1. Introduction
Eurycomalongifolia Jack, from the Simaroubaceae family and identified locally as ‘Tongkat Ali’ or ‘Pasakbumi’ has been commonly prescribed in traditional medicine as a febrifuge and a remedy for dysentery, glandular swelling and fever [
1,
2].
E. longifolia is found in primary and secondary, evergreen and mixed deciduous forests in Burma, Indochina, Thailand, Malaysia, Sumatra, Borneo and The Philippines. It is popularly sought after as a single or an essential component for the treatment of fevers, aches, sexual insufficiency and also as a health supplement. Traditional medicinal users usually take a decoction of the roots in water as a health tonic and anti-stress remedy. Extracts derived from the roots of this plant were also found to demonstrate activity when evaluated with the sarcoma 180 model [
3]. Moreover, anti-malarial [
4,
5,
6,
7,
8] and cytotoxic [
9,
10,
11] activities were also reported being linked to the presence of quassinoids, squalene derivatives, biphenyl-neolignans, tirucallane-type triterpenes, canthine-6-one and carboline alkaloids. Specially, three quassinoids, eurycomanone, its 13 α-(21)-epoxy and 13,21-dihydro analogues were identified as having greater anti-plasmodial activity [
12].
One of the common infections in tropical and subtropical climates is toxoplasmosis caused by
Toxoplasma gondii. It is one of the most widespread protozoan parasites, chronically infecting approximately 30% of the global human population [
13].
T. gondii causes severe neurological deficits in immunosuppressed patients (such as those with AIDS) and lymphadenopathy in healthy adults. It can cross the placenta (generally in women with no or low antibody levels) and cause congenital infections characterized by intra-cerebral calcifications, chorioretinitis, hydrocephaly or microcephaly, and convulsions [
14]. From this perspective, new agents from natural resources that can inhibit the growth of
T. gondii are greatly needed and would enhance the effectiveness of therapy. Our previous study showed that two active fractions of
E. longifolia root, namely TAF355 and TAF401, possessed a good antiparasitic activity against
T. gondii with IC
50 values of 1.125 µg/mL and 1.375 µg/mL, respectively [
15]. The median inhibition concentration (IC
50) value refers to the concentration of the fraction necessary to inhibit at 50% of the control values. The present investigation demonstrates the antiparasitic activity of the
E. longifolia root fractions at the ultra-structural level through TEM and SEM observations at IC
50 concentration of the fractions. Advances in microscopy technique to observed at ultrastructural level of cells morphology enhancing understanding of
in situ antiparasitic activity observation. The microscopy method enables
in situ observations of the effect of anti-parasitic agents on the organisms [
16]. In this study TEM and SEM technique was used to observe the suppression of
T. gondii growth by clindamycin (positive control), TAF 355 and TAF 401.
2. Results and Discussion
The rapidly increasing prevalence of toxoplasma encephalitis in AIDS patients has highlighted the need for more effective therapies and for alternative drugs for the considerable number of patients (up to 60%) who develop allergic reactions or serious side effects during standard therapy with a sulfonamide and trimethoprim or pyrimethamine [
17]. Therefore, the anti-parasitic activity and prevention of
T. gondii infection by local herbal products were emphasized in our present work for the reasons that the management of
T. gondii infections faces a number of problems, including a limited number of effective anti-
T. gondii agents; toxicity of the available drugs; resistance of
T. gondii to commonly used drugs and lack of cost effective anti-
T. gondii agents. Among the different strategies which have been identified to defeat drug resistance, the investigation of new and effective natural products exhibiting antiparasitic activity may possibly play an important role to overcome drug resistance and the side effects.
In this paper, we were especially interested in the morphogenesis of the
T. gondii cells challenged by active fractions from roots of
E. longifolia.
In vitro observation of anti-
T. gondii activity of the active fractions from root of
E. longifolia, was further evaluated in this study by
in situ microscopic methods to examine the possible mechanisms of action in
T. gondii. The morphological observation of
T. gondii cells treated with plant extracts by using electron microscope is considered a gold standard technique to study the
in situ real time anti-
T. gondii activity. The SEM and TEM techniques are useful compared to several other microscopic methods as they are three-dimensional and almost the whole cell of the specimen is sharply focused [
16]. Since the SEM and TEM allow visualization of surface phenomena under high magnification from a three-dimensional perspective, in this study, SEM and TEM observations were utilized to study the anti-
T. gondii activity of active fractions from roots of
E. longifolia.
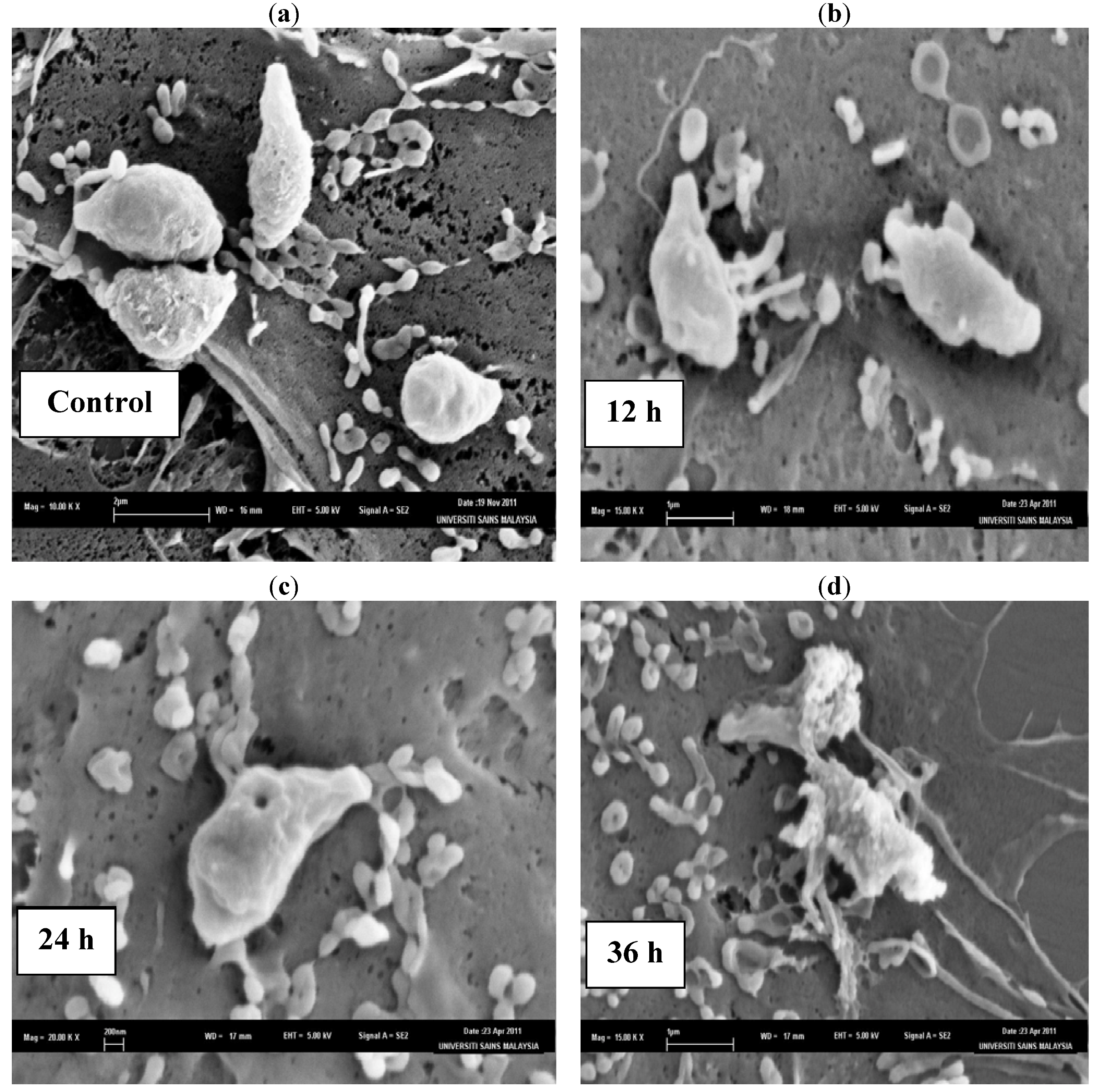
The SEM photomicrographs of the untreated and treated cells of
T. gondii at various times of exposure to clindamycin are shown in
Figure 1. Untreated cells (
Figure 1a) showed crescent-shaped cells and possess two differently structured poles; one is more round, the other is more pointed and probably the site of the conoid. The surface of the untreated tachyzoites is smooth, regular and homogeneous. After 12 h of exposure (
Figure 1b), a mild effect of the clindamycin on
T. gondii was observed, with shape irregularities compared to the untreated control cells. The 24 h treated cells (
Figure 1c) show distorted cell membranes as compared to the normal
T. gondii cells, followed by collapsed cells. After 36 h of exposure (
Figure 1d), completely collapsed and cavitated cells with cytoplasmic eruption were seen. It was believed that at this stage, the cells had lost their metabolic functions completely.
The SEM photomicrographs of the untreated and treated cells of
T. gondii at various times of exposure to TAF355 are shown in
Figure 2. Untreated cells (
Figure 2a) showed crescent-shaped cells and possess two differently structured poles; one is more round, the other is more pointed and probably the site of the conoid. The surface of the untreated tachyzoites is smooth, regular and homogeneous. After 12 h of exposure (
Figure 2b), a mild effect of the TAF355 and elongated cells of
T. gondii were observed with irregular shapes compared to the untreated control cells. The 24 h treated cells (
Figure 2c) show distorted cell membranes with the formation of invaginations as compared to the normal
T. gondii cells. After 36 h of exposure (
Figure 2d), completely collapsed cells with cytoplasmic eruption were observed. It was believed that at this stage, the cells had lost their metabolic functions completely. Huge amounts of vesicular material were equally found scattered in between the
T. gondii cells, was most likely derived from broken cells and presented membrane-limited cytoplasmic residues.
Figure 1.
SEM micrograph of the untreated and clindamycin treated cells of T. gondii after different time intervals. (a) Untreated cells, (b) 12 h of exposure, (c) 24 h treated cells, (d) 36 h of exposure.
Figure 1.
SEM micrograph of the untreated and clindamycin treated cells of T. gondii after different time intervals. (a) Untreated cells, (b) 12 h of exposure, (c) 24 h treated cells, (d) 36 h of exposure.
Figure 2.
SEM micrograph of the untreated and TAF355 treated cells of T. gondii after different time intervals. (a) Untreated cells, (b) 12 h of exposure, (c) 24 h treated cells, (d) 36 h of exposure.
Figure 2.
SEM micrograph of the untreated and TAF355 treated cells of T. gondii after different time intervals. (a) Untreated cells, (b) 12 h of exposure, (c) 24 h treated cells, (d) 36 h of exposure.
The SEM photomicrographs of the untreated and treated cells of
T. gondii at various times of exposure to TAF401 are shown in
Figure 3. Untreated cells (
Figure 3a) showed crescent shapes and possess two differently structured poles; one is more round, the other is more pointed and probably the site of the conoid. The surface of the untreated tachyzoites is smooth, regular and homogeneous. After 12 h of exposure to TAF401 (
Figure 3b), a mild effect with irregular
T. gondii cell shapes was observed, compared to the untreated control cells. The 24 h treated cells (
Figure 3c) had a rough appearance with holes compared to the untreated control cells, with the formation of invaginations. After 36 h of exposure to TAF401 (
Figure 3d), severe alterations of the cell wall with the formation of invaginations were seen and followed by the complete collapse of the cells. The
T. gondii cells lost their symmetrical appearance, they were completely deformed, and deep wrinkles were observed. Cell wall invaginations were also noticed. Leakage of cellular content could be noticed.
Figure 3.
SEM micrograph of the untreated and TAF401 treated cells of T. gondii for different time interval. (a) Untreated cells, (b) 12 h of exposure, (c) 24 h treated cells, (d) 36 h of exposure.
Figure 3.
SEM micrograph of the untreated and TAF401 treated cells of T. gondii for different time interval. (a) Untreated cells, (b) 12 h of exposure, (c) 24 h treated cells, (d) 36 h of exposure.
From the SEM findings, it can be suggested that the
T. gondii cells had undergone some distinct morphological and cytological alterations. Further evidence of these changes was obtained by TEM observations on similarly treated
T. gondii cells. As expected, the TEM observation also reaffirmed the disorganization of
T. gondii cells and destruction of their organelles. The TEM photomicrograph of the untreated cells of
T. gondii is shown in
Figure 4. As the fine structure of tachyzoites of
T. gondii has been reported by numerous investigators, only a brief description of the parasite is presented here. It showed a typically structured cytoplasm with various organelles. The tachyzoite is often crescent shaped, approximately 2 by 6 mm, with a pointed anterior (conoidal) end and a rounded posterior end. Ultrastructurally, the tachyzoite consists of various organelles and inclusion bodies including a pellicle (outer covering), apical rings, polar rings, conoid, rhoptries, micronemes, micropore, mitochondrion, subpellicular microtubules, endoplasmic reticulum, Golgi complex, ribosomes, rough and smooth endoplasmic reticula, micropore, nucleus, dense granules, amylopectin, granules (which may be absent), and a multiple-membrane-bound plastid-like organelle which has also been called a Golgi adjunct or apicoplast [
18].
Figure 4.
TEM micrograph of the control cells of T. gondii. M, mitochondrion; N, nucleus; LB, Lipid body.
Figure 4.
TEM micrograph of the control cells of T. gondii. M, mitochondrion; N, nucleus; LB, Lipid body.
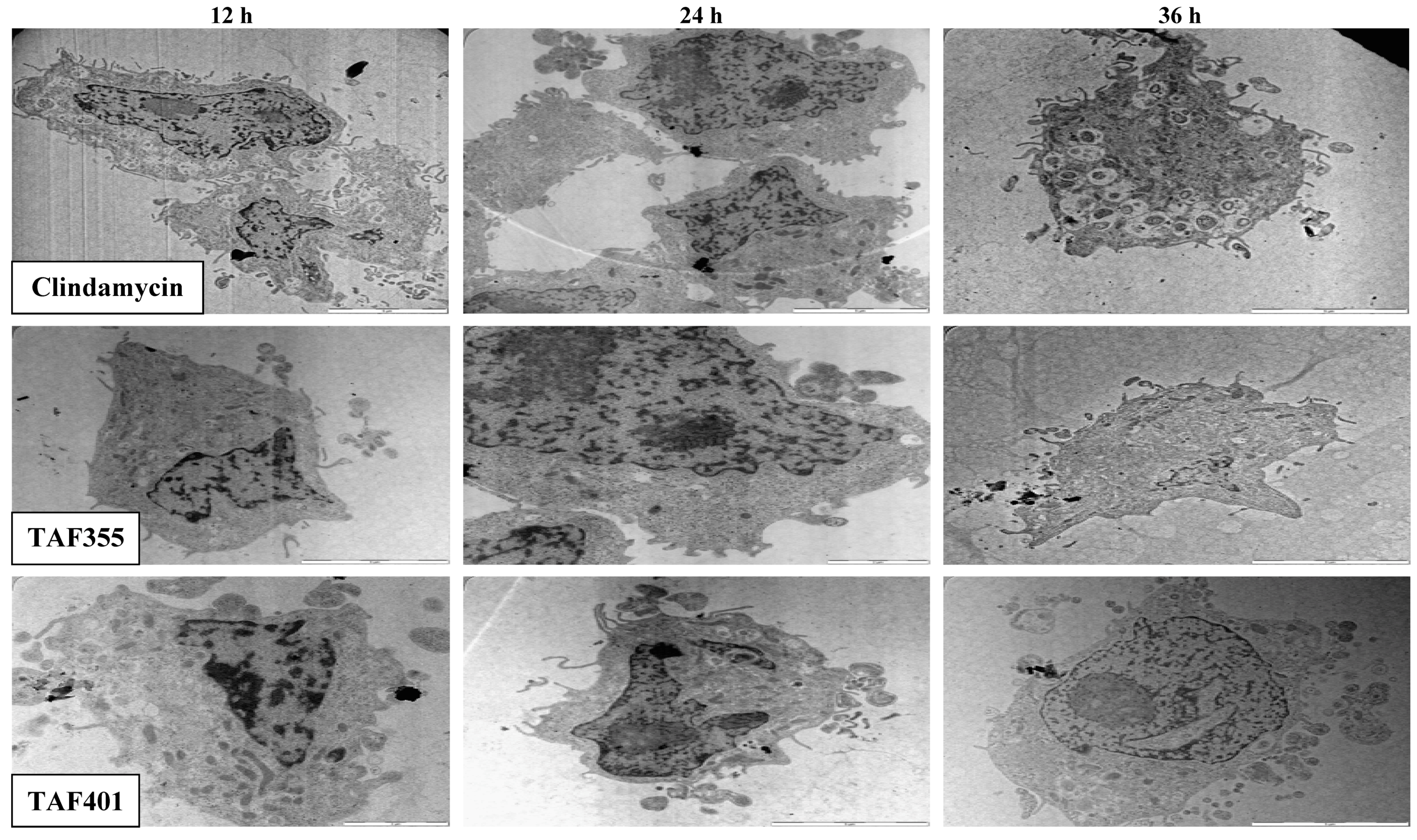
Figure 5 shows the TEM photomicrographs of the longitudinal and transverse sections of the clindamycin, TAF 355 and TAF 401 treated cells of
T. gondi at various time intervals. After 12, 24 and 36 h of exposure to clindamycin, TAF 355 and TAF 401, the cells exhibited notable alterations in the cell cytoplasm.
The cytoplasmic volume decreased, leaving a state of structural disorganization within the cell cytoplasm and destruction of its organelles. It shows that the parasite cells collapsed and lysed. Ultrastructural effects of clindamycin, TAF 355 and TAF 401 were detected as early as 12 h after the start of treatment. General views of thin sections showed completely destroyed parasites after treatment of infected cells with IC50 concentration of clindamycin, TAF 355 and TAF 401, even at 12 h of treatment. This implied that the TAF 355 and TAF 401 fractions have a significant effect on the T. gondii cells or their membranes as early as 12 h after treatment which indicated a rapid antiparasitic activity of these E. longifolia fractions. Of further interest are the inconsistent findings between SEM and TEM observations concerning the viability of the T. gondii cells, i.e., completely collapsed cells only observed after 36 h of treatment with SEM examination, contradictorily upon TEM examination it was found to be completely necrotic even at 12 h of treatment. This means that lytic changes in cell cytoplasm probably appear faster and do not affect the cell walls as observed in this study.
The microscopic examination of
T. gondii using SEM showed that the cells treated with TAF 355 and TAF 401 appeared irregular in shape with cell wall modifications and clear depressions on the cell surface with holes. Also, TEM observation showed irregular cell shape, unclear periplasm and dense cytoplasm without differentiated features. Such modifications may be related to the interference of the
E. longifolia active fractions with enzymatic reactions of metabolism activities of
T. gondi which affects parasite morphogenesis and growth, which deserves further research. Actually, the antiparasitic action of
E. longifolia active fractions may happen in two steps. The first step involves the passive entry of the active fractions into the plasma membrane in order to hit the metabolic activities of
T. gondi in the cytoplasm region. The second stage is the accumulation of
E. longifolia active fractions in the cytoplasm resulting in inhibition of cell growth. With regard to the mechanism of action for TAF 355 and TAF 401 against
T. gondii in the cytoplasm region, we hypothesize that TAF 355 and TAF 401 may produce intracellular oxidative stress by an indirect mechanism [
19]. Mitochondria are the largest source of reactive oxygen species (ROS) within cells [
20]. Moreover, uncontrolled superoxide flashes in mitochondria contribute to global oxidative stress, playing a key role in hypoxia/reoxygenation injury in cells [
21]. This model provides a rational explanation for why TAF 355 and TAF 401 inhibit
T. gondii growth.
Figure 5.
TEM micrographs of the treated treated cells of T. gondii for different time intervals.
Figure 5.
TEM micrographs of the treated treated cells of T. gondii for different time intervals.