Synthesis of Bistetrahydroquinolines as Potential Anticholinesterasic Agents by Double Diels-Alder Reactions
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
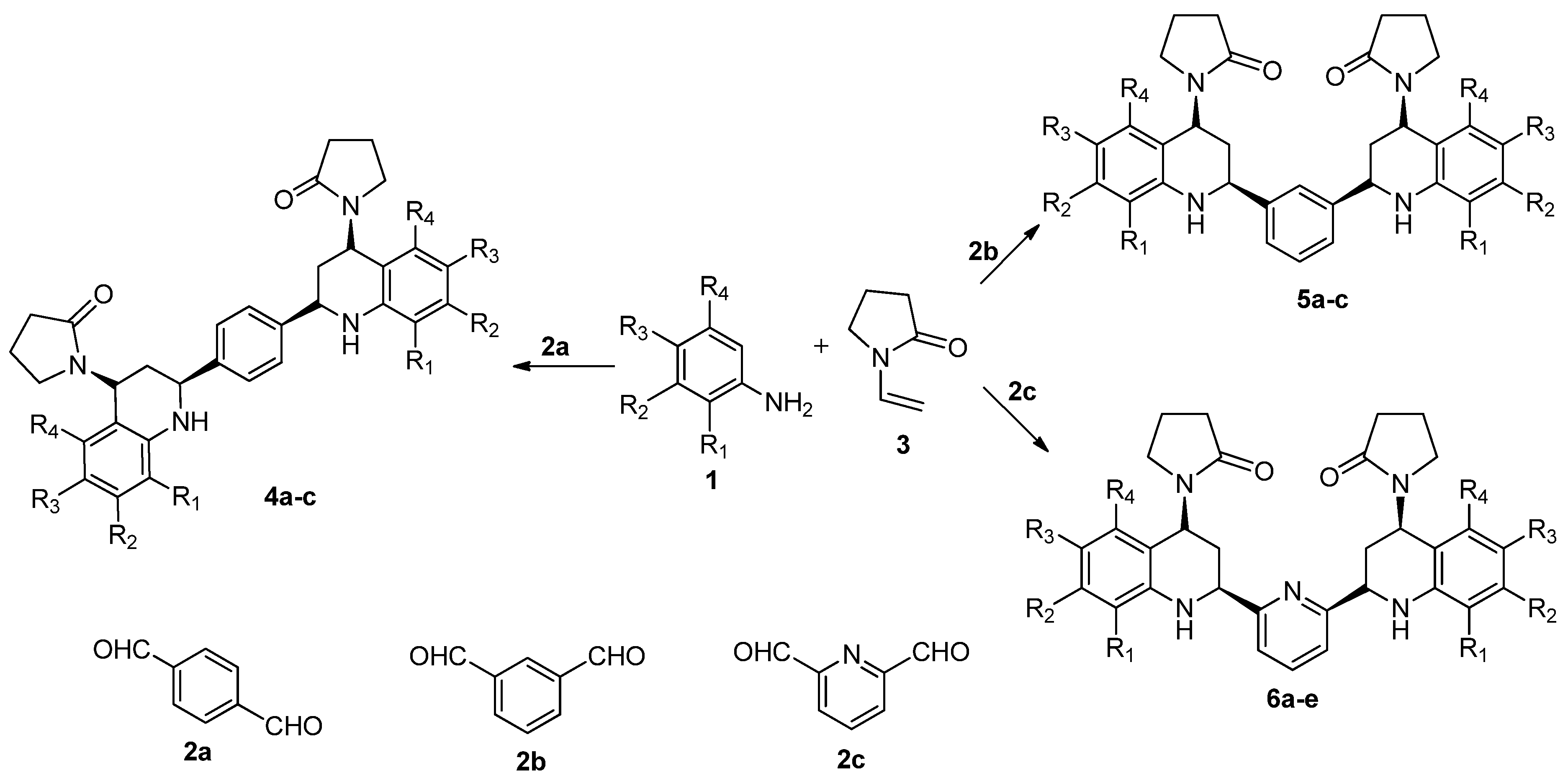
| Compound | R1 | R2 | R3 | R4 | Mp (°C) | Yield (%) | Reaction Time (h) |
|---|---|---|---|---|---|---|---|
| 4a | H | H | H | H | 250–257 | 60 | 7 |
| 4b | H | H | CH3 | H | 295–298 | 75 | 7 |
| 4c | H | H | O-CH3 | H | 218–221 | 22 | 5 |
| 5a | H | H | H | H | 221–225 | 67 | 7 |
| 5b | H | H | CH3 | H | 190–195 | 51 | 8 |
| 5c | H | H | O-CH3 | H | 237–239 | 58 | 6 |
| 6a | H | H | H | H | 155–158 | 46 | 7 |
| 6b | H | H | CH3 | H | 187–190 | 72 | 5 |
| 6c | H | H | O-CH3 | H | 173–178 | 34 | 5 |
| 6d | H | H | NO2 | H | 202–206 | 65 | 10 |
| 6e | H | CH3 | H | CH3 | 157–161 | 25 | 9 |

2.2. Biological Activities
2.3. Molecular Docking and Binding Affinity Calculations
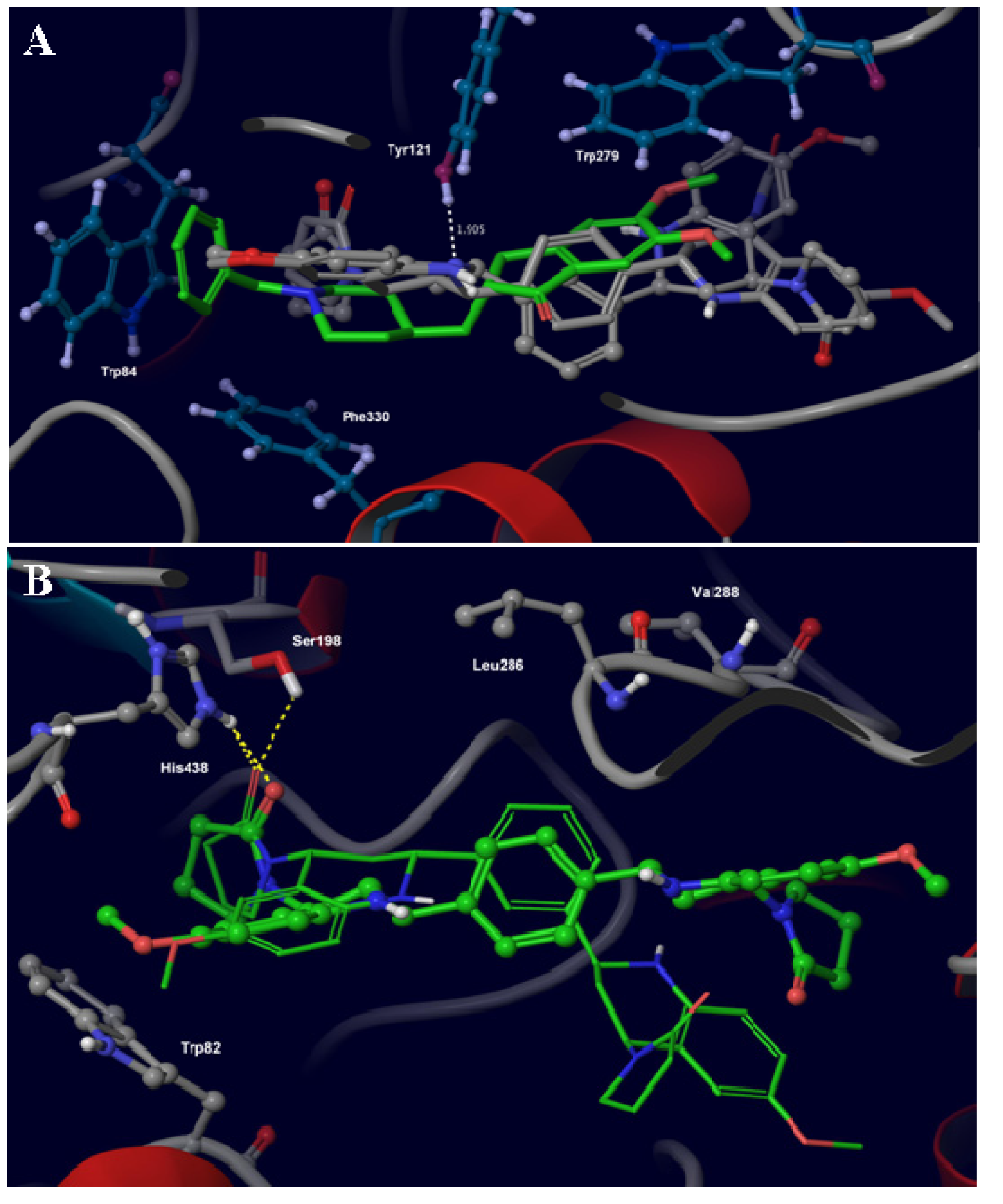
| Glide docking score (kcal mol−1) | Binding Free Energy (kcal mol−1) a | |||
|---|---|---|---|---|
| Target/Ligand | AChE | BuChE | AChE | BuChE |
| 5c | −8.63 | −7.98 | −30.64 | −33.94 |
| 4c | −7.37 | −8.82 | −28.72 | −29.27 |
3. Experimental
3.1. General
3.2. Biological Assays
3.3. Computational Details
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Sankaran, M.; Kumarasamy, C.; Chokkalingam, U.; Mohan, P.S. Synthesis, antioxidant and toxicological study of novel pyrimido quinoline derivatives from 4-hydroxy-3-acylquinolin-2-one. Bioorg. Med. Chem. Lett. 2010, 20, 7147–7151. [Google Scholar] [CrossRef]
- Marella, A.; Prakash, T.O.; Saha, R.; Rahmat, M.A.; Srivastava, S.; Akhter, M.; Shaquiquzzaman, M.; Mumtaz, A. Quinoline: A versatile heterocyclic. Saudi Pharm. J. 2013, 21, 1–12. [Google Scholar] [CrossRef]
- Nesterova, I.N.; Alekseeva, L.M.; Golovira, S.M.; Granik, V.G. Synthesis and study the pharmacological activity of derivatives of 5-dimethylaminopyrano[3,2-c]quinolin-2-ones. Pharm. Chem. J. 1995, 29, 111–114. [Google Scholar] [CrossRef]
- Yamada, N.; Kadowaki, S.; Takahashi, K.; Umezu, K. MY-1250, a major metabolite of the anti-allergic drug repirinast, induces phosphorylation of a 78-kDa protein in rat mast cells. Biochem. Pharmacol. 1992, 44, 1211–1213. [Google Scholar] [CrossRef]
- Wallace, O.B.; Lauwers, K.S.; Jones, S.A.; Dodge, J.A. Tetrahydroquinoline-based selective estrogen receptor modulators (SERMs). Bioorg. Med. Chem. Lett. 2003, 13, 1907–1910. [Google Scholar] [CrossRef]
- Mahajan, A.; Yeh, S.; Nell, M.; Rensburg, C.E.; Chibale, K. Synthesis of new 7-chloroquinolinyl thioureas and their biological investigation as potential antimalarial and anticancer agents. Bioorg. Med. Chem. Lett. 2007, 17, 5683–5685. [Google Scholar] [CrossRef]
- Upadhayaya, R.S.; Vandavasi, J.K.; Vasireddy, N.R.; Sharma, V.; Dixit, S.S.; Chattopadhyaya, J. Design, synthesis, biological evaluation and molecular modelling studies of novel quinoline derivatives against Mycobacterium tuberculosis. Bioorg. Med. Chem. 2009, 17, 2830–2841. [Google Scholar] [CrossRef]
- Kumar, S.; Bawa, S.; Drabu, S.; Panda, B.P. Design and synthesis of 2-chloroquinoline derivatives as non-azoles antimycotic agents. Med. Chem. Res. 2011, 20, 1340–1348. [Google Scholar] [CrossRef]
- Kouznetsov, V.; Meléndez, G.C.; Derita, M.G.; Vetaz, L.; del Olmo, E.; Zacchino, A.S. Synthesis and antifungal activity of diverse C-2 pyridinyl and pyridinylvinyl substituted quinolines. Bioorg. Med. Chem. 2012, 20, 6506–6512. [Google Scholar] [CrossRef]
- Bernotas, R.C.; Singhaus, R.R.; Kaufman, D.H.; Ullrich, J.; Fletcher, H.; Quinet, E.; Nambi, P.; Unwalla, R.; Wilhelmsson, A.; Nilsson, A.G.; et al. Biarylether amide quinolines as liver X receptor agonists. Bioorg. Med. Chem. 2009, 17, 1663–1670. [Google Scholar] [CrossRef]
- Chen, S.; Chen, R.; He, M.; Pang, R.; Tan, Z.; Yang, M. Design, synthesis, and biological evaluation of novel quinoline derivatives as HIV-1 Tat-TAR interaction inhibitors. Bioorg. Med. Chem. 2009, 17, 1948–1956. [Google Scholar] [CrossRef]
- Normand-Bayle, M.; Bénard, C.; Zouhiri, F.; Mouscadet, J.-F.; Leh, H.; Thomas, C.-M.; Mbemba, G.; Desmaële, D.; d’Angelo, J. New HIV-1 replication inhibitors of the styryquinoline class bearing aroyl/acyl groups at the C-7 position: Synthesis and biological activity. Bioorg. Med. Chem. Lett. 2005, 15, 4019–4022. [Google Scholar] [CrossRef]
- Asberom, T.; Bara, T.; Clader, J.W.; Greenlee, W.J.; Guzik, H.S.; Josien, H.B.; Li, W.; Parker, E.M.; Pissarnitski, D.A.; Song, L.; et al. Tetrahydroquinoline sulfonamides as gamma-secretase inhibitors. Bioorg. Med. Chem. Lett. 2007, 17, 205–207. [Google Scholar] [CrossRef]
- Chen, J.J.; Chang, Y.L.; Teng, C.M.; Su, C.C.; Chen, I.S. Quinoline alkaloids and anti-platelet aggregation constituents from the leaves of Melicope semecarpifolia. Planta Med. 2002, 9, 790–793. [Google Scholar]
- Bedoya, L.M.; Abad, M.J.; Calonge, E.; Astudillo, S.L.; Gutierrez, C.M.; Kouznetsov, V.V.; Alcami, J.; Bermejo, P. Quinoline-based compounds as modulators of HIV transcription through NF-kappaB and Sp1 inhibition. Antivir. Res. 2010, 87, 338–344. [Google Scholar] [CrossRef]
- Minati, L.; Edginton, T.; Bruzzone, M.G.; Giaccone, G. Current concepts in Alzheimer’s disease: A multidisciplinary review. Am. J. Alzheimers Dis. Other Dement. 2009, 24, 95–121. [Google Scholar] [CrossRef]
- Munoz-Muriedas, J.; Lopez, J.M.; Orozco, M.; Luque, F.J. Molecular modeling approaches to the design of acetylcholinesterase inhibitors: New challenges for the treatment of Alzheimer’s disease. Curr. Pharm. Design 2004, 10, 3131–3140. [Google Scholar] [CrossRef]
- Kurz, A. The therapeutic potential of tacrine. J. Neural. Transm. Suppl. 1998, 54, 295–299. [Google Scholar] [CrossRef]
- Kryger, G.; Silman, I.; Sussman, J.L. Structure of acetylcholinesterase complexed with E2020 (Aricept): Implications for the design of new anti-Alzheimer drugs. Structure 1999, 7, 297–307. [Google Scholar] [CrossRef]
- Santos, M.D.; Alkondon, M.; Pereira, E.F.R.; Aracava, Y.; Eisenberg, H.M.; Maelicke, A.; Albuquerque, E.X. The nicotinic allosteric potentiating ligand galantamine facilitates synaptic transmission in the mammalian central nervous system. Mol. Pharmacol. 2002, 61, 1222–1234. [Google Scholar] [CrossRef]
- Bar-On, P.; Millard, C.B.; Harel, M.; Dvir, H.; Enz, A.; Sussman, J.L.; Silman, I. Kinetic and structural studies on the interaction of cholinesterases with the anti-Alzheimer drug rivastigmine. Biochemistry 2002, 41, 3555–3564. [Google Scholar] [CrossRef]
- Rizzo, S.; Bartolini, M.; Ceccarini, L.; Piazzi, L.; Gobbi, S.; Cavalli, A.; Recanatini, M.; Andrisano, V.; Rampa, A. Targeting Alzheimer’s disease: Novel indanone hybrids bearing a pharmacophoric fragment of AP2238. Bioorg. Med. Chem. 2010, 18, 1749–1760. [Google Scholar] [CrossRef]
- Kouznetsov, V.; Vargas, L.; Meléndez, C. Recent progress in the synthesis of quinolines. Curr. Org. Chem. 2005, 9, 141–161. [Google Scholar] [CrossRef]
- Kouznetsov, V. Recent synthetic developments in a powerful imino Diels-Alder reaction (Povarov reaction): Application to the synthesis of N-polyheterocycles and related alkaloids. Tetrahedron 2009, 65, 2721–2750. [Google Scholar] [CrossRef]
- Glushkov, V.A.; Tolstikov, A.G. Synthesis of substituted 1,2,3,4-tetrahydroquinones by the Povarov reaction. New potentials of the classical reaction. Russ. Chem. Rev. 2008, 77, 137–159. [Google Scholar] [CrossRef]
- Maiti, G.; Kundu, P. Imino Diels-Alder reactions: An efficient one-pot synthesis of pyrano and furanoquinoline derivatives catalyzed by SbCl3. Tetrahedron Lett. 2006, 47, 5733–5736. [Google Scholar] [CrossRef]
- Grieco, P.A.; Bahsas, A. Role reversal in the cyclocondensation of cyclopentadiene with heterodienophiles derived from aryl amines and aldehydes: Synthesis of novel tetrahydroquinolines. Tetrahedron Lett. 1988, 29, 5855–5858. [Google Scholar] [CrossRef]
- Hadden, M.; Stevenson, P.J. Regioselective synthesis of pyrroloquinolines—Approaches to Martinelline. Tetrahedron Lett. 1999, 40, 1215–1218. [Google Scholar] [CrossRef]
- Kouznetsov, V.; Romero, A.; Stashenko, E. Three-component imino Diels-Alder reaction with essential oil and seeds of anise: Generation of new tetrahydroquinolines. Tetrahedron Lett. 2007, 48, 8855–8860. [Google Scholar] [CrossRef]
- Lenga, R.E. BiCl3 is as Lethal as Sodium Chloride for the Rat. In Sigma-Aldrich Library of Chemical Safety Data, 2nd ed.; Sigma-Aldrich Corporation: Milwaukee, WI, USA, 1988; Volume 1, p. 442. [Google Scholar]
- Mendez, L.Y.; Kouznetsov, V.; Poveda, J.C.; Yolacan, C.; Öcal, N.; Aydongan, F. Transformations of 4-N-arylamino-4-(8-quinolinyl)-i-butenes and 3-aryl-2-(8-quinolinyl)-4-thiazolidinones. Heterocicycl. Commun. 2001, 7, 129–134. [Google Scholar]
- Astudillo, L.; Vallejos, G.; Kouznetsov, V.; Gutiérrez , C.M.; Meléndez, C.M.; Vargas, L; Bermúdez, J. Synthesis of new Diversely linked biquinoline derivates by multicomponent Imino-Diels-Alder Cycloaddition and Intramoleclar Friedel-Crafs Cyclization. Synthesis 2010, 4, 593–600. [Google Scholar]
- Katritzky, A.; Rachwal, B.; Rachwal, S. Reactions of Alkyl-N-phenyl-1H-benzotriazole-1-methanamines with N-Vinilamides and N-Vinylcarbazole. A Convenient Synthesis of 4-(Dialkylamino)tetrahydroquinolines. J. Org. Chem. 1995, 60, 3993–4001. [Google Scholar] [CrossRef]
- Ellman, G.L.; Courtney, K.D.; Andres, V., Jr. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Rainer, M. Clinical studies with galanthamine. Drugs Today 1997, 33, 273–279. [Google Scholar] [CrossRef]
- Sharma, J.; Ramanathan, K.; Sethumadhavan, R. Identification of potencial inhibitors against acetylcholinesterase associated with alzheimer diseases: A molecular docking aproach. J. Comput. Methods Mol. Design 2011, 1, 44–51. [Google Scholar]
- Radic, Z.A.; Pickering, D.C.; Vellom, S.; Camp, P. Three distinct domains in the cholinesterase molecule confer selectivity for acetyl- and butyrylcholinesterase inhibitors. Biochemistry 1993, 32, 12074–12084. [Google Scholar] [CrossRef]
- Bajda, M.; Więckowska, A.; Hebda, M.; Guzior, N.; Sotriffer, C.A.; Malawska, B. Structure-based search for new inhibitors of cholinesterases. Int. J. Mol. Sci. 2013, 14, 5608–5632. [Google Scholar] [CrossRef]
- Kawakami, Y.; Inoue, A.; Kawai, T.; Wakita, M.; Sugimoto, H.; Hopfinger, A.J. The rationale for E2020 as a potent acetylcholinesterase inhibitor. Bioorg. Med. Chem. 1996, 4, 1429–1446. [Google Scholar] [CrossRef]
- Cardozo, M.G.; Imura, Y.; Sugimoto, H.; Yamanishi, Y.; Hopfinger, A.J. QSAR analyses of the substituted indanone and benzylpiperidine rings of a series of indanone-benzylpiperidine inhibitors of acetylcholinesterase. J. Med. Chem. 1992, 35, 584–589. [Google Scholar] [CrossRef]
- Cardozo, M.G.; Kawai, T.; Imura, Y.; Sugimoto, H.; Yamanishi, Y.; Hopfinger, A.J. Conformational analyses and molecular-shape comparisons of a series of indanone-benzylpiperidine inhibitors of acetylcholinesterase. J. Med. Chem. 1992, 35, 590–601. [Google Scholar] [CrossRef]
- Verdonk, M.L.; Boks, G.J.; Kooijman, H.; Kanters, J.A.; Kroon, J. Stereochemistry of charged nitrogen-aromatic interactions and its involvement in ligand-receptor binding. J. Comput. Aided Mol. Design 1993, 7, 173–182. [Google Scholar] [CrossRef]
- Dougherty, D. Cation-π interactions in chemistry and biology: A new view of benzene, Phe, Tyr, and Trp. Science 1996, 271, 163–168. [Google Scholar]
- De la Torre, P.; Astudillo, L.; Caballero, J.; Quiroga, J.; Alzate-Morales, J.; Gutiérrez, M.; Trilleras, J. A novel class of selective acetylcholinesterase inhibitors: Synthesis and evaluation of (E)-2-(Benzo[d]thiazol-2-yl)-3-heteroarylacrylonitriles. Molecules 2012, 17, 12072–12085. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 4a–c, 5a–c and 6a–e are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Duarte, Y.; Gutiérrez, M.; Astudillo, L.; Alzate-Morales, J.; Valdés, N. Synthesis of Bistetrahydroquinolines as Potential Anticholinesterasic Agents by Double Diels-Alder Reactions. Molecules 2013, 18, 12951-12965. https://doi.org/10.3390/molecules181012951
Duarte Y, Gutiérrez M, Astudillo L, Alzate-Morales J, Valdés N. Synthesis of Bistetrahydroquinolines as Potential Anticholinesterasic Agents by Double Diels-Alder Reactions. Molecules. 2013; 18(10):12951-12965. https://doi.org/10.3390/molecules181012951
Chicago/Turabian StyleDuarte, Yorley, Margarita Gutiérrez, Luis Astudillo, Jans Alzate-Morales, and Natalia Valdés. 2013. "Synthesis of Bistetrahydroquinolines as Potential Anticholinesterasic Agents by Double Diels-Alder Reactions" Molecules 18, no. 10: 12951-12965. https://doi.org/10.3390/molecules181012951
APA StyleDuarte, Y., Gutiérrez, M., Astudillo, L., Alzate-Morales, J., & Valdés, N. (2013). Synthesis of Bistetrahydroquinolines as Potential Anticholinesterasic Agents by Double Diels-Alder Reactions. Molecules, 18(10), 12951-12965. https://doi.org/10.3390/molecules181012951





