Antimicrobial and Cytotoxic Isohexenylnaphthazarins from Arnebia euchroma (Royle) Jonst. (Boraginaceae) Callus and Cell Suspension Culture
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isolation of Isohexenylnaphthazarin Pigments

2.2. Antimicrobial Activity
| Zone of inhibition in cm | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| S. aureus | S. epidermidis | P. aeruginosa | K. pneumoniae | E. cloacae | E. coli | C. albicans | C. tropicalis | C. glabrata | |
| A. euchroma (callus n-hexane extract) | 15 | 15 | 12 | 11 | 11 | 11 | 10 | 11 | 12 |
| A. euchroma (cell suspension culture n-hexane extract) | 16 | 16 | 13 | 12 | 12 | 12 | 12 | 13 | 13 |
| 1 | 12 | 12 | 11 | 10 | 11 | 12 | 9 | 10 | 10 |
| 2 | 15 | 14 | 12 | 11 | 11 | 12 | 11 | 12 | 14 |
| 3 | 10 | 11 | 10 | 10 | 10 | 9 | 9 | 10 | 10 |
| 4 | 14 | 13 | 13 | 11 | 11 | 12 | 10 | 12 | 13 |
| 5 | 15 | 15 | 13 | 13 | 12 | 14 | 11 | 12 | 12 |
| 6 | 13 | 12 | 12 | 11 | 11 | 12 | 10 | 11 | 12 |
| 7 | 12 | 13 | 11 | 12 | 12 | 11 | 10 | 11 | 11 |
| 8 | 12 | 11 | 12 | 10 | 10 | 11 | 10 | 11 | 11 |
| 9 | 15 | 14 | 12 | 12 | 11 | 11 | 12 | 11 | 13 |
| Metilmycin | 22 | 24 | 20 | 25 | 23 | 22 | - | - | - |
| Amoxicillin with clavulanic acid | 21 | 21 | 25 | 23 | 22 | 24 | - | - | - |
| 5-Flucytocine | - | - | - | - | - | - | 22 | 24 | 22 |
| Amphotericin B | - | - | - | - | - | - | 23 | 24 | 24 |
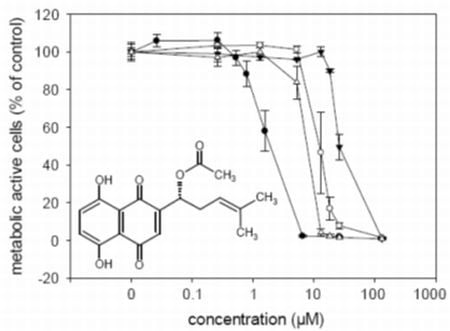
2.3. Cytotoxic Activities

| Compound | IC50 (µM) | |||
|---|---|---|---|---|
| CCRF-CEM | MDA-MB-231 | U251 | HCT 116 | |
| 1 | 2.31 ± 0.27 | 13.34 ± 1.35 | 15.01 ± 1.16 | 8.39 ± 1.20 |
| 2 | 1.67 ± 0.38 | 27.59 ± 8.25 | 41.70 ± 5.85 | 6.53 ± 0.87 |
| 3 | 0.98 ± 0.05 | 6.22 ± 0.46 | 19.17 ± 1.43 | 1.52 ± 0.05 |
| 4 | 0.53 ± 0.02 | 5.60 ± 0.36 | 16.39 ± 0.45 | 5.64 ± 0.14 |
| 5 | 3.32 ± 0.74 | 12.07 ± 0.13 | 69.70 ± 17.67 | 12.50 ± 1.05 |
| 6 | 0.39 ± 0.02 | 4.62 ± 0.14 | 3.17 ± 0.31 | 1.75 ± 0.06 |
| 7 | 1.22 ± 0.24 | 30.05 ± 5.92 | >100 µM | 7.97 ± 0.84 |
| 8 | 1.69 ± 0.15 | 12.21 ± 0.88 | 25.84 ± 0.66 | 6.83 ± 0.85 |
| 9 | 1.01 ± 0.09 | 11.33 ± 1.65 | 15.86 ± 0.67 | 9.02 ± 0.51 |
| VBN | 9.4 × 10−3 ± 2 × 10−4 | 3.1 × 10−2 ± 4.6 × 10−3 | 8.1 × 10−3 ± 1.0 × 10−3 | 8.7 × 10−3 ± 5.0 × 10−4 |
3. Experimental
3.1. Chemicals and Reagents
3.2. Instruments and Methods
3.3. Plant Callus and Cell Suspension Culture
3.4. Extraction and Isolation
3.5. Compound Characterization
3.6. Antimicrobial Activity
3.6.1. Microbial Strains
3.6.2. Antimicrobial Assays
3.7. Cell Culture
3.8. XTT Viability Assay
4. Conclusions
Acknowledgments
- Sample Availability: Samples of all isolated compounds are available from the authors.
References
- Papageorgiou, V.P.; Assimopoulou, A.N.; Couladouros, E.A.; Hepworth, D.; Nicolaou, K.C. The chemistry and biology of alkannin, shikonin and related naphthazarin natural products. Angew. Chem. Int. Ed. Engl. 1999, 38, 270–300. [Google Scholar] [CrossRef]
- Pietrosiuk, A.; Furmanowa, M.; Skopińska-Różewska, E.; Sommer, E.; Skurzak, H.; Bany, J. The effect of acetylshikonin isolated from Lithospermum canescens roots on tumor-induced cutaneous angiogenesis. Acta Pol. Pharm. 2004, 61, 379–382. [Google Scholar]
- Papageorgiou, V.P.; Assimopoulou, A.N.; Samanidou, V.F.; Papadoyannis, I.N. Recent advances in chemistry, biology and biotechnology of alkannins and shikonins. Curr. Org. Chem. 2006, 10, 2123–2142. [Google Scholar] [CrossRef]
- Papageorgiou, V.P.; Assimopoulou, A.N.; Samanidou, V.F.; Papadoyannis, I.N. Analytical methods for the determination of alkannins and shikonins. Curr. Org. Chem. 2006, 10, 583–622. [Google Scholar] [CrossRef]
- Davydenkov, V.N.; Patudin, A.V.; Popov, Y.G.; Rabinovich, S.A.; Miroshnikov, A.I. Cultured Arnebia euchroma (Royle) Jonst. cells, a new source of shikonin production. Khim. Farm. Zh. 1991, 1, 53–55. [Google Scholar]
- Hu, Y.; Jiang, Z.; Leung, K.S.Y.; Zhao, Z. Simultaneous determination of naphthoquinone derivatives in Boraginaceous herbs by high-performance liquid chromatography. Anal. Chim. Acta 2006, 577, 26–31. [Google Scholar] [CrossRef]
- Shukla, Y.N.; Srivastava, A.; Singh, S.C.; Kumar, S. New naphthoquinones from Arnebia hispidissima roots. Planta Med. 2001, 67, 575–576. [Google Scholar] [CrossRef]
- Cho, M.H.; Paik, Y.S.; Hahn, T.R. Physical stability of shikonin derivatives from the roots of Lithospεrmum erythrorhizon cultivated in Korea. J. Agric. Food Chem. 1999, 47, 4117–4120. [Google Scholar] [CrossRef]
- Cho, M.H.; Paik, Y.S.; Hahn, T.R. Propionylshikonin from the roots of Lithospermum erythrorhizon. Arch. Pharm. Res. 1999, 22, 414–416. [Google Scholar] [CrossRef]
- Özgen, U.; Coşkun, M.; Kazaz, C.; Seçen, H. Naphthoquinones from the roots of Onosma argentatum Hub.-Mor. (Boraginaceae). Turk. J. Chem. 2004, 28, 451–454. [Google Scholar]
- Weng, X.C.; Xiang, G.Q.; Jiang, A.L.; Liu, Y.P.; Wu, L.L.; Dong, X.W.; Duan, S. Antioxidant properties of components extracted from puccoon (Lithospermum erythrorizon Sieb. Et Zucc.). Food Chem. 2000, 69, 143–146. [Google Scholar] [CrossRef]
- Pietrosiuk, A.; Wiedenfeld, H. Shikonin derivatives from Lithospermum canescens. Pharm. Biol. 2005, 43, 189–191. [Google Scholar] [CrossRef]
- Shen, C.C.; Syu, W.J.; Li, S.Y.; Lin, C.H.; Lee, G.H.; Sun, C.M. Antimicrobial activities of naphthazarins from Arnebia euchroma. J. Nat. Prod. 2002, 65, 1857–1862. [Google Scholar] [CrossRef]
- Kirimer, N.; Bozan, B.; Başer, K.H.C. A new naphthaquinone from the roots of Arnebia densiflora. Fitoterapia 1995, 66, 499–500. [Google Scholar]
- Singh, B.; Sahu, P.M.; Jain, S.C.; Singh, S. Estimation of naphthaquinones from Arnebia hispidissima (Lehm.) DC. In vivo and In vitro. I. Anti-inflammatory screening. Phytother. Res. 2004, 18, 154–159. [Google Scholar] [CrossRef]
- Bozan, B.; Başer, K.H.C.; Kara, S. Quantitative determination of naphthaquinones of Arnebia densiflora (Nordm.) Ledeb. by an improved high-performance liquid chromatographic method. J. Chromatogr. A 1997, 782, 133–136. [Google Scholar] [CrossRef]
- Pouchert, C.J.; Behnke, J. The Aldrich Library of 13C- and 1H-FT-NMR Spectra, 1st ed; The Aldrich Chemical Company: Milwaukee, WI, USA, 1993; pp. 785–989. [Google Scholar]
- De-Eknamkul, W.; Potduang, B. Biosynthesis of β-sitosterol and stigmasterol in Croton sublyratus proceeds via a mixed origin of isoprene units. Phytochemistry 2003, 62, 389–398. [Google Scholar] [CrossRef]
- Papageorgiou, V.P.; Assimopoulou, A.N. Lipids of the hexane extract from the roots of medicinal Boraginaceous species. Phytochem. Anal. 2003, 14, 251–258. [Google Scholar] [CrossRef]
- Kawata, J.; Kameda, M.; Miyazawa, M. Cyclooxygenase-2 inhibitory effects and composition of the volatile oil from the dried roots of Lithospermum erythrorhizon. J. Nat. Med. 2008, 62, 239–243. [Google Scholar] [CrossRef]
- Santos, A.A.; Clososky, G.C.; Simonelli, F.; Oliveira, A.R.M.; Marques, F.A.; Zarbin, P.H.G. A new approach to the synthesis of (±)-methyl jasmonate and (±)-baclofen via conjugated addition of oxazoline cyanocuprate to Michael acceptors. J. Braz. Chem. Soc. 2001, 12, 673–679. [Google Scholar] [CrossRef]
- Nishiyama, H.; Sakuta, K.; Itoh, K. Conjugate addition of Lithiotrimethylsilyl acetate. A simple synthesis of methyl jasmonate via vicinal double alkylation. Tetrahedron Lett. 1984, 25, 2487–2488. [Google Scholar] [CrossRef]
- Ishida, Y.; Wakamatsu, S.; Yokoi, H.; Ohtani, H.; Tsuge, S. Compositional analysis of polyunsaturated fatty acid oil by one-step thermally assisted hydrolysis and methylation in the presence of trimethylsulfonium hydroxide. J. Anal. Appl. Pyrolysis 1999, 49, 267–276. [Google Scholar] [CrossRef]
- Cui, X.-R.; Tsukada, M.; Suzuki, N.; Shimamura, T.; Gao, L.; Koyanagi, J.; Komada, F.; Saito, S. Comparison of the cytotoxic activities of naturally occurring hydroxyanthraquinones and hydroxynaphthoquinones. Eur. J. Med. Chem. 2008, 43, 1206–1215. [Google Scholar] [CrossRef]
- Papageorgiou, V.P. Naturally occurring isohexenylnaphthazarin pigments: A new class of drugs. Planta Med. 1980, 38, 193–203. [Google Scholar] [CrossRef]
- Lu, Q.; Liu, W.; Ding, J.; Cai, J.; Duan, W. Shikonin derivatives: Synthesis and inhibition of human telomerase. Bioorg. Med. Chem. Lett. 2002, 12, 1375–1378. [Google Scholar] [CrossRef]
- Popova, M.P.; Chinou, Ι.Β.; Marekov, I.N.; Bankova, V.S. Terpenes with antimicrobial activity from Cretan propolis. Phytochemistry 2009, 70, 1262–1271. [Google Scholar] [CrossRef]
- Scudiero, D.A.; Shoemaker, R.H.; Paull, K.D.; Monks, A.; Tierney, S.; Nofziger, T.H.; Currens, M.J.; Seniff, D.; Boyd, M.R. Evaluation of a soluble tetrazolium/formazan assay for cell growth and drug sensitivity in culture using human and other tumor cell lines. Cancer Res. 1988, 48, 4827–4833. [Google Scholar]
- Rinner, B.; Kretschmer, N.; Knausz, H.; Mayer, A.; Boechzelt, H.; Hao, X-J.; Heubl, G.; Efferth, T.; Schaider, H.; Bauer, R. Α petrol ether extract of the roots of Onosma paniculatum induces cell death in a caspase dependent manner. J. Ethnopharmacol. 2010, 129, 182–188. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Damianakos, H.; Kretschmer, N.; Sykłowska-Baranek, K.; Pietrosiuk, A.; Bauer, R.; Chinou, I. Antimicrobial and Cytotoxic Isohexenylnaphthazarins from Arnebia euchroma (Royle) Jonst. (Boraginaceae) Callus and Cell Suspension Culture. Molecules 2012, 17, 14310-14322. https://doi.org/10.3390/molecules171214310
Damianakos H, Kretschmer N, Sykłowska-Baranek K, Pietrosiuk A, Bauer R, Chinou I. Antimicrobial and Cytotoxic Isohexenylnaphthazarins from Arnebia euchroma (Royle) Jonst. (Boraginaceae) Callus and Cell Suspension Culture. Molecules. 2012; 17(12):14310-14322. https://doi.org/10.3390/molecules171214310
Chicago/Turabian StyleDamianakos, Harilaos, Nadine Kretschmer, Katarzyna Sykłowska-Baranek, Agnieszka Pietrosiuk, Rudolf Bauer, and Ioanna Chinou. 2012. "Antimicrobial and Cytotoxic Isohexenylnaphthazarins from Arnebia euchroma (Royle) Jonst. (Boraginaceae) Callus and Cell Suspension Culture" Molecules 17, no. 12: 14310-14322. https://doi.org/10.3390/molecules171214310