NIR Spectroscopic Properties of Aqueous Acids Solutions
Abstract
:1. Introduction
- (1) Penetration of the radiation into sample is deeper than other ranges.
- (2) Water absorbance peaks are less strong and broad than in other ranges and the risk of masking spectral information correlated to low concentration constituents is low.
- (3) The cost of instrumentation for this range is relatively low, it is portable and suitable for process control and for in situ field measurements.
- (5) Lower absorbance at these wavelengths allows transmission optics.
- (6) Moreover, there is strong evidence, that the range from 700–900 nm constitutes a “diagnostic window” in which chemical compositions of samples can be investigated.
| Fruits | Acids | Fruits | Acids |
|---|---|---|---|
| Apples | Malic, Citric | Limes | Citric, Malic, Tartaric, Oxalic |
| Apricots | Malic, Citric | Nectarine | Malic |
| Avocados | Tartaric | Orange Peel | Malic, Citric, Oxalic |
| Bananas | Malic, Citric, Tartaric | Orange | Citric, Malic, Oxalic |
| Blackberries | Malic, Citric, Oxalic | Passionfruit | Malic |
| Blueberries | Citric, Malic | Peaches | Malic, Citric |
| Cherries | Malic, Citric, Tartaric | Pears | Malic, Citric, Tartaric, Oxalic |
| Cranberries | Citric, Malic | Pineapples | Citric, Malic |
| Grapefruit | Citric, Tartaric, Malic, Oxalic | Plums | Malic, Tartaric, Oxalic |
| Grapes | Malic, Tartaric, Citric, Oxalic | Quinces | Malic |
| Kiwifruit | Citric | Strawberries | Citric, Malic |
| Lemons | Citric, Malic, Tartaric, Oxalic | Tangerine | Citric |
2. Materials and Methods
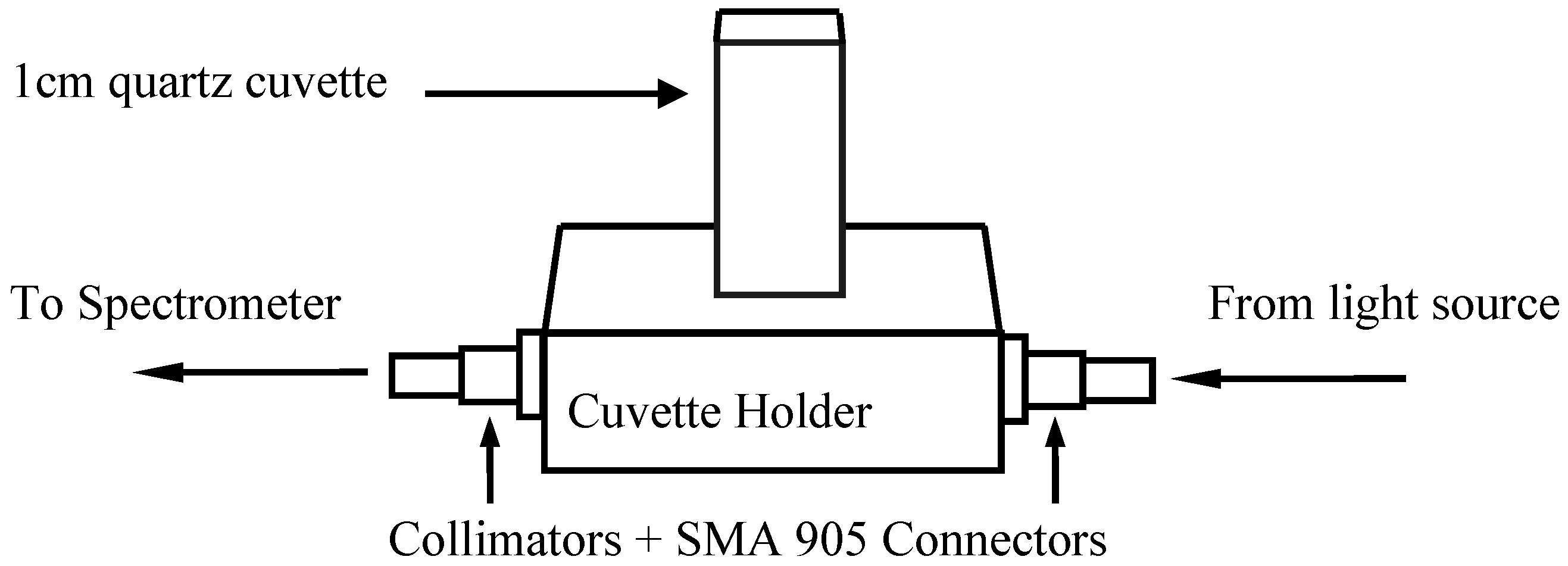
| Acids | Formula | Range (pH) | Mean | n (calibration) | n (prediction) |
|---|---|---|---|---|---|
| Citric | C6H8O7 | 1.88–4.32 | 2.88 | 50 | 50 |
| Tartaric | C4H6O6 | 2.06–4.52 | 3.12 | 50 | 50 |
| Malic | C4H6O5 | 1.99–4.44 | 3.17 | 50 | 50 |
| Oxalic | C2H2O4 | 1.95–4.35 | 3.16 | 50 | 50 |
3. Results and Analysis

- i. Low absorptive with considerable peak at certain wavelengths while accompanied by high scattering for all wavelengths.
- ii. High absorptive for all examined wavelengths with higher absorption peaks at certain wavelengths.






| Acids | ||||||||
|---|---|---|---|---|---|---|---|---|
| Wavelengths (nm) | Citric | Tartaric | Malic | Oxalic | ||||
| R2 | RMSE | R2 | RMSE | R2 | RMSE | R2 | RMSE | |
| 920, 975, 991 | 0.914 | 0.213 | 0.929 | 0.193 | 0.890 | 0.239 | 0.319 | 0.577 |
| 850, 975 | 0.924 | 0.198 | 0.902 | 0.224 | 0.880 | 0.248 | 0.452 | 0.511 |
| 850, 920, 975, 991 | 0.951 | 0.163 | 0.930 | 0.194 | 0.909 | 0.220 | 0.480 | 0.509 |
| 925, 990 | 0.909 | 0.217 | 0.946 | 0.166 | 0.880 | 0.250 | 0.217 | 0.612 |
| 900, 975, 990 | 0.918 | 0.209 | 0.925 | 0.199 | 0.945 | 0.170 | 0.378 | 0.551 |
| 925, 975, 990, 991, 993 | 0.919 | 0.212 | 0.957 | 0.153 | 0.887 | 0.249 | 0.561 | 0.473 |
| 700, 800, 900, 940, 975 | 0.927 | 0.201 | 0.929 | 0.197 | 0.961 | 0.145 | 0.673 | 0.408 |
| 700, 800, 900, 975 | 0.919 | 0.210 | 0.926 | 0.199 | 0.958 | 0.150 | 0.386 | 0.553 |
| 922, 975, 990, 993 | 0.920 | 0.209 | 0.938 | 0.183 | 0.883 | 0.250 | 0.420 | 0.538 |
| 700, 800, 900, 922, 975, 990, 993 | 0.928 | 0.204 | 0.948 | 0.172 | 0.969 | 0.133 | 0.465 | 0.535 |
| 755, 850, 975 | 0.931 | 0.192 | 0.907 | 0.220 | 0.880 | 0.250 | 0.869 | 0.253 |
| 918, 920, 975, 996 | 0.920 | 0.208 | 0.922 | 0.204 | 0.897 | 0.234 | 0.770 | 0.339 |
| 755, 850, 918, 920, 975, 996 | 0.945 | 0.177 | 0.923 | 0.208 | 0.917 | 0.215 | 0.908 | 0.219 |
4. Conclusions
Acknowledgements
- Sample Availability: Contact the authors.
References
- Nicolai, B.M.; Beullens, K.; Bobelyn, E.; Peirs, A.; Saeys, W.; Theron, K.I.; Lammertyn, J. Nondestructive measurement of fruit and vegetable quality by means of NIR spectroscopy: A review. Postharvest Biol. Technol. 2007, 46, 99–118. [Google Scholar] [CrossRef]
- Carlini, P.; Massantini, R.; Mencarelli, F. Measurement of Soluble Solids in Cherry and Apricot by PLS Regression and Wavelength Selection. J. Agric. Food Chem. 2000, 48, 5236–5242. [Google Scholar] [CrossRef]
- McGlone, V.A.; Kawano, S. Firmness, dry matter and soluble solids assessment of postharvest kiwifruit by NIR spectroscopy. Postharvest Biol. Technol. 1998, 13, 131–141. [Google Scholar] [CrossRef]
- Walsh, K.B.; Guthrie, J.A.; Burney, J.W. Application of commercially available, low-cost, miniaturized NIR spectrometers to the assessment of the sugar content of intact fruit. Aust. J. Plant Physiol. 2000, 27, 1175–1186. [Google Scholar]
- Osborne, B.G.; Fearn, T.; Hindle, P.H. Practical NIR spectroscopy with applications. In Food and Beverage Analysis; Osborne, B.G., Fearn, T., Hindle, P.H., Eds.; Longman: Essex, UK, 1993; pp. 11–35. [Google Scholar]
- Abu-Khalaf, N.; Bennedsen, B.; Bjørn, G. Distinguishing Carrot’s Characteristics by Near Infrared (NIR) Reflectance and Multivariate Data Analysis. Agric. Eng. Int. 2004, 6, 1–17. [Google Scholar]
- Cen, H.; He, Y. Theory and application of near infrared reflectance spectroscopy in determination of food quality. Trends Food Sci. Technol. 2007, 18, 72–83. [Google Scholar] [CrossRef]
- Isaksson, T.; Swensen, L.P.; Taylor, R.G.; Fjæra, S.O.; Skjervold, P.O. Non-destructive texture analysis of farmed Atlantic salmon using visual/near-infrared reflectance spectroscopy. J. Sci. Food Agric. 2001, 82, 53–60. [Google Scholar]
- Kelly, J.J.; Barlow, C.H.; Jinguji, T.M.; Callis, J.B. Prediction of gasoline octane numbers from near-infrared spectral features in the range 660–1215 nm. Anal. Chem. 1989, 61, 313–320. [Google Scholar]
- Bakier, S. Application of NIR Spectroscopy for the Analysis of Water-Carbohydrate Interactions in Water Solutions. Acta Agrophys. 2008, 11, 7–21. [Google Scholar]
- Giangiacomo, R. Study of water-sugar interactions at increasing sugar concentration by NIR spectroscopy. Food Chem. 2006, 96, 371–379. [Google Scholar] [CrossRef]
- Buning-Pfaue, H. Analysis of water in food by near infrared spectroscopy. Food Chem. 2003, 82, 107–115. [Google Scholar] [CrossRef]
- Rodriguez-Saona, L.E.; Fry, F.S.; McLaughlin, M.A.; Calvey, E.M. Rapid analysis of sugars in fruit juices by FT-NIR spectroscopy. Carbohydr. Res. 2001, 336, 63–74. [Google Scholar] [CrossRef]
- Berentsen, S.; Stolz, T.; Molt, K. Analysis of aqueous solutions by near-infrared spectrometry (NIRS) IV. One- and two-component systems of organic compounds in water. J. Mol. Struct. 1997, 410–411, 581–585. [Google Scholar]
- Rambla, F.J.; Garrigues, S.; de la Guardia, M. PLS-NIR determination of total sugar, glucose, fructose and sucrose in aqueous solutions of fruit juices. Anal. Chim. Acta 1997, 344, 41–53. [Google Scholar] [CrossRef]
- Luck, W.A.P. Infrared overtone region. In Structure of Water and Aqueous Solutions; Luck, W.A.P., Ed.; Verlag Chemie: Weinheim, Germany, 1974; pp. 248–284. [Google Scholar]
- Gonzalez-Caballero, V.; Sanchez, M.T.; Lopez, M.I.; Perez-Marín, D. First steps towards the development of a non-destructive technique for the quality control of wine grapes during on-vine ripening and on arrival at the winery. J. Food Eng. 2010, 101, 158–165. [Google Scholar] [CrossRef]
- Shao, Y.; He, Y. Nondestructive measurement of the internal quality of bayberry juice using Vis/NIR spectroscopy. J. Food Eng. 2007, 79, 1015–1019. [Google Scholar] [CrossRef]
- Liu, F.; He, Y.; Wang, L.; Pan, H. Feasibility of the use of visible and near infrared spectroscopy to assess soluble solids content and pH of rice wines. J. Food Eng. 2007, 83, 430–435. [Google Scholar] [CrossRef]
- Xie, L.; Ye, X.; Liu, D.; Ying, Y. Application of principal component-radial basis function neural networks (PC-RBFNN) for the detection of water-adulterated bayberry juice by near-infrared spectroscopy. J. Zhejiang Univ.-Sci. B 2008, 9, 982–989. [Google Scholar]
- Li, W.; Goovaerts, P.; Meurens, M. Quantitative analysis of individual sugars and acids in orange juices by near-infrared spectroscopy of dry extract. J. Agric. Food Chem. 1996, 44, 2252–2259. [Google Scholar] [CrossRef]
- Mato, I.; Suarez-Luque, S.; Huidobro, J.F. A review of the analytical methods to determine organic acids in grape juices and wines. Food Res. Int. 2005, 38, 1175–1188. [Google Scholar] [CrossRef]
- Lamikanra, O.; Inyang, I.D.; Leong, S. Distribution and effect of grape maturity on organic acid content of red muscadine grapes. J. Agric. Food Chem. 1995, 43, 3026–3028. [Google Scholar] [CrossRef]
- Hawkins Watts Australia. Natural acids of fruits and vegetables. Available online: http://www.hawkinswatts.com.au/documents/Natural%20Acids%20of%20Fruits%20and%20Vegetables.pdf (accessed on 10 November 2011).
- Tsenkova, R. Aquaphotomics: Water in the biological and aqueous world scrutinised with invisible light. Spectrosc. Eur. 2010, 22, 6–10. [Google Scholar]
- Jenkins, F.A.; White, H.E. Fundamentals of Optics; McGraw-Hill, Inc.: New York, NY, USA, 1976; pp. 455–473. [Google Scholar]
- Cox, A.J.; DeWeerd, A.J.; Linden, J. An experiment to measure Mie and Rayleigh total scattering cross sections. Am. J. Phys. 2002, 70, 620–625. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Omar, A.F.; Atan, H.; MatJafri, M.Z. NIR Spectroscopic Properties of Aqueous Acids Solutions. Molecules 2012, 17, 7440-7450. https://doi.org/10.3390/molecules17067440
Omar AF, Atan H, MatJafri MZ. NIR Spectroscopic Properties of Aqueous Acids Solutions. Molecules. 2012; 17(6):7440-7450. https://doi.org/10.3390/molecules17067440
Chicago/Turabian StyleOmar, Ahmad Fairuz, Hanafi Atan, and Mohd Zubir MatJafri. 2012. "NIR Spectroscopic Properties of Aqueous Acids Solutions" Molecules 17, no. 6: 7440-7450. https://doi.org/10.3390/molecules17067440





