Exposure to Anacardiaceae Volatile Oils and Their Constituents Induces Lipid Peroxidation within Food-Borne Bacteria Cells
Abstract
:1. Introduction
2. Results and Discussion
2.1. Essential Oil Chemical Compositions
| CONSTITUENT | RI * | A. humile | A. occidentale | A. fraxinifolium | M. urundeuva | S. terebinthifolius | |
|---|---|---|---|---|---|---|---|
| March | July | ||||||
| Hydrocarbon Monoterpenes | 29.9 | 4.7 | 67.0 | 91.0 | 30.9 | 46.6 | |
| α-Pinene | 937 | 22.0 ± 0.9 | - | 0.7 ± 0.0 | - | 1.2 ± 0.1 | 4.2 ± 0.1 |
| β-Pinene | 982 | 6.6 ± 1.3 | - | - | - | - | 0.8 ± 0.0 |
| Myrcene | 993 | - | - | 1.9 ± 0.1 | 4.2 ± 0.3 | 15.4 ± 0.9 | 0.8 ± 0.0 |
| δ-2-Carene | 1002 | - | - | 3.6 ± 0.1 | - | - | - |
| α-Phellandrene | 1004 | - | - | - | - | - | 18.2 ± 1.2 |
| δ-3-Carene | 1011 | - | - | - | 78.8 ± 1.7 | - | - |
| ρ-Cymene | 1026 | - | - | - | - | - | 3.3 ± 0.2 |
| Limonene | 1031 | 1.3 ± 0.1 | 4.7 ± 0.3 | 0.4 ± 0.0 | 0.2 ± 0.0 | 12.0 ± 0.6 | 16.7 ± 1.1 |
| β-Phellandrene | 1032 | - | - | - | 3.0 ± 0.2 | - | - |
| ( Z)-β-Ocimene | 1040 | - | - | 1.1 ± 0.1 | - | - | - |
| ( E)-β-Ocimene | 1050 | - | - | 44.1 ± 0.8 | - | 2.3 ± 0.2 | 2.6 ± 0.1 |
| α-Terpinolene | 1089 | - | - | 15.2 ± 0.9 | 4.8 ± 0.6 | - | - |
| Hydrocarbon Sesquiterpenes | 60.9 | 80.7 | 19.0 | 8.1 | 59.4 | 35.4 | |
| δ-Elemene | 1339 | - | - | - | - | 2.4 ± 0.1 | 2.0 ± 0.0 |
| α-Copaene | 1376 | 2.5 ± 0.3 | 10.3 ± 0.9 | 0.4 ± 0.0 | - | 1.7 ± 0.1 | 0.6 ± 0.0 |
| β-Elemene | 1391 | - | - | - | 0.7 ± 0.1 | 4.8 ± 0.3 | 2.1 ± 0.1 |
| α-Gurjunene | 1409 | 1.0 ± 0.2 | - | - | - | - | - |
| ( E)-Caryophyllene | 1418 | 31.0 ± 1.8 | 15.4 ± 1.5 | - | 1.1 ± 0.1 | 14.7 ± 0.8 | 2.7 ± 0.2 |
| Aromadendrene | 1439 | 1.1 ± 0.4 | 1.1 ± 0.2 | 5.6 ± 0.2 | - | - | - |
| α-Humulene | 1454 | 2.9 ± 0.3 | 1.5 ± 0.1 | - | - | 2.5 ± 0.1 | 0.9 ± 0.0 |
| Alloaromadendrene | 1461 | 1.4 ± 0.2 | 2.5 ± 0.7 | 0.5 ± 0.0 | - | - | - |
| β-Camigrene | 1475 | - | 1.0 ± 0.2 | - | 0.5 ± 0.0 | 7.5 ± 1.0 | - |
| Germacrene-D | 1480 | 5.9 ± 1.7 | 11.5 ± 1.2 | 1.4 ± 0.1 | - | 8.8 ± 0.3 | 21.0 ± 1.2 |
| β-Selinene | 1485 | 0.7 ± 0.0 | 1.9 ± 0.3 | - | 2.5 ± 0.2 | 4.3 ± 0.1 | - |
| α-Amorphene | 1485 | - | 3.0 ± 0.2 | 0.5 ± 0.1 | - | - | - |
| Viridiflorene | 1493 | - | - | 9.0 ± 0.4 | 3.0 ± 0.2 | - | - |
| Bicyclogermacrene | 1494 | 7.6 ± 1.2 | 8.2 ± 0.5 | - | - | - | - |
| δ-Selinene | 1495 | - | 1.7 ± 0.2 | - | - | - | - |
| α-Muurolene | 1499 | 1.2 ± 0.4 | 2.3 ± 0.1 | - | - | 2.6 ± 0.1 | 0.9 ± 0.0 |
| Germacrene-A | 1503 | 0.6 ± 0.1 | - | - | 0.3 ± 0.0 | 2.1 ± 0.0 | 1.3 ± 0.0 |
| ( E,E)-α-Farnesene | 1508 | - | - | 0.8 ± 0.1 | - | - | - |
| γ-Cadinene | 1513 | 0.9 ± 0.2 | 2.0 ± 0.1 | 0.8 ± 0.0 | - | 2.3 ± 0.1 | 0.7 ± 0.0 |
| δ-Cadinene | 1524 | 4.1 ± 0.2 | 9.3 ± 0.7 | - | - | 3.6 ± 0.1 | 1.6 ± 0.1 |
| Germacrene B | 1556 | - | 7.3 ± 1.1 | - | - | 2.1 ± 0.1 | 1.6 ± 0.0 |
| Oxygenated Sesquiterpenes | 6.3 | 3.8 | 11.6 | - | 5.8 | 15.5 | |
| Ledol | 1565 | - | - | 0.5 ± 0.0 | - | - | - |
| Spathulenol | 1576 | - | - | 5.7 ± 0.3 | - | 1.1 ± 0.0 | 2.1 ± 0.1 |
| β-Caryophyllene oxide | 1581 | 0.6 ± 0.0 | - | - | - | 0.8 ± 0.0 | 2.6 ± 0.1 |
| Globulol | 1583 | 1.4 ± 0.4 | - | 3.2 ± 0.1 | - | - | - |
| Epiglobulol | 1588 | 1.8 ± 0.2 | - | - | - | - | - |
| Viridiflorol | 1590 | 1.4 ± 0.3 | - | 2.2 ± 0.1 | - | - | 2.5 ± 0.2 |
| δ-Cadinol | 1636 | - | 1.5 ± 0.1 | - | - | 1.3 ± 0.1 | 2.4 ± 0.1 |
| α-Muurolol | 1645 | - | - | - | - | 1.2 ± 0.1 | 2.8 ± 0.1 |
| α-Cadinol | 1653 | 1.1 ± 0.2 | 2.3 ± 0.2 | - | - | 1.4 ± 0.0 | 3.1 ± 0.1 |
| Others | - | 7.2 | - | - | - | - | |
| Tetradecane | 1399 | - | 1.7 ± 0.1 | - | - | - | - |
| Hexadecanoic acid | 1984 | - | 7.2 ± 1.3 | - | - | - | - |
| Total | 97.7 | 96.3 | 97.9 | 99.1 | 96.1 | 97.5 | |
| Yield (%) | 0.3±0.0 | 0.3 ± 0.0 | 0.7 ± 0.1 | 0.6 ± 0.0 | 0.6 ± 0.1 | 0.7 ± 0.1 | |
2.2. Antibacterial Activity of the Essential Oils
| Essential oils | Inhibition Zones Diameter (mm) * | ||
|---|---|---|---|
| Gram-negative | Gram-positive | ||
| E. coli | B. cereus | S. aureus | |
| A.humile | 7 B c | 14 A c | 10 B d |
| A. occidentale | 8 A c | 7 A d | 8 A d |
| A. fraxinifolium | 11 B b | 23 A b | 13 B c |
| M. urundeuva | 14 B b | 22 A b | 22 A b |
| S. terebinthifolius (March) | 6 B c | 15 A c | 14 A c |
| S. terebinthifolius (July) | 14 B b | 25 A b | 20 A b |
| Control (H2O) | 6 A c | 6 A d | 6 A d |
| Chloramphenicol 30 µg | 29 B a | 29 B a | 32 A a |
| Essential Oils/Constituent | Minimum inhibitory Concentration (g L−1) | ||
|---|---|---|---|
| Gram-negative | Gram-positive | ||
| E. coli | B. cereus | S. aureus | |
| S. terebinthifolius (March) | 0.63 | 2.50 | 1.25 |
| S. terebinthifolius (July) | 0.16 | 1.25 | 0.31 |
| A. fraxinifolium | 0.31 | 1.25 | 0.31 |
| M. urundeuva | 0.31 | 0.63 | 0.31 |
| δ-3-Carene | 0.16 | 0.16 | 0.16 |
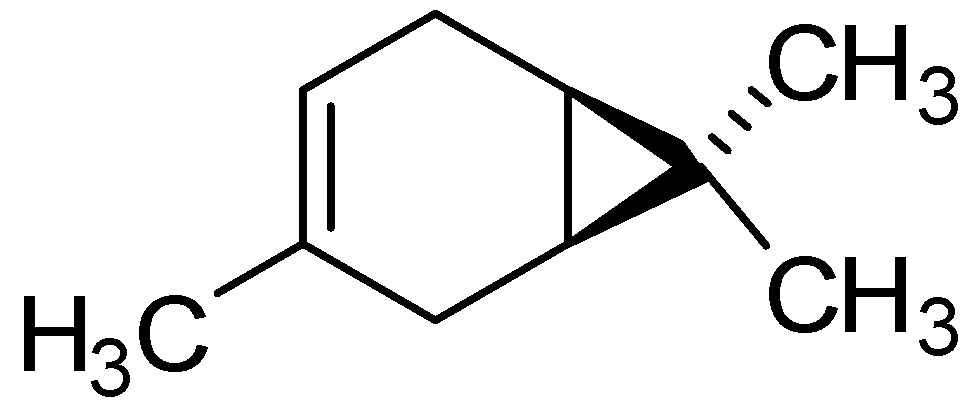
2.3. Lipid Peroxidation within Bacterial Cells
| Essential Oils/Constituents | MDA-TBA2 Concentration (nmol mg−1 Prot) | ||
|---|---|---|---|
| Gram-negative | Gram-positive | ||
| E. coli | B.cereus | S. aureus | |
| S. terebinthifolius (March) | 2.16 A b | 1.85 B b | 1.67 B b |
| S. terebinthifolius (July) | 3.34 A a | 2.14 B b | 2.22 B b |
| A. fraxinifolium | 2.46 A b | 1.51 B b | 1.84 B b |
| M. urundeuva | 2.74 A b | 2.97 A a | 3.21 A a |
| δ-3-Carene | 1.33 B c | 1.92 A b | 1.71 A b |
| Control | 0.69 A d | 0.95 A c | 0.73 A c |
3. Experimental
3.1. Plant Material
3.2. Essential Oil Extraction
3.3. Chemical Analysis of the Essential Oil Extraction—GC-FID and GC-MS
3.4. Antibacterial Assays
3.5. Determinations of Lipid Peroxidation Levels
4. Conclusions
Acknowledgments
References
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef]
- Al-Reza, S.M.; Rahman, A.; Lee, J.; Kang, S.C. Potential roles of essential oil and organic extracts of Zizyphus jujuba in inhibiting food-borne pathogens. Food Chem. 2010, 119, 981–986. [Google Scholar] [CrossRef]
- Roitman, J.N.; Merrill, G.B.; Beck, J.J. Survey of ex situ fruit and leaf volatiles from several Pistacia cultivars grown in California. J. Sci. Food Agric. 2011, 91, 934–942. [Google Scholar]
- Dragull, K.; Beck, J.J.; Merrill, G.B. Essential oil yield and composition of Pistacia vera ‘Kerman’ fruits, peduncles and leaves grown in California. J. Sci. Food Agric. 2010, 90, 664–668. [Google Scholar]
- Pandit, S.S.; Kulkarni, R.S.; Chidley, H.G.; Giri, A.P.; Pujari, K.H.; Köllner, T.G.; Degenhardt, J.; Gershenzon, J.; Gupta, V.S. Changes in volatile composition during fruit development and ripening of ‘Alphonso’ mango. J. Sci. Food Agric. 2009, 89, 2071–2081. [Google Scholar] [CrossRef]
- Tassou, C.C.; Nychas, G.J.E. Antimicrobial activity of the essential oil of mastic gum (Pistacia Zentiscus var. chia) on Gram-positive and Gram-negative bacteria in broth and in model food system. Int. Biodeter. Biodegr. 1995, 36, 411–420. [Google Scholar] [CrossRef]
- El-Massry, K.F.; El-Ghorab, A.H.; Shaaban, H.A.; Shibamoto, T. Chemical compositions and antioxidant/antimicrobial activities of various samples prepared from Schinus terebinthifolius leaves cultivated in Egypt. J. Agric. Food Chem. 2009, 57, 5265–5270. [Google Scholar]
- Erazo, S.; Delporte, C.; Negrete, R.; García, R.; Zaldívar, M.; Iturra, G.; Caballero, E.; López, J.L.; Backhouse, N. Constituents and biological activities of Schinus polygamus. J. Ethnopharmacol. 2006, 107, 395–400. [Google Scholar]
- Hayouni, E.A.; Chraief, I.; Abedrabba, M.; Bouix, M.; Leveau, J.I.; Mohammed, H.; Hamdi, M. Tunisian Salvia officinalis L. and Schinus molle L. essential oils: Their chemical compositions and their preservative effects against Salmonella inoculated in minced beef meat. Int. J. Food Microbiol. 2008, 125, 242–251. [Google Scholar] [CrossRef]
- Guinoiseau, E.; Luciani, A.; Rossi, P.G.; Quilichini, Y.; Ternengo, S.; Bradesi, P.; Berti, L. Cellular effects induced by Inula graveolens and Santolina corsica essential oils on Staphylococcus aureus. Eur. J. Clin. Microbiol. Infect Dis. 2010, 29, 873–879. [Google Scholar]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef]
- Helander, I.M.; Mattila-Sandholm, T. Fluorometric assessment of Gram-negative bacterial permeabilization. J. Appl. Microbiol. 2000, 88, 213–219. [Google Scholar]
- Ultee, A.; Bennik, M.H.J.; Moezelaar, R. The phenolic hydroxyl group of carvacrol is essential for action against the food-borne pathogen Bacillus cereus. Appl. Environ. Microbiol. 2002, 68, 1561–1568. [Google Scholar]
- Cox, S.D.; Mann, C.M.; Karkham, J.L.; Bell, H.C.; Gustafson, J.E.; Warmington, J.R.; Wyllie, S.G. The mode of antimicrobial action of the essential oil of Melaleuca alternifolia (tea tree oil). J. Appl. Microbiol. 2000, 88, 170–175. [Google Scholar]
- Lambert, R.J.W.; Skandamis, P.N.; Coote, P.; Nychas, G.J.E. A study of the minimum inhibitory concentration and mode of action of oregano essential oil, thymol and carvacrol. J. Appl. Microbiol. 2001, 91, 453–462. [Google Scholar]
- Silva, C.J.; Barbosa, L.C.A.; Demuner, A.J.; Montanari, R.M.; Pinheiro, A.L.; Dias, I.; Andrade, N.J. Chemical composition and antibacterial activities from the essential oils of Myrtaceae species planted in Brazil. Quim. Nova 2010, 33, 104–108. [Google Scholar] [CrossRef]
- Silva, C.J.; Barbosa, L.C.A.; Maltha, C.R.A.; Pinheiro, A.L.; Ismail, F.M.D. Comparative study of the essential oils of seven Melaleuca (Myrtaceae) species grown in Brazil. Flavour Fragr. J. 2007, 22, 474–478. [Google Scholar] [CrossRef]
- Martins, F.T.; Santos, M.H.; Polo, M.; Barbosa, L.C.A. Chemical variation in the essential oil of Hyptis suaveolens (L.) Poit, under cultivation conditions. Quim. Nova 2006, 29, 1203–1209. [Google Scholar] [CrossRef]
- Silva, F.; Santos, R.H.S.; Andrade, N.J.; Barbosa, L.C.A.; Casali, V.W.D.; Lima, R.R.; Passarinho, R.V.M. Basil conservation affected by cropping season, harvest time and storage period. Pesq. Agropec. Bras. 2005, 40, 323–328. [Google Scholar]
- Winck, C.R.; Cardoso, C.A.L.; Jeller, A.H.; Re-Poppi, N.; Coelho, R.M.; Schleder, E.J.D. Identification of the Volatile Compounds of Leaf Oil of Anacardium humile (Anacardiaceae). J. Essent. Oil Res. 2010, 22, 11–12. [Google Scholar] [CrossRef]
- Castro, H.G.; Oliveira, L.O.; Barbosa, L.C.A.; Ferreira, F.A.; Silva, D.J.H.; Mosquim, P.R.; Nascimento, E.A. Conttent and composition of the essential oil of mentrasto (Ageratum conyzoides). Quim. Nova 2004, 27, 55–57. [Google Scholar]
- Martins, E.R.; Casali, V.W.D.; Barbosa, L.C.A.; Carazza, F. Essential oil in the taxonomy of Ocimum selloi Benth. J. Braz. Chem. Soc. 1997, 8, 29–32. [Google Scholar]
- Maia, J.G.S.; Andrade, E.H.A.; Zoghbi, M.G.B. Volatile Constituents of the Leaves, Fruits and Flowers of Cashew (Anacardium occidentale L.). J. Food Compos. Anal. 2000, 13, 227–232. [Google Scholar]
- Dzamic, A.; Gbolde, A.; Ristic, M.; Marin, P.D. Essential oil composition of Anacardium occidentale from Nigeria. Chem. Nat. Compd. 2009, 45, 441–442. [Google Scholar] [CrossRef]
- Maia, J.G.S.; Silva, M.H.L.; Andrade, E.H.A.; Zoghbi, M.D.B.; Carreira, L.M.M. Essential oils from Astronium urundeuva (Allemão) Engl. and A. fraxinifolium Schott ex Spreng. Flavour Fragr. J. 2002, 17, 72–74. [Google Scholar]
- Barbosa, L.C.A.; Demuner, A.J.; Clemente, A.D.; Paula, V.F.; Ismail, F.M.D. Composition of volatile oils from Schinus terebinthifolius Raddi. Quim. Nova 2007, 30, 1959–1965. [Google Scholar]
- Martins, F.T.; Santos, M.H.; Polo, M.; Barbosa, L.C.A. Effects of the interactions among macronutrients, plant age and photoperiod in the composition of Hyptis suaveolens (L.) Poit essential oil from Alfenas (MG), Brazil. Flavour Fragr. J. 2007, 22, 123–129. [Google Scholar] [CrossRef]
- Fonsceca, M.C.M.; Barbosa, L.C.A.; Nascimento, E.A.; Casali, V.W.D. Essential oil from leaves and flowers of Porophyllum ruderale (Jacq.) Cassini (Asteraceae). J. Essent. Oil Res. 2006, 18, 345–347. [Google Scholar] [CrossRef]
- Kamada, T.; Casali, V.W.D.; Barbosa, L.C.A.; Fortes, I.C.P.; Finger, F.L. Phenotypic plasticity of the essential oil in Basil accesses (Ocimum basilicum L.). Braz. J. Med. Plants 1999, 1, 13–22. [Google Scholar]
- Shimizu, M.T.; Bueno, L.J.F.; Rodrigues, R.F.O.; Sallowicz, F.A.; Sawaya, A.C.H.F.; Marques, M.O.M. Essential oil of Lithraea molleoides (vell.): Chemical composition and Antimicrobial activity. Braz. J. Microbiol. 2006, 37, 556–560. [Google Scholar]
- Lastbom, A.; Boman, A.; Johnsson, S.; Camner, P.; Ryrfeldt, A. Increased airway responsiveness of a common fragrance component, 3-carene, after skin sensitization—A study in isolated guinea pig lungs. Toxicol. Lett. 2003, 145, 189–196. [Google Scholar] [CrossRef]
- Lastbom, L.; Boman, A.; Camner, P.; Ryrfeldt, A. Increased airway responsiveness after skin sensitization to 3-carene, studied in isolated guinea pig lungs. Toxicology 2000, 147, 209–214. [Google Scholar]
- Lastbom, L.; Boman, A.; Camner, P.; Ryrfeldt, A. Does airway responsiveness increase after skin sensitisation to 3-carene: A study in isolated guinea pig lungs. Toxicology 1998, 125, 59–66. [Google Scholar] [CrossRef]
- Johansson, A.; Lundborg, M. Effects of low concentrations of 3-carene on alveolar macrophages in vitro. Toxicology 1997, 120, 99–104. [Google Scholar] [CrossRef]
- Del’ Rio, D.; Stewart, A.J.; Pellegriniet, N. A review of recent studies on malondialdehyde as toxic molecule and biological marker of oxidative stress. Nutr. Metab. Cardiovas. 2005, 15, 316–328. [Google Scholar]
- Marnett, L.J. Lipid peroxidation—DNA damage by malondialdehyde. Mutat. Res.-Fund. Mol. Mech. 1999, 424, 83–95. [Google Scholar]
- Yoon, S.J.; Park, J.E.; Yang, J.H.; Park, J.W. OxyR Regulon Controls Lipid Peroxidation-mediated Oxidative Stress in Escherichia coli. J. Biochem. Mol. Biol. 2002, 35, 297–301. [Google Scholar]
- Lyon, D.Y.; Alvarez, P.J.J. Fullerene Water Suspension (nC60) Exerts Antibacterial Effects via ROS-Independent Protein Oxidation. Environ. Sci. Technol. 2008, 42, 8127–8132. [Google Scholar]
- Carson, C.F.; Mee, B.J.; Riley, T.V. Mechanism of action of Melaleuca alternifolia (tea tree) oil on Staphylococcus aureus determined by time-kill, lysis, leakage and salt tolerance assays and electron microscopy. Antimicrob. Agents Chemother. 2002, 46, 1914–1920. [Google Scholar]
- Sikkema, J.; de Bont, J.A.M.; Poolman, B. Mechanisms of membrane toxicity of hydrocarbons. Microbiol. Rev. 1995, 59, 201–222. [Google Scholar]
- Wang, Y.W.; Zeng, W.C.; Xu, P.Y.; Lan, Y.J.; Zhu, R.X.; Zhong, K.; Huang, Y.N.; Gao, H. Chemical composition and antibacterial activity of the essential oil of Kumquat (Fortunella crassifolia Swingle) peel. Int. J. Mol. Sci. 2012, 13, 3382–3393. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oils by Gas Chromatography/Mass Spectroscopy, 4th ed; Allured Publishing Corporation: Carol Stream, IL, USA, 2007. [Google Scholar]
- Salustiano, V.C.; Andrade, N.J.; Soares, N.F.F.; Lima, J.C.; Bernardes, P.C.; Luiz, L.M.P.; Fernandes, P.E. Contamination of milk with Bacillus cereus by post-pasteurization surface exposure as evaluated by automated ribotyping. Food Control 2009, 20, 439–442. [Google Scholar]
- National Committee for Clinical Laboratory Standards (NCCLS), Performance Standards for Antimicrobial Disk Susceptibility Tests, Approved Standard, 8th ed; NCCLS: Wayne, PA, USA, 2003; NCCLS document M2-A8 [ISBN 1–56238-485-6].
- GENES Software. Version 2007.0.0. Federal University of Viçosa: Viçosa, MG, Brazil.
- National Committee for Clinical Laboratory Standards (NCCLS), Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically, Approved Standard, 6th ed; NCCLS: Wayne, PA, USA, 2003; NCCLS document M7-A6 [ISBN 1–56238-486-4].
- Bradford, M.M. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar]
- Sample Availability: Samples of the oils are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Montanari, R.M.; Barbosa, L.C.A.; Demuner, A.J.; Silva, C.J.; Andrade, N.J.; Ismail, F.M.D.; Barbosa, M.C.A. Exposure to Anacardiaceae Volatile Oils and Their Constituents Induces Lipid Peroxidation within Food-Borne Bacteria Cells. Molecules 2012, 17, 9728-9740. https://doi.org/10.3390/molecules17089728
Montanari RM, Barbosa LCA, Demuner AJ, Silva CJ, Andrade NJ, Ismail FMD, Barbosa MCA. Exposure to Anacardiaceae Volatile Oils and Their Constituents Induces Lipid Peroxidation within Food-Borne Bacteria Cells. Molecules. 2012; 17(8):9728-9740. https://doi.org/10.3390/molecules17089728
Chicago/Turabian StyleMontanari, Ricardo M., Luiz C. A. Barbosa, Antonio J. Demuner, Cleber J. Silva, Nelio J. Andrade, Fyaz M. D. Ismail, and Maria C. A. Barbosa. 2012. "Exposure to Anacardiaceae Volatile Oils and Their Constituents Induces Lipid Peroxidation within Food-Borne Bacteria Cells" Molecules 17, no. 8: 9728-9740. https://doi.org/10.3390/molecules17089728




