Bioactive Phenylpropanoid Glycosides from Tabebuia avellanedae
Abstract
:1. Introduction
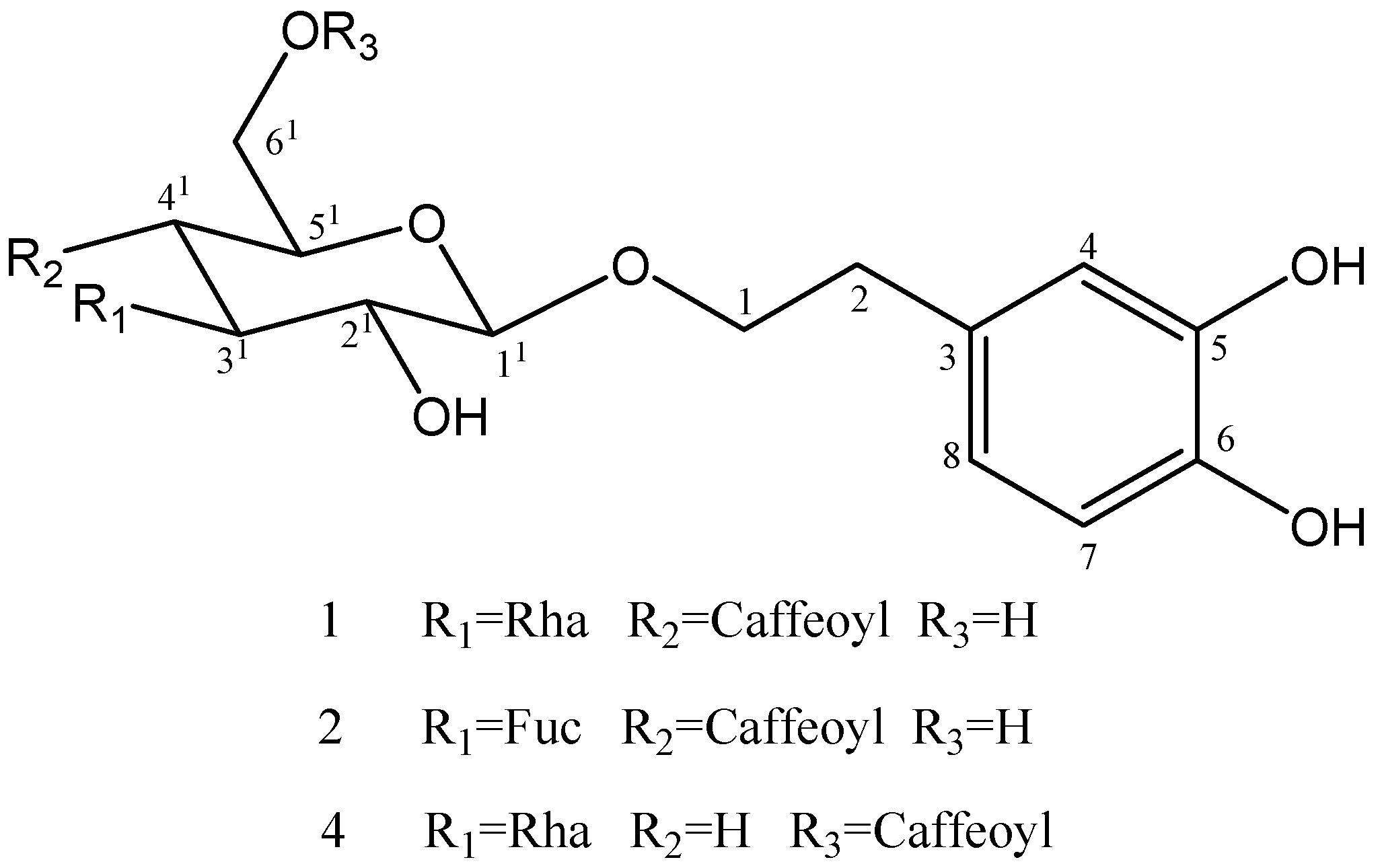
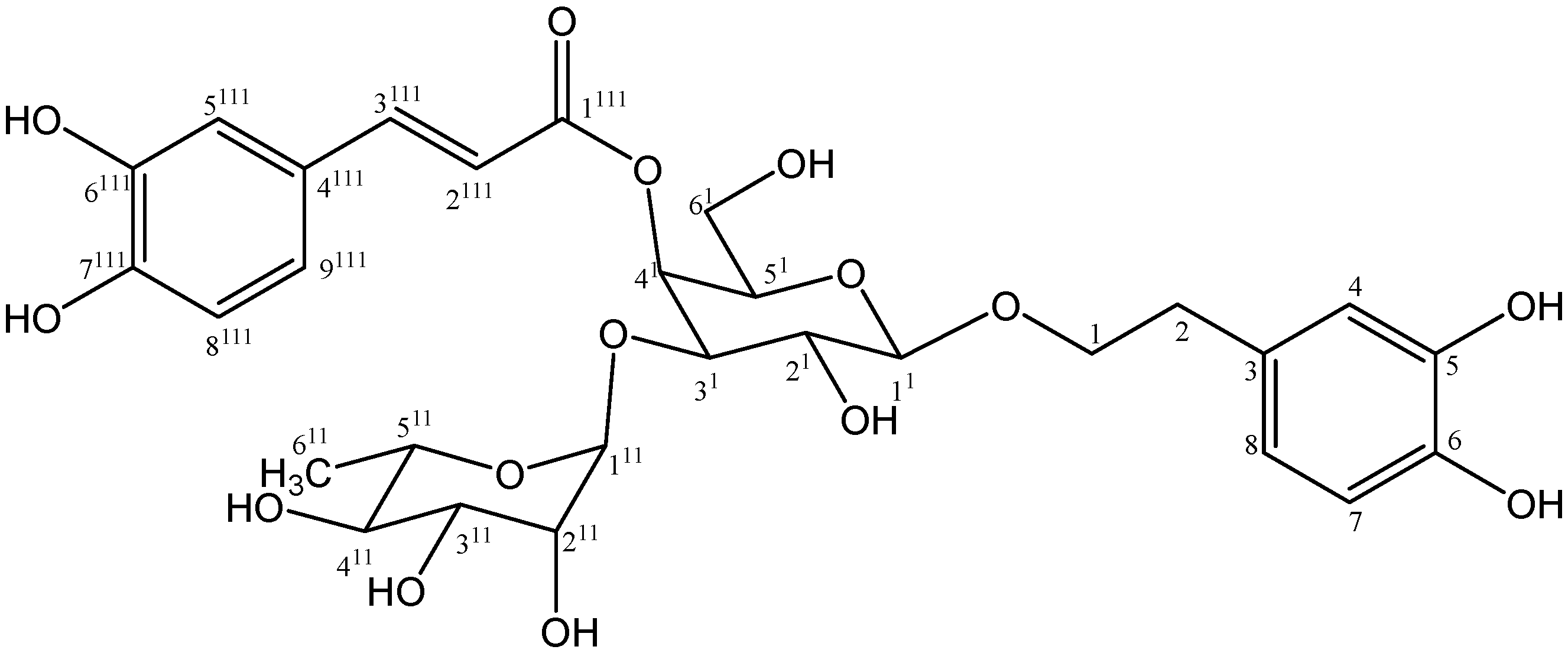
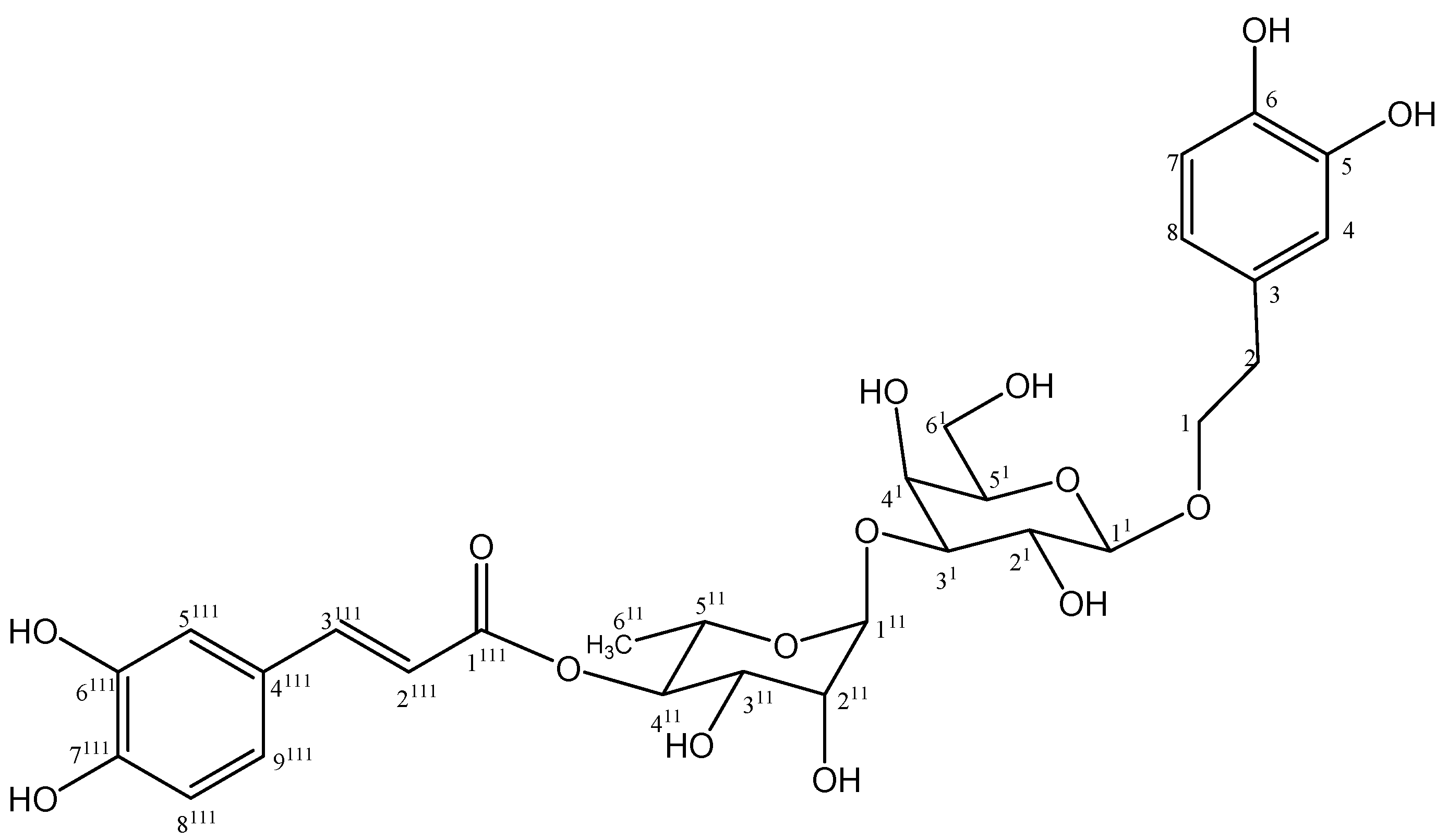
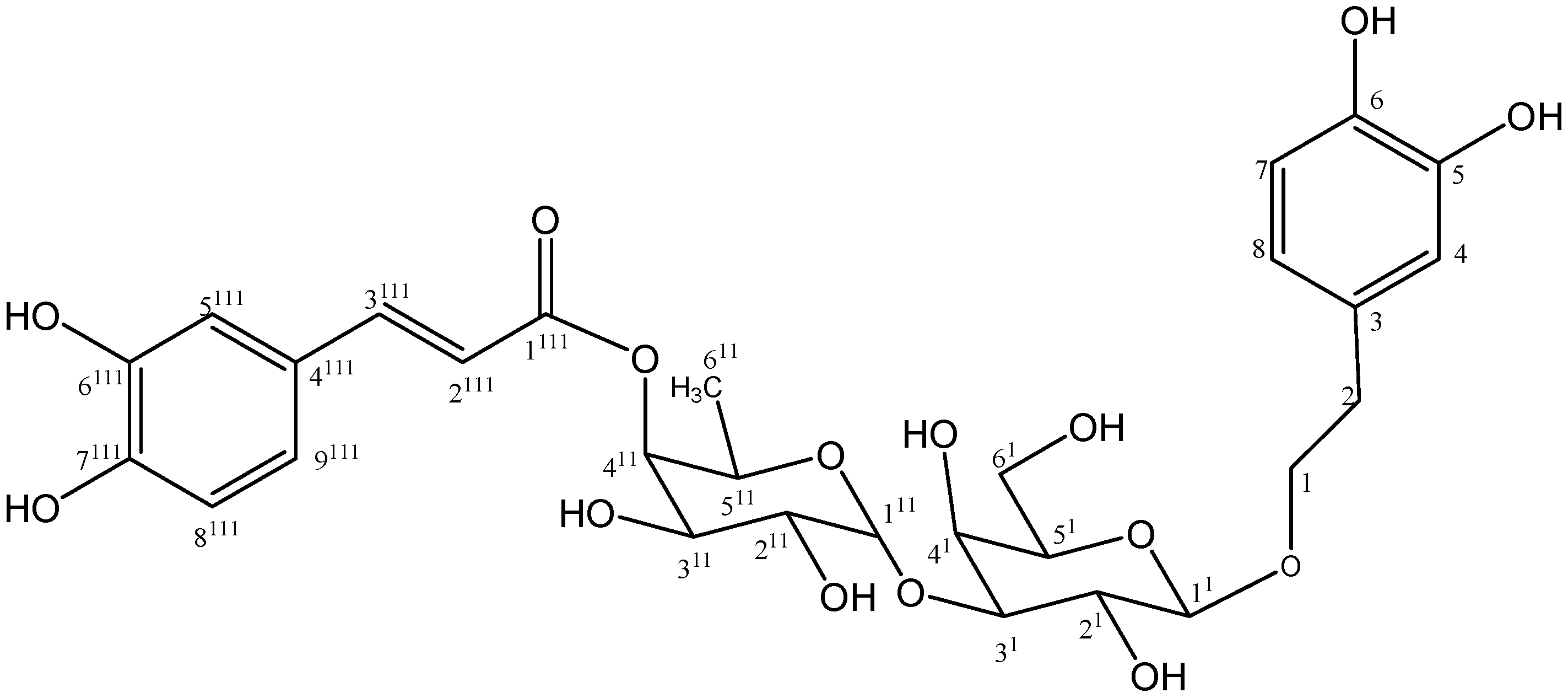
2. Results and Discussion
2.1. Isolation and Chemistry
| No. | 1 | 2 | 3 | |||
|---|---|---|---|---|---|---|
| δ 1H | δ 13C | δ 1H | δ 13C | δ 1H | δ 13C | |
| 1 | 3.91 (1H, m), 4.04 (1H, m) | 72.3 | 3.71 (1H, m), 4.04 (1H, m) | 72.3 | 3.71 (1H, m), 3.96 (1H, m) | 72.4 |
| 2 | 2.78 (2H, m) | 36.5 | 2.78 (2H, m) | 36.6 | 2.78 (2H, m) | 36.6 |
| 3 | 131.4 | 131.4 | 131.2 | |||
| 4 | 6.68 (1H, d, J = 2.4) | 117.1 | 6.68 (1H, d, J = 2.4) | 117.1 | 6.68 (1H, d, J = 2.4) | 117.0 |
| 5 | 146.1 | 146.1 | 146.1 | |||
| 6 | 144.6 | 144.6 | 144.7 | |||
| 7 | 6.66 (1H, d, J = 8.2) | 116.2 | 6.66 (1H, d, J = 7.9) | 116.3 | 6.66 (1H, d, J = 8.3) | 116.2 |
| 8 | 6.55 (1H, dd, J = 2.4, 8.2) | 121.2 | 6.56 (1H, dd, J = 2.4, 7.9) | 121.2 | 6.55 (1H, dd, J = 2.4, 8.2) | 121.2 |
| 1′ | 4.36 (1H, d, J = 7.9) | 104.2 | 4.37 (1H, d, J = 7.6) | 104.1 | 4.39 (1H, d, J = 7.5) | 104.2 |
| 2′ | 3.52 (1H, m) | 76.0 | 3.54 (1H, m) | 76.0 | 3.80 (1H, m) | 73.0 |
| 3′ | 3.81 (1H, m) | 81.6 | 3.85 (1H, m) | 79.9 | 3.82 (1H, m) | 81.3 |
| 4′ | 4.91 (1H, m) | 70.4 | 4.95 (1H, m) | 70.4 | 4.97 (1H, m) | 70.3 |
| 5′ | 3.38 (1H, m) | 76.2 | 3.42 (1H, m) | 76.4 | 3.39 (1H, m) | 76.0 |
| 6′ | 3.50 (1H, dd, J = 5.5, 9.6), 3.62 (1H, br d J = 9.6) | 62.3 | 3.52 (1H, m), 3.63 (1H, br d, J = 5.8) | 62.3 | 4.19 (1H, m), 4.21 (1H, m) | 63.3 |
| 1′′ | 5.18 (1H, d, J = 1.3) | 103.0 | 5.34 (1H, d, J = 1.3) | 102.0 | 5.18 (1H, d, J = 1.3) | 103.1 |
| 2′′ | 3.72 (1H, m) | 72.2 | 3.91 (1H, m) | 72.2 | 3.91 (1H.m) | 72.3 |
| 3′′ | 3.57 (1H, m) | 72.0 | 3.70 (1H, m) | 69.8 | 3.56 (1H, m) | 72.0 |
| 4′′ | 3.28 (1H, m) | 73.7 | 3.55 (1H, m) | 75.9 | 3.28 (1H, m) | 73.7 |
| 5′′ | 3.55 (1H, m) | 70.5 | 3.78 (1H, m) | 67.5 | 3.55 (1H, m) | 70.4 |
| 6′′ | 1.09 (3H, d, J = 6.2) | 18.4 | 1.02 (3H d, J = 6.2) | 18.1 | 1.08 (3H, d, J = 6.2) | 18.4 |
| 1′′′ | 168.3 | 168.3 | 168.0 | |||
| 2′′′ | 6.26 (1H, d, J = 16.2) | 114.6 | 6.25 (1H, d, J = 15.8) | 114.6 | 6.26 (1H, d, J = 16.2) | 114.4 |
| 3′′′ | 7.58 (1H, d, J = 16.2) | 147.9 | 7.58 (1H, d, J = 15.8) | 148.1 | 7.59 (1H, d, J = 16.2) | 148.2 |
| 4′′′ | 127.6 | 127.6 | 127.7 | |||
| 5′′′ | 7.04 ( 1H, d, J = 1.8 ) | 115.2 | 7.04 (1H, d, J = 1.8) | 115.1 | 7.04 (1H, d, J = 1.8) | 115.2 |
| 6′′′ | 146.8 | 146.8 | 146.8 | |||
| 7′′′ | 149.7 | 149.9 | 149.8 | |||
| 8′′′ | 6.77 (1H, d, J = 8.2) | 116.4 | 6.78 (1H, d, J = 8.2) | 116.5 | 6.77 (1H, d, J = 8.2) | 116.4 |
| 9′′′ | 6.95 (1H, dd, J = 1.8, 8.2) | 123.2 | 6.95 (1H, dd, J = 1.8, 8.2) | 123.2 | 6.95 (1H, dd, J = 1.8, 8.2) | 123.2 |
| No. | 4 | 5 | 6 | |||
|---|---|---|---|---|---|---|
| δ 1H | δ 13C | δ 1H | δ 13C | δ 1H | δ 13C | |
| 1 | 3.69 (1H, m), 3.97 (1H, m) | 72.3 | 3.71 (1H, m), 3.93 (1H, m) | 72.4 | 3.69 (1H, m), 3.97 (1H, m) | 72.3 |
| 2 | 2.77 (2H, m) | 36.6 | 2.77 (2H, m) | 36.6 | 2.77 (2H, m) | 36.6 |
| 3 | 131.3 | 131.4 | 131.3 | |||
| 4 | 6.66 (1H, d, J = 2.4) | 117.0 | 6.68 (1H, d, J = 2.4) | 117.0 | 6.66 (1H, d, J = 2.4) | 117.0 |
| 5 | 146.1 | 146.1 | 146.1 | |||
| 6 | 144.6 | 144.6 | 144.6 | |||
| 7 | 6.62 (1H, d, J = 8.2) | 116.3 | 6.66 (1H, d, J = 8.2) | 116.3 | 6.62 (1H, d, J = 8.2) | 116.3 |
| 8 | 6.52 (1H, dd, J = 2.4, 8.2) | 121.2 | 6.55 (1H, dd, J = 2.4, 8.2) | 121.2 | 6.52 (1H, dd, J = 2.4, 8.2) | 121.2 |
| 1′ | 4.32 (1H, d, J = 7.9) | 104.3 | 4.31 (1H, d, J = 7.8) | 104.4 | 4.34 (1H, d, J = 7.9) | 104.3 |
| 2′ | 3.54 (1H, m) | 75.4 | 3.52 (1H, m) | 75.4 | 3.54 (1H, m) | 75.5 |
| 3′ | 3.52 (1H, m) | 83.9 | 3.54 (1H, m) | 83.3 | 3.55 (1H, m) | 83.1 |
| 4′ | 3.99 (1H, m) | 70.3 | 4.10 (1H, m) | 70.2 | 3.73 (1H, m) | 72.2 |
| 5′ | 3.32 (1H, m) | 75.7 | 3.34 (1H, m) | 75.8 | 3.32 (1H, m) | 75.9 |
| 6′ | 4.35 (1H, dd, J = 5.9, 12.1), 4.49 (1H, dd, J = 2.1, 11.7) | 64.6 | 4.32 (1H, m), 4.47 (1H, dd, J = 2.1, 11.7) | 64.4 | 4.32 (1H, br d, J = 8.5), 4.47 (1H, dd, J = 2.3, 11.7) | 64.6 |
| 1′′ | 5.17 (1H, d, J = 1.8) | 102.7 | 5.23 (1H, d, J = 1.4) | 102.2 | 5.25 (1H, d, J = 1.8) | 102.3 |
| 2′′ | 3.71 (1H, m) | 72.2 | 3.90 (1H, m) | 71.1 | 3.89 (1H, m ) | 70.0 |
| 3′′ | 3.94 (1H, m) | 72.2 | 3.67 (1H, m) | 72.4 | 3.41 (1H, m) | 70.3 |
| 4′′ | 3.38 (1H, m) | 73.9 | 5.05 (1H, m) | 75.5 | 4.89 (1H, m) | 76.0 |
| 5′′ | 3.41 (1H, m) | 70.0 | 3.44 (1H, m) | 70.0 | 4.26 (1H, m) | 67.4 |
| 6′′ | 1.23 (3H, d, J = 6.2) | 17.8 | 1.25 (3H, d, J = 7.0) | 17.8 | 1.15 (3H, d, J = 5.9) | 17.6 |
| 1′′′ | 169.1 | 168.3 | 169.1 | |||
| 2′′′ | 6.28 (1H, d, J = 15.8) | 114.8 | 6.26 (1H, d, J = 15.8) | 114.6 | 6.28 (1H, d, J = 15.8) | 114.8 |
| 3′′′ | 7.56 (1H, d, J = 15.8) | 147.2 | 7.58 (1H, d, J = 15.8) | 147.9 | 7.56 (1H, d, J = 15.8) | 147.2 |
| 4′′′ | 127.6 | 127.6 | 127.6 | |||
| 5′′′ | 7.03 (1H, d, J = 2.0) | 115.0 | 7.04 (1H, d, J = 2.0) | 115.2 | 7.03 (1H, d, J = 2.0) | 115.0 |
| 6′′′ | 146.7 | 146.8 | 146.7 | |||
| 7′′′ | 149.6 | 149.7 | 149.6 | |||
| 8′′′ | 6.62 (1H, d, J = 8.2) | 116.5 | 6.77 (1H, d, J = 8.2) | 116.4 | 6.62 (1H, d, J = 8.2) | 116.5 |
| 9′′′ | 6.88 (1H, dd, J = 2.0, 8.2) | 123.1 | 6.95 (1H, dd, J = 2.0, 8.2) | 123.2 | 6.88 (1H, dd, J = 2.0, 8.2) | 123.1 |
2.2. Bioactivity
| Compound | IC50 (µM) ± SD |
|---|---|
| 1 | 2.33 ± 0.06 |
| 2 | 1 ± 0.02 |
| 3 | 0.66 ± 0.04 |
| 4 | 1.15 ± 0.04 |
| 5 | 0.12 ± 0.03 |
| 6 | 0.24 ± 0.01 |
| Ascorbic acid | 3.08 ± 0.06 |
| Compound | IC50 (µM) ± SD |
|---|---|
| 1 | 46.27 ± 1.53 |
| 2 | 43.3 ± 1.79 |
| 3 | 37.73 ± 2.11 |
| 4 | 91.7 ± 5.44 |
| 5 | 22.49 ± 1.65 |
| 6 | 15.1 ± 2.43 |
| Ketoconazole | 6.44 ± 0.56 |
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
 −128 (MeOH c 0.45); UV
−128 (MeOH c 0.45); UV  nm (log ε): 238 (4.40), 352 (4.24) nm; IR (KBr) cm−1, 3130, 3028, 2401, 2318,1753, 1720, 1697, 1548, 1506, 766; 1H-NMR and 13C-NMR spectra data see Table 1; HR-FABMS m/z 625.2126 [M+H]+ (calculated for C29H37O15 625.2203).
nm (log ε): 238 (4.40), 352 (4.24) nm; IR (KBr) cm−1, 3130, 3028, 2401, 2318,1753, 1720, 1697, 1548, 1506, 766; 1H-NMR and 13C-NMR spectra data see Table 1; HR-FABMS m/z 625.2126 [M+H]+ (calculated for C29H37O15 625.2203). −105 ( MeOH c 0.1 ); UV
−105 ( MeOH c 0.1 ); UV  nm (log ε): 236 (4.39), 282 (4.33), 346 (4.37) nm; IR (KBr) cm−1, 3125, 1701, 1525, 1510, 1045; 1H-NMR and 13C-NMR spectral data see Table 2; HR-FABMS m/z 625.2135 [M+H]+ (calculated for C29H37O15 625.2169).
nm (log ε): 236 (4.39), 282 (4.33), 346 (4.37) nm; IR (KBr) cm−1, 3125, 1701, 1525, 1510, 1045; 1H-NMR and 13C-NMR spectral data see Table 2; HR-FABMS m/z 625.2135 [M+H]+ (calculated for C29H37O15 625.2169).  65 (MeOH c 0.1); UV
65 (MeOH c 0.1); UV  nm (log ε): 236 (4.31), 346 (4.29) nm; IR (KBr) cm−1, 3120, 1701, 1552, 1525, 1510; 1H-NMR and 13C-NMR spectral data see Table 2; HR-FABMS m/z 625.2144 [M+H]+ (calculated for C29H37O15 625.2183).
nm (log ε): 236 (4.31), 346 (4.29) nm; IR (KBr) cm−1, 3120, 1701, 1552, 1525, 1510; 1H-NMR and 13C-NMR spectral data see Table 2; HR-FABMS m/z 625.2144 [M+H]+ (calculated for C29H37O15 625.2183).3.4. Acid Hydrolysis of Compounds 2, 5, 6
3.5. Antioxidant Assay
3.6. Inhibitory Activity on Cytochrome CYP3A4 Enzyme
4. Conclusions
Conflicts of Interest
References
- Casinovi, C.G.; Marini, G.B.; Limaog, D.A.; Daliamaia, M.E. On quinones isolated from the wood of Tabebuia Avellanedae Lor. Ex Griseb. Rend. Ist. Super. Sanità 1963, 26, 5–10. [Google Scholar]
- De Santana, C.F.; de Lima, O.; d’Albuquerque, I.L.; Lacerda, A.L.; Martins, D.G. Antitumoral and toxicological properties of extracts of bark and various wood components of Pau d’arco (Tabebuia avellanedae). Rev. Inst. Antibiot. 1968, 8, 89–94. [Google Scholar]
- Hashimoto, G. Illustrated cyclopedia of brazilian medical plants. Aboc-sha Kamakura 1996, 158. [Google Scholar]
- Woo, H.J.; Choi, Y.H. Growth inhibition of A549 human lung carcinoma cells by beta-lapachone through induction of apoptosis and inhibition of telomerase activity. Int. J. Oncol. 2005, 26, 1017–1023. [Google Scholar]
- Kreher, B.; Lotter, H.; Cordell, G.A.; Wagner, H. New furanonaphthoquinones and other constituents of Tabebuia avellanedae and their immunomodulating activities in vitro. Planta Med. 1988, 54, 562–563. [Google Scholar]
- Awale, S.; Kawakami, T.; Tezuka, Y.; Ueda, J.Y.; Tanaka, K.; Kadota, S. Nitric oxlde(NO) production inhibitory constituents of Tabebuia avetlanedae from Brazil. Chem. Pharm. Bull. 2005, 53, 710–713. [Google Scholar] [CrossRef]
- Ueda, S.; Umemura, T.; Dohguchi, K.; Matsuzaki, T.; Tokuda, H.; Nishino, H.; Iwashima, A. Production of anti-tumour promoting furanonaphthoquinones in Tabebuia avellanedae cell cultures. Phytochemistry 1994, 36, 323–325. [Google Scholar]
- Pereira, E.M.; Machado, T.B.; Leal, I.C.; Jesus, D.M.; Damaso, C.R.; Pinto, A.V.; Giambiagi-de, M.; Kuster, R.M.; Santos, K.R. Tabebuia avellanedae naphthoquinones: Activity against methicillin-resistant staphylococcal strains, cytotoxic activity and in vivo dermal irritability analysis. Anna. Clini. Microbiol. Antimicrol. 2006, 5, 5–11. [Google Scholar] [CrossRef]
- Kim, S.O.; Kwon, J.I.; Jeong, Y.K.; Kim, G.Y.; Kim, N.D.; Choi, Y.H. Induction of Egr-1 is associated with anti-metastatic and anti-invasive ability of beta-lapachone in human hepatocarcinoma cells. Biosci. Biotechnol. Biochem. 2007, 7, 2169–2176. [Google Scholar]
- López-Munguía, A.; Hernández-Romero, Y.; Pedraza-Chaverri, J.; Miranda-Molina, A.; Regla, I.; Martínez, A.; Castillo, E. Phenylpropanoid glycoside analogues: Enzymatic synthesis, antioxidant activity and theoretical study of their free radical scavenger mechanism. PLoS One 2011, 6, e20115. [Google Scholar] [CrossRef]
- Booker, F.L.; Burkey, K.O.; Jones, A.M. Re-evaluating the role of ascorbic acid and phenolic glycosides in ozone scavenging in the leaf apoplast of Arabidopsis thaliana L. Plant Cell Environ. 2012, 35, 1456–1466. [Google Scholar] [CrossRef]
- Abdel-Mageed, W.M.; Backheet, E.Y.; Khalifa, A.A.; Ibraheim, Z.Z.; Ross, S.A. Antiparasitic antioxidant phenylpropanoids and iridoid glycosides from Tecoma mollis. Fitoteria 2012, 83, 500–507. [Google Scholar] [CrossRef]
- Liu, Z.; Jia, Z.J. Phenylpropanoid and iridoid glycosides from pedicularis striata. Phytochemistry 1991, 30, 1341–1345. [Google Scholar] [CrossRef]
- Andary, C.; Wylde, R.; Laffite, C.; Privat, G.; Wintemitz, F. Structures of verbascoside and orobanchoside, caffeic acid sugar esters from Orobanche rapum-genistae. Phytochemistry 1982, 21, 1123–1126. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Suo, M.; Ohta, T.; Takano, F.; Jin, S. Bioactive Phenylpropanoid Glycosides from Tabebuia avellanedae. Molecules 2013, 18, 7336-7345. https://doi.org/10.3390/molecules18077336
Suo M, Ohta T, Takano F, Jin S. Bioactive Phenylpropanoid Glycosides from Tabebuia avellanedae. Molecules. 2013; 18(7):7336-7345. https://doi.org/10.3390/molecules18077336
Chicago/Turabian StyleSuo, Maorong, Tomihisa Ohta, Fumihide Takano, and Shouwen Jin. 2013. "Bioactive Phenylpropanoid Glycosides from Tabebuia avellanedae" Molecules 18, no. 7: 7336-7345. https://doi.org/10.3390/molecules18077336
APA StyleSuo, M., Ohta, T., Takano, F., & Jin, S. (2013). Bioactive Phenylpropanoid Glycosides from Tabebuia avellanedae. Molecules, 18(7), 7336-7345. https://doi.org/10.3390/molecules18077336




