Repeated Dose 28-Days Oral Toxicity Study of Carica papaya L. Leaf Extract in Sprague Dawley Rats
Abstract
:1. Introduction
2. Results and Discussion
2.1. Phytochemical Analysis
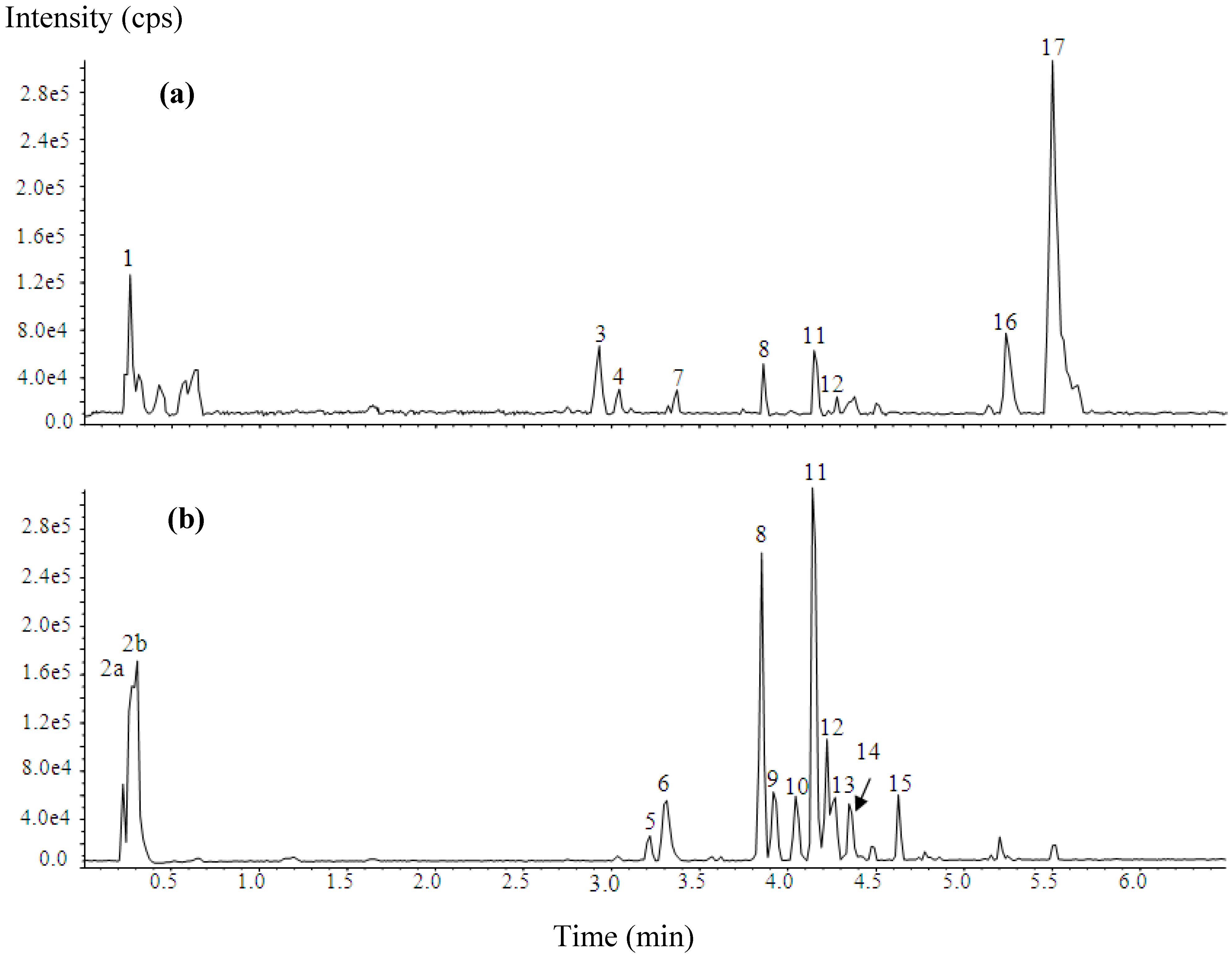
| No | tR (min) | [M−H]−/[M−H]+ (m/z) | MS2 (m/z) | Assignment |
|---|---|---|---|---|
| 1 | 0.26 | nd/381.0846 | LOW INTENSITY | Unidentified |
| 2a | 0.29 | 133.016/nd | 133, 115, 89, 87, 73 | Malic acid |
| 2b | 0.30 | 191.0214/nd | 191, 173, 149, 129, 111, 87, 85 | Quinic acid |
| 3 | 2.93 | nd/256.1935 | 256, 238, 220, 218, 122, 108 | Unidentified |
| 4 | 3.04 | LOW INTENSITY | nd | Unidentified |
| 5 | 3.22 | 447.1533/nd | LOW INTENSITY | Unidentified |
| 6 | 3.32 | 295.0484/nd | 295, 277, 195, 179, 146, 135, 133, 115, 89 | Caffeoyl malate |
| 7 | 3.37 | LOW INTENSITY | nd | Unidentified |
| 8 | 3.86 | 755.2078/757.2274 | 755, 301, 300, 271, 255, 179, 151 | Quercetin-3-O-(2'',6''-di- O-rhamnopyranosyl)glucopyranoside (manghaslin) |
| 9 | 3.94 | 279.0531/nd | 279, 163, 133, 119, 115, 93 | p-Coumaroyl malate (Isomer 1) |
| 10 | 4.06 | 279.0535/nd | 279, 163, 133, 119, 115, 93 | p-Coumaroyl malate (Isomer 2) |
| 11 | 4.16 | 739.2128/741.2327 | 739, 284, 285, 255, 227, 151, 133 | Kaempferol-3- O-(2'',6''-di-O-rhamnopyranosyl)glucopyranoside (clitorin) |
| 12 | 4.24 | 609.1479/611.1684 | 609, 301, 300, 271, 255, 179, 151 | Quercetin-3- O-rutinoside (rutin) |
| 13 | 4.28 | 309.0630/nd | 309, 291, 247, 197, 193, 149, 134, 133, 115 | Feruloyl malate (Isomer 1) |
| 14 | 4.37 | 309.0637/nd | 309, 291, 247, 197, 193, 149, 134, 133, 115 | Feruloyl malate (Isomer 2) |
| 15 | 4.64 | 593.1533/nd | 593, 284, 285, 255, 227, 151, 133 | Kaempferol-3- O-rutinoside (nicotiflorin) |
| 16 | 5.25 | nd/386.2125 a | LOW INTENSITY | Unidentified |
| 17 | 5.51 | nd/479.3898 | 479, 240, 222 | Carpaine |
2.2. Sub Acute Study
2.2.1. Clinical Observations

2.2.2. Hematology, Clinical Biochemistry, Organs Weight and Histopathology Analysis
| Sex | Parameter measured | Study group | ||||||
|---|---|---|---|---|---|---|---|---|
| Control | 0.01 g/kg BW | 0.14 g/kg BW | 2 g/kg BW | |||||
| Male | WBC (10³/μL) | 3.42 ± 3.60 | 7.14 ± 2.34 | 3.80 ± 4.80 | 7.40 ± 1.74 | |||
| RBC (106×μL) | 6.46 ± 0.34 | 6.97 ± 0.31 | 7.13 ± 0.41 | 6.97 ± 0.36 | ||||
| HGB (g/dL) | 13.70 ± 0.59 | 14.62 ± 0.56 | 14.38 ± 0.79 | 14.50 ± 0.54 | ||||
| HCT (%) | 41.22 ± 2.34 | 43.62 ± 1.88 | 43.38 ± 1.82 | 44.00 ± 1.90 | ||||
| MCV (fL) | 63.82 ± 0.92 | 62.56 ± 1.00 | 60.93 ± 1.05 * | 63.20 ± 2.24 | ||||
| MCH (pg) | 21.2 ± 0.40 | 20.98 ± 0.40 | 20.18 ± 0.13 | 20.84 ± 0.80 | ||||
| MCHC (g/dL) | 33.26 ± 0.70 | 33.52 ± 0.54 | 33.13 ± 0.48 | 32.94 ± 0.36 | ||||
| Lymphocyte % | 84.70 ± 6.60 | 87.42 ± 1.99 | 82.05 ± 3.93 | 83.54 ± 6.65 | ||||
| Lymno | 3.72 ± 1.57 | 6.22 ± 2.00 | 4.25 ± 2.49 | 6.18 ± 1.56 | ||||
| PLT(10³/μL) | 1012.20 ± 220.64 | 1033.20 ± 139.96 | 997.25 ± 237.45 | 1023.00 ± 129.89 | ||||
| Female | WBC (10³/μL) | 6.786 ± 2.021 | 5.880 ± 0.981 | 4.26 ± 4.297 | 6.70 ± 1.806 | |||
| RBC (106×μL) | 6.592 ± 0.515 | 6.266 ± 0.285 | 6.32 ± 0.164 | 6.612 ± 0.461 | ||||
| HGB (g/dL) | 13.92 ± 0.729 | 13.60 ± 0.32 | 13.578 ± 0.519 | 13.86 ± 0.83 | ||||
| HCT (%) | 41.00 ± 2.952 | 39.46 ± 0.976 | 39.36 ± 1.226 | 40.16 ± 2.772 | ||||
| MCV (fL) | 62.28 ± 2.411 | 63.04 ± 2.00 | 62.28 ± 1.04 | 60.78 ± 1.73 | ||||
| MCH (pg) | 21.16 ± 0.100 | 21.72 ± 0.701 | 21.46 ± 0.27 | 20.98 ± 0.593 | ||||
| MCHC (g/dL) | 34.00 ± 0.82 | 34.46 ± 0.19 | 34.46 ± 0.71 | 34.54 ± 0.38 | ||||
| Lymphocyte % | 81.60 ± 10.13 | 85.60 ± 1.685 | 82.78 ± 5.296 | 81.00 ± 6.841 | ||||
| Lymno | 5.40 ± 0.99 | 5.02 ± 0.85 | 4.26 ± 1.80 | 5.48 ± 1.75 | ||||
| PLT (10³/μL) | 1029.20 ± 435.12 | 1076.0 ± 101.53 | 1152.2 ± 301.92 | 1073.40 ± 283.31 | ||||
| Sex | Parameters measured | Study Groups | ||||
|---|---|---|---|---|---|---|
| Control | 0.01 g/kg BW | 0.14 g/kg BW | 2 g/kg BW | |||
| Male | Glucose (mmol/L) | 6.25 ± 1.60 | 10.09 ± 4.93 | 11.56 ± 9.51 | 8.13 ± 4.99 | |
| Liver profile | Total Protein (g/L) | 44.00 ± 7.25 | 48.60 ± 5.32 | 52.00 ± 1.41 | 46.20 ± 3.27 | |
| Albumin g/L | 28.74 ± 4.55 | 31.58 ± 2.97 | 32.78 ± 1.75 | 30.94 ± 1.99 | ||
| ALP (U/L) | 253.20 ± 65.12 | 368.80 ± 116.41 | 421.75 ± 108.00 * | 331.80 ± 32.67 | ||
| AST (U/L) | 198.80 ± 41.44 | 226.00 ± 20.15 | 249.25 ± 75.98 | 232.60 ± 25.85 | ||
| ALT (U/L) | 57.80 ± 11.71 | 81.40 ± 18.02 * | 84.50 ± 5.26 * | 65.80 ± 6.38 | ||
| Renal profile | Urea (mmol/L) | 6.11 ± 1.13 | 6.44 ± 1.61 | 7.34 ± 0.80 | 6.56 ± 0.62 | |
| Creatinine (μmol/L) | 49.40 ± 9.69 | 53.80 ± 8.32 | 56.50 ± 16.46 | 51.20 ± 5.54 | ||
| Cardiac profile | CK (U/L) | 1017.60 ± 190.33 | 1002.20 ± 241.34 | 1187.75 ± 674.08 | 739.40 ± 194.35 | |
| LDH (U/L) | 2555.80 ± 339.72 | 2526.80 ± 384.92 | 2069.25 ± 559.93 | 2327.80 ± 304.06 | ||
| Lipid profile | HDL-Cholesterol(mmol/L) | 17.19 ± 3.79 | 15.39 ± 1.85 | 14.54 ± 1.48 | 14.47 ± 5.48 | |
| Cholestrol (mmol/L) | 1.29 ± 0.23 | 1.33 ± 0.14 | 1.53 ± 0.11 | 1.43 ± 0.29 | ||
| Triglycerides (mmol/L) | 0.53 ± 0.11 | 0.72 ± 0.22 | 0.93 ± 0.15 * | 0.85 ± 0.07 * | ||
| Female | Glucose (mmol/L) | 11.12 ± 4.46 | 12.43 ± 4.54 | 13.00 ± 5.00 | 11.47 ± 5.15 | |
| Liver profile | Total Protein (g/L) | 63.20 ± 5.40 | 63.80 ± 2.49 | 78.00 ± 6.08 * | 52.50 ± 9.00 | |
| Albumin g/L | 49.54 ± 15.62 | 42.20 ± 1.72 | 48.28 ± 3.19 | 34.76 ± 5.99 | ||
| ALP (U/L) | 236.80 ± 70.05 | 257.20 ± 64.66 | 303.80 ± 119.89 | 225.00 ± 83.47 | ||
| AST (U/L) | 43.20 ± 32.74 | 38.40 ± 41.37 | 312.80 ± 73.75 * | 263.60 ± 116.75 * | ||
| ALT (U/L) | 52.52 ± 21.60 | 60.40 ± 4.67 | 88.00 ± 8.00 | 72.4 ± 41.42 | ||
| Renal profile | Urea (mmol/L) | 7.39 ± 0.51 | 8.09 ± 1.38 | 8.29 ± 1.53 | 6.38 ± 1.24 | |
| Creatinine (μmol/L) | 63.60 ± 5.17 | 71.00 ± 10.49 | 64.20 ± 4.32 | 49.00 ± 6.48 | ||
| Cardiac profile | CK (U/L) | 1220.20 ± 525.63 | 1207.60 ± 422.03 | 1505.40 ± 723.17 | 1220.50 ± 566.09 | |
| LDH (U/L) | 1756.00 ± 828.12 | 1699.20 ± 841.18 | 2174.00 ± 805.54 | 2848.75 ± 189.43 | ||
| Lipid profile | HDL-Cholesterol(mmol/L) | 19.74 ± 3.41 | 22.98 ± 3.52 | 31.08 ± 1.71 * | 19.17 ± 4.32 | |
| Cholestrol (mmol/L) | 1.68 ± 0.25 | 1.78 ± 0.32 | 2.07 ± 0.06 | 1.34 ± 0.25 | ||
| Triglycerides (mmol/L) | 0.92 ± 0.26 | 1.01 ± 0.25 | 1.33 ± 0.32 | 1.07 ± 0.34 | ||
| Sex | Organ | Study Groups | |||
|---|---|---|---|---|---|
| Control | 0.01 g/kg BW | 0.14 g/kg BW | 2 g/kg BW | ||
| Male | Lung | 0.52 ± 0.14 | 0.48 ± 0.11 | 0.44 ± 0.05 | 0.49 ± 0.02 |
| Heart | 0.34 ± 0.03 | 0.33 ± 0.03 | 0.34 ± 0.00 | 0.32 ± 0.02 | |
| Liver | 4.07 ± 0.36 | 3.56 ± 0.36 | 3.97 ± 0.28 | 3.78 ± 0.22 | |
| Stomach | 0.53 ± 0.06 | 0.51 ± 0.05 | 0.53 ± 0.05 | 0.53 ± 0.02 | |
| Spleen | 0.21 ± 0.05 | 0.12 ± 0.02 | 0.19 ± 0.03 | 0.22 ± 0.03 | |
| GIT | 0.56 ± 0.16 | 0.44 ± 0.06 | 0.45 ± 0.18 | 0.55 ± 0.09 | |
| Kidney Left | 0.38 ± 0.04 | 0.38 ± 0.05 | 0.39 ± 0.03 | 0.40 ± 0.05 | |
| Kidney Right | 0.38 ± 0.03 | 0.38 ± 0.04 | 0.40 ± 0.03 | 0.41 ± 0.04 | |
| Testis Left | 0.39 ± 0.16 | 0.49 ± 0.07 | 0.50 ± 0.04 | 0.52 ± 0.06 | |
| Testis Right | 0.42 ± 0.18 | 0.50 ± 0.07 | 0.50 ± 0.04 | 0.53 ± 0.04 | |
| Adrenal Left | 0.01 ± 0.01 | 0.01 ± 0.00 | 0.01 ± 0.00 | 0.01 ± 0.01 | |
| Adrenal Right | 0.01 ± 0.01 | 0.01 ± 0.01 | 0.01 ± 0.01 | 0.01 ± 0.00 | |
| Female | Lung | 0.50 ± 0.04 | 0.49 ± 0.05 | 0.49 ± 0.05 | 0.46 ± 0.07 |
| Heart | 0.37 ± 0.04 | 0.34 ± 0.01 | 0.35 ± 0.03 | 0.33 ± 0.03 | |
| Liver | 4.02 ± 0.21 | 3.87 ± 0.18 | 4.04 ± 0.24 | 3.85 ± 0.27 | |
| Stomach | 0.64 ± 0.09 | 0.54 ± 0.04 | 0.58 ± 0.10 | 0.56 ± 0.06 | |
| Spleen | 0.23 ± 0.02 | 0.21 ± 0.02 | 0.20 ± 0.02 | 0.22 ± 0.03 | |
| GIT | 0.67 ± 0.12 | 0.60 ± 0.13 | 0.61 ± 0.07 | 0.61 ± 0.09 | |
| Kidney Left | 0.37 ± 0.02 | 0.40 ± 0.01 | 0.39 ± 0.04 | 0.39 ± 0.03 | |
| Kidney Right | 0.39 ± 0.02 | 0.40 ± 0.03 | 0.39 ± 0.04 | 0.39 ± 0.03 | |
| Ovary Left | 0.02 ± 0.01 | 0.03 ± 0.01 | 0.03 ± 0.01 | 0.03 ± 0.02 | |
| Ovary Right | 0.02 ± 0.01 | 0.03 ± 0.01 | 0.03 ± 0.01 | 0.03 ± 0.01 | |
| Adrenal Left | 0.01 ± 0.00 | 0.01 ± 0.01 | 0.02 ± 0.00 | 0.01 ± 0.01 | |
| Adrenal Right | 0.01 ± 0.00 | 0.01 ± 0.01 | 0.01 ± 0.00 | 0.01 ± 0.00 | |
| Urinary bladder | 0.04 ± 0.01 | 0.03 ± 0.01 | 0.03 ± 0.01 | 0.03 ± 0.01 | |
3. Experimental
3.1. Plant Material and Extraction
3.2. Phytochemical Analysis
3.2.1. Chemicals
3.2.2. UPLC-TripleTOF-ESI-MS Instrumentation and Conditions
3.2.3. Isolation and Characterization of Major Flavonol Glycosides
3.2.4.Quercetin-3-O-(2'',6''-di-O-rhamnopyranosyl) glucopyranoside (manghaslin) (Peak 8)
3.2.5. Kaempferol-3-O-(2'',6''-di-O-rhamnopyranosyl) glucopyranoside (clitorin) (Peak 11)
3.3. Sub Acute Study
3.3.1. Test System and Husbandry
3.3.2. Experimental Design
3.3.3. Selection of Doses
3.3.4. Oral Administration of the Extracts
3.3.5 Parameters Measured during the Study

3.3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgements
References and Notes
- Ong, H.; Chua, S.; Milow, P. Ethno-medicinal plants used by the Temuan villagers in Kampung Jeram Kedah, Negeri Sembilan, Malaysia. Ethno Med. 2011, 5, 95–100. [Google Scholar]
- Otsuki, N.; Dang, N.H.; Kumagai, E.; Kondo, A.; Iwata, S.; Morimoto, C. Aqueous extract of Carica papaya leaves exhibits anti-tumor activity and immunomodulatory effects. J. Ethnopharmacol. 2010, 127, 760–767. [Google Scholar]
- Vigneron, M.; Deparis, X.; Deharo, E.; Bourdy, G. Antimalarial remedies in French Guiana: A knowledge attitudes and practices study. J. Ethnopharmacol. 2005, 98, 351–360. [Google Scholar]
- Thomás, G.E.; Rodolfo, H.G.; Juan, M.D.; Georgina, S.F.; Luis, C.G.; Ingrid, R.B.; Santiago, G.T. Proteolytic activity in enzymatic extracts from Carica papaya L. cv. Maradol harvest by-products. Process Biochem. 2009, 44, 77–82. [Google Scholar] [CrossRef]
- Khuzhaev, V.; Aripova, S. Pseudocarpaine from Carica papaya. Chem. Nat. Compd. 2000, 36, 418–418. [Google Scholar] [CrossRef]
- Canini, A.; Alesiani, D.; D’Arcangelo, G.; Tagliatesta, P. Gas chromatography-mass spectrometry analysis of phenolic compounds from Carica papaya L. leaf. J. Food Compos. Anal. 2007, 20, 584–590. [Google Scholar] [CrossRef]
- De Oliveira, J.G.; Vitoria, A.P. Papaya: Nutritional and pharmacological characterization and quality loss due to physiological disorders. An overview. Food Res. Int. 2011, 44, 1306–1313. [Google Scholar] [CrossRef]
- Nakamura, Y.; Yoshimoto, M.; Murata, Y.; Shimoishi, Y.; Asai, Y.; Park, E.Y.; Sato, K. Papaya seed represents a rich source of biologically active isothiocyanate. J. Agric. Food Chem. 2007, 55, 4407–4413. [Google Scholar]
- Jiao, Z.; Deng, J.; Li, G.; Zhang, Z.; Cai, Z. Study on the compositional differences between transgenic and non-transgenic papaya (Carica papaya L.). J. Food Compos. Anal. 2010, 23, 640–647. [Google Scholar] [CrossRef]
- Seigler, D.S.; Pauli, G.F.; Nahrstedt, A.; Leen, R. Cyanogenic allosides and glucosides from Passiflora edulis and Carica papaya. Phytochemistry 2002, 60, 873–882. [Google Scholar]
- Halim, S.; Abdullah, N.R.; Afzan, A.; Rashid, B.A.A.; Jantan, I.; Ismail, Z. Study of acute toxicity of Carica papaya leaf extract in Sprague Dawley rats. J. Med. Plants Res. 2011, 5, 1867–1872. [Google Scholar]
- Oduola, T.; Bello, I.; Idowu, T.; Avwioro, G.; Adeosun, G.; Olatubosun, L.H. Histopathological changes in Wistar albino rats exposed to aqueous extract of unripe Carica papaya. N. Am. J. Med. Sci. 2010, 2, 234–237. [Google Scholar]
- Lohiya, N.K.; Manivannan, B.; Garg, S. Toxicological investigations on the methanol sub-fraction of the seeds of Carica papaya as a male contraceptive in albino rats. Reprod. Toxicol. 2006, 22, 461–468. [Google Scholar] [CrossRef]
- Bylund, D.; Norström, S.H.; Essén, S.A.; Lundström, U.S. Analysis of low molecular mass organic acids in natural waters by ion exclusion chromatography tandem mass spectrometry. J. Chromatogr. A 2007, 1176, 89–93. [Google Scholar] [CrossRef]
- Bastos, D.H.; Saldanha, L.A.; Catharino, R.R.; Sawaya, A.; Cunha, I.B.; Carvalho, P.O.; Eberlin, M.N. Phenolic antioxidants identified by ESI-MS from yerba mate (Ilex paraguariensis) and green tea (Camelia sinensis) extracts. Molecules 2007, 12, 423–432. [Google Scholar] [CrossRef]
- Harbaum, B.; Hubbermann, E.M.; Wolff, C.; Herges, R.; Zhu, Z.; Schwarz, K. Identification of flavonoids and hydroxycinnamic acids in pak choi varieties (Brassica campestris L. ssp. chinensis var. communis) by HPLC-ESI-MS n and NMR and their quantification by HPLC-DAD. J. Agric. Food Chem. 2007, 55, 8251–8260. [Google Scholar]
- Gomez-Romero, M.; Zurek, G.; Schneider, B.; Baessmann, C.; Segura-Carretero, A.; Fernandez-Gutierrez, A. Automated identification of phenolics in plant-derived foods by using library search approach. Food Chem. 2011, 124, 379–386. [Google Scholar]
- Harbaum, B.; Hubbermann, E.M.; Zhu, Z.; Schwarz, K. Impact of fermentation on phenolic compounds in leaves of pak choi (Brassica campestris L. ssp. chinensis var. communis) and Chinese leaf mustard (Brassica juncea Coss). J. Agric. Food Chem. 2008, 56, 148–157. [Google Scholar]
- Engels, C.; Gräter, D.; Esquivel, P.; Jiménez, V.M.; Gänzle, M.G.; Schieber, A. Characterization of phenolic compounds in Jocote (Spondias purpurea L.) peels by ultra high-performance liquid chromatography/electrospray ionization mass spectrometry. Food Res. Int. 2011. [Google Scholar]
- Simirgiotis, M.J.; Caligari, P.D.S.; Schmeda-Hirschmann, G. Identification of phenolic compounds from the fruits of the mountain papaya Vasconcellea pubescens A. DC. grown in Chile by liquid chromatography-UV detection-mass spectrometry. Food Chem. 2009, 115, 775–784. [Google Scholar] [CrossRef]
- Kazuma, K.; Noda, N.; Suzuki, M. Malonylated flavonol glycosides from the petals of Clitoria ternatea. Phytochemistry 2003, 62, 229–237. [Google Scholar]
- Ogan, A.U. The basic constituents of the leaves of Carica papaya. Phytochemistry 1971, 10, 2544–2547. [Google Scholar] [CrossRef]
- Rivera-Pastrana, D.M.; Yahia, E.M.; González-Aquilar, G.A. Phenolic and carotenoid profiles of papaya fruit (Carica papaya L.) and their contents under low temperature storage. J. Sci. Food Agric. 2010, 90, 2358–2365. [Google Scholar] [CrossRef]
- Cano, M.P.; Tarja, E.; Marín, M.A.; Cámara, M. A simple ion exchange chromatographic determination of non volatile organic acids in some spanish exotic fruits. Zeitschrift für Lebensmitteluntersuchung und-Forschung A 1994, 199, 214–218. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar]
- Harwood, M.; Danielewska-Nikiel, B.; Borzelleca, J.F.; Flamm, G.W.; Williams, G.M.; Lines, T.C. A critical review of the data related to safety of quercetin and lack of evidence of in vivo toxicity, including lack of genotoxicity/carcinogeginc properties. Food Chem. Toxicol. 2007, 45, 2179–2205. [Google Scholar] [CrossRef]
- Calderón-Montaño, J.M.; Burgos-Morón, E.; Pérez-Guerrero, C.; López-Lázaro, M. A review on the dietry flavonoid kaempferol. Mini-Rev. Med. Chem. 2011, 11, 298–344. [Google Scholar] [CrossRef]
- Russo, M.; Spagnuolo, C.; Tedesco, I.; Bilotto, S.; Russo, G.L. The flavonoid quercetin in disease prevention and therapy: Facts and fancies. Biochem. Pharmacol. 2012, 83, 6–15. [Google Scholar]
- Olson, H.; Betton, G.; Robinson, D.; Thomas, K.; Monro, A.; Kolaja, G.; Lilly, P.; Sanders, J.; Sipes, G.; Bracken, W. Concordance of the toxicity of pharmaceuticals in humans and in animals. Regul. Toxicol. Pharmacol. 2000, 32, 56–67. [Google Scholar]
- Giknis, M.; Clifford, C. Clinical Laboratory Parameters for Crl: CD(SD)Rats; Charles River Laboratories: Wilmington, MA, USA, 2006. [Google Scholar]
- Orhan, D.D.; Orhan, N.; Ergun, E.; Ergun, F. Hepatoprotective effect of Vitis vinifera L. leaves on carbon tetrachloride-induced acute liver damage in rats. J. Ethnopharmacol. 2007, 112, 145–151. [Google Scholar] [CrossRef]
- Rasekh, H.R.; Nazari, P.; Kamli-Nejad, M.; Hosseinzadeh, L. Acute and subchronic oral toxicity of Galega officinalis in rats. J. Ethnopharmacol. 2008, 116, 21–26. [Google Scholar] [CrossRef]
- MOH (Ministry of Health) Malaysia, Principles and Guide to Ethical Use of Laboratory Animals; Institute for Medical Research: Kuala Lumpur, Malaysia, 2000; pp. 1–54.
- OECD (Organization for Economic Cooperation and Development), Guidelines for Testing of Chemicals. No 407: Repeated Dose 28-Day Oral Toxicity Study in Rodents; Paris, France, 27 July 1995.
- Sample Availability: Samples of the compounds are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Afzan, A.; Abdullah, N.R.; Halim, S.Z.; Rashid, B.A.; Semail, R.H.R.; Abdullah, N.; Jantan, I.; Muhammad, H.; Ismail, Z. Repeated Dose 28-Days Oral Toxicity Study of Carica papaya L. Leaf Extract in Sprague Dawley Rats. Molecules 2012, 17, 4326-4342. https://doi.org/10.3390/molecules17044326
Afzan A, Abdullah NR, Halim SZ, Rashid BA, Semail RHR, Abdullah N, Jantan I, Muhammad H, Ismail Z. Repeated Dose 28-Days Oral Toxicity Study of Carica papaya L. Leaf Extract in Sprague Dawley Rats. Molecules. 2012; 17(4):4326-4342. https://doi.org/10.3390/molecules17044326
Chicago/Turabian StyleAfzan, Adlin, Noor Rain Abdullah, Siti Zaleha Halim, Badrul Amini Rashid, Raja Hazlini Raja Semail, Noordini Abdullah, Ibrahim Jantan, Hussin Muhammad, and Zakiah Ismail. 2012. "Repeated Dose 28-Days Oral Toxicity Study of Carica papaya L. Leaf Extract in Sprague Dawley Rats" Molecules 17, no. 4: 4326-4342. https://doi.org/10.3390/molecules17044326




