Scholar] [CrossRef]
- World Health Organization, Global Report on Antimalarial Drug Efficacy and Drug Resistance: 2000–2010; World Health Organization: Geneva, Switzerland, 2010; p. 115.
- Young, M.D.; Moore, D.V. Chloroquine resistance in Plasmodium falciparum. Am. J. Trop. Med. Hyg. 1961, 10, 317–320. [Google Scholar]
- Harinasuta, T.; Migasen, S.; Boonag, D. Chloroquine Resistance in Plasmodium Falciparum in Thailand; UNESCO First Regional Symposium on Scientific Knowledge of Tropical Parasites: Singapore University, Singapore, 1962. [Google Scholar]
- Peters, W. Chemotherapy and Drug Resistance in Malaria, 2nd ed.; Academic: London, UK, 1987. [Google Scholar]
- Roper, C.; Pearce, R.; Nair, S.; Sharp, B.; Nosten, F.; Anderson, T. Intercontinental spread of pyrimethamine-resistant malaria. Science 2004, 305, 1124. [Google Scholar] [CrossRef]
- Enserink, M. Combating malaria. Malaria treatment: ACT two. Science 2007, 318, 560–563. [Google Scholar] [CrossRef]
- World Health Organization; Roll Back Malaria Dept. Guidelines for the Treatment of Malaria; World Health Organization: Geneva, Switzerland, 2006; p. 253. [Google Scholar]
- Da Mata, A.A. Flora Médica Brasiliense. Valer Editora: Manaus, Brazil, 2003. [Google Scholar]
- Elisabetsky, E.; Shanley, P. Ethnopharmacology in the Brazilian Amazon. Pharmacol. Therap. 1994, 64, 201–214. [Google Scholar] [CrossRef]
- Rodrigues, E. Plants and animals utilized as medicines in the Jau National Park (JNP), Brazilian Amazon. Phytother. Res. 2006, 20, 378–391. [Google Scholar] [CrossRef]
- Santos, J.F.; Pagani, E.; Ramos, J.; Rodrigues, E. Observations on the therapeutic practices of riverine communities of the Unini River, AM, Brazil. J. Ethnopharmacol. 2012, 142, 503–515. [Google Scholar] [CrossRef]
- Suffredini, I.B.; Paciencia, M.L.; Varella, A.D.; Younes, R.N. Antibacterial activity of Brazilian Amazon plant extracts. Braz. J. Infect. Dis. 2006, 10, 400–402. [Google Scholar]
- Carneiro, A.L.; Teixeira, M.F.; Oliveira, V.M.; Fernandes, O.C.; Cauper, G.S.; Pohlit, A.M. Screening of Amazonian plants from the Adolpho Ducke forest reserve, Manaus, state of Amazonas, Brazil, for antimicrobial activity. Mem. Inst. Oswaldo Cruz 2008, 103, 31–38. [Google Scholar]
- Pohlit, A.M.; Lopes, N.P.; Gama, R.A.; Tadei, W.P.; Neto, V.F. Patent literature on mosquito repellent inventions which contain plant essential oils-A review. Planta Med. 2011, 77, 598–617. [Google Scholar] [CrossRef]
- Pohlit, A.M.; Rezende, A.R.; Lopes Baldin, E.L.; Lopes, N.P.; Neto, V.F. Plant extracts, isolated phytochemicals, and plant-derived agents which are lethal to arthropod vectors of human tropical diseases-A review. Planta Med. 2011, 77, 618–630. [Google Scholar] [CrossRef]
- Schmidt, T.J.; Khalid, S.A.; Romanha, A.J.; Alves, T.M.; Biavatti, M.W.; Brun, R.; Da Costa, F.B.; de Castro, S.L.; Ferreira, V.F.; de Lacerda, M.V.; et al. The potential of secondary metabolites from plants as drugs or leads against protozoan neglected diseases-part I. Curr. Med. Chem. 2012, 19, 2128–2175. [Google Scholar]
- Achan, J.; Talisuna, A.O.; Erhart, A.; Yeka, A.; Tibenderana, J.K.; Baliraine, F.N.; Rosenthal, P.J.; D'Alessandro, U. Quinine, an old anti-malarial drug in a modern world: role in the treatment of malaria. Malar. J. 2011, 10, 144. [Google Scholar] [CrossRef]
- Bray, P.G.; Ward, S.A.; O'Neill, P.M. Quinolines and artemisinin: chemistry, biology and history. Curr. Top. Microbiol. Immunol. 2005, 295, 3–38. [Google Scholar] [CrossRef]
- Pohlit, A.M.; Jabor, V.A. P.; Amorim, R.C. N.; Costa e Silva, E.C.; Lopes, N.P. LC-ESI-MS Determination of Quassinoids Isobrucein B and Neosergeolide in Picrolemma sprucei Stem Infusions. J. Braz. Chem. Soc. 2009, 20, 1065–1070. [Google Scholar] [CrossRef]
- de Andrade-Neto, V.F.; Pohlit, A.M.; Pinto, A.C.; Silva, E.C.; Nogueira, K.L.; Melo, M.R.; Henrique, M.C.; Amorim, R.C.; Silva, L.F.; Costa, M.R.; et al. In vitro inhibition of plasmodium falciparum by substances isolated from Amazonian antimalarial plants. Mem. Inst. Oswaldo Cruz 2007, 102, 359–365. [Google Scholar]
- Pinto, A.C.; Chaves, F.C.; dos Santos, P.A.; Nunez, C.V.; Tadei, W.P.; Pohlit, A.M. Piper peltatum: Biomass and 4-nerolidylcatechol production. Planta Med. 2010, 76, 1473–1476. [Google Scholar] [CrossRef]
- Pinto, P.S. Estudo químico e atividade biológica de frações do extrato etanólico da raiz de Cassia spruceana Benth. (Leguminosae: Caesalpinioideae). M.Sc. Thesis, Universidade Federal do Amazonas, Manaus, Brazil, 2006. [Google Scholar]
- Henrique, M.C.; Nunomura, S.M.; Pohlit, A.M. Indole alkaloids from the bark of Aspidosperma vargasii and A. desmanthum. Quim. Nova 2010, 33, 284–287. [Google Scholar] [CrossRef]
- Rocha e Silva, L.F.; Montoia, A.; Amorim, R.C.; Melo, M.R.; Henrique, M.C.; Nunomura, S.M.; Costa, M.R.; Andrade Neto, V.F.; Costa, D.S.; Dantas, G.; et al. Comparative in vitro and in vivo antimalarial activity of the indole alkaloids ellipticine, olivacine, cryptolepine and a synthetic cryptolepine analog. Phytomed. Int. J. Phytother. Phytopharmacol. 2012, 20, 71–76. [Google Scholar] [CrossRef]
- Dos Santos Torres, Z.E.; Silveira, E.R.; Rocha, E.S.L.F.; Lima, E.S.; de Vasconcellos, M.C.; de Andrade Uchoa, D.E.; Filho, R.B.; Pohlit, A.M. Chemical Composition of Aspidosperma ulei Markgr and Antiplasmodial Activity of Selected Indole Alkaloids. Molecules 2013, 18, 6281–6297. [Google Scholar] [CrossRef]
- Oliveira, A.B.; Dolabela, M.F.; Póvoa, M.M.; Santos, C.A.M.; Varotti, F.P. Antimalarial activity of ulein and proof of its action on the Plasmodium falciparum digestive vacuole. Malar. J. 2010. [Google Scholar] [CrossRef]
- Tanaka, Y.; Sakamoto, A.; Inoue, T.; Yamada, T.; Kikuchi, T.; Kajimoto, T.; Muraoka, O.; Sato, A.; Wataya, Y.; Kim, H.-S.; et al. Andirolides H–P from the flower of andiroba (Carapa guianensis, Meliaceae). Tetrahedron 2012, 68, 3669–3677. [Google Scholar] [CrossRef]
- Marti, G.; Eparvier, V.; Moretti, C.; Susplugas, S.; Prado, S.; Grellier, P.; Retailleau, P.; Gueritte, F.; Litaudon, M. Antiplasmodial benzophenones from the trunk latex of Moronobea coccinea (Clusiaceae). Phytochemistry 2009, 70, 75–85. [Google Scholar] [CrossRef]
- Pinto, A.C.; Silva, L.F.; Cavalcanti, B.C.; Melo, M.R.; Chaves, F.C.; Lotufo, L.V.; de Moraes, M.O.; de Andrade-Neto, V.F.; Tadei, W.P.; Pessoa, C.O.; et al. New antimalarial and cytotoxic 4-nerolidylcatechol derivatives. Eur. J. Med. Chem. 2009, 44, 2731–2735. [Google Scholar] [CrossRef]
- Bertani, S.; Houel, E.; Stien, D.; Chevolot, L.; Jullian, V.; Garavito, G.; Bourdy, G.; Deharo, E. Simalikalactone D is responsible for the antimalarial properties of an Amazonian traditional remedy made with Quassia amara L. (Simaroubaceae). J. Ethnopharmacol. 2006, 108, 155–157. [Google Scholar] [CrossRef]
- Cachet, N.; Hoakwie, F.; Bertani, S.; Bourdy, G.; Deharo, E.; Stien, D.; Houel, E.; Gornitzka, H.; Fillaux, J.; Chevalley, S.; et al. Antimalarial activity of simalikalactone E, a new quassinoid from Quassia amara L. (Simaroubaceae). Antimicrob. Agents Chemother. 2009, 53, 4393–4398. [Google Scholar] [CrossRef]
- Muhammad, I.; Bedir, E.; Khan, S.I.; Tekwani, B.L.; Khan, I.A.; Takamatsu, S.; Pelletier, J.; Walker, L.A. A new antimalarial quassinoid from Simaba orinocensis. J. Nat. Prod. 2004, 67, 772–777. [Google Scholar] [CrossRef]
- Morais, S.K.R.; Silva, S.G.; Portela, C.N.; Nunomura, S.M.; Quignard, E.L.J.; Pohlit, A.M. Bioactive dihydroxyfuranonaphthoquinones from the bark of Tabebuia incana A.H. Gentry (Bignoniaceae) and HPLC analysis of commercial pau d'arco and certified T. incana bark infusions. Acta Amaz. 2007, 37, 99–102. [Google Scholar] [CrossRef]
- Pérez, H.; Diaz, F.; Medina, J.D. Chemical investigation and in vitro antimalarial activity of Tabebuia ochracea ssp. neochrysantha. Pharm. Biol. 1997, 35, 227–235. [Google Scholar] [CrossRef]
- Rocha e Silva, L.F.; Lima, E.S.; Vasconcellos, M.C.; Aranha, E.S. P.; Costa, D.S.; Santos, E.V.M.; Silva, T.C.M.; Morais, S.K.R.; Quignard, E.L.J.; Alecrim, M.G.C.; et al. In vitro and in vivo antimalarial activity and cytotoxicity of extracts, fractions and a substance isolated from the Amazonian plant Tachia grandiflora (Gentianaceae). Mem. Inst. Oswaldo Cruz 2013, 108, 501–507. [Google Scholar]
- Pohlit, A.M.; Rocha e Silva, L.F.; Henrique, M.C.; Montoia, A.; Amorim, R.C.; Nunomura, S.M.; Andrade-Neto, V.F. Antimalarial activity of ellipticine. Phytom. Int. J. Phytother. Phytopharmacol. 2012, 19, 1049. [Google Scholar] [CrossRef]
- Roumy, V.; Fabre, N.; Portet, B.; Bourdy, G.; Acebey, L.; Vigor, C.; Valentin, A.; Moulis, C. Four anti-protozoal and anti-bacterial compounds from Tapirira guianensis. Phytochem. 2009, 70, 305–311. [Google Scholar] [CrossRef]
- Jullian, V.; Bourdy, G.; Georges, S.; Maurel, S.; Sauvain, M. Validation of use of a traditional antimalarial remedy from French Guiana, Zanthoxylum rhoifolium Lam. J. Ethnopharmacol. 2006, 106, 348–352. [Google Scholar] [CrossRef]
- Saraiva Nunomura, R.C.; Pinto, A.C.; Nunomura, S.M.; Pohlit, A.M.; Fernandes Amaral, A.C. Chemical constitutents from stems of Simaba guianensis subesp ecaudata (Cronquist). Quim. Nova 2012, 35, 2153–2158. [Google Scholar] [CrossRef]
- Montoia, A. Semi-síntese de derivados da elipticina e atividade antimalárica de isolados e infusões de Aspidosperma vargasii. M.Sc. Thesis, Universidade Federal do Amazonas, Manaus, Brazil, 2013. [Google Scholar]
- Camargo, M.R.M. Avaliação da Atividade Antimalárica e Antimicrobiana de Geissospermum argenteum e Minquartia guianensis, coletadas em Roraima. M.Sc. Thesis, Universidade Federal de Rondônia, Brazil, 2011. [Google Scholar]
- Pinto, P.S.; Rocha e Silva, L.F.; Amorim, R.C.N.; Melo, M.R.S.; Nunomura, S.M.; Pohlit, A.M.Q.N. Phenolic constituents from the roots of Cassia spruceana Benth. (Fabaceae). Quim. Nova 2013. submitted for publish.
- Cavalcanti, B.C.; da Costa, P.M.; Carvalho, A.A.; Rodrigues, F.A.; Amorim, R.C.; Silva, E.C.; Pohlit, A.M.; Costa-Lotufo, L.V.; Moraes, M.O.; Pessoa, C. Involvement of intrinsic mitochondrial pathway in neosergeolide-induced apoptosis of human HL-60 leukemia cells: the role of mitochondrial permeability transition pore and DNA damage. Pharm. Biol. 2012, 50, 980–993. [Google Scholar] [CrossRef]
- Cabral, J.A.; McChesney, J.D.; Milhous, W.K. A new antimalarial quassinoid from Simaba guianensis. J. Nat. Prod. 1993, 56, 1954–1961. [Google Scholar] [CrossRef]
- Passemar, C.; Salery, M.; Soh, P.N.; Linas, M.D.; Ahond, A.; Poupat, C.; Benoit-Vical, F. Indole and aminoimidazole moieties appear as key structural units in antiplasmodial molecules. Phytomed. Int. J. Phytother. Phytopharmacol. 2011, 18, 1118–1125. [Google Scholar] [CrossRef]
- Chong, C.R.; Sullivan, D.J., Jr. Inhibition of heme crystal growth by antimalarials and other compounds: implications for drug discovery. Biochem. Pharmacol. 2003, 66, 2201–2212. [Google Scholar] [CrossRef]
- Rocha, E.S.L.F.; Silva Pinto, A.C.; Pohlit, A.M.; Quignard, E.L.; Vieira, P.P.; Tadei, W.P.; Chaves, F.C.; Samonek, J.F.; Lima, C.A.; Costa, M.R.; et al. In vivo and in vitro antimalarial activity of 4-nerolidylcatechol. Phytother. Res. 2011, 25, 1181–1188. [Google Scholar] [CrossRef]
- Silva Lima, E.; Silva Pinto, A.C.; Nogueira, K.L.; Rocha e Silva, L.F.; Oliveira de Almeida, P.D.; Carvalho de Vasconcellos, M.; Chaves, F.C.; Tadei, W.P.; Pohlit, A.M. Stability and antioxidant activity of semi-synthetic derivatives of 4-nerolidylcatechol. Molecules 2012, 18, 178–189. [Google Scholar] [CrossRef]
- Mota, M.L.; Lobo, L.T.; Costa, J.M.; Costa, L.S.; Rocha, H.A.; Rocha e Silva, L.F.; Pohlit, A.M.; Neto, V.F. In vitro and in vivo antimalarial activity of essential oils and chemical components from three medicinal plants found in northeastern Brazil. Planta Med. 2012, 78, 658–664. [Google Scholar] [CrossRef]
- Baird, J.K. Eliminating malaria--all of them. Lancet 2010, 376, 1883–1885. [Google Scholar] [CrossRef]
- Schrader, F.C.; Barho, M.; Steiner, I.; Ortmann, R.; Schlitzer, M. The antimalarial pipeline-An update. Int. J. Med. Microbiol. 2012, 302, 165–171. [Google Scholar] [CrossRef]
- Rieckmann, K.H.; McNamara, J.V.; Frischer, H.; Stockert, T.A.; Carson, P.E.; Powell, R.D. Effects of chloroquine, quinine, and cycloguanil upon the maturation of asexual erythrocytic forms of two strains of Plasmodium falciparum in vitro. Am. J. Trop. Med. Hyg. 1968, 17, 661–671. [Google Scholar]
- Rieckmann, K.H.; Campbell, G.H.; Sax, L.J.; Mrema, J.E. Drug sensitivity of plasmodium falciparum. An in vitro microtechnique. Lancet 1978, 1, 22–23. [Google Scholar]
- Noedl, H.; Wongsrichanalai, C.; Wernsdorfer, W.H. Malaria drug-sensitivity testing: New assays, new perspectives. Trends Parasitol. 2003, 19, 175–181. [Google Scholar] [CrossRef]
- Trager, W.; Jensen, J.B. Human malaria parasites in continuous culture. Science 1976, 193, 673–675. [Google Scholar]
- Desjardins, R.E.; Canfield, C.J.; Haynes, J.D.; Chulay, J.D. Quantitative assessment of antimalarial activity in vitro by a semiautomated microdilution technique. Antimicrob. Agents Chemother. 1979, 16, 710–718. [Google Scholar] [CrossRef]
- Aguiar, A.C.; Rocha, E.M.; Souza, N.B.; Franca, T.C.; Krettli, A.U. New approaches in antimalarial drug discovery and development: a review. Mem. Inst. Oswaldo Cruz 2012, 107, 831–845. [Google Scholar] [CrossRef]
- Makler, M.T.; Ries, J.M.; Williams, J.A.; Bancroft, J.E.; Piper, R.C.; Gibbins, B.L.; Hinrichs, D.J. Parasite lactate dehydrogenase as an assay for Plasmodium falciparum drug sensitivity. Am. J. Trop. Med. Hyg. 1993, 48, 739–741. [Google Scholar]
- Druilhe, P.; Moreno, A.; Blanc, C.; Brasseur, P.H.; Jacquier, P. A colorimetric in vitro drug sensitivity assay for Plasmodium falciparum based on a highly sensitive double-site lactate dehydrogenase antigen-capture enzyme-linked immunosorbent assay. Am. J. Trop. Med. Hyg. 2001, 64, 233–241. [Google Scholar]
- Noedl, H.; Wernsdorfer, W.H.; Miller, R.S.; Wongsrichanalai, C. Histidine-rich protein II: a novel approach to malaria drug sensitivity testing. Antimicrob. Agents Chemother. 2002, 46, 1658–1664. [Google Scholar] [CrossRef]
- Plouffe, D.; Brinker, A.; McNamara, C.; Henson, K.; Kato, N.; Kuhen, K.; Nagle, A.; Adrian, F.; Matzen, J.T.; Anderson, P.; et al. In silico activity profiling reveals the mechanism of action of antimalarials discovered in a high-throughput screen. Proc. Natl. Acad. Sci. USA 2008, 105, 9059–9064. [Google Scholar] [CrossRef]
- Cervantes, S.; Stout, P.E.; Prudhomme, J.; Engel, S.; Bruton, M.; Cervantes, M.; Carter, D.; Tae-Chang, Y.; Hay, M.E.; Aalbersberg, W.; et al. High content live cell imaging for the discovery of new antimalarial marine natural products. BMC Infect. Dis. 2012, 12, 1. [Google Scholar] [CrossRef]
- van Vianen, P.H.; Thaithong, S.; Reinders, P.P.; van Engen, A.; van der Keur, M.; Tanke, H.J.; van der Kaay, H.J.; Mons, B. Automated flow cytometric analysis of drug susceptibility of malaria parasites. Am. J. Trop. Med. Hyg. 1990, 43, 602–607. [Google Scholar]
- Saito-Ito, A.; Akai, Y.; He, S.; Kimura, M.; Kawabata, M. A rapid, simple and sensitive flow cytometric system for detection of Plasmodium falciparum. Parasitol. Int. 2001, 50, 249–257. [Google Scholar] [CrossRef]
- Wilson, D.W.; Crabb, B.S.; Beeson, J.G. Development of fluorescent Plasmodium falciparum for in vitro growth inhibition assays. Malar. J. 2010, 9, 152. [Google Scholar] [CrossRef]
- Che, P.; Cui, L.; Kutsch, O.; Cui, L.; Li, Q. Validating a firefly luciferase-based high-throughput screening assay for antimalarial drug discovery. Assay Drug Dev. Technol. 2012, 10, 61–68. [Google Scholar] [CrossRef]
- Cui, L.; Miao, J.; Wang, J.; Li, Q.; Cui, L. Plasmodium falciparum: development of a transgenic line for screening antimalarials using firefly luciferase as the reporter. Exp. Parasitol. 2008, 120, 80–87. [Google Scholar] [CrossRef]
- Krettli, A.U.; Adebayo, J.O.; Krettli, L.G. Testing of natural products and synthetic molecules aiming at new antimalarials. Curr. Drug Targets 2009, 10, 261–270. [Google Scholar]
- Gething, P.W.; Elyazar, I.R.; Moyes, C.L.; Smith, D.L.; Battle, K.E.; Guerra, C.A.; Patil, A.P.; Tatem, A.J.; Howes, R.E.; Myers, M.F.; et al. A long neglected world malaria map: Plasmodium vivax endemicity in 2010. PLoS neglect Trop. Dis. 2012, 6, e1814. [Google Scholar] [CrossRef]
- Guerra, C.A.; Howes, R.E.; Patil, A.P.; Gething, P.W.; Van Boeckel, T.P.; Temperley, W.H.; Kabaria, C.W.; Tatem, A.J.; Manh, B.H.; Elyazar, I.R.; et al. The international limits and population at risk of Plasmodium vivax transmission in 2009. PLoS Negl. Trop. Dis. 2010, 4, e774. [Google Scholar] [CrossRef]
- Boyd, M.F.; Stratman-Thomas, W.K.; Muench, H. The occurrence of gametocytes of Plasmodium vivax during the primary attack. Am. J. Trop. Med. Hyg. 1935, 16, 133–138. [Google Scholar]
- Feachem, R.G.; Phillips, A.A.; Hwang, J.; Cotter, C.; Wielgosz, B.; Greenwood, B.M.; Sabot, O.; Rodriguez, M.H.; Abeyasinghe, R.R.; Ghebreyesus, T.A.; et al. Shrinking the malaria map: progress and prospects. Lancet 2010, 376, 1566–78. [Google Scholar] [CrossRef]
- Rodriguez, J.C.; Uribe, G.A.; Araujo, R.M.; Narvaez, P.C.; Valencia, S.H. Epidemiology and control of malaria in Colombia. Mem. Inst. Oswaldo Cruz 2011, 106, 114–122. [Google Scholar]
- Gama, B.E.; Lacerda, M.V.; Daniel-Ribeiro, C.T.; Ferreira-da-Cruz Mde, F. Chemoresistance of Plasmodium falciparum and Plasmodium vivax parasites in Brazil: consequences on disease morbidity and control. Mem. Inst. Oswaldo Cruz 2011, 106, 159–166. [Google Scholar] [CrossRef]
- Oliveira-Ferreira, J.; Lacerda, M.V.; Brasil, P.; Ladislau, J.L.; Tauil, P.L.; Daniel-Ribeiro, C.T. Malaria in Brazil: An overview. Malar. J. 2010, 9, 115. [Google Scholar] [CrossRef]
- World Health Organization, World Malaria Report 2011; World Health Organization: Geneva, Switzerland, 2011; p. 246.
- Price, R.N.; Tjitra, E.; Guerra, C.A.; Yeung, S.; White, N.J.; Anstey, N.M. Vivax malaria: Neglected and not benign. Am. J. Trop. Med. Hyg. 2007, 77, 79–87. [Google Scholar]
- Baird, J.K. Neglect of Plasmodium vivax malaria. Trends Parasitol. 2007, 23, 533–539. [Google Scholar] [CrossRef]
- Tjitra, E.; Anstey, N.M.; Sugiarto, P.; Warikar, N.; Kenangalem, E.; Karyana, M.; Lampah, D.A.; Price, R.N. Multidrug-resistant Plasmodium vivax associated with severe and fatal malaria: A prospective study in Papua, Indonesia. PLoS Med. 2008, 5, e128. [Google Scholar] [CrossRef]
- Genton, B.; D'Acremont, V.; Rare, L.; Baea, K.; Reeder, J.C.; Alpers, M.P.; Muller, I. Plasmodium vivax and mixed infections are associated with severe malaria in children: a prospective cohort study from Papua New Guinea. PLoS Med. 2008, 5, e127. [Google Scholar] [CrossRef]
- Beg, M.A.; Sani, N.; Mehraj, V.; Jafri, W.; Khan, M.A.; Malik, A.; Menezes, E.; Hussain, R.; Smego, R., Jr. Comparative features and outcomes of malaria at a tertiary care hospital in Karachi, Pakistan. Int. J. Infect. Dis. 2008, 12, 37–42. [Google Scholar] [CrossRef]
- Barcus, M.J.; Basri, H.; Picarima, H.; Manyakori, C.; Sekartuti; Elyazar, I.; Bangs, M.J.; Maguire, J.D.; Baird, J.K. Demographic risk factors for severe and fatal vivax and falciparum malaria among hospital admissions in northeastern Indonesian Papua. Am. J. Trop. Med. Hyg. 2007, 77, 984–991. [Google Scholar]
- Lampah, D.A.; Yeo, T.W.; Hardianto, S.O.; Tjitra, E.; Kenangalem, E.; Sugiarto, P.; Price, R.N.; Anstey, N.M. Coma associated with microscopy-diagnosed Plasmodium vivax: a prospective study in Papua, Indonesia. PLoS Negl. Trop. Dis. 2011, 5, e1032. [Google Scholar] [CrossRef]
- Kochar, D.K.; Das, A.; Kochar, S.K.; Saxena, V.; Sirohi, P.; Garg, S.; Kochar, A.; Khatri, M.P.; Gupta, V. Severe Plasmodium vivax malaria: a report on serial cases from Bikaner in northwestern India. Am. J. Trop. Med. Hyg. 2009, 80, 194–198. [Google Scholar]
- Kochar, D.K.; Saxena, V.; Singh, N.; Kochar, S.K.; Kumar, S.V.; Das, A. Plasmodium vivax malaria. Emerg. Infect. Dis. 2005, 11, 132–134. [Google Scholar] [CrossRef]
- Tan, L.K.; Yacoub, S.; Scott, S.; Bhagani, S.; Jacobs, M. Acute lung injury and other serious complications of Plasmodium vivax malaria. Lancet Infect. Dis. 2008, 8, 449–454. [Google Scholar] [CrossRef]
- Bassat, Q.; Alonso, P.L. Defying malaria: Fathoming severe Plasmodium vivax disease. Nat. Med. 2011, 17, 48–49. [Google Scholar] [CrossRef]
- Anstey, N.M.; Russell, B.; Yeo, T.W.; Price, R.N. The pathophysiology of vivax malaria. Trends Parasitol. 2009, 25, 220–227. [Google Scholar] [CrossRef]
- Costa, F.T.; Lopes, S.C.; Albrecht, L.; Ataide, R.; Siqueira, A.M.; Souza, R.M.; Russell, B.; Renia, L.; Marinho, C.R.; Lacerda, M.V. On the pathogenesis of Plasmodium vivax malaria: perspectives from the Brazilian field. Int. J. Parasitol. 2012, 42, 1099–1105. [Google Scholar] [CrossRef]
- Lacerda, M.V.; Fragoso, S.C.; Alecrim, M.G.; Alexandre, M.A.; Magalhaes, B.M.; Siqueira, A.M.; Ferreira, L.C.; Araujo, J.R.; Mourao, M.P.; Ferrer, M.; et al. Postmortem characterization of patients with clinical diagnosis of Plasmodium vivax malaria: to what extent does this parasite kill? Clin. Infect. Dis. 2012, 55, e67–e74. [Google Scholar] [CrossRef]
- Alexandre, M.A.; Ferreira, C.O.; Siqueira, A.M.; Magalhaes, B.L.; Mourao, M.P.; Lacerda, M.V.; Alecrim, M. Severe Plasmodium vivax malaria, Brazilian Amazon. Emerg. Infect. Dis. 2010, 16, 1611–1614. [Google Scholar] [CrossRef]
- de Lacerda, M.V.; Zackiewicz, C.; Alecrim, W.D.; Alecrim, M. The neglected Plasmodium vivax: are researchers from endemic areas really concerned about new treatment options? Rev. Soc. Bras. Med. Trop. 2007, 40, 489–490. [Google Scholar] [CrossRef]
- Price, R.N.; Douglas, N.M.; Anstey, N.M.; von Seidlein, L. Plasmodium vivax treatments: What are we looking for? Curr. Opin.Infect. Dis. 2011, 24, 578–585. [Google Scholar] [CrossRef]
- Carlton, J.M.; Sina, B.J.; Adams, J.H. Why Is Plasmodium vivax a neglected tropical disease? PLoS Negl. Trop. Dis. 2011, 5, e1160. [Google Scholar] [CrossRef]
- Rieckmann, K.H.; Davis, D.R.; Hutton, D.C. Plasmodium vivax resistance to chloroquine? Lancet 1989, 2, 1183–1184. [Google Scholar] [CrossRef]
- Harinasuta, T.; Suntharasamai, P.; Viravan, C. Chloroquine-resistant falciparum malaria in Thailand. Lancet 1965, 2, 657–660. [Google Scholar] [CrossRef]
- Sumawinata, I.W.; Bernadeta; Leksana, B.; Sutamihardja, A.; Purnomo; Subianto, B.; Sekartuti; Fryauff, D.J.; Baird, J.K. Very high risk of therapeutic failure with chloroquine for uncomplicated Plasmodium falciparum and P. vivax malaria in Indonesian Papua. Am. J. Trop. Med. Hyg. 2003, 68, 416–420. [Google Scholar]
- Baird, J.K.; Wiady, I.; Fryauff, D.J.; Sutanihardja, M.A.; Leksana, B.; Widjaya, H.; Kysdarmanto; Subianto, B. In vivo resistance to chloroquine by Plasmodium vivax and Plasmodium falciparum at Nabire, Irian Jaya, Indonesia. Am. J. Trop. Med. Hyg. 1997, 56, 627–631. [Google Scholar]
- Sutanto, I.; Suprijanto, S.; Nurhayati; Manoempil, P.; Baird, J.K. Resistance to chloroquine by Plasmodium vivax at Alor in the Lesser Sundas Archipelago in eastern Indonesia. Am. J. Trop. Med. Hyg. 2009, 81, 338–342. [Google Scholar]
- Ratcliff, A.; Siswantoro, H.; Kenangalem, E.; Wuwung, M.; Brockman, A.; Edstein, M.D.; Laihad, F.; Ebsworth, E.P.; Anstey, N.M.; Tjitra, E.; et al. Therapeutic response of multidrug-resistant Plasmodium falciparum and P. vivax to chloroquine and sulfadoxine-pyrimethamine in southern Papua, Indonesia. Trans. R.oyal Soc. Trop. Med. Hyg. 2007, 101, 351–359. [Google Scholar] [CrossRef]
- Young, M.D.; Burgess, R.W. Pyrimethamine resistance in Plasmodium vivax malaria. Bull. WorldHealth Org. 1959, 20, 27–36. [Google Scholar]
- Baird, J.K. Resistance to therapies for infection by Plasmodium vivax. Clin. Microbiol. Rev. 2009, 22, 508–534. [Google Scholar] [CrossRef]
- Russell, B.; Chalfein, F.; Prasetyorini, B.; Kenangalem, E.; Piera, K.; Suwanarusk, R.; Brockman, A.; Prayoga, P.; Sugiarto, P.; Cheng, Q.; et al. Determinants of in vitro drug susceptibility testing of Plasmodium vivax. Antimicrob. Agents Chemother. 2008, 52, 1040–1045. [Google Scholar] [CrossRef]
- Sharrock, W.W.; Suwanarusk, R.; Lek-Uthai, U.; Edstein, M.D.; Kosaisavee, V.; Travers, T.; Jaidee, A.; Sriprawat, K.; Price, R.N.; Nosten, F.; et al. Plasmodium vivax trophozoites insensitive to chloroquine. Malar. J. 2008, 7, 94. [Google Scholar] [CrossRef]
- Price, R.N.; Auburn, S.; Marfurt, J.; Cheng, Q. Phenotypic and genotypic characterisation of drug-resistant Plasmodium vivax. Trends Parasitol. 2012, 28, 522–529. [Google Scholar] [CrossRef]
- Chotivanich, K.; Udomsangpetch, R.; Chierakul, W.; Newton, P.N.; Ruangveerayuth, R.; Pukrittayakamee, S.; Looareesuwan, S.; White, N.J. In vitro efficacy of antimalarial drugs against Plasmodium vivax on the western border of Thailand. Am. J. Trop. Med. Hyg. 2004, 70, 395–397. [Google Scholar]
- Kerlin, D.H.; Boyce, K.; Marfurt, J.; Simpson, J.A.; Kenangalem, E.; Cheng, Q.; Price, R.N.; Gatton, M.L. An analytical method for assessing stage-specific drug activity in Plasmodium vivax malaria: implications for ex vivo drug susceptibility testing. PLoS Negl. Trop. Dis. 2012, 6, e1772. [Google Scholar] [CrossRef] [Green Version]
- Marfurt, J.; Chalfein, F.; Prayoga, P.; Wabiser, F.; Kenangalem, E.; Piera, K.A.; Fairlie, D.P.; Tjitra, E.; Anstey, N.M.; Andrews, K.T.; et al. Ex vivo activity of histone deacetylase inhibitors against multidrug-resistant clinical isolates of Plasmodium falciparum and P. vivax. Antimicrob. Agents Chemother. 2011, 55, 961–966. [Google Scholar] [CrossRef] [Green Version]
- Tasanor, O.; Noedl, H.; Na-Bangchang, K.; Congpuong, K.; Sirichaisinthop, J.; Wernsdorfer, W.H. An in vitro system for assessing the sensitivity of Plasmodium vivax to chloroquine. Acta Trop. 2002, 83, 49–61. [Google Scholar] [CrossRef]
- Mazier, D.; Renia, L.; Snounou, G. A pre-emptive strike against malaria's stealthy hepatic forms. Nat. Rev. Drug Discov. 2009, 8, 854–864. [Google Scholar] [CrossRef]
- Hollingdale, M.R.; Collins, W.E.; Campbell, C.C. In vitro culture of exoerythrocytic parasites of the North Korean strain of Plasmodium vivax in hepatoma cells. Am. J. Trop. Med. Hyg. 1986, 35, 275–276. [Google Scholar]
- Hollingdale, M.R.; Collins, W.E.; Campbell, C.C.; Schwartz, A.L. In vitro culture of two populations (dividing and nondividing) of exoerythrocytic parasites of Plasmodium vivax. Am. J. Trop. Med. Hyg. 1985, 34, 216–222. [Google Scholar]
- Mazier, D.; Collins, W.E.; Mellouk, S.; Procell, P.M.; Berbiguier, N.; Campbell, G.H.; Miltgen, F.; Bertolotti, R.; Langlois, P.; Gentilini, M. Plasmodium ovale: In vitro development of hepatic stages. Exp. Parasitol. 1987, 64, 393–400. [Google Scholar] [CrossRef]
- Mazier, D.; Landau, I.; Druilhe, P.; Miltgen, F.; Guguen-Guillouzo, C.; Baccam, D.; Baxter, J.; Chigot, J.P.; Gentilini, M. Cultivation of the liver forms of Plasmodium vivax in human hepatocytes. Nature 1984, 307, 367–369. [Google Scholar] [CrossRef]
- Hastings, M.D.; Porter, K.M.; Maguire, J.D.; Susanti, I.; Kania, W.; Bangs, M.J.; Sibley, C.H.; Baird, J.K. Dihydrofolate reductase mutations in Plasmodium vivax from Indonesia and therapeutic response to sulfadoxine plus pyrimethamine. J. Infect. Dis. 2004, 189, 744–750. [Google Scholar] [CrossRef]
- Krudsood, S.; Tangpukdee, N.; Wilairatana, P.; Phophak, N.; Baird, J.K.; Brittenham, G.M.; Looareesuwan, S. High-dose primaquine regimens against relapse of Plasmodium vivax malaria. Am. J. Trop. Med. Hyg. 2008, 78, 736–740. [Google Scholar]
- Ku, M.J.; Dossin, F.M.; Choi, Y.; Moraes, C.B.; Ryu, J.; Song, R.; Freitas-Junior, L.H. Quantum dots: A new tool for anti-malarial drug assays. Malar. J. 2011, 10, 118. [Google Scholar] [CrossRef]
- Karl, S.; Wong, R.P.; St Pierre, T.G.; Davis, T.M. A comparative study of a flow-cytometry-based assessment of in vitro Plasmodium falciparum drug sensitivity. Malar. J. 2009, 8, 294. [Google Scholar] [CrossRef]
- Malleret, B.; Claser, C.; Ong, A.S.; Suwanarusk, R.; Sriprawat, K.; Howland, S.W.; Russell, B.; Nosten, F.; Renia, L. A rapid and robust tri-color flow cytometry assay for monitoring malaria parasite development. Sci. Rep. 2011, 1, 118. [Google Scholar]
- Meister, S.; Plouffe, D.M.; Kuhen, K.L.; Bonamy, G.M.; Wu, T.; Barnes, S.W.; Bopp, S.E.; Borboa, R.; Bright, A.T.; Che, J.; et al. Imaging of Plasmodium liver stages to drive next-generation antimalarial drug discovery. Science 2011, 334, 1372–1377. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pohlit, A.M.; Lima, R.B.S.; Frausin, G.; Silva, L.F.R.e.; Lopes, S.C.P.; Moraes, C.B.; Cravo, P.; Lacerda, M.V.G.; Siqueira, A.M.; Freitas-Junior, L.H.; et al. Amazonian Plant Natural Products: Perspectives for Discovery of New Antimalarial Drug Leads. Molecules 2013, 18, 9219-9240. https://doi.org/10.3390/molecules18089219
Pohlit AM, Lima RBS, Frausin G, Silva LFRe, Lopes SCP, Moraes CB, Cravo P, Lacerda MVG, Siqueira AM, Freitas-Junior LH, et al. Amazonian Plant Natural Products: Perspectives for Discovery of New Antimalarial Drug Leads. Molecules. 2013; 18(8):9219-9240. https://doi.org/10.3390/molecules18089219
Chicago/Turabian StylePohlit, Adrian Martin, Renata Braga Souza Lima, Gina Frausin, Luiz Francisco Rocha e Silva, Stefanie Costa Pinto Lopes, Carolina Borsoi Moraes, Pedro Cravo, Marcus Vinícius Guimarães Lacerda, André Machado Siqueira, Lucio H. Freitas-Junior, and et al. 2013. "Amazonian Plant Natural Products: Perspectives for Discovery of New Antimalarial Drug Leads" Molecules 18, no. 8: 9219-9240. https://doi.org/10.3390/molecules18089219





