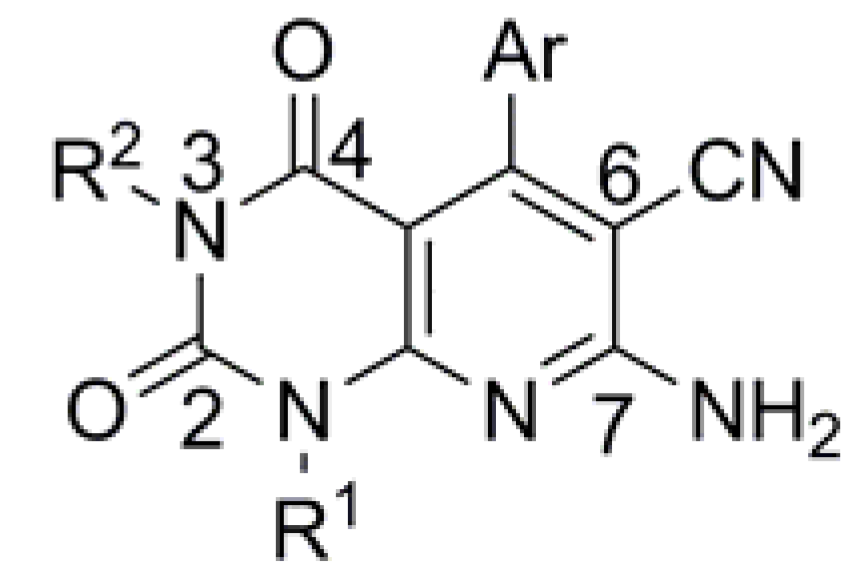
3.4. Syntheses of Compounds 3a–s
The corresponding compounds 2 (1 mmol) and anhydrous potassium carbonate were stirred at 60 °C (for 3f–q, room temperature) in DMF (5 mL), then R2-X (1.1 mmol) was added. After completion of the reaction monitored by TLC, the mixture was poured into water (25 mL). It was extracted with dichloromethane (for 3f–q, ethyl acetate was used). The combined organic layer was washed with water, dried with Na2SO4, evaporated to obtain a residue, and purified by column chromatography.
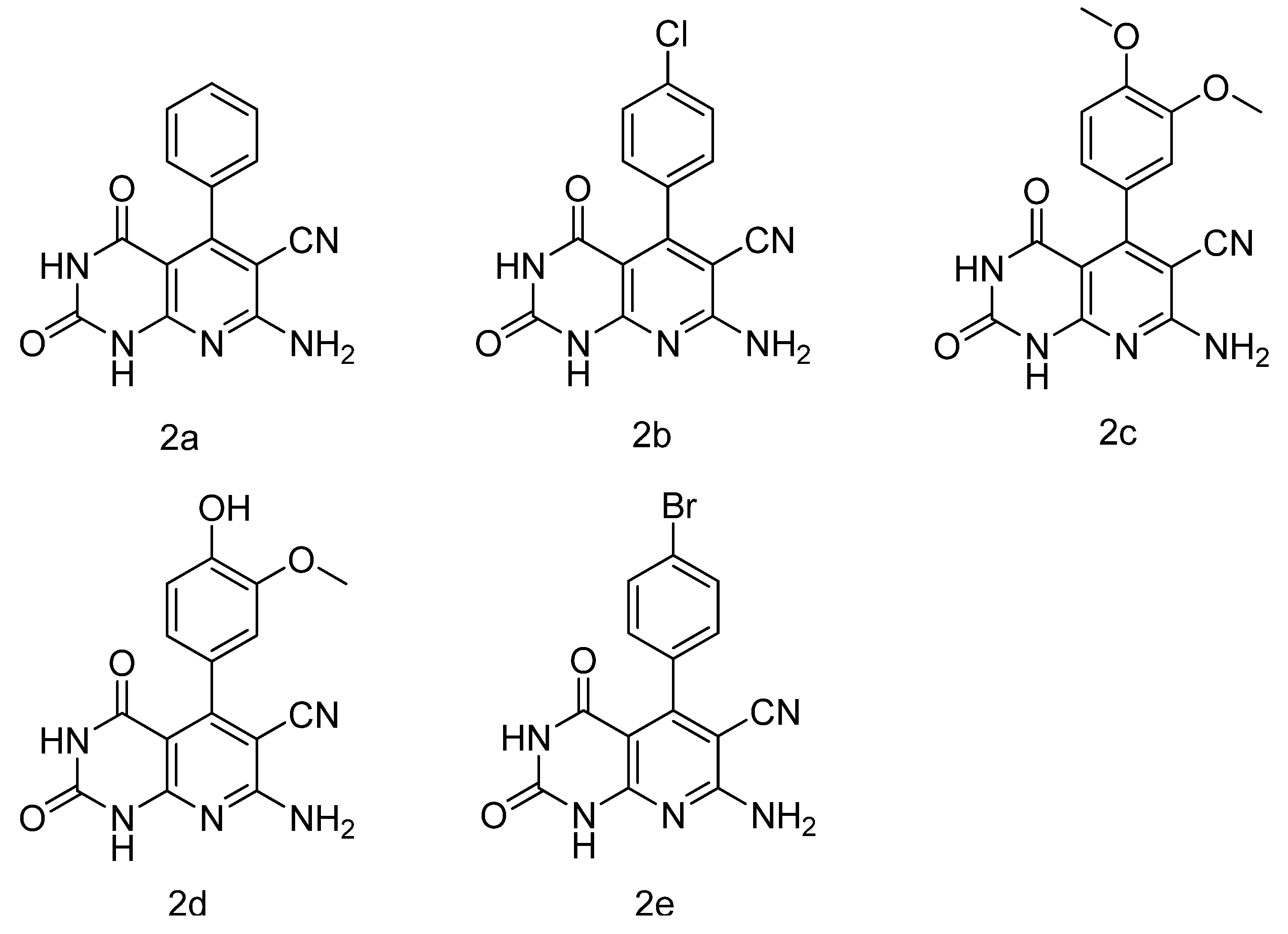
7-Amino-3-(2-(3,7-dimethyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-1-yl)ethyl)-2,4-dioxo-5-phenyl-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3a) White solid; Yield: 40.1%; 1H-NMR (DMSO-d6) δ 11.11 (s, 1H), 8.00(s, 1H), 7.65 (s, 2H), 7.41 (d, J = 2.4 Hz, 2H), 7.26 (t, J = 2.8 Hz, 1H), 7.15 (t, J = 3.2 Hz, 2H), 4.47 (s, 2H), 4.26 (s, 2H), 3.79 (s, 3H), 3.35 (s, 3H); 13C-NMR (DMSO-d6): δ 160.78, 159.91, 158.91, 155.46, 155.00, 154.56, 150.99, 150.54, 150.19, 148.17, 142.84, 136.65, 128.23, 127.65, 127.37, 115.24, 106.37, 98.89, 98.22, 88.61, 87.80, 33.08, 29.34; HRMS: calcd. for C23H19N9O4+ [M+Na+]: 508.1458, found: 508.1486.
7-Amino-5-(4-chlorophenyl)-3-(2-(3,7-dimethyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-1-yl)ethyl)-2,4-dioxo-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3b) White solid; Yield: 57.3%; 1H-NMR (DMSO-d6) δ 11.17 (s, 1H), 8.00 (s, 1H), 7.69 (s, 2H), 7.49 (d, J = 8.8 Hz, 2H), 7.19 (d, J = 8.8 Hz, 2H), 4.46 (d, J = 3.2 Hz, 2H), 4.26 (d, J = 3.2 Hz, 2H), 3.79 (s, 3H), 3.35 (s, 3H); 13C-NMR (DMSO-d6): δ 159.83, 158.82, 157.56, 154.99 154.55, 150.98, 150.49, 148.18, 142.84, 135.53, 133.06, 129.41, 127.78, 114.93, 106.37, 98.84, 87.62, 67.19, 60.70, 59.72, 33.08, 29.25, 19.93; HRMS: calcd. for C23H18ClN9O4+ [M+Na+]: 542.1068, 544.1038, found: 542.1012, 544.1064.
7-Amino-5-(3,4-dimethoxyphenyl)-3-(2-(3,7-dimethyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-1-yl)-ethyl)-2,4-dioxo-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3c) White solid; Yield: 32.2%; 1H-NMR (DMSO-d6) δ 11.09 (s, 1H), 8.00 (s, 1H), 7.58 (s, 2H), 6.99 (d, J = 8.4 Hz, 1H), 6.76 (d, J = 2 Hz,1H), 6.71 (dd, J = 8 Hz, J = 2 Hz, 1H), 4.46 (m, 2H), 4.25 (s, 2H), 3.81 (s, 3H), 3.79 (s, 3H), 3.72 (s, 3H), 3.35 (s, 3H); 13C-NMR (DMSO-d6): δ 159.87, 158.73, 155.00, 154.56, 151.00, 150.53, 148.93, 148.71, 147.82, 142.82, 128.73, 120.18, 115.34, 111.79, 110.88, 106.38, 99.03, 88.07, 67.19, 67.15, 60.70, 55.45, 33.07, 29.23, 19.92; HRMS: calcd. for C25H23N9O6+ [M+Na+]: 568.1669, found: 568.1703.
7-Amino-3-(2-(3,7-dimethyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-1-yl)ethyl)-5-(4-hydroxy-3-methoxyphenyl)-2,4-dioxo-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3d) White solid; Yield: 30.5%; 1H-NMR (DMSO-d6) δ 11.07 (s, 1H), 9.25 (s, 1H), 7.53 (s, 2H), 6.80 (d, J = 8 Hz, 1H), 6.73 (d, J = 1.6 Hz, 1H), 6.58 (dd, J = 7.6 Hz, J = 1.2 Hz, 1H), 4.46 (d, J = 19.6 Hz, 2H), 4.25 (s, 2H), 3.79 (s, 3H), 3.73 (s, 3H), 3.35 (s, 3H); 13C-NMR (DMSO-d6): δ 159.88, 159.05, 158.75, 155.02(2C), 150.98, 150.54, 148.18(2C), 146.95, 146.76, 142.82, 127.23, 120.61, 114.83(2C), 112.32, 106.37, 99.03, 88.11, 55.67(2C), 33.07, 29.33; HRMS: calcd. for C24H21N9O6+ [M+Na+]: 554.1512, found: 554.1580.
7-Amino-5-(4-bromophenyl)-3-(2-(3,7-dimethyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-1-yl)ethyl)-2,4-dioxo-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3e) White solid; Yield: 27.7%; 1H-NMR (DMSO-d6) δ 11.18 (s, 1H), 8.00 (s, 1H), 7.64 (s, 2H), 7.63 (d, J = 8.4 Hz, 1H), 7.13 (d, J = 8.4 Hz, 1H), 4,46 (s, 2H), 4.25 (s, 2H), 3.79 (s, 3H), 3.36 (s, 3H); 13C-NMR (DMSO-d6): δ 159.83, 158.83, 157.57, 155.00, 154.56, 150.99, 150.50, 148.19, 142.86, 135.94, 130.76, 129.57, 121.72, 114.93, 106.37, 98.79, 87.53, 59.72(2C), 33.09, 29.34, 20.73, 14.05; HRMS: calcd. for C23H18BrN9O4+ [M-H+]: 562.0587, 564.0566, found: 562.0511, 564.0612.
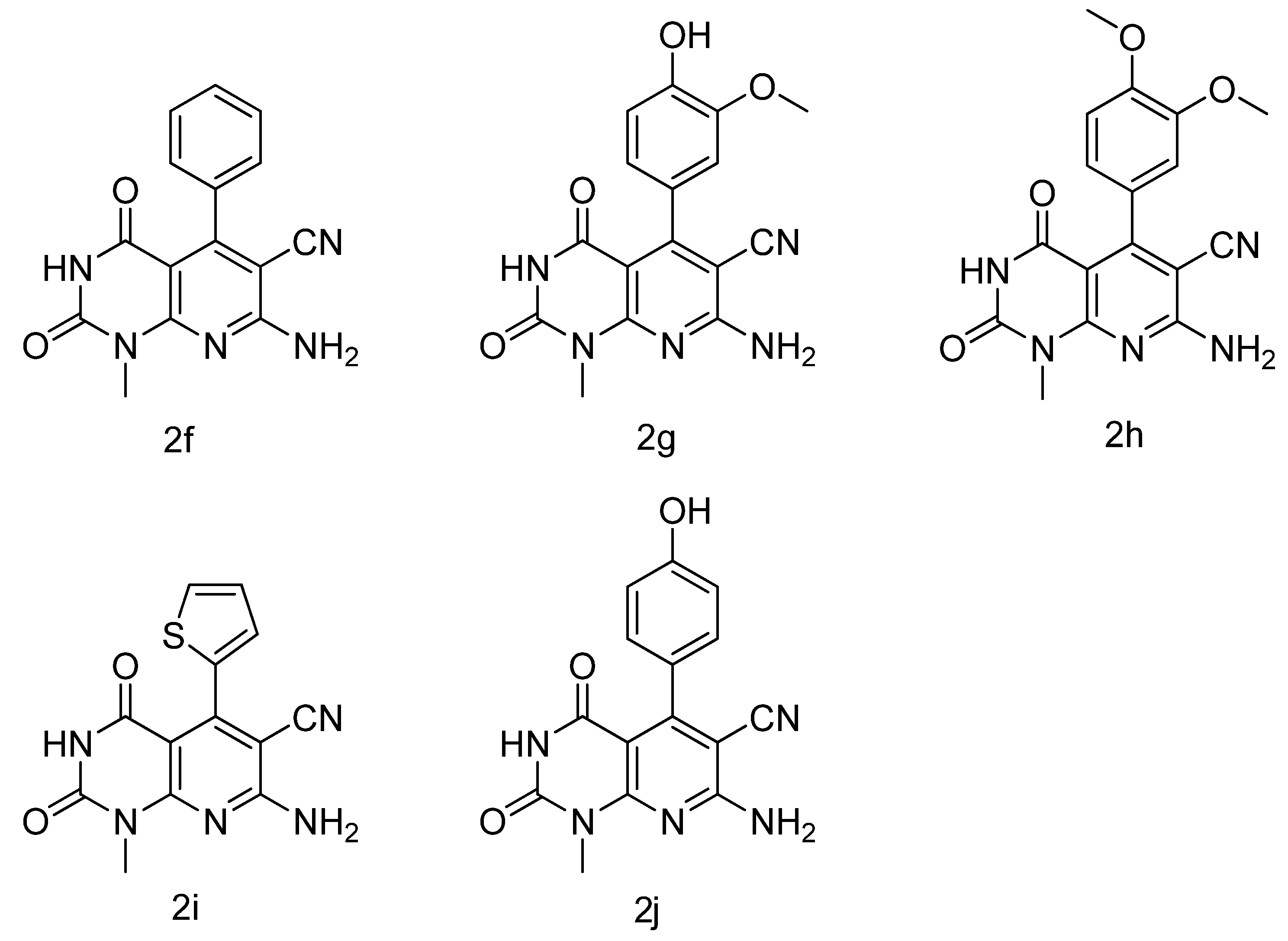
7-Amino-3-(2-(3,7-dimethyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-1-yl)ethyl)-1-methyl-2,4-dioxo-5-phenyl-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3f) White solid; Yield: 57.8%; 1H-NMR (DMSO-d6) δ 8.08 (s, 1H), 7.89 (s, 2H), 7.34 (t, J = 7.6 Hz, 1H), 7.25 (t, J = 7.6 Hz, 2H), 6.83 (d, J = 7.2 Hz, 2H), 4.06 (m, 4H).3.76 (s, 3H), 3.46 (s, 3H), 3.30 (s, 3H); 13C-NMR (DMSO-d6): δ 160.75, 159.89, 158.84, 155.09(2C), 154.15, 151.51, 151.26(2C), 148.81, 143.41, 136.90, 128.49, 127.96, 127.30, 115.68, 106.96, 98.56(2C), 88.90(2C), 33.52, 29.96, 29.74; HRMS: calcd. for C24H21N9O4+ [M+Na+]: 522.1614, found: 522.1642.
7-Amino-3-(2-(3,7-dimethyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-1-yl)ethyl)-5-(4-hydroxy-3-methoxyphenyl)-1-methyl-2,4-dioxo-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3g) White solid; Yield: 52.1%; 1H-NMR (DMSO-d6) δ 9.21 (s, 1H), 8.04 (s, 1H), 7.83 (s, 2H), 6.73 (s, 1H), 6.62 (d, J = 8.4 Hz, 1H), 6.20 (dd, J = 8.4 Hz, J = 1.6 Hz, 1H), 4.18 (m, 2H), 3.98 (m, 2H), 3.77 (s, 3H), 3.68 (s, 3H), 3.43 (s, 3H), 3.31(s, 3H); 13C-NMR (DMSO-d6): δ 160.77, 160.21, 159.08, 155.08, 154.65, 152.05(2C), 148.79, 147.30(2C), 143.96, 127.96, 121.33, 115.10(2C), 112.95, 107.05, 98.81, 88.78, 79.64, 56.08(2C), 33.53, 29.95, 29.77; HRMS: calcd. for C25H23N9O6+ [M+Na+]: 568.1669, found: 568.1605.
7-Amino-5-(3,4-dimethoxyphenyl)-3-(2-(3,7-dimethyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-1-yl) ethyl)-1-methyl-2,4-dioxo-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3h) White solid; Yield: 49.5%; 1H-NMR (DMSO-d6) δ 8.06 (s, 1H), 7.87 (s, 2H), 7.77 (s, 1H), 6.76 (d, J = 8.4 Hz, 1H), 6.35 (dd, J = 1.6 Hz, J = 8 Hz, 1H), 4.15 (m, 2H), 4.22 (m, 2H), 3.79 (s, 3H), 3.78 (s, 3H), 3.67 (s, 3H), 3.43 (s, 3H), 3.31 (s, 3H); 13C-NMR (DMSO-d6): δ160.43, 159.84, 158.55, 154.87, 154.13, 151.59, 151.35, 148.81, 148.38, 143.56, 129.52, 120.22, 115.67(2C), 112.13, 106.99, 98.87, 88.40, 79.22, 55.98, 55.86, 33.55, 29.96, 29.78, 21.22, 14.55; HRMS: calcd. for C26H25N9O6+ [M+Na+]: 582.1825, found: 582.1877.
7-Amino-3-(2-(3,7-dimethyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-1-yl)ethyl)-1-methyl-2,4-dioxo-5-(thiophen-2-yl)-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3i) White solid; Yield: 60.3%; 1H-NMR (DMSO-d6) δ 8.04 (s, 1H), 7.96 (s, 2H), 7.59 (d, J = 4.8 Hz, 1H), 7.01 (t, J = 3.2 Hz, 1H), 6.81 (d, J = 2.4 Hz, 1H), 4.08 (d, J = 5.2 Hz, 4H), 3.77 (s, 3H), 3.44 (s, 3H), 3.31 (s, 3H); 13C-NMR (DMSO-d6): δ 161.03, 158.95, 154.12, 151.05, 149.88, 148.62, 137.76, 136.25, 130.89, 128.43, 128.02, 123.52, 116.83, 112.04, 111.80, 101.62, 90.85, 56.66, 55.89, 45.73, 30.68, 16.98; HRMS: calcd. for C22H19N9O4S+ [M+Na+]: 528.1178, found: 528.1096.
7-Amino-5-(3,4-dimethoxyphenyl)-1-methyl-3-(2-methylbenzyl)-2,4-dioxo-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3j) White solid; Yield: 57.8%; 1H-NMR (CDCl3) δ 7.12 (s, 1H), 7.11 (d, J = 1.2 Hz, 1H), 7.06 (m, 1H), 6.96 (d, J = 8 Hz, 2H), 6.90 (dd, J = 8 Hz, J = 2 Hz, 1H), 6.73 (d, J = 2 Hz, 1H), 5.70 (s, 2H), 5.10 (s, 2H), 3.92 (s, 3H), 3.80 (s, 3H), 3.64 (s, 3H); 13C-NMR (DMSO-d6): δ 160.16(2C), 158.77, 154.22, 151.05, 149.88, 148.62, 135.76, 134.44, 130.30, 128.23, 127.02, 125.84, 125.59, 120.20, 115.53, 111.24, 110.70, 100.42, 90.35, 55.89, 55.75, 42.23, 30.27, 19.26; HRMS: calcd. for C25H23N5O4+ [M+Na+]: 480.1648, found: 480.1604.
7-Amino-5-(3-methoxy-4-(2-methylbenzyloxy)phenyl)-1-methyl-3-(2-methylbenzyl)-2,4-dioxo-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3k) White solid; Yield: 28.8%; 1H-NMR (CDCl3) δ 7.46 (d, J = 7.2 Hz, 1H), 7.26–7.21 (m, 3H), 7.13 (t, J = 2.8 Hz, 2H), 7.10–7.07 (m, 1H), 7.02 (d, J = 8.8 Hz, 1H), 6.97 (d, J = 7.6 Hz, 1H), 6.88 (dd, J = 8 Hz, J = 2Hz, 1H), 6.78 (d, J = 1.6 Hz, 1H), 5.71 (s, 2H), 5.15(s, 2H), 5.13 (s, 2H), 3.80 (s, 3H), 3.66 (s, 3H), 2.41 (s, 3H), 2.39 (s, 3H); 13C-NMR (DMSO-d6): δ 160.88, 159.84, 158.73, 154.40, 151.28(2C), 148.71, 148.59, 137.28, 135.40, 135.29, 135.26, 130.56, 130.26, 129.97, 129.33, 128.63, 126.91, 126.26, 125.04, 120.46, 116.11, 112.76, 112.47, 99.29, 89.36, 68.96, 55.98, 42.23, 30.24, 19.13, 18.94; HRMS: calcd. for C32H29N5O4+ [M-H+]: 546.2141, found: 546.2157.
7-Amino-1-methyl-3-(2-methylbenzyl)-2,4-dioxo-5-phenyl-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3l) White solid; Yield: 65.8%; 1H-NMR (CDCl3) δ 7.48 (t, J = 3.2 Hz, 3H), 7.28 (d, J = 3.6 Hz, 2H), 7.11 (d, J = 3.6 Hz, 2H), 7.07 (m, 1H), 6.90 (d, J = 7.6 Hz, 1H), 5.71 (s, 2H), 5.08 (s, 2H), 3.65 (s, 3H), 2.35 (s, 3H); 13C-NMR (DMSO-d6): δ 160.49, 159.92, 158.82, 154.24, 151.32, 150.06, 148.77, 136.18, 135.63, 134.28, 130.26, 129.19, 128.32, 127.07, 126.99, 125.91, 125.35, 115.19, 100.56, 90.60, 42.20, 30.26, 19.21; HRMS: calcd. for C23H19N5O2+ [M+H+]: 398.1617, found: 398.1685.
7-Amino-3-(2-fluorobenzyl)-1-methyl-2,4-dioxo-5-phenyl-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3m) White solid; Yield: 60.2%; 1H-NMR (CDCl3) δ 7.51 (t, J = 2.8 Hz, 3H), 7.38 (dd, J = 8.4 Hz, J = 5.2 Hz, 2H), 7.26 (s, 2H), 6.93 (t, J = 8.8 Hz, 2H), 5.68 (s, 2H), 5.03 (s, 2H), 3.62 (s, 3H), 2.18 (s, 3H); 13C-NMR (DMSO-d6): δ 160.49, 159.94, 159.87, 158.70, 154.12, 150.80, 136.14, 131.03, 130.98, 129.25, 128.95, 128.92, 128.35, 127.10, 123.98(2C), 115.42, 115.14, 100.37, 90.26, 43.84, 38.67, 30.21; HRMS: calcd. for C22H16FN5O2+ [M-H+]: 400.1210, found: 400.1248.
7-Amino-3-(3-fluorobenzyl)-1-methyl-2,4-dioxo-5-phenyl-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3n) White solid; Yield: 66.7%; 1H-NMR (CDCl3) δ 7.51 (t, J = 3.2 Hz, 3H), 7.28–7.19 (m, 3H), 7.14 (d, J = 7.6 Hz, 1H), 7.05 (d, J = 10 Hz, 1H), 6.94–6.89 (m, 1H), 5.70 (s, 2H), 5.05 (s, 2H), 3.63 (s, 3H); 13C-NMR (DMSO-d6): δ 163.94, 161.49, 160.47, 159.95, 158.68, 154.07, 150.98, 139.06, 136.21, 129.82, 129.24, 128.35, 127.11, 124.43, 115.62, 115.13, 115.38, 114.41, 100.39, 90.31, 44.09, 30.22; HRMS: calcd. for C22H16FN5O2+ [M-H+]: 400.1210, found: 400.1296.
7-Amino-3-(2-(4-fluorophenyl)-2-oxoethyl)-1-methyl-2,4-dioxo-5-phenyl-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3o) Yellow solid; Yield: 48.0%; 1H-NMR (CDCl3) δ 7.95 (dd, J = 8.8, J = 5.2 Hz, 2H), 7.46 (t, J = 3.6Hz, 3H), 7.26 (d, J = 9.2Hz, 2H), 7.12 (t, J = 8.6 Hz, 2H), 5.74 (s, 2H), 5.32 (s, 2H), 3.66 (s, 3H); 13C-NMR (DMSO-d6): δ 191.68, 166.53, 160.90, 160.12, 158.37, 154.23, 151.00, 137.43, 131.60, 131.53, 128.64, 128.23(2C), 127.68(3C), 116.55, 116.41, 115.65, 98.69, 89.53, 47.82, 30.20; HRMS: calcd. for C23H16FN5O3+ [M-H+]: 428.1159, found: 428.1105.
7-Amino-3-(2-(4-methoxyphenyl)-2-oxoethyl)-1-methyl-2,4-dioxo-5-phenyl-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3p) Yellow solid; Yield: 45.2%; 1H-NMR (CDCl3) δ 7.90 (d, J = 8.8 Hz, 2H), 7.46 (t, J = 4 Hz, 3H), 7.27 (d, J = 9.2 Hz, 2H), 6.91 (d, J = 8.8 Hz, 2H), 5.73 (s, 2H), 5.32 (s, 2H), 3.86 (s, 3H), 3.67 (s, 3H); 13C-NMR (DMSO-d6): δ 191.11, 164.13, 160.88, 160.11, 158.40, 154.22, 151.03, 137.46, 130.77(2C), 128.63, 128.23(2C), 127.69(2C), 115.68, 114.57(2C), 98.72, 89.50, 56.07(2C), 47.56, 30.18; HRMS: calcd. for C24H19N5O4+ [M-H+]: 440.1359, found: 440.1375.
7-Amino-1-methyl-2,4-dioxo-5-phenyl-3-(prop-2-ynyl)-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3q) White solid; Yield: 65.7%; 1H-NMR (DMSO-d6) δ 7.95 (s, 2H), 7.44 (t, J = 3 Hz, 3H), 7.25–7.23 (m, 2H), 7.44 (d, J = 1.6 Hz, 2H), 3.52 (s, 3H), 3.07 (s, 1H); 13C-NMR (DMSO-d6): δ 160.82, 160.13, 157.85, 154.17, 150.50, 137.52, 128.67, 128.28(2C), 127.69(2C), 115.66, 98.86, 89.41, 79.67, 73.45, 30.67, 30.18; HRMS: calcd. for C18H13N5O2+ [M-H+]: 330.0991, found: 330.0916.
7-Amino-3-cyclopentyl-1-methyl-2,4-dioxo-5-phenyl-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (3r) White solid; Yield: 48.3%; 1H-NMR (CDCl3) δ 7.51–7.49 (m, 3H), 7.25 (t, J = 4 Hz, 2H), 5.66 (s, 2H), 5.26–5.21 (m, 1H), 3.61 (s, 3H), 2.05–1.99 (m, 2H), 1.90–1.85 (m, 2H), 1.79–1.71 (m, 2H), 1.52–1.48 (m, 2H); 13C-NMR (DMSO-d6): δ 160.73, 160.02, 159.12, 154.08, 150.70, 137.96, 128.48, 128.22(2C), 127.64(2C), 115.80, 99.37, 89.10, 52.83, 29.88, 28.37(2C), 25.66(2C); HRMS: calcd. for C20H19N5O2+ [M-H+]: 360.1460, found: 360.1436.
7-Amino-3-butyl-1-methyl-2,4-dioxo-5-phenyl-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbo-nitrile (3s) White solid; Yield: 51.9%; 1H-NMR (CDCl3) δ 7.50 (t, J = 3.6 Hz, 3H), 7.25 (d, J = 2.8 Hz, 2H), 5.67 (s, 2H), 3.86 (t, J = 7.8 Hz, 2H), 3.64 (s, 3H), 1.56–1.48 (m, 2H), 1.31–1.25 (m, 2H), 0.89–0.86 (m, 3H); 13C-NMR (DMSO-d6): δ 160.23, 159.80, 158.64, 154.01, 151.00, 136.37, 129.14, 128.31(2C), 127.00(2C), 115.26, 100.59, 90.06, 41.69, 30.10, 29.74, 20.15, 13.75; HRMS: calcd. for C19H19N5O2+ [M-H+] m/z 348.1460, found: 348.1488.
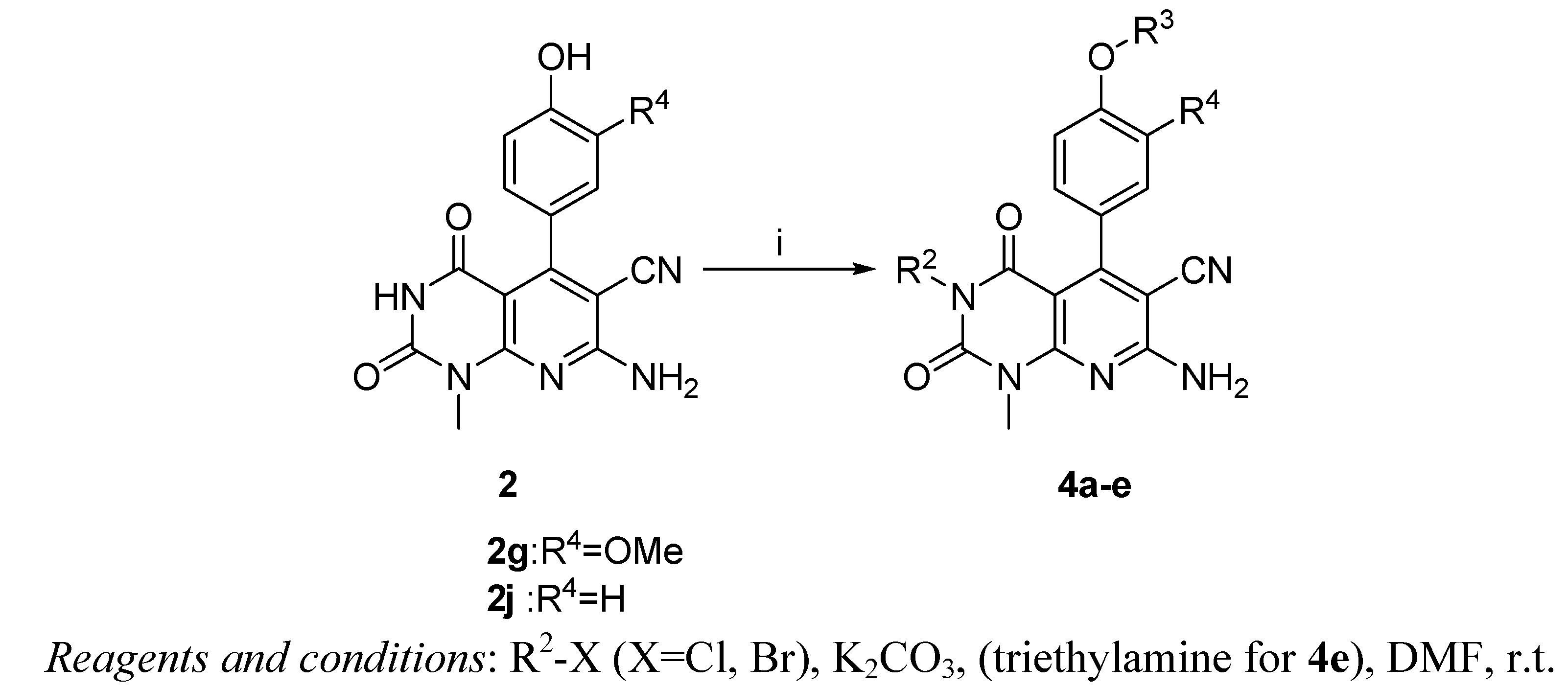
3.5. Synthesis of Compounds 4a–d
Compounds 2g or 2j (1 mmol) and anhydrous potassium carbonate were stirred at room temperature in DMF (5 mL). Reagents 1-(bromomethyl)-4-fluorobenzene or 2-bromo-1-(4-methoxyphenyl) ethanone (2.1 mmol) were added. After monitoring the completion of the reaction by TLC, the mixture was poured into water (25 mL). It was extracted with EA. The combined organic layer was washed with water, dried with Na2SO4, evaporated to obtain a residue, that was purified by column chromatography.
7-Amino-3-(4-fluorobenzyl)-5-(4-(4-fluorobenzyloxy)-3-methoxyphenyl)-1-methyl-2,4-dioxo-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (4a) White solid; Yield: 27.2%; 1H-NMR (CDCl3) δ 7.47–7.40 (m, 4H), 7.08 (t, J = 8.6 Hz, 2H), 7.00 (d, J = 8.4 Hz, 1H), 6.94 (t, J = 8.8 Hz, 2H), 6.84 (dd, J = 8.4 Hz, J = 2 Hz, 1H), 7.75 (d, J = 2 Hz, 1H), 5.67 (s, 2H), 5.15 (s, 2H), 5.05 (s, 2H), 3.83 (s, 3H), 3.62 (s, 3H); 13C-NMR (DMSO-d6): δ 163.04, 162.44, 160.62, 160.33, 160.03, 159.29, 158.17, 153.77, 150.78, 148.18, 147.93, 133.38, 133.24, 133.21, 130.25, 130.17, 129.64, 119.97, 115.56, 115.33, 115.12, 114.99, 114.78, 112.36, 112.06, 98.74, 88.91, 69.13, 55.51, 29.69; HRMS: calcd. for C30H23F2N5O4+ [M+H+]: 556.1796, found: 556.1856.
7-Amino-3-(4-fluorobenzyl)-5-(4-(4-fluorobenzyloxy)phenyl)-1-methyl-2,4-dioxo-1,2,3,4-tetrahydropyrido-[2,3-d]pyrimidine-6-carbonitrile (4b) White solid; Yield: 24.6%; 1H-NMR (CDCl3) δ 7.55 (t, J = 7.6 Hz, 1H), 7.36–7.00 (m, 11H), 5.72 (s, 2H), 5.20 (s, 2H), 5.19 (s, 2H), 3.64 (s, 3H); 13C-NMR (DMSO-d6): δ 161.47, 160.30, 159.71, 158.82, 154.20, 150.79, 129.95, 129.88, 129.82, 129.04, 128.95, 128.43, 124.34, 123.98, 123.96, 123.88, 123.78, 123.64, 123.54, 115.48, 115.34, 115.32, 114.44, 100.40, 90.39, 63.64, 63.61, 38.71, 30.24; HRMS: calcd. for C29H21F2N5O3+ [M+H+]: 526.1691, found: 526.1675.
7-Amino-5-(3-methoxy-4-(2-(4-methoxyphenyl)-2-oxoethoxy)phenyl)-3-(2-(4-methoxyphenyl)-2-oxo-ethyl)-1-methyl-2,4-dioxo-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (4c) White solid; Yield: 28.1%; 1H-NMR (DMSO-d6) δ 8.01 (dd, J = 8.8 Hz, J = 6.8 Hz, 4H), 7.07 (dd, J = 8.4 Hz, J = 5.2 Hz, 4H), 6.90–6.85 (m, 2H), 6.73(dd, J = 8.8 Hz, J = 2 Hz, 1H), 5.50 (s, 2H), 5.23 (s, 2H), 3.85 (s, 6H), 3.74 (s, 3H), 3.53 (s, 3H); 13C-NMR (DMSO-d6): δ 193.22, 191.18, 164.14, 164.02, 160.90, 159.84, 158.36, 154.22, 151.04, 148.50, 148.10, 130.77(4C), 130.01, 127.77, 127.71, 120.37, 115.97, 114.59(2C), 114.50(2C), 112.97, 112.64, 98.88, 89.67, 70.87, 56.13, 56.07, 47.61, 30.18; HRMS: calcd. for C34H29N5O8+ [M+Na+]: 658.1914, found: 658.1896.
7-Amino-5-(4-(2-(4-methoxyphenyl)-2-oxoethoxy)phenyl)-3-(2-(4-methoxyphenyl)-2-oxoethyl)-1-methyl-2,4-dioxo-1,2,3,4-tetrahydropyrido[2,3-d]pyrimidine-6-carbonitrile (4d) White solid; Yield: 21.7%; 1H-NMR (DMSO-d6) δ 8.01 (t, J = 9.2 Hz, 4H), 7.17 (d, J = 8.8 Hz, 2H), 7.10–7.03 (m, 4H), 6.96 (d, J = 8.8 Hz, 2H), 5.53 (s, 2H), 5.22 (s, 2H), 3.86 (s, 6H), 3.53 (s, 3H); 13C-NMR (DMSO-d6): δ 193.14, 191.15, 164.13, 164.03, 160.90, 160.50, 159.94, 158.58, 158.45, 158.14, 154.26, 151.02, 130.79(2C), 130.73(2C), 129.63, 129.46, 129.39, 127.72, 127.68, 114.98, 114.54(2C), 114.30(2C), 98.82, 89.66, 70.33, 56.07(2C), 47.58, 30.19; HRMS: calcd. for C33H27N5O7+ [M+H+]:606.1989, found: 606.1971.

 ; b NT denotes not tested; c Cis denotes cisplatin.
; b NT denotes not tested; c Cis denotes cisplatin.