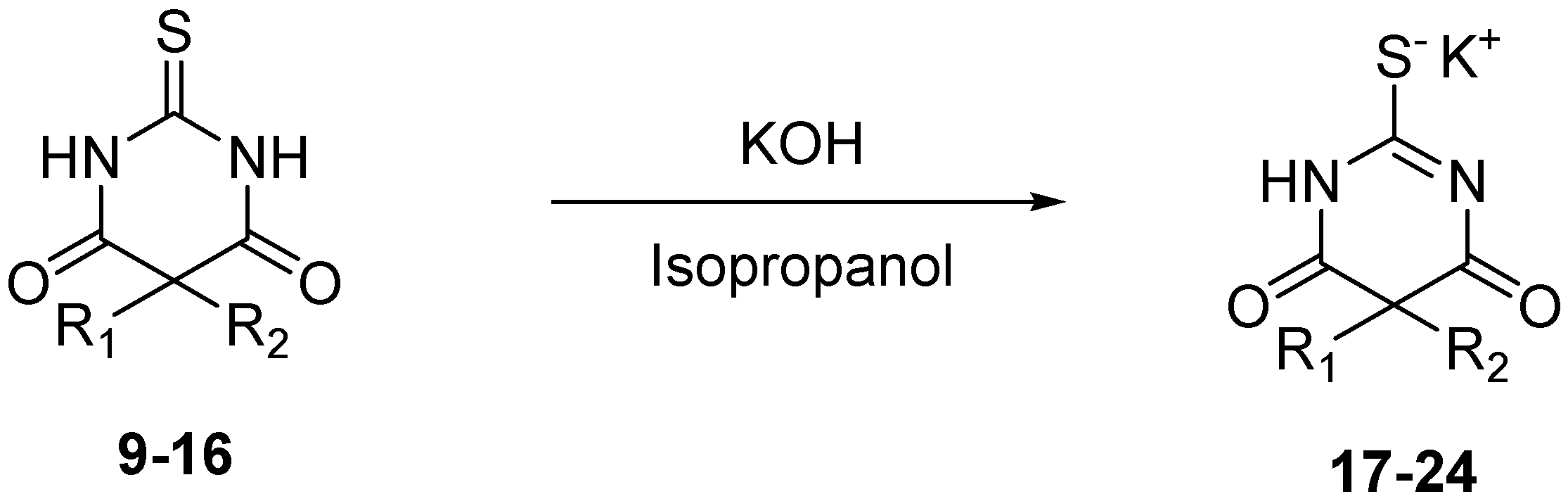3. Experimental
3.1. General
Microwave-assisted reactions were performed in a multimode microwave oven (ETHOS Synth Lab Station, Ethos start, Milestone Inc., Shelton, CT, USA). Melting points were determined with a B-540 Büchi melting point apparatus. 1H-NMR (200 MHz) and 13C-NMR (50 MHz) spectra were recorded on a Bruker ARX 200 spectrometer in CDCl3 or D2O at the Service interuniversitaire de RMN de la Faculté de Pharmacie de Marseille. The 1H-NMR chemical shifts are reported as parts per million downfield from tetramethylsilane (Me4Si), and the 13C-NMR chemical shifts were referenced to the solvent peaks: CDCl3 (76.9 ppm) or DMSO-d6 (39.6 ppm). Absorptions are reported with the following notations: s, singlet; bs, broad singlet; d, doublet; t, triplet; q, quartet; m, a more complex multiplet or overlapping multiplets. Elemental analysis and mass spectra which were run on an API-QqToF mass spectrometer were carried out at the Spectropole de la Faculté des Sciences Saint-Jérôme site. Silica gel 60 (Merck, particle size 0.040–0.063 nm, 70–230 mesh ASTM) was used for flash column chromatography. TLC were performed on 5 cm × 10 cm aluminium plates coated with silica gel 60 F-254 (Merck, Gernsteim, Germany) in an appropriate solvent.
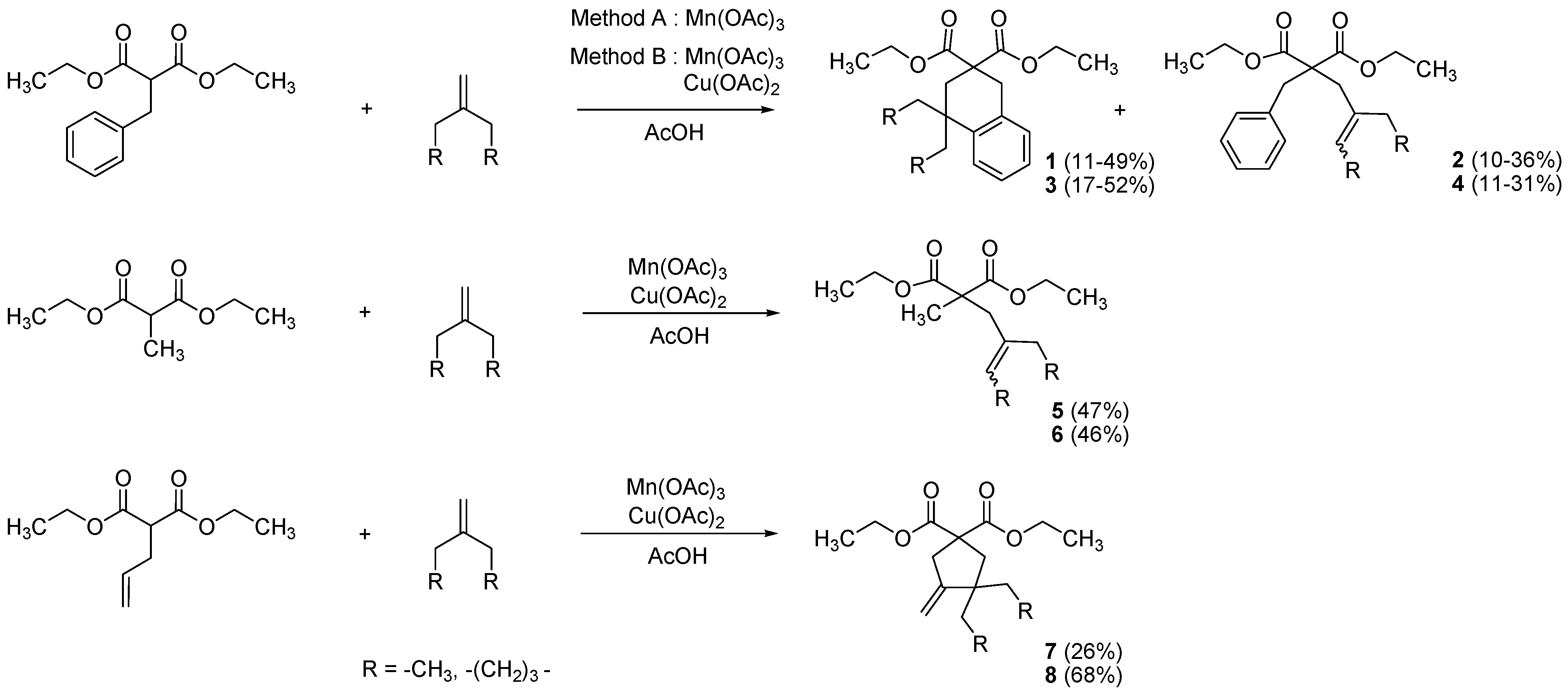
3.2. General Procedure for the Synthesis of Substituted Malonates 1–8
Method A: A solution of manganese(III) acetate dihydrate (1.68 mmol, 0.45 g) in glacial acetic acid (55 mL) was heated under microwave irradiation (200 W, 80 °C) for 15 min, until dissolution. Then, the reaction mixture was cooled down to 60 °C, and a solution of malonate (3.99 mmol, 1 equiv.) and alkene (11.97 mmol, 3 equiv.) in glacial acetic acid (5 mL) was added. The mixture was heated under microwave irradiation (200 W, 80 °C) for 20 min. Then, the reaction mixture was cooled down to 60 °C once more, and a second portion of manganese(III) acetate dihydrate (1.68 mmol, 0.45 g) was added. The mixture was heated under microwave irradiation (200 W, 80 °C) for 20 min. The addition of manganese(III) acetate dihydrate (1.68 mmol, 0.45 g) was repeated three times under the same conditions every 20 min. successively. The reaction mixture was poured into cold water (100 mL), and extracted with chloroform (3 × 70 mL). The organic extracts were collected, washed with saturated aqueous NaHCO3 (3 × 50 mL) and brine (3 × 50 mL), dried over MgSO4, filtrated, and concentrated under vacuum. The crude product was purified by silica gel chromatography with ethyl acetate/petroleum ether (0.5/9.5) to give corresponding compounds 1–4.
Method B: A solution of manganese(III) acetate dihydrate (8.38 mmol, 2.24 g, 2.1 equiv.) and copper(II) acetate monohydrate (3.99 mmol, 0.80 g, 1 equiv.) in glacial acetic acid (55 mL) was heated under microwave irradiation (200 W, 80 °C) for 15 min, until dissolution. Then, the reaction mixture was cooled down to 60 °C, and a solution of malonate (3.99 mmol, 1 equiv.) and alkene (7.98 mmol, 3 equiv.) in glacial acetic acid (5 mL) was added. The mixture was heated under microwave irradiation (200 W, 80 °C) for 60 min. The reaction mixture was poured into cold water (100 mL), and extracted with chloroform (3 × 70 mL). The organic extracts were collected, washed with saturated aqueous NaHCO3 (3 × 50 mL) and brine (3 × 50 mL), dried over MgSO4, filtrated, and concentrated under vacuum. The crude product was purified by silica gel chromatography with ethyl acetate/petroleum ether (0.5/9.5) to give corresponding compounds 1–8.
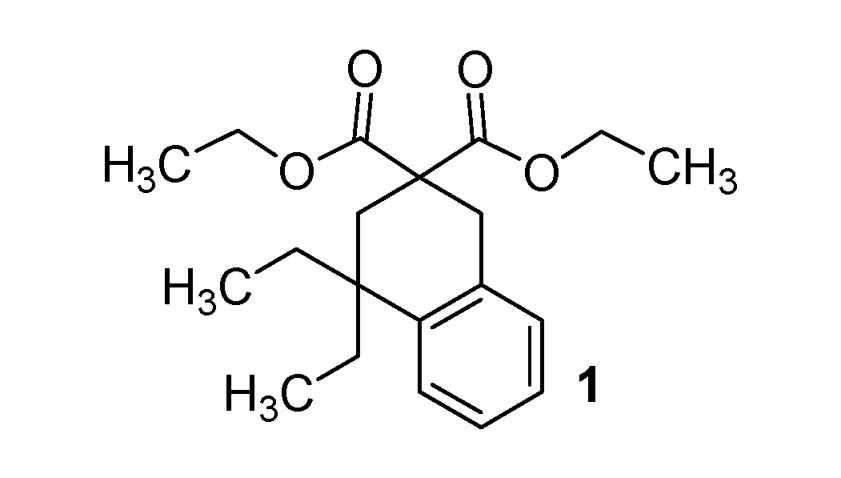
Diethyl 4,4-diethyl-3,4-dihydronaphthalene-2,2(1H)-dicarboxylate (1). Colorless oil; yields: 49% (method A), 11% (method B); 1H-NMR (CDCl3) δH 0.77 (t, J = 7.3, 6H, 2CH3), 1.22 (t, J = 7.2, 6H, 2CH3), 1.52–1.68 (m, 4H, 2CH2), 2.32 (s, 2H, CH2), 3.17 (s, 2H, CH2), 4.08–4.21 (m, 4H, 2CH2), 7.10–7.18 (m, 4H, 4CH). 13C-NMR (CDCl3) δC 8.3 (2CH3), 13.8 (2CH3), 33.1 (CH2), 33.3 (2CH2), 35.4 (CH2), 40.2 (C), 52.5 (C), 61.2 (2CH2), 125.5 (CH), 126.2 (CH), 126.5 (CH), 128.6 (CH), 134.2 (C), 141.5 (C), 172.9 (2C). HMRS (ESI): m/z calcd for C20H28O4[M+H+]: 333.2060. Found: 333.2061.
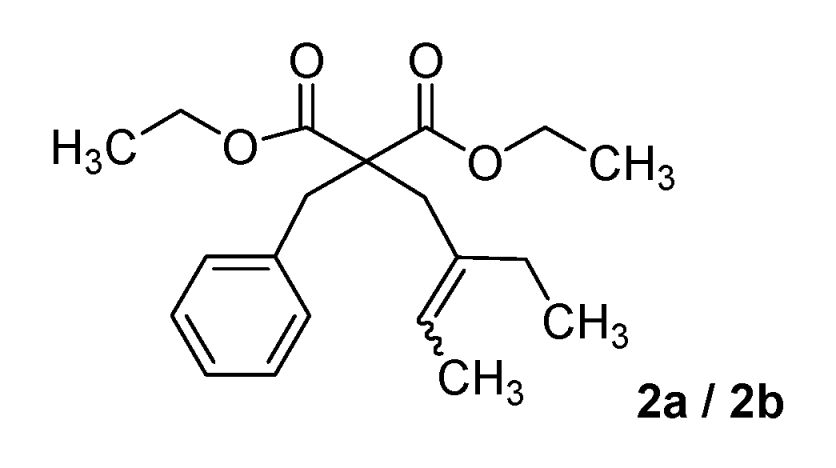
Diethyl 2-benzyl-2-(2-ethylbut-2-enyl)malonate (2a/2b) (50:50 inseparable mixture of Z/E isomers). Colorless oil; yields: 10% (method A), 36% (method B); 1H-NMR (CDCl3) δH 0.89–0.99 (m, 3H, CH3), 1.12–1.22 (m, 6H, 2CH3), 1.54–1.64 (m, 3H, CH3), 1.93–2.04 (m, 2H, CH2), 2.63 and 2.80 (s, 2H, CH2), 3.24 and 3.26 (s, 2H, CH2), 4.03–4.15 (m, 4H, 2CH2), 5.26–5.42 (m, 1H, CH), 7.11–7.36 (m, 5H, 5CH). 13C-NMR (CDCl3) δC 12.7 (CH3), 12.8 and 13.2 (CH3), 13.8 and 13.9 (2CH3), 23.3 and 29.6 (CH2), 33.5 and 40.6 (CH2), 39.1 and 39.2 (CH2), 58.9 and 59.0 (C), 61.1 (2CH2), 122.2 and 123.0 (CH), 126.7 (CH), 128.0 (2CH), 130.1 (2CH), 130.2 (C), 136.8 and 137.3 (C), 171.5 and 171.6 (2C). HMRS (ESI): m/z calcd for C20H28O4 [M+H+]: 333.2060. Found: 333.2063.
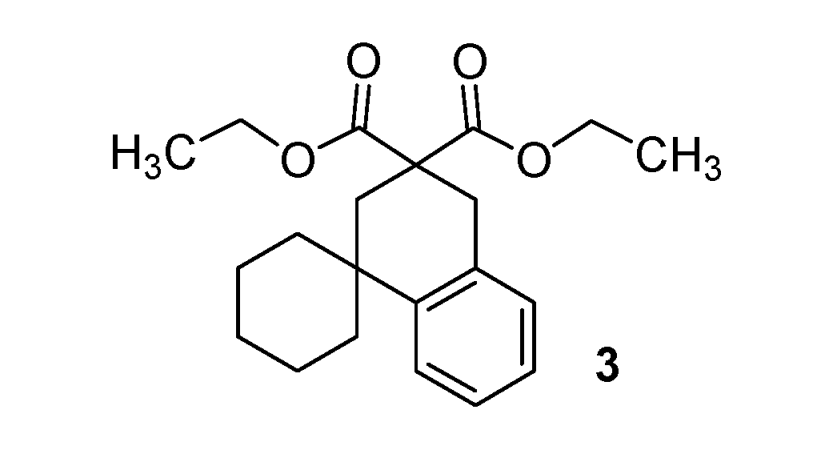
Diethyl 2'H-spiro[cyclohexane-1,
1'-naphtalene]-3',
3'(4'H)-dicarboxylate (
3). [
34] Colorless oil; yields: 52% (
method A), 17% (
method B);
1H-NMR (CDCl
3)
δH 1.22 (t,
J = 7.1, 6H, 2CH
3), 1.47–1.80 (m, 10H, 5CH
2), 2.46 (s, 2H, CH
2), 3.19 (s, 2H, CH
2), 4.14 (q,
J = 7.1, 2H, CH
2), 4.15 (q,
J = 7.1, 2H, CH
2), 7.10–7.23 (m, 3H, 3CH), 7.35–7.39 (m, 1H, 1CH).
13C-NMR (CDCl
3)
δC 13.9 (2CH
3), 21.9 (2CH
2), 25.9 (CH
2), 34.9 (CH
2), 35.6 (CH
2), 36.8 (C), 39.6 (2CH
2), 52.4 (C), 61.26 (2CH
2), 125.8 (CH), 126.1 (CH), 126.5 (CH), 128.7 (CH), 133.4 (C), 144.0 (C), 171.8 (2C). Anal. Calcd for C
21H
28O
4: C, 73.23; H, 8.19. Found: C, 73.40; H, 8.50.
Diethyl 2-benzyl-2-(cyclohexenylmethyl)malonate (
4). [
34] Colorless oil; yields: 11% (
method A), 31% (
method B);
1H-NMR (CDCl
3)
δH 1.20 (t,
J = 7.1, 6H, 2CH
3), 1.55–1.59 (m, 4H, 2CH
2), 1.90–2.00 (m, 4H, 2CH
2), 2.58 (s, 2H, CH
2), 3.26 (s, 2H, CH
2), 4.12 (q,
J = 7.1, 4H, 2CH
2), 5.52 (s, 1H, 1CH), 7.11–7.24 (m, 5H, 5CH).
13C-NMR (CDCl
3)
δC 13.9 (2CH
3), 22.1 (CH
2), 23.0 (CH
2), 25.5 (CH
2), 29.2 (CH
2), 39.0 (CH
2), 41.4 (CH
2), 58.7 (C), 61.0 (2CH
2), 126.4 (CH), 126.7 (CH), 128.0 (2CH), 130.1 (2CH), 133.1 (C), 136.7 (C), 171.4 (2C). Anal. Calcd for C
21H
28O
4: C, 73.23; H, 8.19. Found: C, 72.95; H, 8.35.
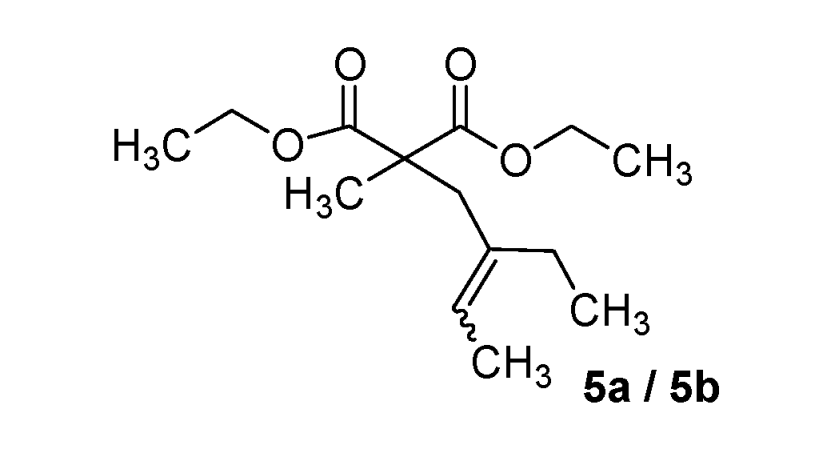
Diethyl 2-(2-ethylbut-2-enyl)-2-methylmalonate (5a/5b) (50:50 inseparable mixture of Z/E isomers). Colorless oil; yields: 47% (method B); 1H-NMR (CDCl3) δH 0.81–0.93 (m, 3H, CH3), 1.14–1.21 (m, 6H, 2CH3), 1.27 (s, 3H, CH3), 1.48–1.53 (m, 3H, CH3), 1.65–1.96 (m, 2H, CH2), 2.57 and 2.71 (s, 2H, CH2), 4.04–4.15 (m; 4H, 2CH2), 5.13 and 5.34 (m, 1H, 1CH). 13C-NMR (CDCl3) δC 12.4 (CH3), 12.6 and 12.9 (CH3), 13.7 and 13.8 (CH3), 19.2 and 19.7 (CH3), 22.9 and 29.7 (CH2), 33.6 and 40.8 (CH2), 53.2 and 53.4 (C), 60.9 and 61.0 (2CH2), 122.4 and 123.4 (CH), 136.6 and 136.8 (C), 172.3 and 172.5 (2C). HMRS (ESI): m/z calcd for C14H24O4 [M+H+]: 257.1747. Found: 257.1743.
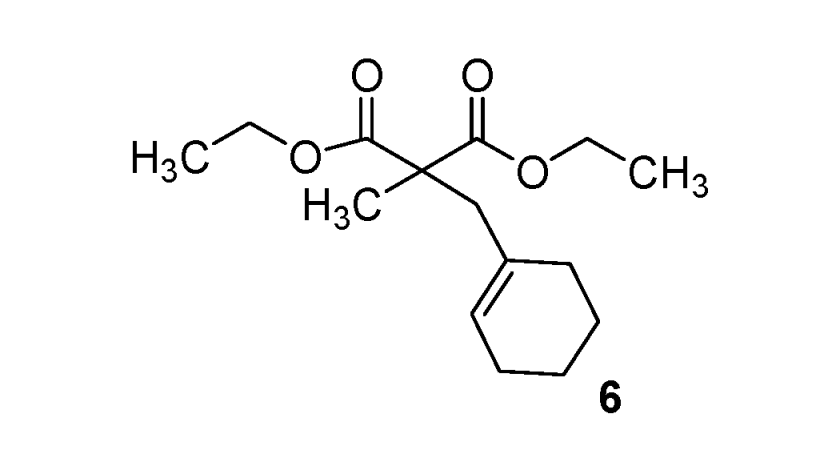
Diethyl 2-(cyclohexenylmethyl)-2-methylmalonate (6). Colorless oil; yields: 46% (method B); 1H-NMR (CDCl3) δH 1.23 (t, J = 7.1 Hz, 6H, 2CH3), 1.34 (s, 3H, CH3), 1.44–1.58 (m, 4H, 2CH2), 1.73–2.03 (m, 4H, 2CH2), 2.58 (s, 2H, CH2), 4.15 (q, J = 7.1, 2CH2), 5.43 (s, 1H, 1CH). 13C-NMR (CDCl3) δC 14.0 (2CH3), 19.9 (CH3), 22.0 (CH2), 22.9 (CH2), 25.4 (CH2), 29.2 (CH2), 43.7 (CH2), 53.3 (C), 61.1 (2CH2), 126.6 (CH), 132.9 (C), 172.6 (2C). HMRS (ESI): m/z calcd for C15H24O4 [M+H+]: 269.1747. Found: 269.1754.
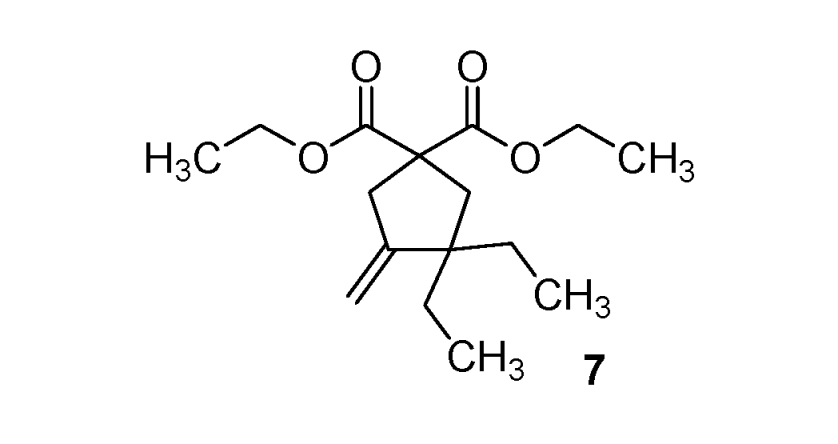
Diethyl 3,3-diethyl-4-methylenecyclopentane-1,1-dicarboxylate (7). Colorless oil; yields: 26% (method B); 1H-NMR (CDCl3) δH 0.79 (t, J = 7.3, 6H, 2CH3), 1.24 (t, J = 7.1, 6H, 2CH3), 1.33–1.41 (m, 4H, 2CH2), 2.29 (s, 2H, CH2), 2.98–3.00 (m, 2H, CH2), 4.17 (q, J = 7.1, 4H, 2CH2), 4.65 (bs, 1H, CH), 4.95 (bs, 1H, CH). 13C-NMR (CDCl3) δC 8.6 (2CH3), 14.0 (2CH3), 29.9 (2CH2), 41.8 (CH2), 43.3 (CH2), 48.5 (C), 57.3 (C), 61.4 (2CH2), 106.0 (CH2), 154.8 (C), 172.3 (2C). HMRS (ESI): m/z calcd for C16H26O4 [M+H+]: 283.1904. Found: 283.1906.
Diethyl 4-methylenespiro[4.5]decane-2,2-dicarboxylate (8). Colorless oil; yields: 68% (method B); 1H-NMR (CDCl3) δH 1.22 (t, J = 7.2, 6H, 2CH3), 1.33–1.66 (m, 10H, 5CH2), 2.33 (s, 2H, CH2), 3.01 (bs, 2H, CH2), 4.15 (q, J = 7.1, 4H, 2CH2), 4.77 (bs, 1H, CH), 4.87 (bs, 1H, CH). 13C-NMR (CDCl3) δC 13.9 (2CH3), 23.2 (2CH2), 25.8 (CH2), 38.0 (2CH2), 40.8 (CH2), 42.6 (CH2), 45.6 (C), 57.9 (C), 61.4 (2CH2), 104.6 (CH2), 158.4 (C), 172.1 (2C). HMRS (ESI): m/z calcd for C17H26O4[M+H+]: 295.1904. Found: 295.1903.
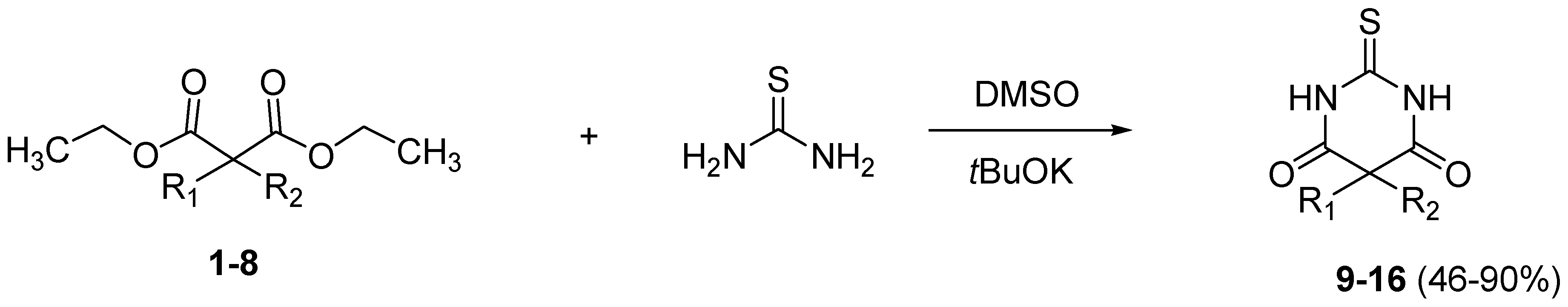
3.3. General Procedure for the Synthesis of Thiobarbituric Acids 9–16
Thiourea (1.25 g, 16.38 mmol, 6 equiv.) was added to a solution of malonate 1–8 (2.73 mmol, 1 equiv.) in dry DMSO (3 mL). Then, a solution 1M of potassium tert-butoxide (0.67 g, 6.0 mmol, 2.2 equiv.) was added dropwise. The solution was stirred for 4 h under inert atmosphere and at rt (starting from malonates 1, 3, 7, 8) or at 50 °C (starting from malonates 2, 4, 5, 6). The solution was diluted with ethyl acetate (15 mL) and washed with a solution of 1 N hydrochloric acid. The layers were separated and the aqueous phase was extracted with ethyl acetate. The collected organic phase was washed with brine, dried over anhydrous Na2SO4, filtered and the solvent was removed in vacuo. The residue was purified with column chromatography (CH2Cl2/petroleum ether, 8:2), affording the corresponding thiobarbituric acids 9–16.
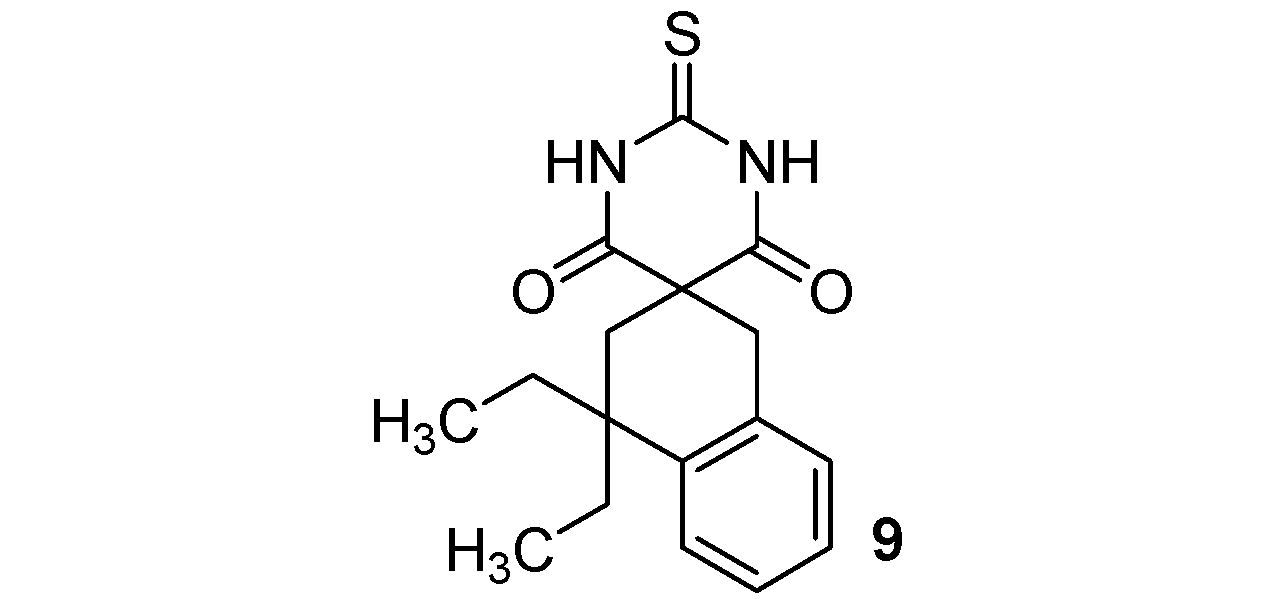
4,4-Diethyl-2'-thioxo-3,4-dihydro-1H,2'H-spiro[naphthalene-2,5'-pyrimidine]-4',6'(1'H,3'H)-dione (9). White solid; m.p. 151 °C (cyclohexane); yields: 53% 1H-NMR (CDCl3) δH 0.76 (t, J = 7.4, 6H, 2CH3), 1.67–1.80 (m, 4H, 2CH2), 2.23 (s, 2H, CH2), 3.28 (s, 2H, CH2), 7.12–7.36 (m, 4H, 4CH), 8.99 (bs, 2H). 13C-NMR (CDCl3) δC 8.4 (2CH3), 31.5 (2CH2), 34.3 (CH2), 38.2 (CH2), 52.2 (C), 53.4 (C), 126.0 (CH), 126.2 (CH), 126.8 (CH), 128.5 (CH), 132.4 (C), 140.9 (C), 170.4 (2C), 176.0 (C). HMRS (ESI): m/z calcd for C17H20N2O2S [M+H+]: 317.1318. Found: 317.1317.
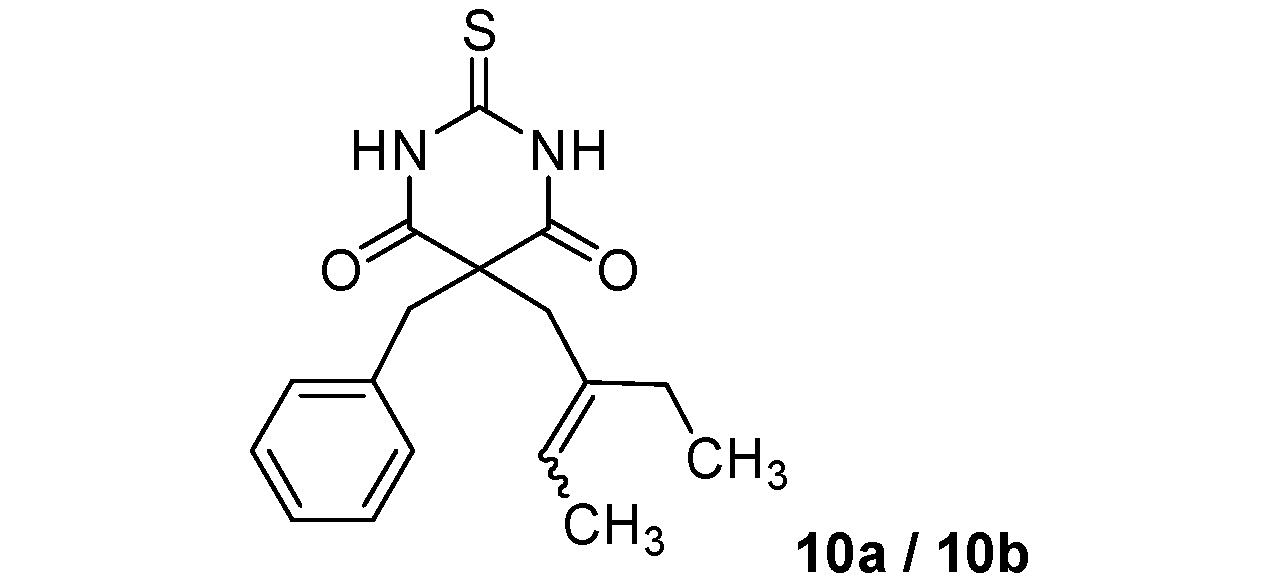
5-Benzyl-5-(2-ethylbut-2-enyl)-2-thioxo-dihydropyrimidine-4,6(1H,5H)-dione (10a/10b) (50:50 inseparable mixture of Z/E isomers). White solid; m.p. 182 °C (cyclohexane); yields: 46% 1H-NMR (CDCl3) δH 0.90–0.99 (m, 3H, CH3), 1.53–1.66 (m, 3H, CH3), 1.85–2.02 (m, 2H, CH2), 2.87 and 3.00 (s, 2H, CH2), 3.30 and 3.38 (s, 2H, CH2), 5.19–5.30 and 5.41–5.52 (m, 1H, CH), 7.07–7.24 (m, 5H, 5CH), 8.84 (bs, 2H). 13C-NMR (CDCl3) δC 12.6 and 13.0 (CH3), 13.4 and 13.7 (CH3), 23.4 and 29.9 (CH2), 39.1 and 44.9 (CH2), 45.0 and 45.2 (CH2), 58.0 and 59.0 (C), 124.6 and 124.8 (CH), 127.9 (CH), 128.9 (2CH), 129.5 and 129.6 (2CH), 134.2 and 134.3 (C), 134.7 and 135.7 (C), 169.6 (2C), 175.3 (C). m/z calcd for C17H20N2O2S [M+H+]: 317.1318. Found: 317.1323.
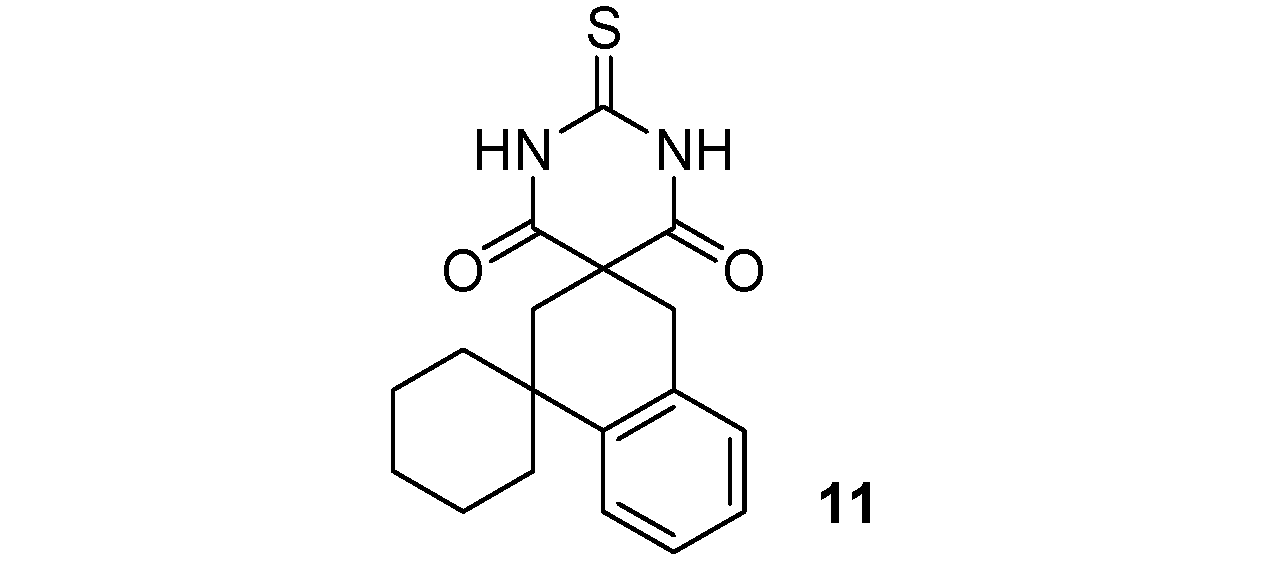
2"-Thioxo-2"H,4'H-dispiro[cyclohexane-1,1'-naphtalene-3',5"-pyrimidine]-4",6"(1"H,3"H)-dione (11). White solid; m.p. 200–202 °C (ethyl alcohol); yields: 64% 1H-NMR (CDCl3) δH 1.49–1.84 (m, 10H, 5CH2), 2.35 (s, 2H, CH2), 3.31 (s, 2H, CH2), 7.12–7.41 (m, 4H, 4CH), 9.33 (bs, 2H, 2NH). 13C-NMR (CDCl3) δC 22.0 (2CH2), 25.7 (CH2), 33.6 (CH2), 37.8 (C), 38.1 (2CH2), 38.3 (CH2), 52.2 (C), 125.1 (CH), 126.1 (CH), 127.2 (CH), 128.5 (CH), 132.1 (C), 143.8 (C), 170.2 (2C), 176.0 (C). HMRS (ESI): m/z calcd for C18H20N2O2S [M+H+]: 329.1318. Found: 329.1317.
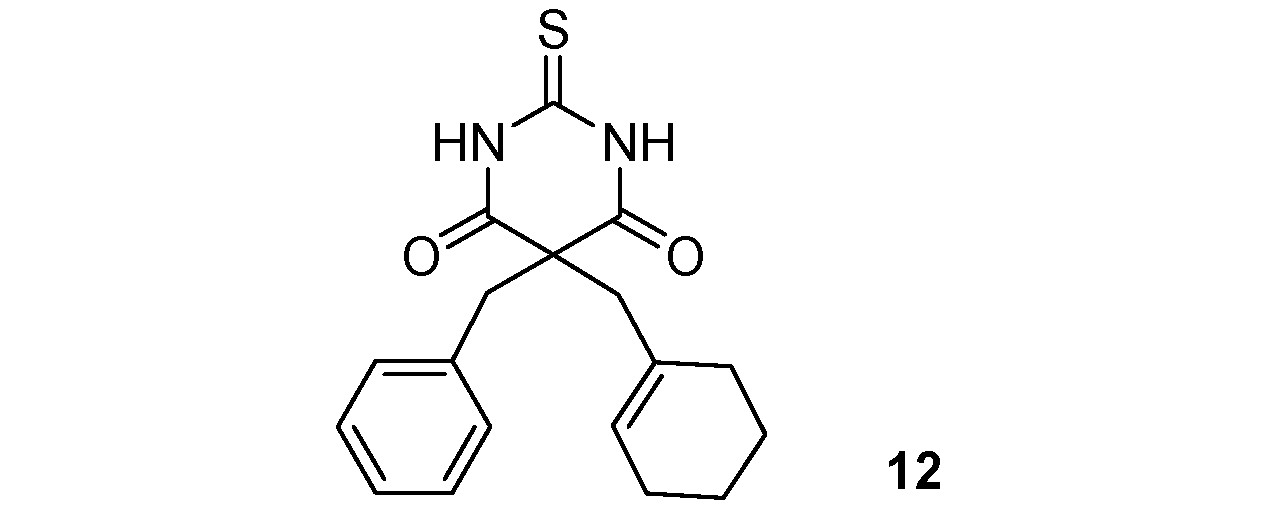
5-Benzyl-5-(cyclohexenylmethyl)-2-thioxo-dihydropyrimidine-4,6(1H,5H)-dione (12). Colorless oil; yields: 88% 1H-NMR (CDCl3) δH 1.35–2.04 (m, 8H, 4CH2), 2.82 (s, 2H, CH2), 3.31 (s, 2H, CH2), 5.50 (s, 1H, 1CH), 7.13–7.26 (m, 5H, 5CH), 8.98 (bs, 2H, 2NH). 13C-NMR (CDCl3) δC 21.9 (CH2), 22.8 (CH2), 23.6 (CH2), 29.8 (CH2), 44.5 (CH2), 47.6 (CH2), 58.9 (C), 127.7 (CH), 127.8 (CH), 128.8 (2CH), 129.5 (2CH), 131.5 (C), 134.3 (C), 169.7 (2C), 175.4 (C). HMRS (ESI): m/z calcd for C18H20N2O2S [M+NH4+]: 346.1584. Found: 346.1579.
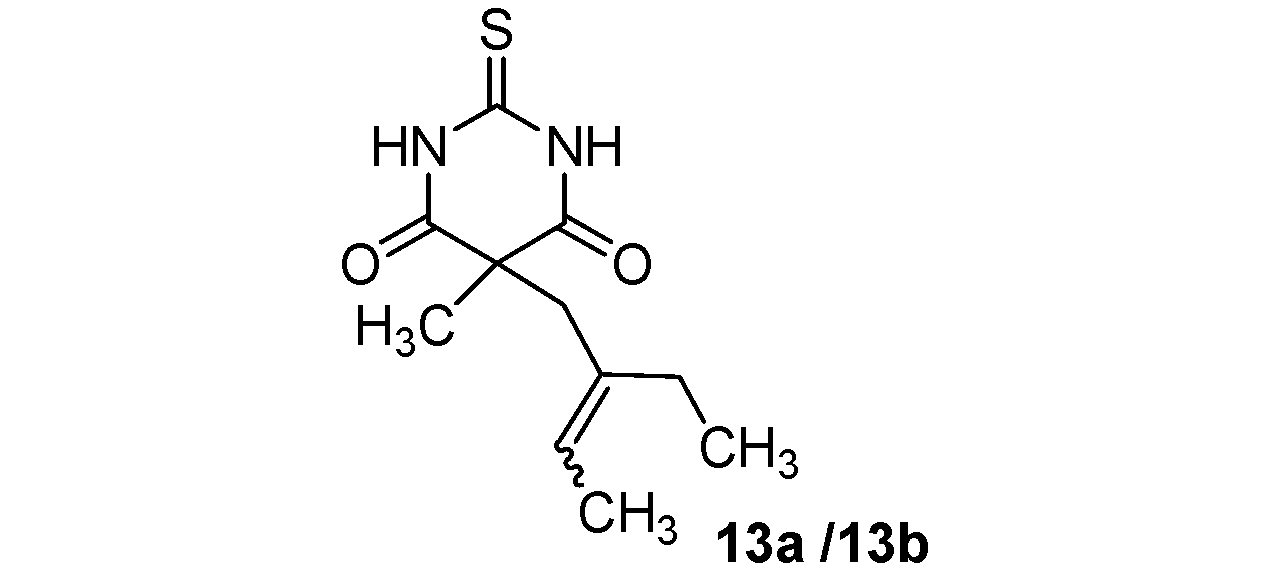
5-(2-Ethylbut-2-enyl)-5-methyl-2-thioxo-dihydropyrimidine-4,6(1H,5H)-dione (13a/13b) (50:50 inseparable mixture of Z/E isomers). Colorless oil; yields: 75% 1H-NMR (CDCl3) δH 0.87–0.97 (m, 3H, CH3), 1.54–1.61 (m, 3H, CH3), 1.57 (s, 3H, CH3), 1.80–2.01 (m, 2H, CH2), 2.70 and 2.82 (s, 2H, CH2), 5.18 and 5.47 (m, 1H, CH), 9.05 (bs, 2H, 2NH). 13C-NMR (CDCl3) δC 12.6 and 13.0 (CH3), 13.3 and 13.9 (CH3), 23.1 and 23.3 (CH3), 23.5 and 29.9 (CH2), 40.4 and 46.2 (CH2), 51.0 and 51.9 (C), 124.5 and 125.0 (CH), 134.8 and 135.9 (C), 170.5 and 170.6 (2C), 176.0 (C). Anal. Calcd for C11H16N2O2S: C, 54.98; H, 6.71; N, 11.66. Found: C, 55.15; H, 6.86; N, 11.63.
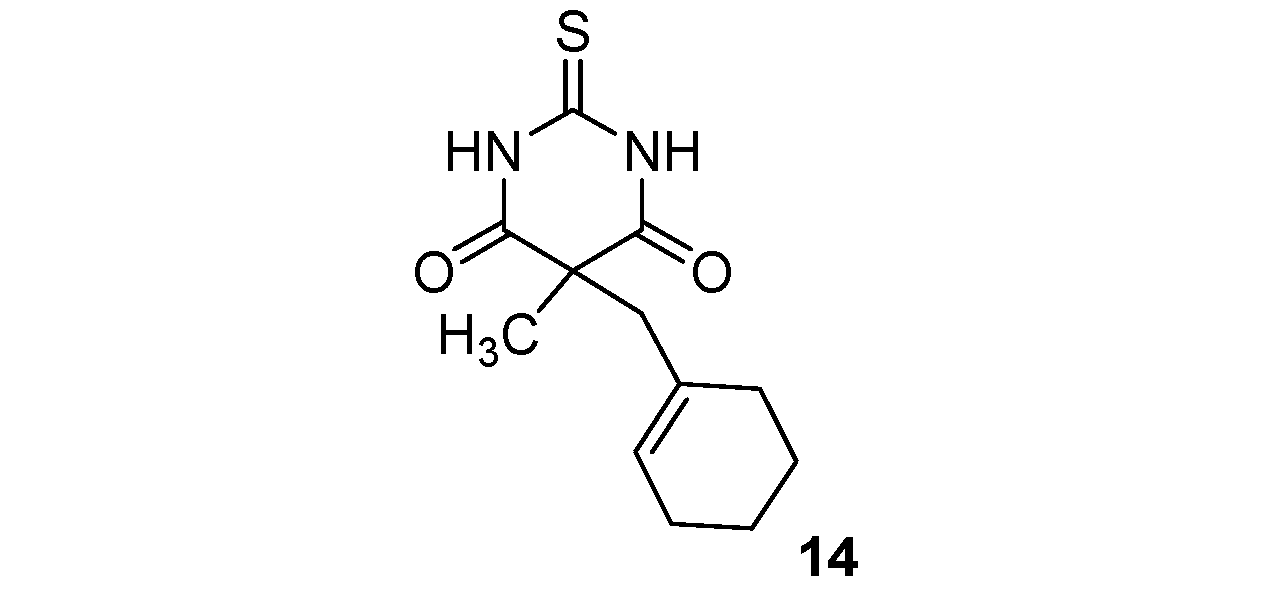
5-(Cyclohexenylmethyl)-5-methyl-2-thioxo-dihydropyrimidine-4,6(1H,5H)-dione (14). White solid; m.p. 160–164 °C (ethyl alcohol); yields: 90% 1H-NMR (CDCl3) δH 1.37–1.52 (m, 4H, 2CH2), 1.57 (s, 3H, CH3), 1.76–1.98 (m, 4H, 2CH2), 2.65 (s, 2H, CH2), 5.44 (s, 1H, 1CH), 9.61 (bs, 2H, 2NH). 13C-NMR (CDCl3) δC 21.8 (CH2), 22.8 (CH2), 23.0 (CH3), 25.4 (CH2), 29.7 (CH2), 48.5 (CH2), 51.8 (C), 127.5 (CH), 131.6 (C), 170.9 (2C), 176.2 (C). HMRS (ESI): m/z calcd for C12H16N2O2S [M+H+]: 253.1005. Found: 253.1007.
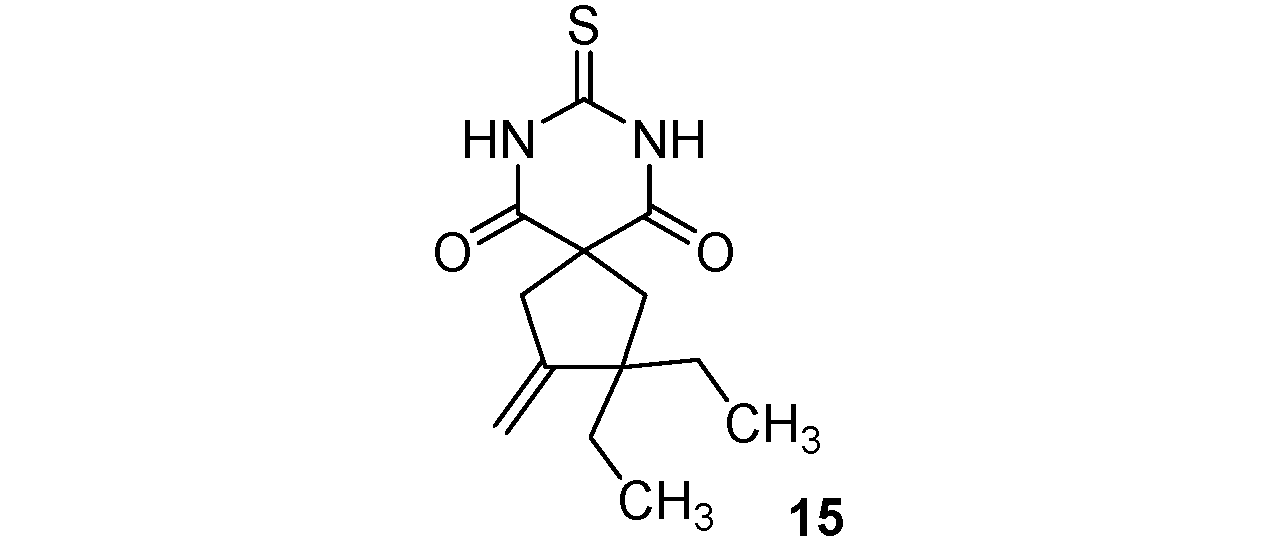
2,2-Diethyl-3-methylene-8-thioxo-7,9-diazaspiro[4.5]decane-6,10-dione (15). White solid; m.p. 194–196 °C (cyclohexane); yields: 70% 1H-NMR (CDCl3) δH 0.83 (t, J = 7.4, 6H, 2CH3), 1.43–1.70 (m, 4H, 2CH2), 2.27 (s, 2H, CH2), 3.03 (bs, 2H, CH2), 4.77 (bs, 1H, CH), 5.01 (bs, 1H, CH), 8.96 (bs, 2H, 2NH). 13C-NMR (CDCl3) δC 8.7 (2CH3), 29.0 (2CH2), 44.4 (CH2), 47.2 (CH2), 49.9 (C), 54.3 (C), 107.4 (CH2), 153.4 (C), 170.7 (2C), 176.1 (C). HMRS (ESI): m/z calcd for C13H18N2O2S [M+NH4+]: 284.1427. Found: 284.1434.
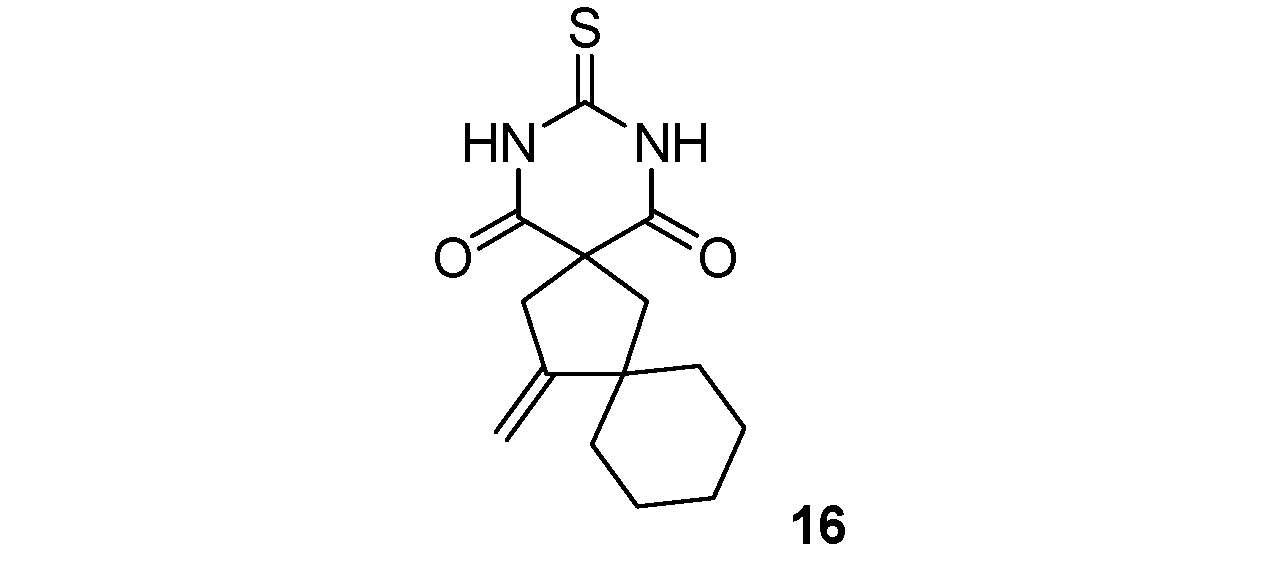
14-Methylene-3-thioxo-2,4-diazadispiro[5.1.5.2]pentadecane-1,5-dione (16). White solid; m.p. 177 °C (isopropanol); yields: 54% 1H-NMR (CDCl3) δH 1.22–1.47 (m, 6H, 2CH3), 1.66–1.77 (m, 4H, 2CH2), 2.33 (s, 2H, CH2), 3.06 (s, 2H, CH2), 4.89–4.93 (m, 2H, CH2), 9.09 (bs, 2H, 2NH). 13C-NMR (CDCl3) δC 23.2 (2CH2), 25.7 (CH2), 37.5 (2CH2), 44.0 (CH2), 45.2 (CH2), 46.8 (C), 55.0 (C), 105.4 (CH2), 157.4 (C), 170.7 (2C), 176.2 (C). HMRS (ESI): m/z calcd for C14H18N2O2S [M+NH4+]: 296.1427. Found: 296.1422.
3.4. General Procedure for Salification of Barbituric Acids to Barbiturate Potassium Salts 17–24
A suspension of potassium hydroxide (0.02 g, 0.36 mmol, 1 equiv.) in isopropanol (5 mL) was stirred under inert atmosphere. The corresponding barbituric acid 9–16 (0.36 mmol, 1 equiv.) was added, and reaction was monitored by TLC until the barbituric acid disappeared. Isopropanol was removed in vacuo, and corresponding barbiturates 17–24 were obtained without further purification.
Potassium 4,4-diethyl-4',6'-dioxo-1',3,4,6'-tetrahydro-1H,4'H-spiro[naphthalene-2,5'-pyrimidine]-2'-thiolate (17). White solid; m.p. 161–163 °C (isopropanol); yields: 77%; 1H-NMR (D2O) δH 0.72 (s, 3H, CH3), 0.99 (s, 3H, CH3), 1.42–1.79 (m, 4H, 2CH2), 2.38 (d, J = 15.4, 1H, CH2), 2.53 (d, J = 15.4, 1H, CH2), 3.13 (d, J = 16.4, 1H, CH2), 3.40 (d, J = 16.4, 1H, CH2), 7.35–7.41 (m, 4H, 4CH). 13C-NMR (D2O) δC 8.4 (CH3), 8.5 (CH3), 32.9 (CH2), 35.1 (CH2), 35.2 (CH2), 35.8 (CH2), 41.2 (C), 57.2 (C), 126.5 (CH), 127.0 (CH), 127.9 (CH), 129.1 (CH), 136.2 (C), 142.8 (C), 177.0 (2C), 178.9 (C). HMRS (ESI): m/z calcd for C17H19N2O2S− M: 315.1173. Found: 315.1183.
Potassium 5-benzyl-5-[2-ethylbut-2-en-1-yl]-4,6-thioxo-1,4,5,6-tetrahydropyrimidine-2-thiolate (18a/ 18b) (50:50 inseparable mixture of Z/E isomers). White solid; m.p. 142–144 °C (isopropanol); yields: 78%; 1H-NMR (D2O) δH 0.98–1.05 (m, 3H, CH3), 1.61–1.73 (m, 3H, CH2), 1.93–2.12 (m, 2H, CH2), 2.89 and 3.01 (s, 2H, CH2), 3.28 and 3.38 (s, 2H, CH2), 5.11 and 5.51 (bs, 1H, 1CH), 7.22–7.39 (m, 5H, 5CH). 13C-NMR (D2O) δC 12.6 and 12.7 (CH3), 13.2 and 13.3 (CH3), 23.6 and 29.8 (CH2), 39.3 and 44.8 (CH2), 45.0 and 45.9 (CH2), 57.6 (C), 122.7 and 123.8 (CH), 128.0 (CH), 129.1 (2CH), 129.9 (2CH), 135.9 (C), 138.0 (C), 172.9 (C), 179.6 (2C). HMRS (ESI): m/z calcd for C17H19N2O2S− M: 315.1173. Found: 315.1180.
Potassium 4",6"-dioxo-1",6"-dihydro-4'H,4"H-dispiro[cyclohexane-1,1'-naphtalene-3',5"-pyrimidine]-2"-thiolate (19). White solid; m.p. 216–218 °C (isopropanol); yields: 70%; 1H-NMR (D2O) δH 1.38–2.25 (m, 10H, 5CH2), 2.40 (bs, 1H, CH2), 3.08–3.68 (m, 3H, CH2), 7.40–7.58 (m, 3H, 3CH), 7.72–7.78 (m, 1H, 1CH). 13C-NMR (D2O) δC 22.1 (CH2), 22.4 (CH2), 26.0 (CH2), 35.8 (CH2), 37.6 (CH2), 37.7 (C), 38.2 (CH2), 42.0 (CH2), 56.9 (C), 126.8 (CH), 127.2 (CH), 127.3 (CH), 129.3 (CH), 135.3 (C), 144.7 (C), 176.5 (C), 178.8 (C), 181.5 (C). HMRS (ESI): m/z calcd for C18H19N2O2S− M: 327.1173. Found: 327.1184.
Potassium 5-benzyl-5-(cyclohex-1-en-1-ylmethyl)-4,6-dioxo-1,4,5,6-tetrahydropyrimidine-2-thiolate (20). White solid; m.p. 143 °C (isopropanol); yields: 84% 1H-NMR (D2O) δH 1.36–1.60 (m, 4H, 2CH2), 1.74–2.00 (m, 4H, 2CH2), 2.70 (s, 2H, CH2), 3.18 (s, 2H, CH2), 5.39 (s, 1H, 1CH), 7.06–7.11 (m, 2H, 2CH), 7.26–7.30 (m, 3H, 3CH). 13C-NMR (D2O) δC 22.3 (CH2), 23.3 (CH2), 25.7 (CH2), 29.8 (CH2), 45.5 (CH2), 47.6 (CH2), 57.3 (C), 126.2 (CH), 127.8 (CH), 129.1 (2CH), 129.9 (2CH), 134.0 (C), 136.6 (C), 181.5 (2C), 192.6 (C). HMRS (ESI): m/z calcd for C18H19N2O2S− M: 327.1173. Found: 327.1173.
Potassium 5-[2-ethylbut-2-en-1-yl]-5-methyl-4,6-dioxo-1,4,5,6-tetrahydropyrimidine-2-thiolate (21a/ 21b) (50:50 inseparable mixture of Z/E isomers). White solid; m.p. 174–176 °C (isopropanol); yields: 28% 1H-NMR (D2O) δH 0.82–0.98 (m, 3H, CH3), 1.32–1.42 (m, 3H, CH3), 1.49–1.56 (m, 3H, CH3), 1.76–2.05 (m, 2H, CH2), 2.54–2.69 (m, 2H, CH2), 5.01 and 5.45 (bs, 1H, 1CH). 13C-NMR (D2O) δC 12.8 and 13.0 (CH3), 13.2 and 14.0 (CH3), 21.0 and 22.7 (CH3), 23.7 and 30.2 (CH2), 38.5 and 44.8 (CH2), 56.7 and 57.0 (C), 123.1 and 123.8 (CH), 138.3 and 139.1 (C), 177.8 and 177.9 (C), 180.0 and 180.1 (C), 181.5 and 181.6 (C). HMRS (ESI): m/z calcd for C11H15N2O2S− M: 239.0860. Found: 239.0857.
Potassium 5-(cyclohex-1-en-1-ylmethyl)-5-methyl-4,6-dioxo-1,4,5,6-tetrahydropyrimidine-2-thiolate (22). White solid; m.p. 177 °C (isopropanol); yields: 69% 1H-NMR (D2O) δH 1.47 (s, 3H, CH3), 1.45–1.61 (m, 4H, 2CH2), 1.84–2.09 (m, 4H, 2CH2), 2.55 (s, 2H, CH2), 5.41 (s, 1H, 1CH). 13C-NMR (D2O) δC 22.4 (CH2), 22.5 (CH3), 23.3 (CH2), 25.7 (CH2), 29.6 (CH2), 47.4 (CH2), 56.8 (C), 126.7 (CH), 134.9 (C), 177.9 (2C), 181.6 (C). HMRS (ESI): m/z calcd for C12H15N2O2S− M: 251.0860. Found: 251.0859.
Potassium 2,2-diethyl-3-methylene-6,10-dioxo-7,9-diazaspiro[4.5]dec-7-ene-8-thiolate (23). White solid; decomp. 270 °C (isopropanol); yields: 88% 1H-NMR (D2O) δH 0.72–0.83 (m, 6H, 2CH3), 1.14–1.53 (m, 4H, 2CH2), 2.27 (s, 2H, CH2), 2.84 (d, J = 16.3, 1H, CH2), 3.04 (d, J = 16.3, 1H, CH2), 4.72 (bs, 1H, CH), 5.01 (bs, 1H, CH). 13C-NMR (D2O) δC 8.6 (CH3), 8.7 (CH3), 30.5 (CH2), 31.0 (CH2), 41.7 (CH2), 45.0 (CH2), 49.1 (C), 62.5 (C), 105.8 (CH2), 157.2 (C), 176.7 (C), 179.1 (C), 182.1 (C). HMRS (ESI): m/z calcd for C13H17N2O2S− M: 265.1016. Found: 265.1025.
Potassium 14-methylene-1,5-dioxo-2,4-diazaspiro[5.1.5.2]pentadec-2-ene-3-thiolate (24). White solid; m.p. 174–176 °C (isopropanol); yields: 53% 1H-NMR (D2O) δH 1.13–1.65 (m, 10H, 5CH2), 2.25 (d, J = 14.0, 1H, CH2), 2.40 (d, J = 14.0, 1H, CH2), 2.88 (d, J = 16.4, 1H, CH2), 3.04 (d, J = 16.4, 1H, CH2), 4.86 (bs, CH), 4.96 (bs, CH). 13C-NMR (D2O) δC 22.8 (CH2), 22.9 (CH2), 37.6 (CH2), 38.6 (CH2), 40.1 (CH2), 44.0 (CH2), 45.7 (C), 62.3 (C), 104.0 (CH2), 160.7 (C), 175.9 (C), 178.3 (C). 1C not observed in these conditions. HMRS (ESI): m/z calcd for C14H17N2O2S− M: 277.1016. Found: 277.1009.