A Facile Synthesis of Deaza-Analogues of the Bisindole Marine Alkaloid Topsentin
Abstract
:1. Introduction
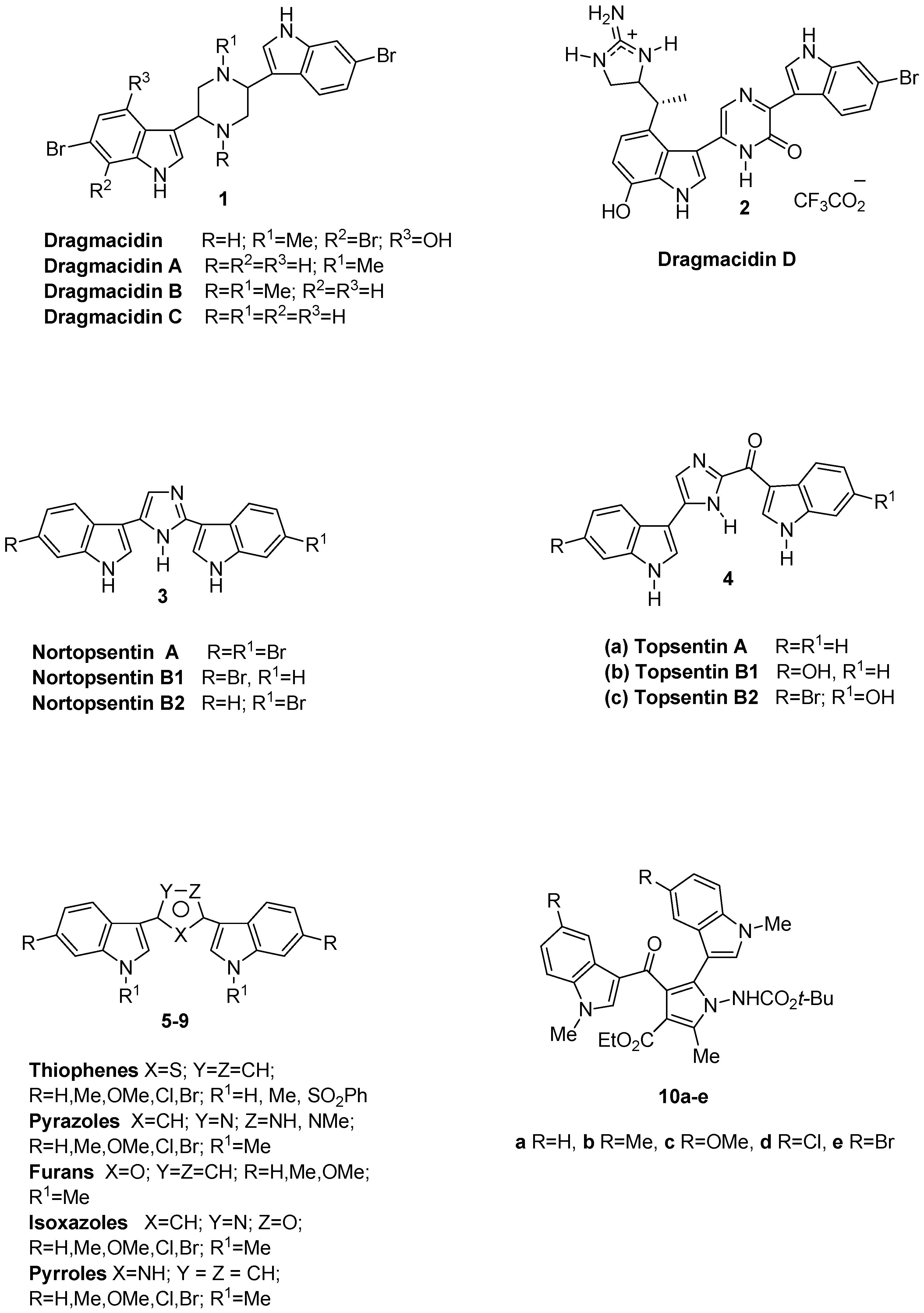
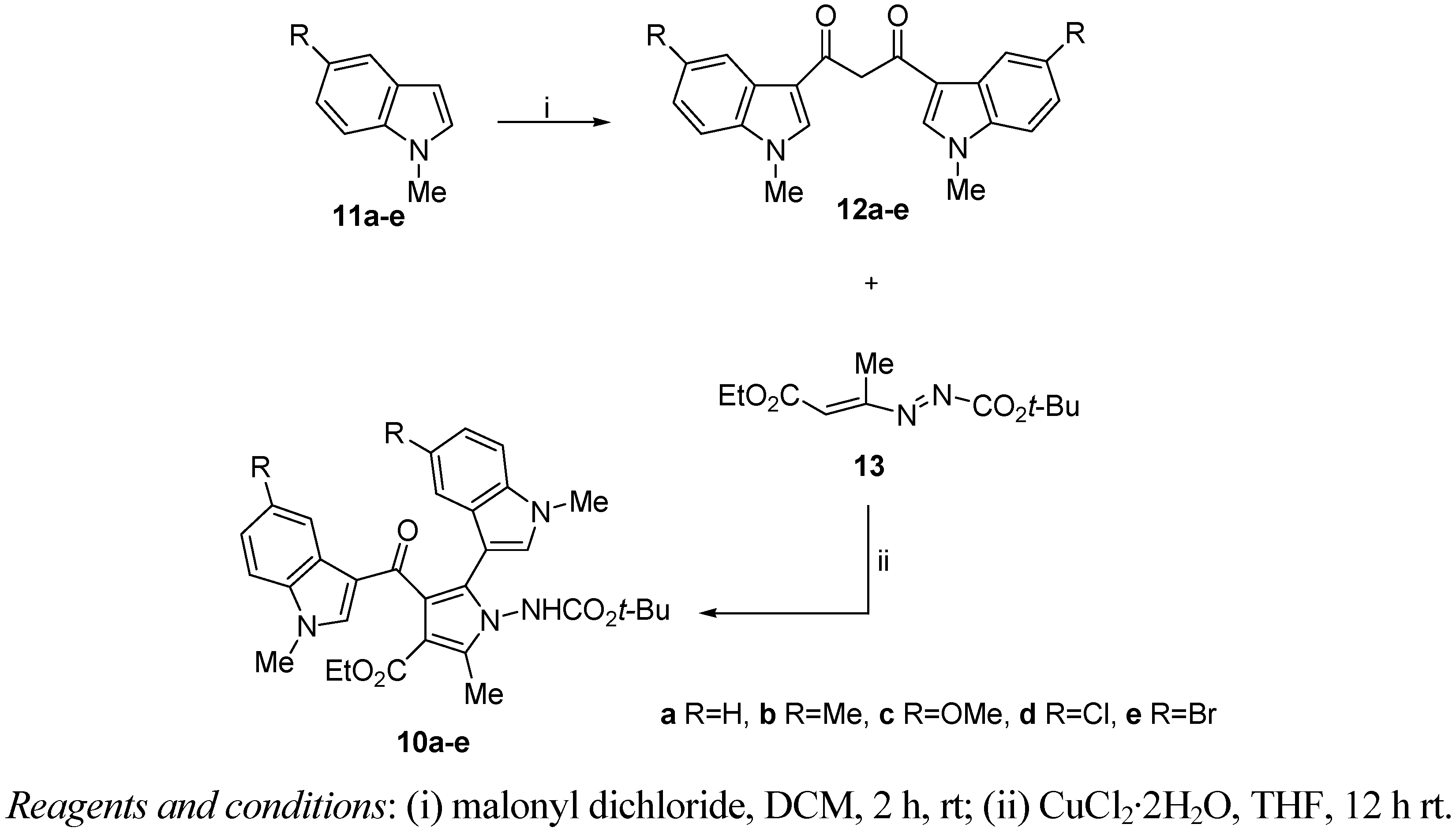
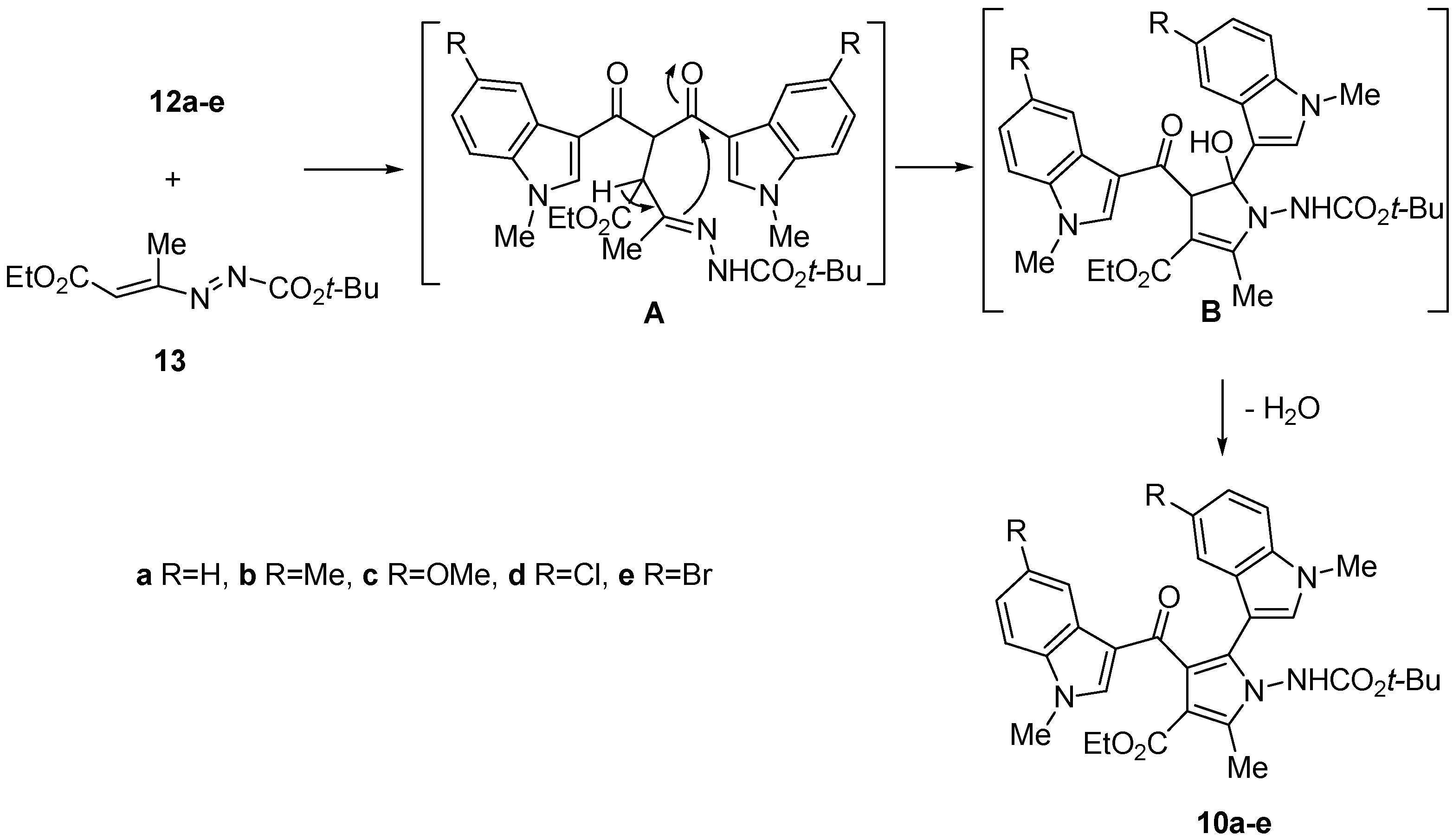
2. Results and Discussion
3. Experimental
3.1. Chemistry
3.1.1 General Procedures
3.1.1.1. General Procedure for the Preparation of 1,3-bis(indol-3-yl)propane-1,3-diones 12a–e
3.1.1.2 General Procedure for the Preparation of Ethyl 1-[(tert-butoxycarbonyl)amino]-2-methyl-5-(1-methyl-1H-indol-3-yl)-4-[(1-methyl-1H-indol-3-yl)carbonyl]-1H-pyrrole-3-carboxylates 10a–e
3.2. Biology
3.2.1. Methodology of the in Vitro Cancer Screen
4. Conclusions
Acknowledgments
References and Notes
- Shin, J.; Seo, Y.; Cho, K.W.; Rho, J.R.; Sim, C. New Bis(indole)Alkaloids of the Topsentin class from the sponge Spongosorites genitrix. J. Nat. Prod. 1999, 62, 647–649. [Google Scholar] [CrossRef]
- Casapullo, A.; Bifulco, G.; Bruno, I.; Riccio, R. Hamacanthin Classes from the Mediterranean Marine Sponge Raphisia lacazei. J. Nat. Prod. 2000, 63, 447–451. [Google Scholar] [CrossRef]
- Bao, B.; Sun, Q.; Yao, X.; Hong, J.; Lee, C.; Sim, C.J.; Jung, J.H. Cytotoxic Bisindole Alkaloids from a Marine Sponge Spongosorites sp. J. Nat. Prod. 2005, 68, 711–715. [Google Scholar] [CrossRef]
- Gul, W.; Hamann, M.T. Indole alkaloid marine natural products: An established source of cancer drug leads with considerable promise for the control of parasitic, neurological and other diseases. Life Sci. 2005, 78, 442–453. [Google Scholar] [CrossRef]
- Kohmoto, S.; Kashman, Y.; McConnell, O.J.; Rinehart, K.L., Jr.; Wright, A.; Koehn, F. The first total synthesis of Dragmacidin D. J. Org. Chem. 1988, 53, 3116–3118. [Google Scholar]
- Morris, S.A.; Andersen, R.J. Brominated bis(indole)alkaloids from the marine sponge Hexadella sp. Tetrahedron 1990, 46, 715–720. [Google Scholar] [CrossRef]
- Fahy, E.; Potts, B.C.M.; Faulkner, D.J.; Smith, K. 6-Bromotryptamine Derivatives from the Gulf of California Tunicate Didemnum candidum. J. Nat. Prod. 1991, 54, 564–569. [Google Scholar] [CrossRef]
- Wright, A.E.; Pomponi, S.A.; Cross, S.S.; McCarthy, P. A new bis-(indole)alkaloids from a deep-water marine sponge of the genus Spongosorites. J. Org. Chem. 1992, 57, 4772–4775. [Google Scholar] [CrossRef]
- Capon, R.J.; Rooney, F.; Murray, L.M.; Collins, E.; Sim, A.T.R.; Rostas, J.A.P.; Butler, M.S.; Carroll, A.R. Dragmacidins: New protein phosphatase inhibitors from a Southern Australian deep-water Marine Sponge Spongosorites sp. J. Nat. Prod. 1998, 61, 660–662. [Google Scholar] [CrossRef]
- Alvarez, M.; Salas, M. Marine, nitrogen-containing heterocyclic natural products-structures and syntheses of compounds containing indole units. Heterocycles 1991, 32, 1391–1429. [Google Scholar] [CrossRef]
- Sakem, S.; Sun, H.H. Cytotoxic and antifungal imidazolediylbis[indoles] from the sponge Spongosorites ruetzleri. J. Org. Chem. 1991, 56, 4304–4307. [Google Scholar] [CrossRef]
- Bartik, K.; Braekman, J.-C.; Daloze, D.; Stoller, C. Topsentin, new toxic bis-indole alkaloids from the marine sponge Topsentia genitrix. Can. J. Chem. 1987, 65, 2118–2121. [Google Scholar] [CrossRef]
- Burres, N.S.; Barber, D.A.; Gunasekera, S.P.; Shen, L.L.; Clement, J. Antitumor activity and biochemical effects of topsentin. Biochem. Pharmacol. 1991, 42, 745–751. [Google Scholar]
- Tsuji, S.; Rinehart, K.L.; Gunasekera, S.P.; Kashman, Y.; Cross, S.S.; Lui, M.S.; Pomponi, S.A.; Diaz, M.C. Topsentin, Bromotopsentin, and Dihydrodeoxybromotopsentin: Antiviral and Antitumor bis (indolyl)imidazoles from Caribbean deep-sea sponge of the family Halichondriidae. Structural and synthetic studies. J. Org. Chem. 1988, 53, 5446–5453. [Google Scholar]
- Gu, X.; Wan, X.; Jiang, B. Syntheses and biological activities of bis(3-indolyl)thiazoles, analogues of marine bis(indole)alkaloid nortopsentin. Bioorg. Med. Chem. Lett. 1999, 9, 569–572. [Google Scholar] [CrossRef]
- Jiang, B.; Gu, X.H. Syntheses and cytotoxicity evaluation of bis(indolyl)thiazole, bis(indolyl)pyrazinone and bis(indolyl)pyrazine: analogues of cytotoxic marine bis(indole) alkaloid. Bioorg. Med. Chem. Lett. 2000, 8, 363–371. [Google Scholar] [CrossRef]
- Jiang, B.; Xiong, X.; Yang, C. Synthesis and cytotoxicity evaluation of novel indolylpyrimidines and indolylpyrazines as potential antitumor agents. Bioorg. Med. Chem. 2001, 9, 1149–1154. [Google Scholar] [CrossRef]
- Jiang, B.; Xiong, X.; Yang, C. Synthesis and antitumor evaluation of novel monoindolyl-4-trifluoromethylpyridines and bisindolyl-4-trifluoromethylpyridines. Bioorg. Med. Chem. Lett. 2001, 11, 475–477. [Google Scholar] [CrossRef]
- Xiong, W.; Yang, C.; Jiang, B. Synthesis of novel analogues of marine indole alkaloids: mono(indolyl)-4-trifluoromethylpyridines and bis(indolyl)-4-trifluoromethylpyridines as potential anticancer agents. Bioorg. Med. Chem. 2001, 9, 1773–1770. [Google Scholar] [CrossRef]
- Diana, P.; Carbone, A.; Barraja, P.; Montalbano, A.; Martorana, A.; Dattolo, G.; Gia, O.; Dalla Via, L.; Cirrincione, G. Synthesis and antitumor properties of 2,5-bis(3’-indolyl)thiophenes: analogues of marine alkaloid nortopsentin. Bioorg. Med. Chem. Lett. 2007, 17, 2342–2346. [Google Scholar]
- Diana, P.; Carbone, A.; Barraja, P.; Martorana, A.; Gia, O.; Dalla Via, L.; Cirrincione, G. 3,5-Bis(3’-indolyl)pyrazoles, analogues of marine alkaloid nortopsentin: synthesis and antitumor properties. Bioorg. Med. Chem. Lett. 2007, 17, 6134–6137. [Google Scholar] [CrossRef]
- Diana, P.; Carbone, A.; Barraja, P.; Kelter, H.H.; Fiebig, G.; Cirrincione, G. Synthesis and antitumor activity of 2,5-bis(3’-indolyl)-furans and 3,5-bis(3’-indolyl)-isoxazoles, nortopsentin analogues. Bioorg. Med. Chem. 2010, 18, 4524–4529. [Google Scholar] [CrossRef]
- Carbone, A.; Parrino, B.; Barraja, P.; Spanò, V.; Cirrincione, G.; Diana, P.; Maier, A.; Kelter, G.; Fiebig, H-H. Synthesis and antiproliferative activity of 2,5-bis(3’-Indolyl)pyrroles, analogues of the marine alkaloid Nortopsentin. Marine Drugs 2013, 11. [Google Scholar] [CrossRef]
- Recent review on the chemistry of 1,2-diaza-1,3-dienes, see:Attanasi, O.A.; De Crescentini, L.; Favi, G.; Filippone, P.; Mantellini, F.; Perrulli, F.R.; Santeusanio, S. Cultivating the passion to build heterocycles from 1,2-diaza-1,3-dienes: the Force of immagination. Eur. J. Org. Chem. 2009, 3109–3127. [Google Scholar]
- Report on copper-catalyzed synthesis of pyrroles from 1,2-diaza-1,3-dienes, see: (b)Attanasi, O.A.; Grossi, M.; Serra-Zanetti, F.; Foresti, E. Effect of metal ions in organic synthesis. Part XXXIII. 2-hydroxy-4-pyrrolines as stable intermediates in the synthesis of 1-aminopyrrole derivatives. Tetrahedron 1987, 43, 4249–4256. [Google Scholar]
- Monks, A.; Scudiero, D.; Skehan, P.; Shoemaker, R.; Paull, K.; Vistica, D.; Hose, C.; Langely, J.; Cronise, P.; Vaigro-Wolff, A.; et al. Feasibility of a high-flux anticancer drug screen using a diverse panel of culture human tumor cell lines. J. Natl. Cancer Inst. 1991, 83, 757–766. [Google Scholar] [CrossRef]
- Sommer, S. Inverse diels-alder-reaktionen von azo-alkenen mit aliene. Tetrahedron Lett. 1977, 18, 117–120. [Google Scholar] [CrossRef]
- Attanasi, O.A.; Filippone, P.; Mei, A.; Santeusanio, S. Effect of metal ions in organic synthesis; Part XXIII. Easy and high-yield direct synthesis of 3-aminocarbonyl-1-ureidopyrroles by the copper(II) chloride-catalyzed reaction of aminocarbonylazoalkenes with 3-oxoalkanamides. Synthesis 1984, 671–672. [Google Scholar]
- Attanasi, O.A.; Filippone, P.; Mei, A.; Santeusanio, S. Effect of metal ions in organic. Synthesis; Part XXIV. Facile one-flask synthesis of 1-alkoxycarbonylamino by reaction of alkoxycarbonylamino-3-aminocarbonylpyrroles by reaction of alkoxycarbonylazoalkenes with 3-oxoalkanamides under copper(II) chloride catalysis. Synthesis 1984, 873–874. [Google Scholar]
- NCI-60 DTP Human tumor cell line screen. Available online: http://dtp.nci.nih.gov/branches/btb/ivclsp.html (accessed on 12 March 2012).
- Sample Availability: Samples of the compounds 10a–e are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Carbone, A.; Spanò, V.; Parrino, B.; Ciancimino, C.; Attanasi, O.A.; Favi, G. A Facile Synthesis of Deaza-Analogues of the Bisindole Marine Alkaloid Topsentin. Molecules 2013, 18, 2518-2527. https://doi.org/10.3390/molecules18032518
Carbone A, Spanò V, Parrino B, Ciancimino C, Attanasi OA, Favi G. A Facile Synthesis of Deaza-Analogues of the Bisindole Marine Alkaloid Topsentin. Molecules. 2013; 18(3):2518-2527. https://doi.org/10.3390/molecules18032518
Chicago/Turabian StyleCarbone, Anna, Virginia Spanò, Barbara Parrino, Cristina Ciancimino, Orazio A. Attanasi, and Gianfranco Favi. 2013. "A Facile Synthesis of Deaza-Analogues of the Bisindole Marine Alkaloid Topsentin" Molecules 18, no. 3: 2518-2527. https://doi.org/10.3390/molecules18032518
APA StyleCarbone, A., Spanò, V., Parrino, B., Ciancimino, C., Attanasi, O. A., & Favi, G. (2013). A Facile Synthesis of Deaza-Analogues of the Bisindole Marine Alkaloid Topsentin. Molecules, 18(3), 2518-2527. https://doi.org/10.3390/molecules18032518




