A Phytoanticipin Derivative, Sodium Houttuyfonate, Induces in Vitro Synergistic Effects with Levofloxacin against Biofilm Formation by Pseudomonas aeruginosa
Abstract
:1. Introduction
2. Results and Discussion
2.1. MICs of LFX and SH on Pseudomonas aeruginosa
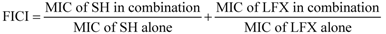
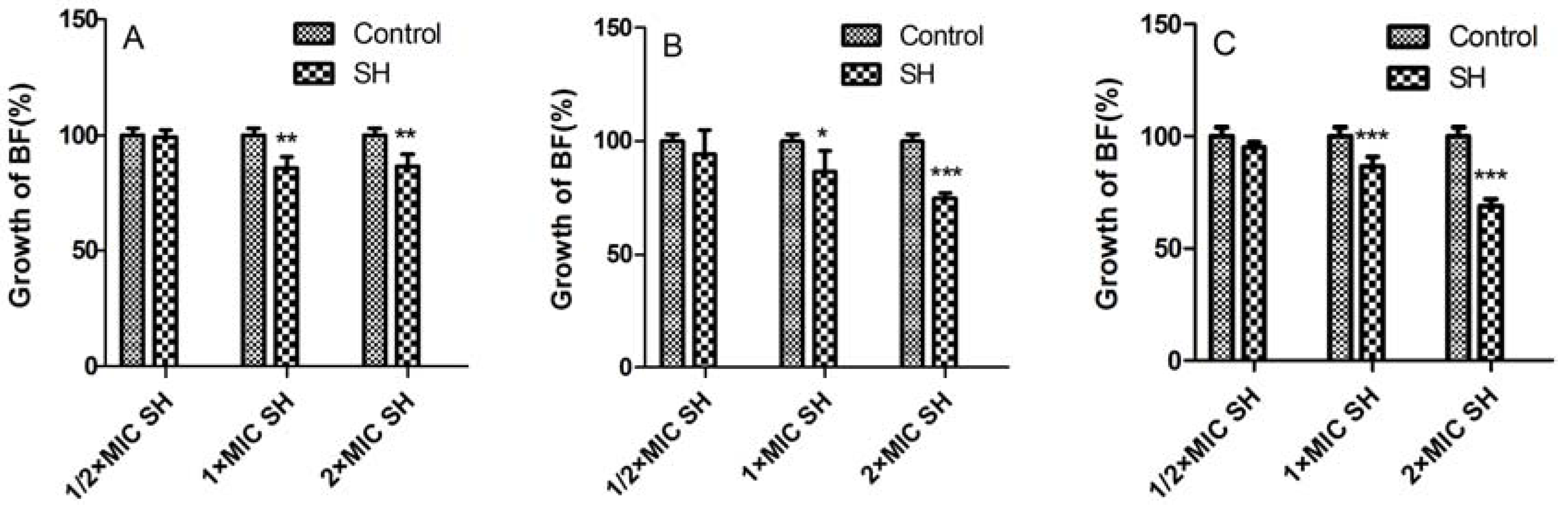
2.2. Biofilm Suppression by SH Alone and in Combination with LFX
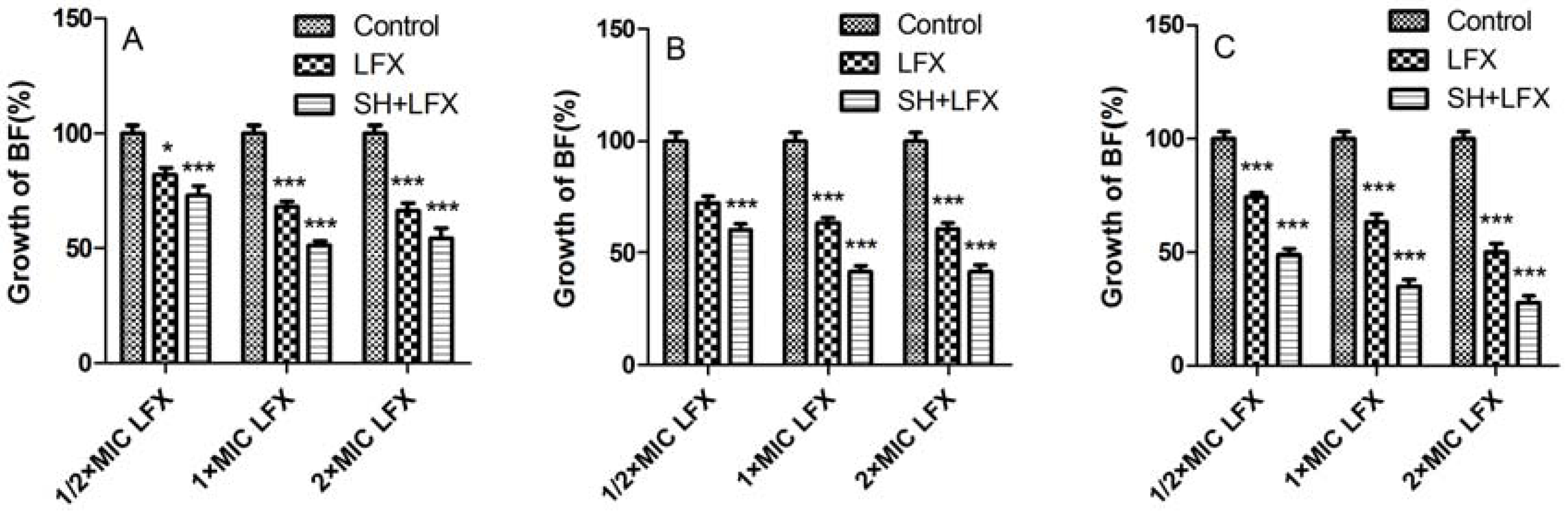
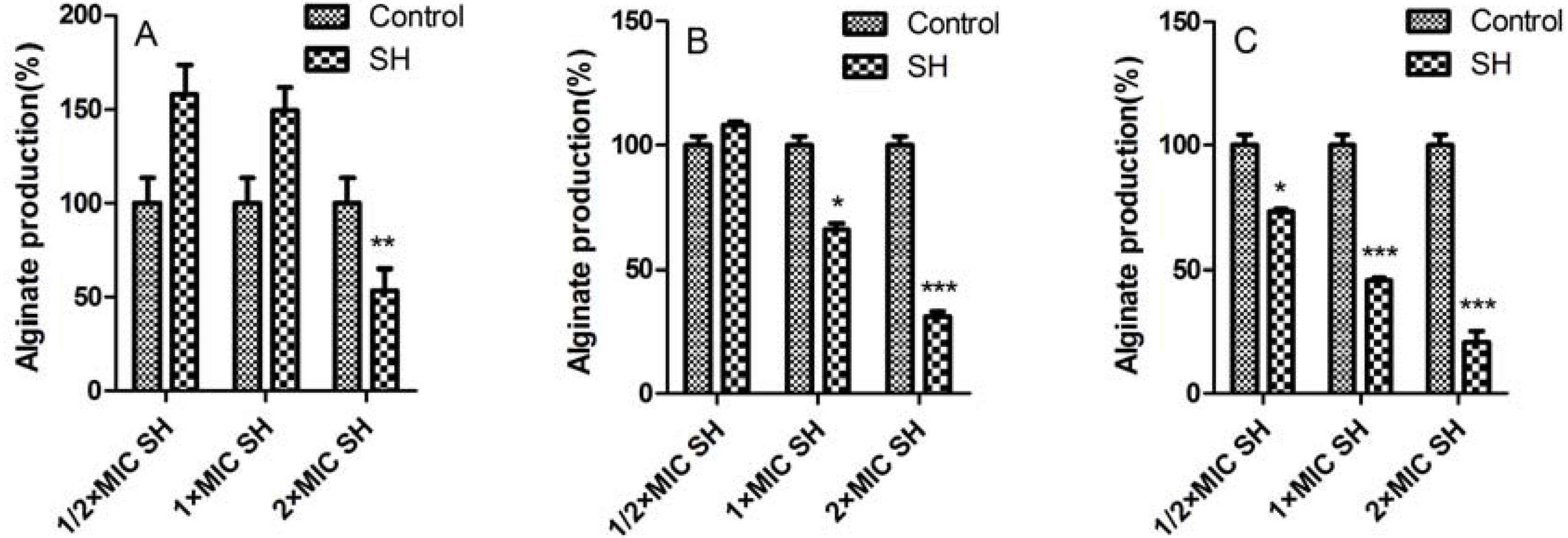
2.3. Alginate Removal by SH Alone and in Combination with LFX
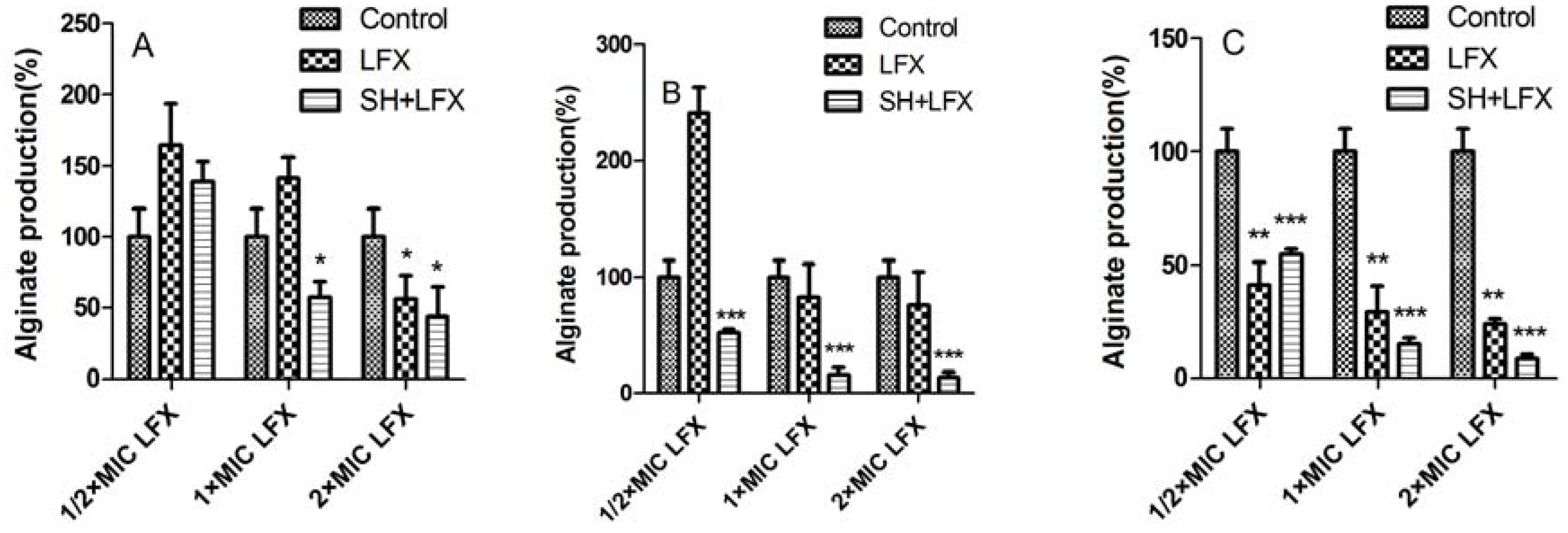
2.4. In Vitro Antimicrobial Role of SH and Its Synergistic Activity with LFX
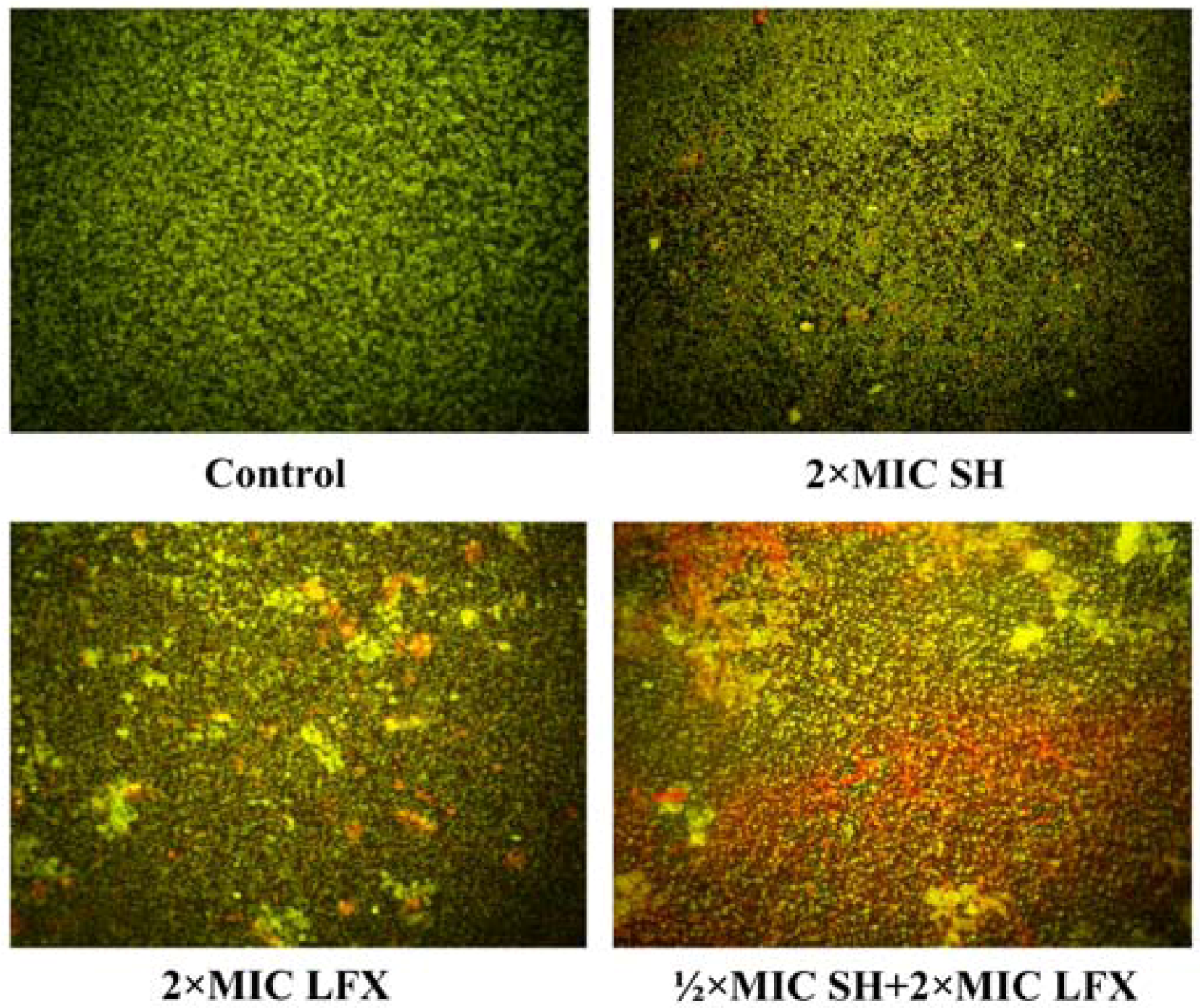
2.5. Observation of Pseudomonas aeruginosa by Fluorescence Microscope
2.6. Observation of Pseudomonas aeruginosa Biofilm Morphology by SEM

3. Experimental
3.1. Strains and Materials

3.2. Cultivation of Pseudomonas aeruginosa
3.3. Preparation of SH and LFX Solutions
3.4. MIC Measurement
3.5. Effects of SH Alone and SH + LFX in Combination on Anti-Biofilm Formation
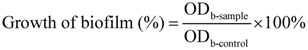
3.6. Effects of SH Alone and SH + LFX in Combination on Anti-Alginate Production
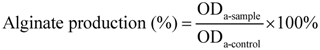
3.7. Fluorescence Microscopy of Bacteria
3.8. Scanning Electric Microscope of Biofilm Morphology
4. Conclusions
Acknowledgments
Conflict of interest
References
- Costerton, J. Introduction to biofilm. Int. J. Antimicrob. Agents 1999, 11, 217–221. [Google Scholar] [CrossRef]
- Hall-Stoodley, L.; Costerton, J.W.; Stoodley, P. Bacterial biofilms: From the natural environment to infectious diseases. Nat. Rev. Microbiol. 2004, 2, 95–108. [Google Scholar] [CrossRef]
- Kirketerp-Møller, K.; Jensen, P.Ø.; Fazli, M.; Madsen, K.G.; Pedersen, J.; Moser, C.; Tolker-Nielsen, T.; Høiby, N.; Givskov, M.; Bjarnsholt, T. Distribution, organization, and ecology of bacteria in chronic wounds. J. Clin. Microbiol. 2008, 46, 2717–2722. [Google Scholar] [CrossRef]
- Bjarnsholt, T.; Jensen, P.O.; Fiandaca, M.J.; Pedersen, J.; Hansen, C.R.; Andersen, C.B.; Pressler, T.; Givskov, M.; Hoiby, N. Pseudomonas aeruginosa biofilms in the respiratory tract of cystic fibrosis patients. Pediatr. Pulmonol. 2009, 44, 547–558. [Google Scholar] [CrossRef]
- Harmsen, M.; Yang, L.; Pamp, S.J.; Tolker-Nielsen, T. An update on pseudomonas aeruginosa biofilm formation, tolerance, and dispersal. FEMS Immunol. Med. Microbiol. 2010, 59, 253–268. [Google Scholar]
- Carneiro, V.A.; Santos, H.S.; Arruda, F.V.; Bandeira, P.N.; Albuquerque, M.R.; Pereira, M.O.; Henriques, M.; Cavada, B.S.; Teixeira, E.H. Casbane diterpene as a promising natural antimicrobial agent against biofilm-associated infections. Molecules 2011, 16, 190–201. [Google Scholar]
- Hengzhuang, W.; Wu, H.; Ciofu, O.; Song, Z.; Hoiby, N. In vivo pharmacokinetics/pharmacodynamics of colistin and imipenem in pseudomonas aeruginosa biofilm infection. Antimicrob. Agents Chemother. 2012, 56, 2683–2690. [Google Scholar] [CrossRef]
- Saleh, S.; Taha, M.O.; Haddadin, R.N.; Marzooqa, D.; Hodali, H. Preparation of silver- and zinc-doped mullite-based ceramics showing anti-bacterial biofilm properties. Molecules 2011, 16, 2862–2870. [Google Scholar] [CrossRef]
- Saleh, S.; Sweileh, B.; Taha, S.O.; Mahmoud, R.; Taha, M.O. Preparation of polyester-based metal-cross linked polymeric composites as novel materials resistant to bacterial adhesion and biofilm formation. Molecules 2011, 16, 933–950. [Google Scholar] [CrossRef]
- Hengzhuang, W.; Wu, H.; Ciofu, O.; Song, Z.; Hoiby, N. Pharmacokinetics/pharmacodynamics of colistin and imipenem on mucoid and nonmucoid pseudomonas aeruginosa biofilms. Antimicrob. Agents Chemother. 2011, 55, 4469–4474. [Google Scholar] [CrossRef]
- VanEtten, H.D.; Mansfield, J.W.; Bailey, J.A.; Farmer, E.E. Two classes of plant antibiotics: Phytoalexins versus “phytoanticipins”. Plant Cell 1994, 6, 1191–1192. [Google Scholar]
- Dixon, R.A. Natural products and plant disease resistance. Nature 2001, 411, 843–847. [Google Scholar] [CrossRef]
- Zeng, X.; Liu, X.; Bian, J.; Pei, G.; Dai, H.; Polyak, S.W.; Song, F.; Ma, L.; Wang, Y.; Zhang, L. Synergistic effect of 14-alpha-lipoyl andrographolide and various antibiotics on the formation of biofilms and production of exopolysaccharide and pyocyanin by pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2011, 55, 3015–3017. [Google Scholar] [CrossRef]
- Kosuge, T. Structure of an antimicrobial substance isolated from houttuynia cordata thunb. J. Pharm. Sac. Jpn. 1952, 72, 12271–12275. [Google Scholar]
- Liu, G.; Xiang, H.; Tang, X.; Zhang, K.; Wu, X.; Wang, X.; Guo, N.; Feng, H.; Wang, G.; Liu, L.; et al. Transcriptional and functional analysis shows sodium houttuyfonate-mediated inhibition of autolysis in staphylococcus aureus. Molecules 2011, 16, 8848–8865. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhou, M.; Zhang, D.; Zhang, H.; Zhang, L. Immune response of aa broilers to ibv h120 vaccine and sodium new houttuyfonate. Res. Vet. Sci. 2010, 89, 373–377. [Google Scholar] [CrossRef]
- Pan, P.; Wang, Y.J.; Han, L.; Liu, X.; Zhao, M.; Yuan, Y.F. Effects of sodium houttuyfonate on expression of nf-kappab and mcp-1 in membranous glomerulonephritis. J. Ethnopharmacol. 2010, 131, 203–209. [Google Scholar] [CrossRef]
- Gao, J.P.; Chen, C.X.; Wang, Y.; Lu, J.; Gu, W.L. Effect of sodium houttuyfonate on myocardial hypertrophy in mice and rats. J. Pharm. Pharmacol. 2009, 61, 677–683. [Google Scholar]
- Ye, X.; Li, X.; Yuan, L.; Ge, L.; Zhang, B.; Zhou, S. Interaction of houttuyfonate homologues with the cell membrane of gram-positive and gram-negative bacteria. Colloids Surf. A Physicochem. Eng. Aspects 2007, 301, 412–418. [Google Scholar] [CrossRef]
- Johnson, D.; Jones, R. In-vitro activity of a combination of two oral beta-lactams (cefpodoxime and amoxycillin) against streptococcus pneumoniae isolates with reduced susceptibilities to penicillin. J. Antimicrob. Chemother. 1998, 42, 555–557. [Google Scholar] [CrossRef]
- Sauer, K.; Camper, A.K.; Ehrlich, G.D.; Costerton, J.W.; Davies, D.G. Pseudomonas aeruginosa displays multiple phenotypes during development as a biofilm. J. Bacteriol. 2002, 184, 1140–1154. [Google Scholar] [CrossRef]
- Evans, L.R.; Linker, A. Production and characterization of the slime polysaccharide of pseudomonas aeruginosa. J. Bacteriol. 1973, 116, 915–924. [Google Scholar]
- Simpson, J.A.; Smith, S.E.; Dean, R.T. Alginate inhibition of the uptake of pseudomonas aeruginosa by macrophages. J. Gen. Microbiol. 1988, 134, 29–36. [Google Scholar]
- Simpson, J.A.; Smith, S.E.; Dean, R.T. Scavenging by alginate of free radicals released by macrophages. Free Radic. Biol. Med. 1989, 6, 347–353. [Google Scholar] [CrossRef]
- Simpson, J.; Smith, S.; Dean, R. Alginate may accumulate in cystic fibrosis lung because the enzymatic and free radical capacities of phagocytic cells are inadequate for its degradation. Biochem. Mol. Biol. Int. 1993, 30, 1021–1034. [Google Scholar]
- Mann, E.E.; Wozniak, D.J. Pseudomonas biofilm matrix composition and niche biology. FEMS Microbiol. Rev. 2012, 36, 893–916. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, G.; Li, Y.; Liu, Y.; Song, Y.; Zheng, W.; Zhang, N.; Hu, X.; Yan, S.; Jia, J. In vitro interactions between aspirin and amphotericin b against planktonic cells and biofilm cells of candida albicans and c. Parapsilosis. Antimicrob. Agents Chemother. 2012, 56, 3250–3260. [Google Scholar] [CrossRef]
- O’Toole, G.A. Microtiter dish biofilm formation assay. J. Vis. Exp. 2011, 47, e2437. [Google Scholar]
- Sample Availability: Samples of Sodium Houttuyfonate are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Shao, J.; Cheng, H.; Wang, C.; Wang, Y. A Phytoanticipin Derivative, Sodium Houttuyfonate, Induces in Vitro Synergistic Effects with Levofloxacin against Biofilm Formation by Pseudomonas aeruginosa. Molecules 2012, 17, 11242-11254. https://doi.org/10.3390/molecules170911242
Shao J, Cheng H, Wang C, Wang Y. A Phytoanticipin Derivative, Sodium Houttuyfonate, Induces in Vitro Synergistic Effects with Levofloxacin against Biofilm Formation by Pseudomonas aeruginosa. Molecules. 2012; 17(9):11242-11254. https://doi.org/10.3390/molecules170911242
Chicago/Turabian StyleShao, Jing, Huijuan Cheng, Changzhong Wang, and Yan Wang. 2012. "A Phytoanticipin Derivative, Sodium Houttuyfonate, Induces in Vitro Synergistic Effects with Levofloxacin against Biofilm Formation by Pseudomonas aeruginosa" Molecules 17, no. 9: 11242-11254. https://doi.org/10.3390/molecules170911242
APA StyleShao, J., Cheng, H., Wang, C., & Wang, Y. (2012). A Phytoanticipin Derivative, Sodium Houttuyfonate, Induces in Vitro Synergistic Effects with Levofloxacin against Biofilm Formation by Pseudomonas aeruginosa. Molecules, 17(9), 11242-11254. https://doi.org/10.3390/molecules170911242



