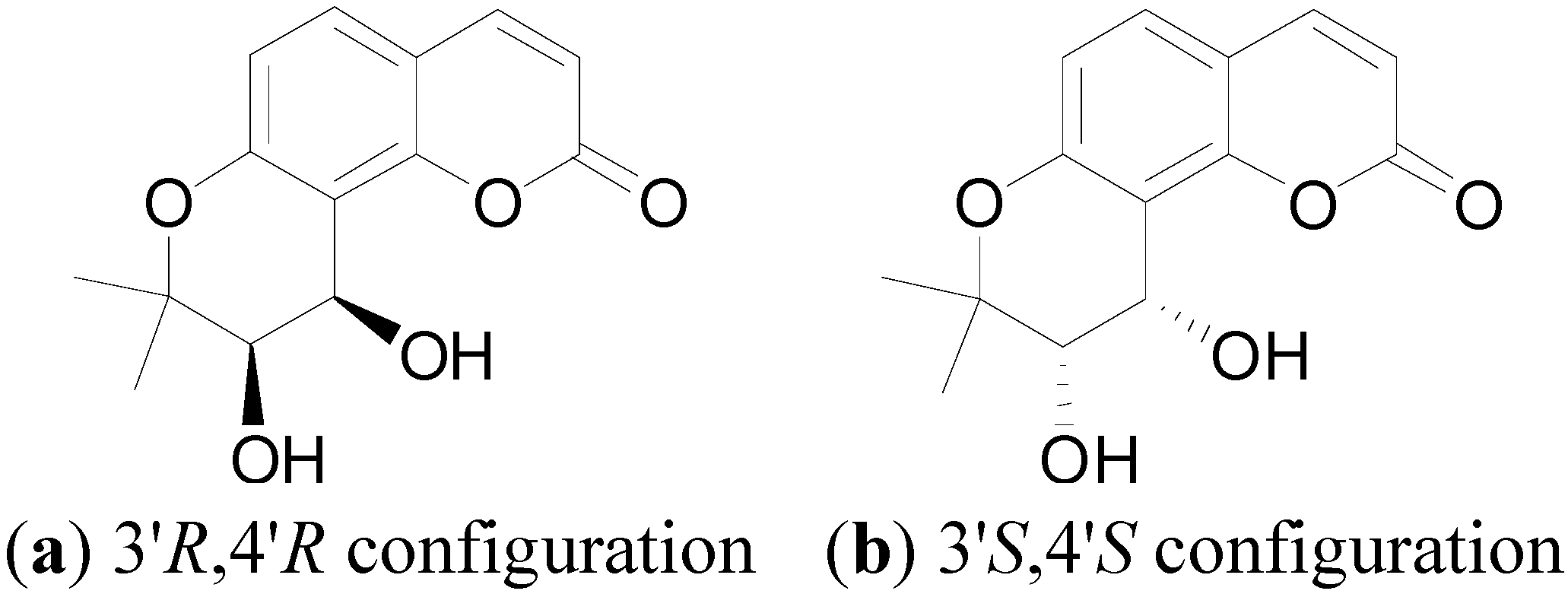
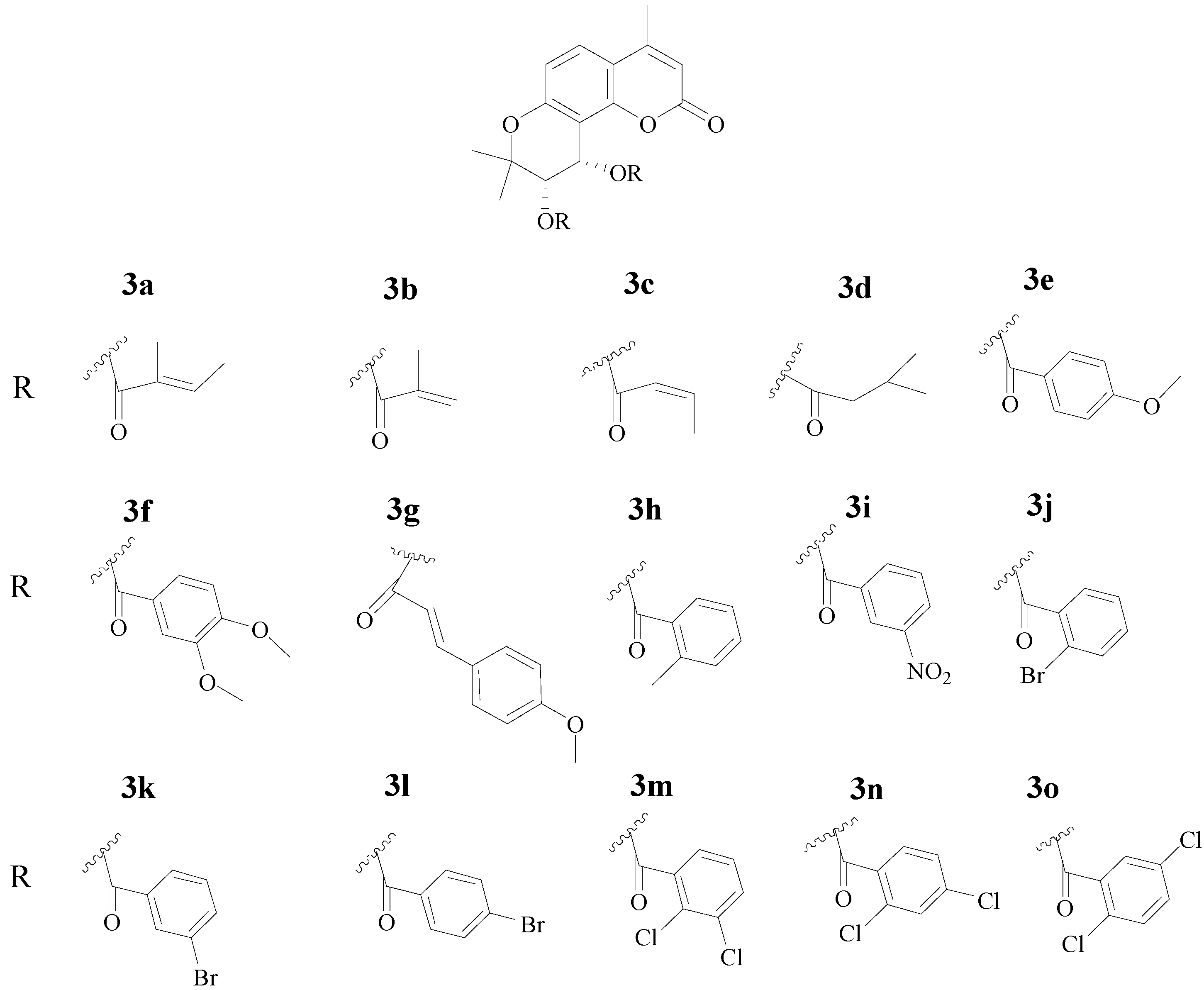
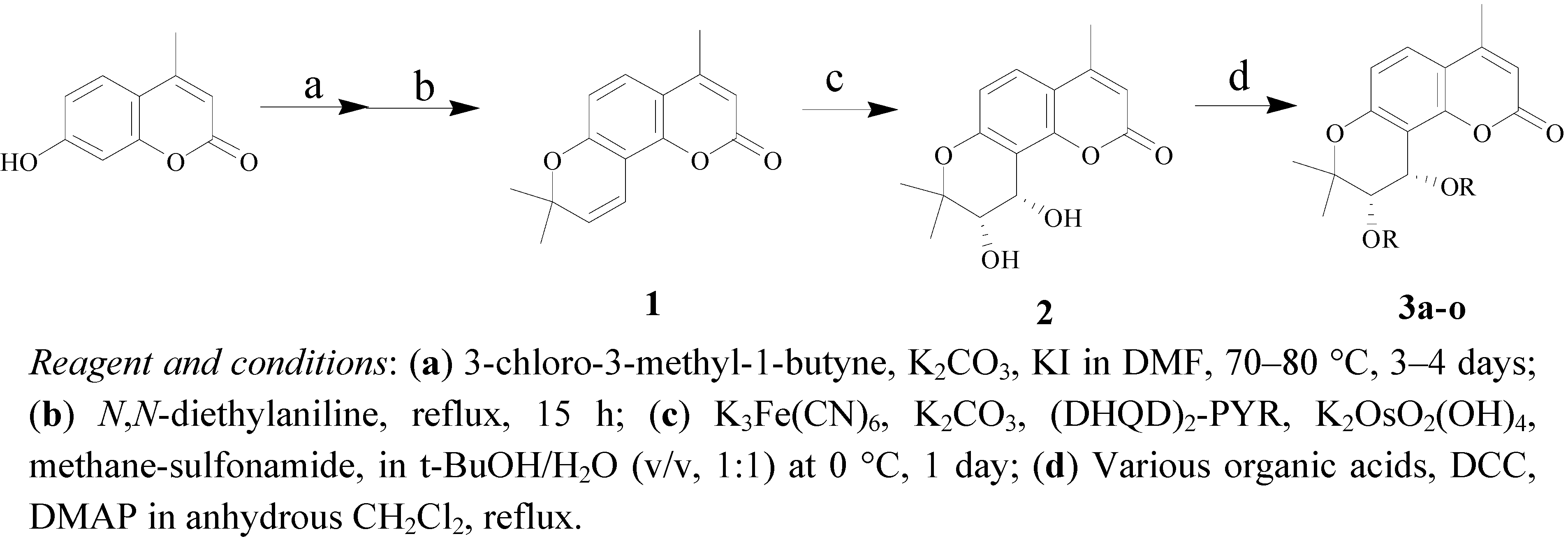
3.4. Procedure for the Synthesis of 4-Methyl-(3'S,4'S)-cis-Khellactone Derivatives 3a–o
To a solution of crude compound 2 (138 mg, 0.5 mmol) in anhydrous CH2Cl2 (10 mL) tiglic acid (6 mmol), angelic acid (6 mmol), crotonic acid (6 mmol), isovaleric acid (4 mmol), 4-methoxybenzoic acid (3 mmol), 3,4-dimethoxybenzoic acid (3 mmol), 4-methoxycinnamic acid (2 mmol), o-toluic acid (3 mmol), 3-nitrobenzoic acid (3 mmol), 2-bromobenzoic acid (3 mmol), 3-bromobenzoic acid (3 mmol), 4-bromobenzoic acid (3 mmol), 2,3-dichlorobenzoic acid (2 mmol), 2,4-dichlorobenzoic acid (2 mmol) or 2,5-dichlorobenzoic acid (2 mmol) were added, respectively, followed by N,N’-dicyclo-hexylcarbodiimide (412 mg, 2 mmol) and 4-dimethylaminopyridine (8 mg, 0.064 mmol). The mixture was heated to reflux until the reaction was complete as monitored by TLC. After cooling to room temperature, filtered, and the filtrate was separated and purified by column chromatography (petroleum ether/acetone, 10:1) to give pure target compounds 3a–o.
(3'S,4'S)-Di-O-tigloyl-4-methyl-(+)-cis-khellactone (3a). Molecular formula (MW): C25H28O7 (440.49 g/mol); white solid; 23% Yield; mp: 84–86 °C; [α: +3.8 (c 0.1, CH2Cl2). 1H-NMR (DMSO-d6) δ 1.39 (3H, s, C-2'-CH3), 1.43 (3H, s, C-2'-CH3), 1.71 (3H, br s, H-5''), 1.72 (3H, br s, H-5'''), 1.74 (3H, br d, H-4''), 1.76 (3H, br d, H-4'''), 2.39 (3H, s, C-4-CH3), 5.32 (1H, d, J = 4.92 Hz, H-3'), 6.21 (1H, s, H-3), 6.50 (1H, d, J = 4.92 Hz, H-4'), 6.66 (1H, m, H-3''), 6.72 (1H, m, H-3'''), 6.94 (1H, d, J = 8.86 Hz, 7.72 (1H, d, J = 8.86 Hz, H-5), H-6); 13C-NMR (CDCl3) δ 11.87, 12.05, 14.25, 14.33, 18.63, 22.27, 25.40, 60.64, 70.50, 77.36, 107.48, 112.06, 113.51, 113.92, 125.91, 127.96, 128.31, 137.08, 138.16, 152.04, 153.46, 156.47, 159.72, 166.50, 166.56; MS (ESI) m/z 462.7,463.9 ([M+Na]+).
(3'S,4'S)-Di-O-angeloyl-4-methyl-(+)-cis-khellactone (3b). Molecular formula (MW): C25H28O7 (440.49 g/mol); white solid; 31% Yield; mp: 151–153 °C; [α: +4.2 (c 0.1, CH2Cl2). 1H-NMR (CDCl3) δ 1.42 (3H, s, C-2'-CH3), 1.48 (3H, s, C-2'-CH3), 1.73 (3H, br d, H-4''), 1.75 (3H, br d, H-4'''), 1.78 (3H, br s, H-5''), 1.80 (3H, br s, H-5'''), 2.36 (3H, s, C-4-CH3), 5.39 (1H, d, J = 4.92 Hz, H-3'), 6.08 (1H, s, H-3), 6.65 (1H, d, J = 4.92 Hz, H-4'), 6.74 (1H, m, H-3''), 6.78 (1H, m, H-3'''), 6.83 (1H, d, J = 8.84 Hz, H-6), 7.49 (1H, d, J = 8.84 Hz, H-5); 13C-NMR (CDCl3) δ 11.91, 12.08, 14.28, 14.35, 18.65, 22.40, 25.41, 60.76, 70.54, 77.47, 107.60, 112.16, 113.58, 113.93, 125.87, 128.04, 128.41, 137.09, 138.16, 151.96, 153.58, 156.57, 159.74, 166.55, 166.62; MS (ESI) m/z 462.9 ([M+Na]+).
(3'S,4'S)-Di-O-crotonoyl-4-methyl-(+)-cis-khellactone (3c). Molecular formula (MW): C23H24O7 (412.43 g/mol); white solid; 45% Yield; mp: 63–65 °C; [α: +5.1 (c 0.1, CH2Cl2). 1H-NMR (CDCl3) δ 1.40 (3H, s, C-2'-CH3), 1.45 (3H, s, C-2'-CH3), 1.83 (3H, m, H-4'''), 1.85 (3H, m, H-4''), 2.35 (3H, s, C-4-CH3), 5.36 (1H, d, J = 4.88 Hz, H-3'), 5.78–5.84 (2H, m, H-2'', H-2'''), 6.07 (1H, s, H-3), 6.60 (1H, d, J = 4.88 Hz, H-4'), 6.82 (1H, d, J = 8.84 Hz, H-6), 6.90–6.98 (2H, m, H-3'', H-3'''), 7.49 (1H, d, J = 8.84 Hz, H-5); 13C-NMR (CDCl3) δ 17.98, 18.00, 18.64, 22.27, 25.23, 60.52, 70.20, 77.41, 107.23, 112.05, 113.50, 113.94, 121.79, 122.03, 125.95, 145.18, 145.96, 152.09, 153.41, 156.46, 159.73, 164.91, 165.06; MS (ESI) m/z 434.9,435.8 ([M+Na]+).
(3'S,4'S)-Di-O-isovaleryloxy-4-methyl-(+)-cis-khellactone (3d). Molecular formula (MW): C25H32O7 (444.52 g/mol); light yellow oil; 52% Yield; [α: +4.5 (c 0.1, CH2Cl2). 1H-NMR (CDCl3) δ 0.95–0.98 (12H, m, CH3×4), 1.41 (3H, s, C-2'-CH3), 1.43 (3H, s, C-2'-CH3), 2.10–2.30 (6H, m, COCH2 × 2, CH × 2), 2.37 (3H, s, C-4-CH3), 5.31 (1H, d, J = 4.79 Hz, H-3'), 6.10 (1H, s, H-3), 6.54 (1H, d, J = 4.79 Hz, H-4'), 6.81 (1H, d, J = 8.85 Hz, H-6), 7.49 (1H, d, J = 8.85 Hz, H-5); 13C-NMR (CDCl3) δ 18.67, 22.36, 22.41, 25.31, 25.43, 25.47, 43.05, 43.23, 60.49, 70.37, 107.24, 112.03, 113.50, 113.94, 125.98, 152.13, 153.32, 156.36, 159.72, 171.78; MS (ESI) m/z 467.3 ([M+Na]+).
(3'S,4'S)-Di-O-p-methoxybenzoyl-4-methyl-(-)-cis-khellactone (3e). Molecular formula (MW): C31H28O9 (544.55 g/mol); white solid; 19% Yield; mp: 177–179 °C; [α: −35.9 (c 0.1, CH2Cl2). 1H-NMR (CDCl3) δ 1.49 (3H, s, C-2'-CH3), 1.62 (3H, s, C-2'-CH3), 2.37 (3H, s, C-4-CH3), 3.82 (3H, s, OCH3), 3.83 (3H, s, OCH3), 5.64 (1H, d, J = 4.92 Hz, H-3'), 6.06 (1H, s, H-3), 6.79–6.82(4H, m, Ar-H), 6.90 (1H, d, J = 8.88 Hz, H-6), 6.92 (1H, d, J = 4.92 Hz, H-4'), 7.54 (1H, d, J = 8.88 Hz, H-5), 7.81–7.86 (4H, m, Ar-H); 13C-NMR (CDCl3) δ 18.67, 22.44, 25.62, 55.35, 55.40, 61.01, 70.93, 77.53, 107.49, 112.25, 113.50, 113.62, 113.99, 121.83, 122.40, 126.09, 131.84, 131.89, 151.88, 153.63, 156.60, 159.58, 163.29, 163.60, 164.88, 164.96; MS (ESI) m/z 566.8 ([M+Na]+).
(3'S,4'S)-bis-O-(3,4-Dimethoxybenzoyl)-4-methyl-(-)-cis-khellactone (3f). Molecular formula (MW): C33H32O11 (604.60 g/mol); white solid; 28% Yield; mp : 207–209 °C; [α: −10.0 (c 0.1, CH2Cl2). 1H-NMR (CDCl3) δ 1.51 (3H, s, C-2'-CH3), 1.65 (3H, s, C-2'-CH3), 2.38 (3H, s, C-4-CH3), 3.66 (3H, s, OCH3), 3.76 (3H, s, OCH3), 3.89 (6H, s, OCH3), 5.65 (1H, d, J = 4.92 Hz, H-3'), 6.08 (1H, s, H-3), 6.76 (1H, d, J = 8.49 Hz, Ar-H), 6.79(1H, d, J = 8.49 Hz, Ar-H), 6.91 (1H, d, J = 8.90 Hz, H-6), 6.95 (1H, d, J = 4.92 Hz, H-4'), 7.38 (1H, d, J = 1.92 Hz, Ar-H), 7.43 (1H, d, J = 1.92 Hz, Ar-H), 7.50 (1H, m, Ar-H), 7.55 (1H, d, J = 8.90 Hz, H-5), 7.56 (1H, m, Ar-H); 13C-NMR (CDCl3) δ 18.66, 22.25, 25.73, 55.59, 55.82, 55.96, 60.98, 71.02, 77.40, 107.33, 110.22, 112.19, 112.23, 112.45, 113.60, 114.00, 121.79, 122.41, 123.71, 123.92, 126.19, 129.70, 129.96, 148.62, 151.97, 153.00, 153.31, 153.58, 156.55, 159.63, 164.85, 164.89; MS (ESI) m/z 626.8 ([M+Na]+).
(3'S,4'S)-bis-O-p-Methoxycinnamoyl-4-methyl-(-)-cis-khellactone (3g). Molecular formula (MW): C35H32O9 (596.62 g/mol); white solid; 34% Yield; mp: 120–122 °C; [α: −59.5 (c 0.1, CH2Cl2). 1H-NMR (CDCl3) δ 1.47 (3H, s, C-2'-CH3), 1.56 (3H, s, C-2'-CH3), 2.37 (3H, s, C-4-CH3), 3.81 (3H, s, OCH3), 3.82 (3H, s, OCH3), 5.51 (1H, d, J = 5.00 Hz, H-3'), 6.10 (1H, s, H-3), 6.31 (2H, d, J = 15.93 Hz, 2 × ArCH=), 6.77 (1H, d, J = 5.00 Hz, H-4'), 6.82-6.85 (4H, m, Ar-H), 6.88 (1H, d, J = 8.92 Hz, H-6), 7.37–7.41 (4H, m, Ar-H), 7.52 (1H, d, J = 8.92 Hz, H-5), 7.62 (2H, d, J = 15.93 Hz, 2 × -COCH=); 13C-NMR (CDCl3) δ 18.70, 22.67, 25.22, 55.31, 55.34, 60.82, 70.35, 77.63, 107.39, 112.20, 113.29, 113.43, 113.65, 113.98, 114.16, 114.25, 114.53, 114.99, 125.96, 126.98, 127.26, 129.82, 129.96, 132.33, 132.41, 145.07, 145.70, 151.97, 153.56, 156.59, 159.75, 161.29, 161.53, 165.72, 166.02; MS (ESI) m/z 618.8 ([M+Na]+).
(3'S,4'S)-Di-O-o-methylbenzoyl-4-methyl-(-)-cis-khellactone (3h). Molecular formula (MW): C31H28O7 (512.55 g/mol); white solid; 47% Yield; mp: 196–198 °C; [α: −36.7 (c 0.1, CH2Cl2). 1H-NMR (CDCl3) δ 1.54 (3H, s, C-2'-CH3), 1.61 (3H, s, C-2'-CH3), 2.38 (3H, s, C-4-CH3), 2.43 (3H, s, CH3), 2.52 (3H, s, CH3), 5.66 (1H, d, J = 4.95 Hz, H-3'), 6.09 (1H, s, H-3), 6.89 (1H, d, J = 8.88 Hz, H-6), 6.95 (1H, d, J = 4.95 Hz, H-4'), 7.06 (1H, m, Ar-H), 7.13 (2H, m, Ar-H), 7.20 (1H, m, Ar-H), 7.30 (1H, m, Ar-H), 7.36 (1H, m, Ar-H), 7.53 (1H, d, J = 8.88 Hz, H-5), 7.65 (1H, m, Ar-H), 7.80 (1H, m, Ar-H); 13C-NMR (CDCl3) δ 18.69, 20.92, 21.56, 22.48, 25.61, 60.99, 71.01, 77.43, 107.41, 112.18, 113.64, 114.04, 125.31, 125.58, 126.11, 128.74, 129.61, 129.87, 130.50, 131.36, 131.57, 131.71, 132.24, 140.53, 140.94, 152.04, 153.55, 156.54, 159.66, 165.96, 166.25; MS (ESI) m/z 534.5 ([M+Na]+).
(3'S,4'S)-Di-O-m-nitrobenzoyl-4-methyl-(-)-cis-khellactone (3i). Molecular formula (MW): C29H22N2O11 (574.49 g/mol); white solid; 34% Yield; mp: 187–189 °C; [α: −10.0 (c 0.1, CH2Cl2). 1H-NMR (CDCl3) δ 1.54 (3H, s, C-2'-CH3), 1.65 (3H, s, C-2'-CH3), 2.40 (3H, s, C-4-CH3), 5.74 (1H, d, J = 4.95 Hz, H-3'), 6.07 (1H, s, H-3), 6.96 (1H, d, J = 8.93 Hz, H-6), 6.98 (1H, d, J = 4.95 Hz, H-4'), 7.58–7.63 (2H, m, Ar-H), 7.61 (1H, d, J = 8.93 Hz, H-5), 8.27 (2H, m, Ar-H), 8.39 (2H, m, Ar-H), 8.56 (1H, m, Ar-H), 8.60 (1H, m, Ar-H); 13C-NMR (CDCl3) δ 18.78, 22.78, 25.04, 62.48, 71.82, 77.30, 105.94, 112.22, 113.93, 114.21, 124.30, 124.46, 126.76, 127.69, 128.00, 129.78, 129.91, 130.71, 131.07, 135.41, 135.71, 148.07, 148.21, 152.29, 153.42, 156.40, 159.47, 163.46, 163.57; MS (ESI) m/z 596.7 ([M+Na]+).
(3'S,4'S)-Di-O-o-bromobenzoyl-4-methyl-(-)-cis-khellactone (3j). Molecular formula (MW): C29H22Br2O7 (642.29 g/mol); white solid; 42% Yield; mp: 178–180 °C; [α: −62.1 (c 0.1, CH2Cl2). 1H-NMR (600 MHz, CDCl3) δ 1.59 (3H, s, C-2'-CH3), 1.60 (3H, s, C-2'-CH3), 2.39 (3H, s, C-4-CH3), 5.73 (1H, d, J = 4.51 Hz, H-3'), 6.12 (1H, s, H-3), 6.87 (1H, d, J = 8.57 Hz, H-6), 6.94 (1H, d, J = 4.51 Hz, H-4'), 7.22 (1H, d, J = 7.31 Hz, Ar-H), 7.25 (1H, d, J = 7.31 Hz, Ar-H), 7.31 (2H, m, Ar-H), 7.53 (2H, m, Ar-H), 7.61 (1H, d, J = 7.57 Hz, Ar-H), 7.71 (1H, d, J = 7.57 Hz, Ar-H), 7.84 (1H, d, J = 8.57 Hz, H-5); 13C-NMR (150 MHz, CDCl3) δ 18.73, 22.25, 25.97, 62.13, 72.11, 77.45, 106.84, 112.19, 113.61, 114.12, 120.96, 121.41, 126.42, 127.06, 127.20, 131.03, 131.77, 132.05, 132.10, 132.68, 132.76, 133.74, 134.08, 152.16, 153.48, 156.43, 159.61, 165.20, 165.67; MS (ESI) m/z 662.0, 664.0, 666.5 ([M+Na]+).
(3'S,4'S)-Di-O-m-bromobenzoyl-4-methyl-(-)-cis-khellactone (3k). Molecular formula (MW): C29H22Br2O7 (642.29 g/mol); white solid; 36% Yield; mp: 201–203 °C; [α: −17.8 (c 0.1, CH2Cl2). 1H-NMR (CDCl3) δ 1.51 (3H, s, C-2'-CH3), 1.63 (3H, s, C-2'-CH3), 2.39 (3H, s, C-4-CH3), 5.66 (1H, d, J = 5.00 Hz, H-3'), 6.09 (1H, s, H-3), 6.92 (1H, d, J = 8.52 Hz, H-6), 6.93 (1H, d, J = 5.00 Hz, H-4'), 7.24–7.27 (2H, m, Ar-H), 7.58 (1H, d, J = 8.52 Hz, H-5), 7.63–7.68 (2H, m, Ar-H), 7.81–7.86 (2H, m, Ar-H), 7.95 (2H, m, Ar-H); 13C-NMR (CDCl3) δ 18.70, 22.49, 25.36, 61.70, 71.46, 77.31, 106.59, 112.31, 113.80, 114.08, 122.38, 122.53, 126.47, 128.35, 128.46, 129.96, 130.02, 131.08, 131.54, 132.55, 132.64, 135.97, 136.37, 152.01, 153.52, 156.45, 159.46, 164.04, 164.07; MS (ESI) m/z 662.1, 664.3, 666.5 ([M+Na]+).
(3'S,4'S)-Di-O-p-bromobenzoyl-4-methyl-(-)-cis-khellactone (3l). Molecular formula (MW): C29H22Br2O7 (642.29 g/mol); white solid; 40% Yield; mp: 192–194 °C; [α: −20.0 (c 0.1, CH2Cl2). 1H-NMR (CDCl3) δ 1.50 (3H, s, C-2'-CH3), 1.60 (3H, s, C-2'-CH3), 2.38 (3H, s, C-4-CH3), 5.64 (1H, d, J = 4.82 Hz, H-3'), 6.08 (1H, s, H-3), 6.91 (1H, d, J = 8.98 Hz, H-6), 6.92 (1H, d, J = 4.82 Hz, H-4'), 7.49 (4H, m, Ar-H), 7.56 (1H, d, J = 8.98 Hz, H-5), 7.72 (4H, m, Ar-H); 13C-NMR (CDCl3) δ 18.70, 22.49, 25.36, 61.68, 71.35, 77.35, 106.71, 112.24, 113.76, 114.13, 126.41, 128.10, 128.21, 128.52, 128.62, 129.62, 131.17, 131.57, 131.68, 131.81, 132.15, 152.17, 153.48, 156.49, 159.67, 164.60, 164.69; MS (ESI) m/z 662.6, 664.5, 666.5 ([M+Na]+).
(3'S,4'S)-bis-O-(2,3-dichlorobenzoyl)-4-methyl-(-)-cis-khellactone (3m). Molecular formula (MW): C29H20Cl4O7 (622.28 g/mol); white solid; 29% Yield; mp: 197–199 °C; [α: −34.6 (c 0.1, CH2Cl2). 1H-NMR (CDCl3) δ 1.55 (3H, s, C-2'-CH3), 1.61 (3H, s, C-2'-CH3), 2.40 (3H, s, C-4-CH3), 5.71 (1H, d, J = 4.88 Hz, H-3'), 6.14 (1H, s, H-3), 6.88 (1H, d, J = 8.85 Hz, H-6), 6.92 (1H, d, J = 4.88 Hz, H-4'), 7.18 (1H, m, Ar-H), 7.26 (1H, m, Ar-H), 7.49 (1H, m, Ar-H), 7.55 (1H, d, J = 8.85 Hz, H-5), 7.58 (1H, m, Ar-H), 7.61 (1H, m, Ar-H), 7.72 (1H, m, Ar-H); 13C-NMR (CDCl3) δ 18.73, 21.90, 25.99, 62.44, 72.49, 77.28, 106.58, 112.19, 113.67, 114.23, 126.60, 127.32, 127.36, 128.63, 129.67, 130.62, 131.19, 132.41, 132.60, 133.29, 133.45, 133.95, 134.33, 152.33, 153.42, 156.38, 159.66, 164.55, 165.12; MS (ESI) m/z 642.1, 644.2 ([M+Na]+).
(3'S,4'S)-bis-O-(2,4-dichlorobenzoyl)-4-methyl-(-)-cis-khellactone (3n). Molecular formula (MW): C29H20Cl4O7 (622.28 g/mol); white solid; 41% Yield; mp: 140–142 °C; [α: −73.1 (c 0.1, CH2Cl2). 1H-NMR (CDCl3) δ 1.56 (3H, s, C-2'-CH3), 1.57 (3H, s, C-2'-CH3), 2.39 (3H, s, C-4-CH3), 5.69 (1H, d, J = 5.18 Hz, H-3'), 6.12 (1H, s, H-3), 6.88 (1H, d, J = 5.18 Hz, H-4'), 6.89 (1H, d, J = 8.95 Hz, H-6), 7.21 (1H, m, Ar-H), 7.27 (1H, m, Ar-H), 7.37 (1H, J = 1.92 Hz, Ar-H), 7.44 (1H, J = 1.92 Hz, Ar-H), 7.55 (1H, d, J = 8.95 Hz, H-5), 7.71 (1H, d, J = 8.45 Hz, Ar-H), 7.82 (1H, d, J = 8.45 Hz, Ar-H); 13C-NMR (CDCl3) δ 18.72, 22.23, 25.65, 62.28, 72.12, 77.28, 106.57, 112.19, 113.67, 114.14, 126.50, 127.02, 127.12, 127.69, 128.69, 130.51, 130.89, 132.24, 133.11, 134.21, 134.73, 138.00, 138.71, 152.22, 153.44, 156.38, 159.53, 163.86, 164.38; MS (ESI) m/z 642.0, 644.2 ([M+Na]+).
(3'S,4'S)-bis-O-(2,5-dichlorobenzoyl)-4-methyl-(-)-cis-khellactone (3o). Molecular formula (MW): C29H20Cl4O7 (622.28 g/mol); white solid; 28% Yield; mp: 116–118 °C; [α: −71.2 (c 0.1, CH2Cl2). 1H-NMR (CDCl3) δ 1.58 (6H, s, 2 × C-2'-CH3), 2.40 (3H, s, C-4-CH3), 5.69 (1H, d, J = 4.95 Hz, H-3'), 6.14 (1H, s, H-3), 6.90 (1H, d, J = 8.88 Hz, H-6), 6.91 (1H, d, J = 4.95 Hz, H-4'), 7.29–7.40 (4H, m, Ar-H), 7.57 (1H, d, J = 8.88 Hz, H-5), 7.72 (1H, d, J = 2.30 Hz, Ar-H), 7.81 (1H, d, J = 2.30 Hz, Ar-H); 13C-NMR (CDCl3) δ 18.73, 22.17, 25.70, 62.32, 72.32, 106.38, 112.28, 113.73, 114.12, 126.61, 130.61, 131.08, 131.27, 131.72, 131.75, 131.87, 131.92, 132.13, 132.34, 132.60, 132.82, 152.13, 153.45, 156.33, 159.44, 163.56, 163.87; MS (ESI) m/z 642.0, 644.2 ([M+Na]+).