Isolation and Partial Characterization of an Antifungal Protein Produced by Bacillus licheniformis BS-3
Abstract
:1. Introduction
2. Results
2.1. Purification of the Antifungal Protein
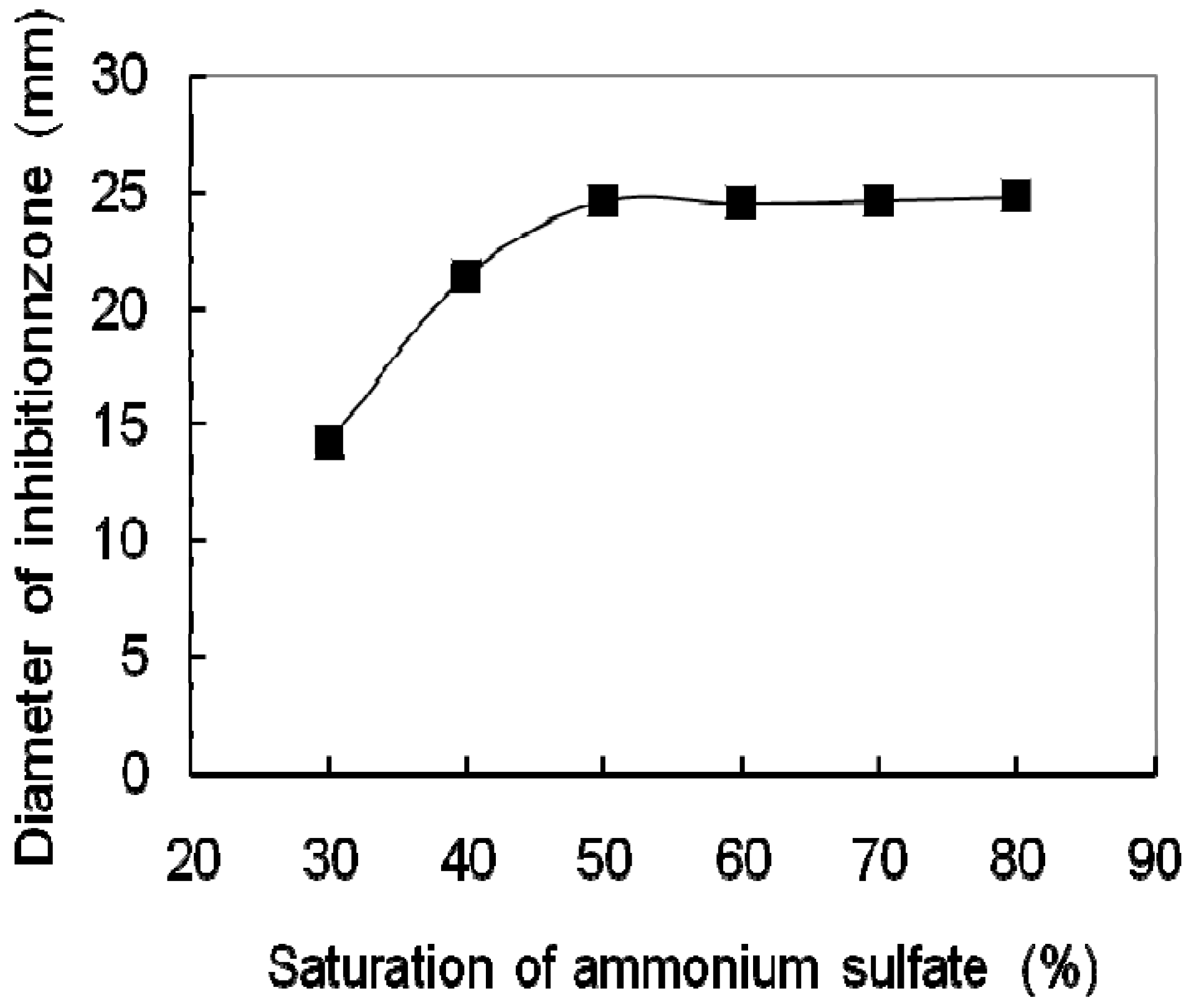
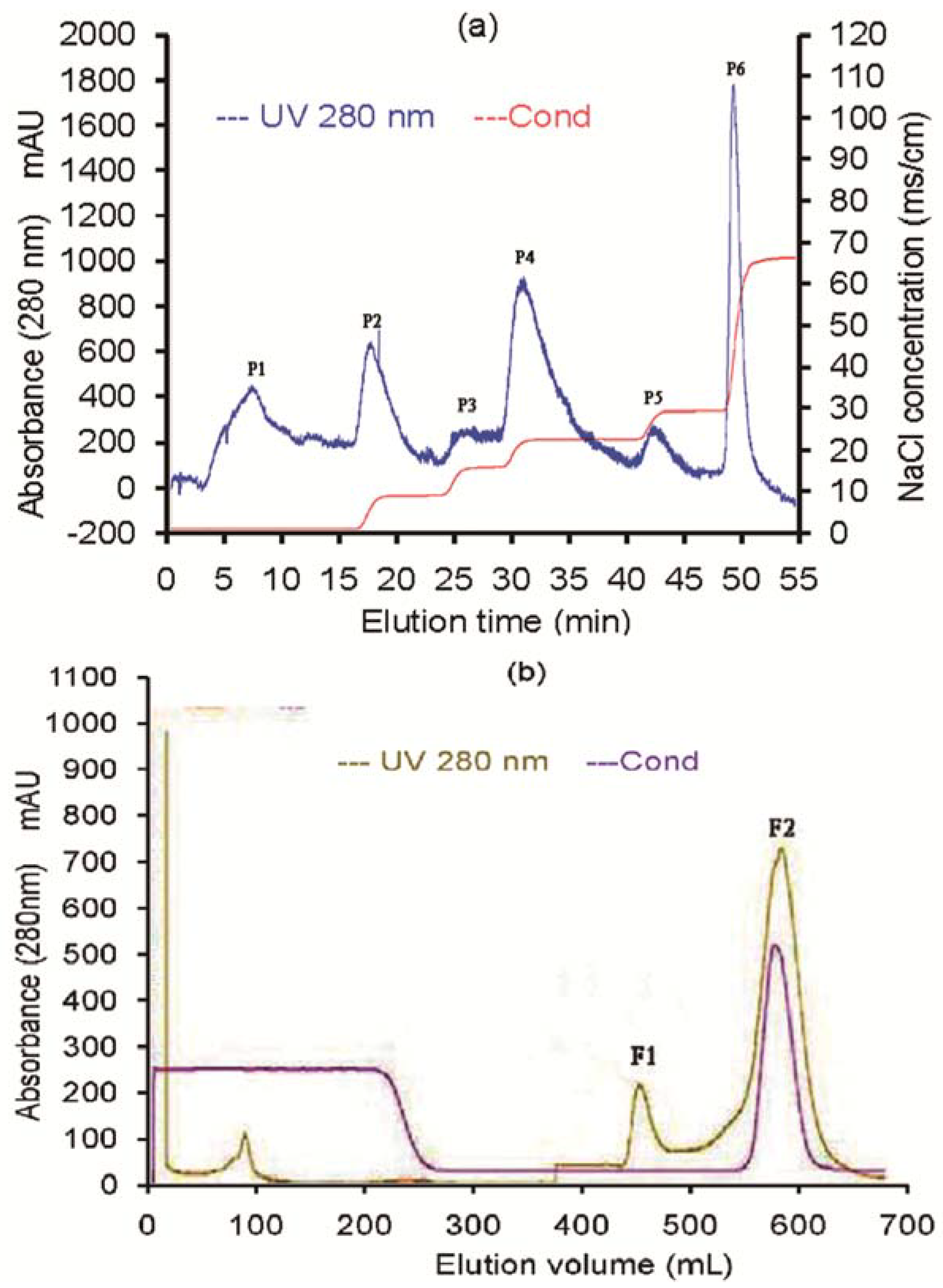
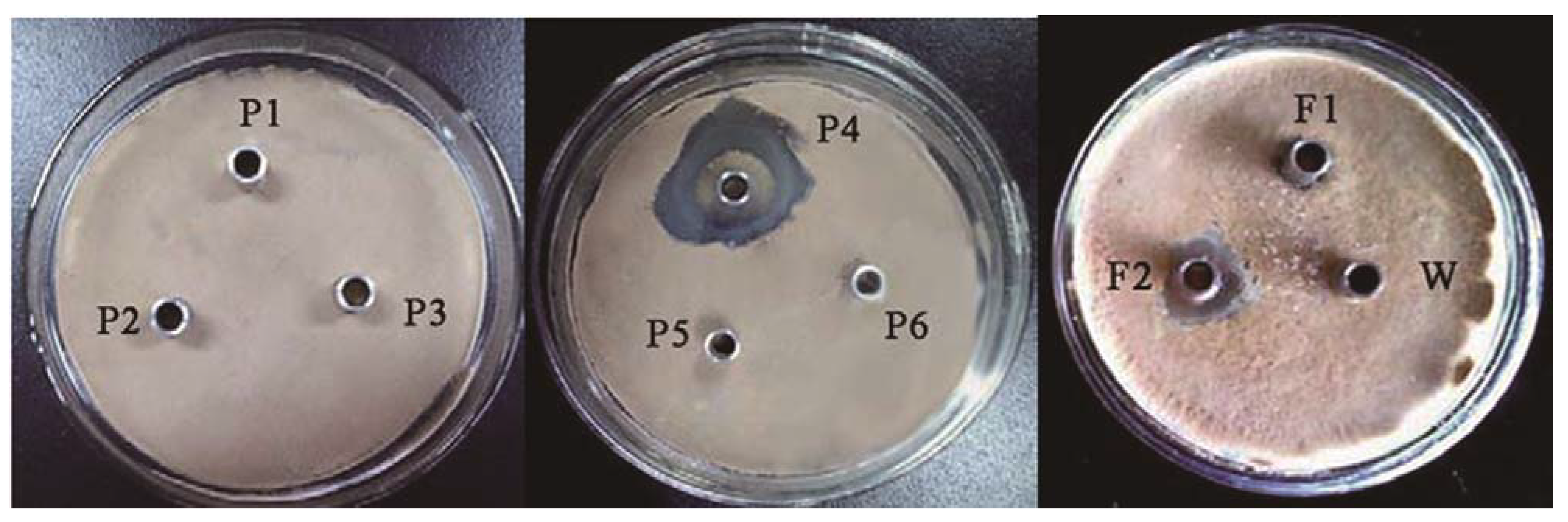

| Step | Amount of proteins (mg) | Total activity (AU) | Specific activity (AU/mg) | Recovery (%) | Purification (-fold) |
|---|---|---|---|---|---|
| Crude supernatant | 747.24 | 53,770 | 71.96 | 100.00 | 1.00 |
| Ammonium sulfate precipitation | 186.86 | 50,482 | 270.16 | 93.89 | 3.75 |
| DEAE cation exchange | 113.67 | 43,940 | 386.56 | 81.72 | 5.40 |
| Gel filtration chromatography | 74.31 | 23,200 | 1352.85 | 43.15 | 18.80 |
2.2. Inhibitory Spectrum
2.3. Effects of Enzymes, Heat, and pH
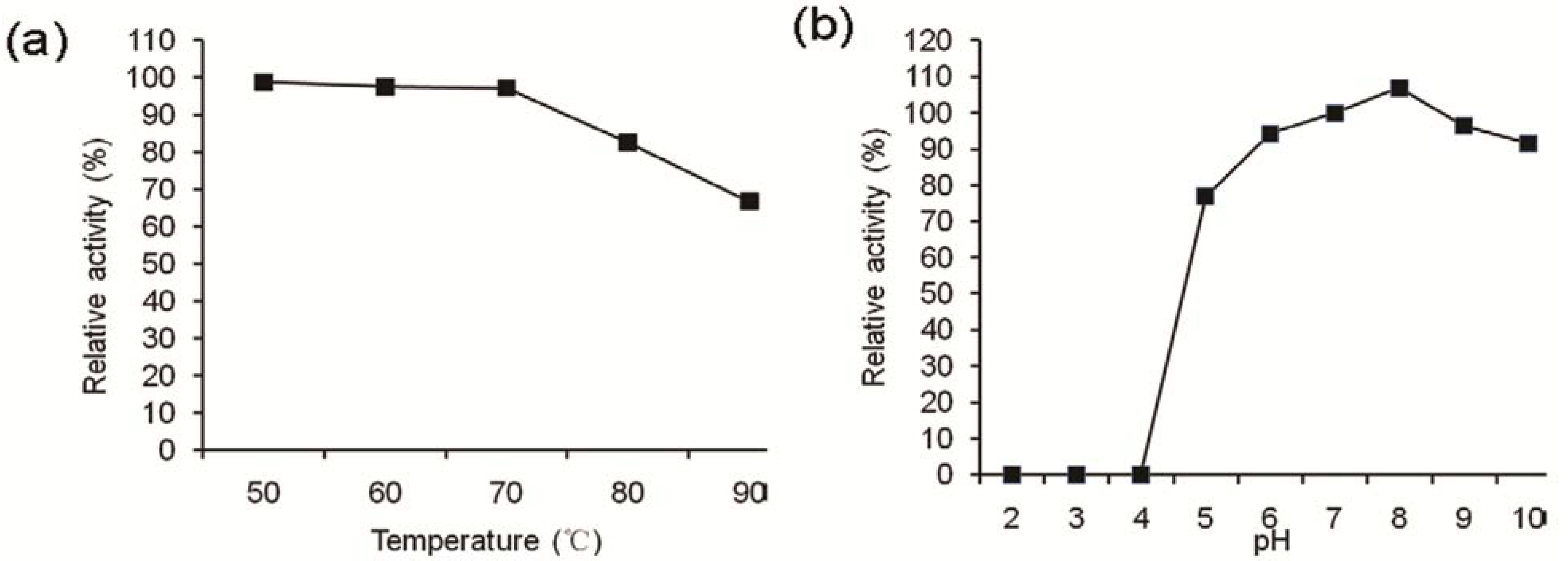
2.4. Enzymatic Activities
| Enzymes | Substrates | Concentration | Enzyme activity (U/mL) |
|---|---|---|---|
| Chitinase | Colloidal chitin | 1% | n.d. |
| Cellulase | CM-cellulose | 1.25% | n.d. |
| Xylanase | Xylan | 0.5% | n.d. |
| Protease | Casein | 1% | 5,140 |
| Lyozyme | M. lysodeikticus | Optical density of 1.5 | n.d. |
| β- N-acetyl-glucosaminidase | p-Nitrophenyl-N-acetylglucosaminide | 5 mM | n.d. |
3. Discussion
4. Experimental
4.1. Antifungal Strain
4.2. Indicator Strains
4.3. Growth and Antifungal Protein Production
4.4. Quantification of Antifungal Activity
4.5. Extraction and Purification of Antifungal Protein
4.6. Electrophoresis and HPLC
4.7. Effects of Enzymes, Heat, and pH
4.8. Measurement of Enzyme Activities
5. Conclusions
Acknowledgements
- Sample Availability: Samples of the F2 protein are available from the authors.
References and notes
- Cladera-Olivera, F.; Caron, G.R.; Brandelli, A. Bacteriocin-like peptide production by Bacillus licheniformis strain P4. Lett. Appl. Microbiol. 2004, 38, 251–252. [Google Scholar] [CrossRef]
- Haq, I.U.; Ashraf, H.; Ali, S.; Qadeer, M.A. Kinetic characterization of extracellular α-amylase from a derepressed mutant of Bacillus licheniformis. Appl. Biochem. Biotechnol. 2007, 141, 251–264. [Google Scholar] [CrossRef]
- Çalık, P.; Arifoğlu, M.; Çalık, G. Oxygen transfer effects in β-lactamase fermentation by Bacillus licheniformis in a glucose-based defined mediu. J. Chem. Technol. Biotechnol. 2005, 80, 1062–1071. [Google Scholar] [CrossRef]
- Tendulkar, S.R.; Saikumari, Y.K.; Patel, V.; Raghotama, S.; Munshi, T.K.; Balaram, P.; Chattoo, B.B. Isolation, purification and characterization of an antifungal molecule produced by Bacillus licheniformis BC98, and its effect on phytopathogen Magnaporthe grise. J. Appl. Microbiol. 2007, 103, 2331–2339. [Google Scholar] [CrossRef]
- Chen, Y.C.; Chiang, T.J.; Liang, T.W.; Wang, I.L.; Wang, S.L. Reclamation of squid pen by Bacillus licheniformis TKU004 for the production of thermally stable and antimicrobial biosurfactan. Biocatal. Agric. Biotechnol. 2012, 1, 62–69. [Google Scholar]
- Manocha, B.; Margaritis, A. A novel method for the selective recovery and purification of γ-polyglutamic acid from Bacillus licheniformis fermentation Brot. Biotechnol. Prog. 2010, 3, 734–742. [Google Scholar] [CrossRef]
- Mendo, S.; Faustino, N.A.; Sarmento, A.C.; Amado, F.; Moir, A.J.G. Purification and characterization of a new peptide antibiotic produced by a thermotolerant Bacillus licheniformis strai. Biotechnol. Lett. 2004, 26, 115–119. [Google Scholar] [CrossRef]
- Pattnaik, P.; Kaushik, J.K.; Grover, S.; Batish, V.K. Purification and characterization of a bacteriocin coumpond (Lichenin) produced anaerobically by Bacillus licheniformis isolated from water buffalo. J. Appl. Microbiol. 2001, 91, 636–645. [Google Scholar] [CrossRef]
- Martirani, L.; Varcamonti, M.; Naclerio, G.; de Felice, M. Purification and partial characterization of bacillocin 490, a novel bacteriocin produced by a thermophilic strain of Bacillus licheniformis. Microb. Cell Fact. 2002, 1, 1–5. [Google Scholar] [CrossRef]
- Kayalvizhi, N.; Gunasekaran, P. Production and characterization of a low-molecular-weight bacteriocin from Bacillus licheniformis MKU3. Lett. Appl. Microbiol. 2008, 47, 600–607. [Google Scholar] [CrossRef]
- He, L.L.; Chen, W.L.; Liu, Y. Production and partial characterization of bacteriocin-like pepitdes by Bacillus licheniformis ZJU12. Microbiol. Res. 2006, 161, 321–326. [Google Scholar] [CrossRef]
- Elad, Y.; Malathrakis, N.E.; Dik, A.J. Biological control of Botrytis incited diseases and powdery mildews in greenhouse crop. Crop Prot. 1996, 15, 229–240. [Google Scholar] [CrossRef]
- Emmert, E.A.B.; Handelsman, J. Biocontrol of plant disease: A (Gram-) positive perspective. FEMS Microbiol. Lett. 1999, 171, 1–9. [Google Scholar] [CrossRef]
- Moon, B.J.; Kim, C.S.; Song, J.H.; Kim, H.J.; Lee, J.P.; Park, H.C.; Shin, D.B. Biological control of gray mold rot of perilla caused by Botrytis cinerea. II. Formulation of antagonistic bacteria and is control effect. Res. Plant Dis. 2002, 8, 184–188. [Google Scholar] [CrossRef]
- Lee, J.P.; Lee, S.W.; Kim, C.S.; Son, J.H.; Song, J.H.; Lee, K.Y.; Kim, H.J.; Jung, S.J.; Moon, B.J. Evaluation of formulations of Bacillus licheniformis for the biological control of tomato gray mold caused by Botrytis cinerea. Biol. Control 2006, 37, 329–337. [Google Scholar] [CrossRef]
- Lim, J.H.; Kim, S.D. Biocontrol of phytophthora blight of red pepper caused by phytophthora capsici using Bacillus subtilis AH18 and B. licheniformis K11 formulations. J. Korean Soc. Appl. Biol. Chem. 2010, 6, 766–773. [Google Scholar]
- Abdel-Mohsein, H.S.; Sasaki, T.; Tada, C.; Nakai, Y. Characterization and partial purification of a bacteriocin-like substance produced by thermophilic Bacillus licheniformis H1 isolated from cow manure compost. Anim. Sci. J. 2011, 82, 340–351. [Google Scholar] [CrossRef]
- Batrakov, S.G.; Rodoniova, T.A.; Esipov, S.E.; Polyakov, N.B.; Sheichenko, V.I.; Shekhovtsova, N.V.; Lukin, S.M.; Panikov, N.S.; Nikolaev, Y.A. A novel lipopeptide, an inhibitor of bacterial adhesion, from the thermophilic and halotolerant subsurface Bacillus licheniformis strain 603. Biochim. Biophys. Atca 2003, 1634, 107–115. [Google Scholar]
- Xiao, L.; Xie, C.C.; Cai, J.; Lin, Z.J.; Chen, Y.H. Identification and characterization of a chitinase-produced Bacillus showing significant antifungal activity. Curr. Microbiol. 2009, 58, 528–533. [Google Scholar]
- Koschorreck, K.; Schmid, R.D.; Urlacher, V.B. Improving the functional expression of a Bacillus licheniformis laccase by random and site-directed mutagenesis. BMC Biotechnol. 2009, 9, 12–13. [Google Scholar] [CrossRef]
- Anthony, T.; Rajesh, T.; Kayalvizhi, N.; Gunasekaran, P. Influence of medium components and fermentation conditions on the production of bacteriocin(s) by Bacillus licheniformis AnBa9. Bioresour. Technol. 2009, 100, 872–877. [Google Scholar] [CrossRef]
- Yen, Y.H.; Li, P.L.; Wang, C.L.; Wang, S.L. An antifungal protease produced by Pseudomonas aeruginosa M-1001 with shrimp and crab shell powder as a carbon source. Enzyme Microb. Technol. 2006, 39, 311–317. [Google Scholar] [CrossRef]
- Dunne, C.; Moenne-Loccoz, Y.; de Bruijin, F.J.; O’Gara, F. Overproduction of an inducible extracellular serine protease improves biological control of Pythium ultimum by Stenotromonas maltophilia strain W81. Microbiologica 2000, 146, 2069–2078. [Google Scholar]
- Singh, A.K.; Chhatpar, H.S. Purification, characterization and thermodynamics of antifungal protease from Streptomyces sp. A6. J. Basic Microbiol. 2011, 51, 424–432. [Google Scholar] [CrossRef]
- Desai, S.S.; Hegde, S.; Inamdar, P.; Sake, N.; Aravind, M.S. Isolation of keratinase from bacterial isolates of poultry soil for waste degradation. Eng. Life. Sci. 2010, 4, 361–367. [Google Scholar]
- Toyokawa, Y.; Takahara, H.; Reungsang, A.; Fukuta, M.; Hachimine, Y.; Tachibana, S.; Yasuda, M. Purification and characterization of a halotolerant serine proteinase from thermotolerant Bacillus licheniformis RKK-04 isolated from Thai fish sauce. Appl. Microbiol. Biotechnol. 2010, 86, 1867–187. [Google Scholar] [CrossRef]
- Wang, S.L.; Shih, I.L.; Liang, T.W.; Wang, C.H. Purification and characterization of two antifungal chitinases extracellularly produced by Bacillus amyloliquefaciens V656 in a shrimp and crab shell powder medium. J. Agric. Food Chem. 2002, 50, 2241–2248. [Google Scholar]
- Imoto, T.; Yagishita, K. A simple activity measurement of lysozyme. Agric. Biol. Chem. 1971, 5, 1154–1156. [Google Scholar]
- Wang, S.L.; Chang, W.T. Purification and characterization of two bifunctional chitinase/lysozymes extracellularly produced by Pseudomonas aeruginosa K-187 in a shrimp and crab shell powder medium. Appl. Environ. Microbiol. 1997, 63, 380–386. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cui, T.-B.; Chai, H.-Y.; Jiang, L.-X. Isolation and Partial Characterization of an Antifungal Protein Produced by Bacillus licheniformis BS-3. Molecules 2012, 17, 7336-7347. https://doi.org/10.3390/molecules17067336
Cui T-B, Chai H-Y, Jiang L-X. Isolation and Partial Characterization of an Antifungal Protein Produced by Bacillus licheniformis BS-3. Molecules. 2012; 17(6):7336-7347. https://doi.org/10.3390/molecules17067336
Chicago/Turabian StyleCui, Tang-Bing, Hai-Yun Chai, and Li-Xiang Jiang. 2012. "Isolation and Partial Characterization of an Antifungal Protein Produced by Bacillus licheniformis BS-3" Molecules 17, no. 6: 7336-7347. https://doi.org/10.3390/molecules17067336
APA StyleCui, T.-B., Chai, H.-Y., & Jiang, L.-X. (2012). Isolation and Partial Characterization of an Antifungal Protein Produced by Bacillus licheniformis BS-3. Molecules, 17(6), 7336-7347. https://doi.org/10.3390/molecules17067336




