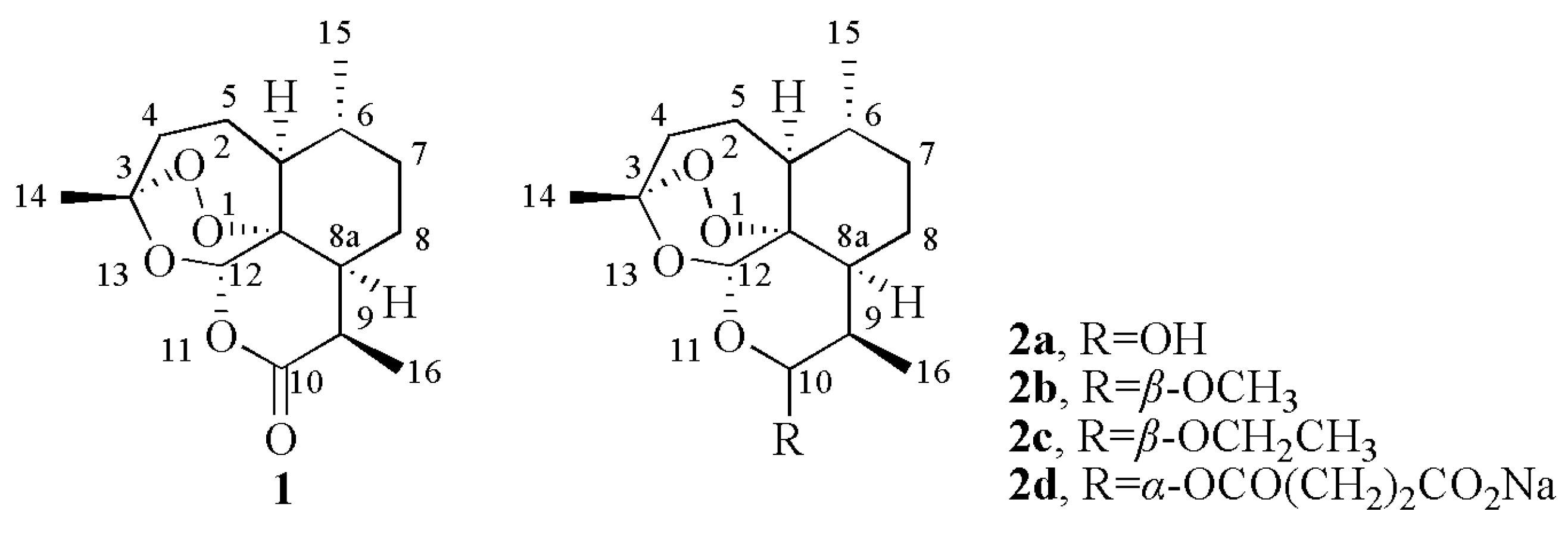
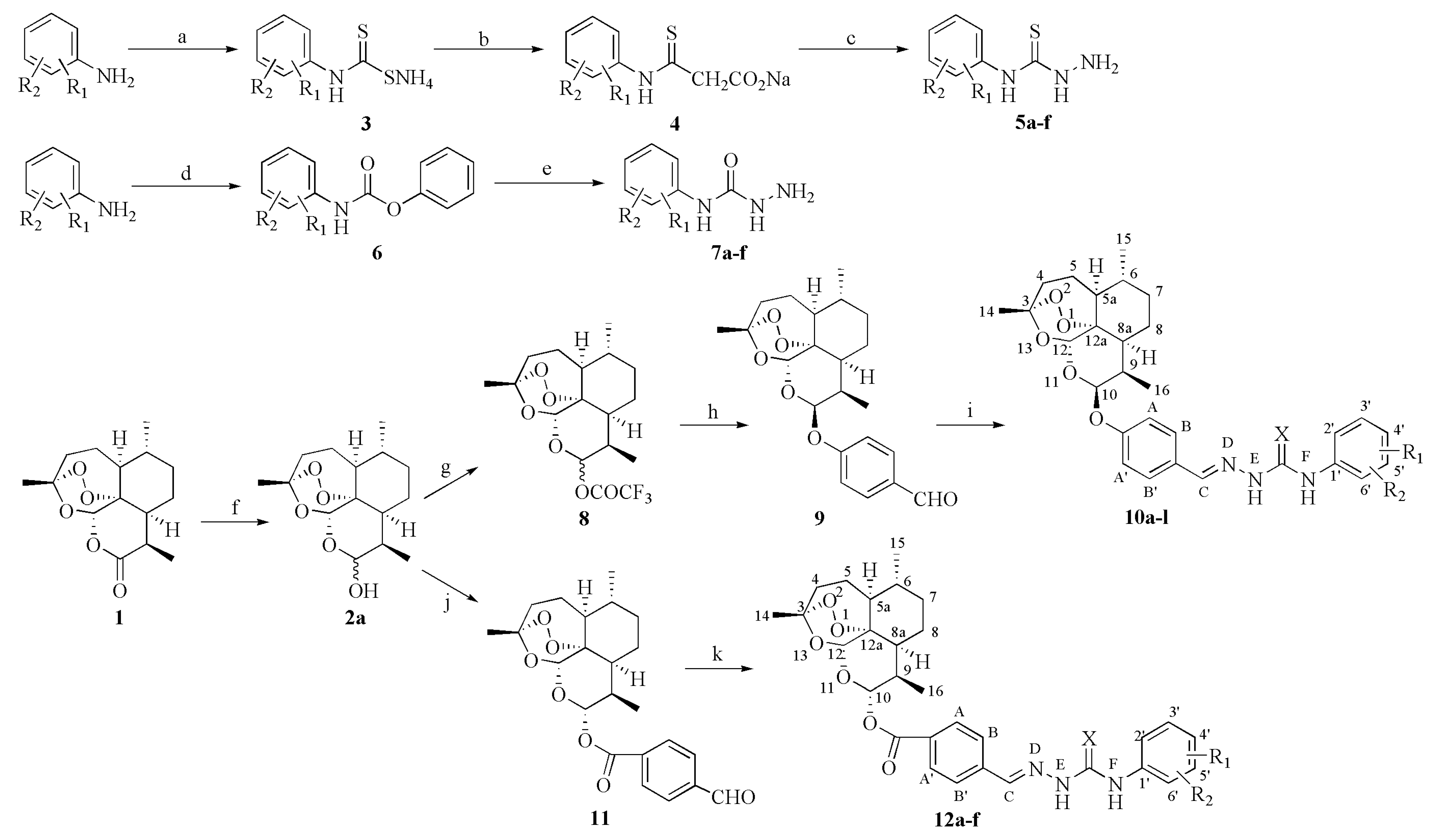
3.5. General Procedure for the Synthesis of 4-[(10S)-Dihydroartemisinin-10-oxy]benzaldehyde N4-(substitued phenyl)(thio)semicarbazones 10a-l
To a CH2Cl2 solution (50 mL) of dihydroartemisinin (2a, 11.36 g, 40 mmol) and triethylamine (11.08 mL, 80 mmol), TFAA (11.12 mL, 80 mmol) in CH2Cl2 (30 mL) was added dropwise at −5–0 °C. After 1 d, 4-hydroxylbenzaldehyde (9.76 g, 80 mmol) was added and stirred for 12 h at the same temperature. The reaction solution was quenched with saturated NaHCO3 solution and washed by saturated NaHCO3 solution (50 mL × 5) and water (50 mL × 5), respectively. The organic layer was dried (Na2SO4) and concentrated in vacuo. The residue was purified on silica gel column with petroleum ether/ethyl acetate (8:1) to afford 9. Yield 40.29%, m.p. 88–90 °C. MS (ESI) m/z: 411 (M+Na); 1H-NMR (300 MHz, CDCl3) δ: 0.97 (3H, d, J = 6.0 Hz, 16-CH3), 1.03 (3H, d, J = 7.2 Hz, 15-CH3), 1.45 (3H, s, 14-CH3), 2.34–2.44 (1H, m, 4-H), 2.83–2.88 (1H, m, 9-H), 5.44 (1H, s, 12-H), 5.62 (1H, d, J = 3.3 Hz, 10-H), 7.23 (2H, d, J = 8.7 Hz, AA’-2H), 7.84 (2H, d, J = 8.7 Hz, BB’-2H), 9.90 (1H, s, C-H); 13C-NMR (75 MHz, CDCl3) δ: 191.17, 162.65, 132.20, 131.03, 116.95, 104.63, 100.40, 88.65, 81.11, 52.74, 44.52, 37.69, 36.57, 34.85, 31.10, 26.29, 24.88, 24.67, 20.56, 13.13. Equimolar quantities of 9 (2 mmol) and 5a-f (2 mmol) or 7a-f (2 mmol) was dissolved in EtOH (20 mL) catalyzed by glacial acetic acid (3 drops) and stirred for 2 h at room temperature. The precipitate was filtered and purified on silica gel column with petroleum ether/ethyl acetate (3:1) to afford 10a-f or with CH2Cl2/CH3OH (80:1) to afford 10g-l.
4-[(10S)-Dihydroartemisinin-10-oxy]benzaldehyde N4-(2-fluorophenyl)thiosemicarbazone (10a). Yield 19.90%, m.p. 108–109 °C. MS (ESI) m/z: 556 (M+H); 1H-NMR (300 MHz, CDCl3) δ: 0.97 (3H, d, J = 6.0 Hz, 16-CH3), 1.03 (3H, d, J = 7.2 Hz, 15-CH3), 1.45 (3H, s, 14-CH3), 2.34–2.44 (1H, m, 4-H), 2.82–2.87 (1H, m, 9-H), 5.47 (1H, s, 12-H), 5.57 (1H, d, J = 3.0 Hz, 10-H), 7.16 (2H, d, J = 8.7 Hz, AA’-2H), 7.63 (2H, d, J = 9.0 Hz, BB’-2H), 7.84 (1H, s, C-H), 9.37 (1H, s, E-H), 9.53 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 176.32, 159.78, 143.30, 136.74, 134.31, 130.96, 129.30, 127.53, 127.22, 126.77, 117.25, 104.63, 100.58, 88.63, 81.25, 52.79, 44.61, 37.72, 36.63, 34.90, 31.21, 26.36, 24.92, 24.73, 20.62, 13.22.
4-[(10S)-Dihydroartemisinin-10-oxy]benzaldehyde N4-(4-fluorophenyl)thiosemicarbazone (10b). Yield 42.29%, m.p. 117–118 °C. MS (ESI) m/z: 556 (M+H); 1H-NMR (300 MHz, CDCl3) δ: 0.97 (3H, d, J = 6.0 Hz, 16-CH3), 1.03 (3H, d, J = 7.2 Hz, 15-CH3), 1.44 (3H, s, 14-CH3), 2.34–2.44 (1H, m, 4-H), 2.82-2.87 (1H, m, 9-H), 5.46 (1H, s, 12-H), 5.57 (1H, d, J = 3.0 Hz, 10-H), 7.16 (2H, d, J = 8.7 Hz, AA’-2H), 7.62 (2H, d, J = 9.0 Hz, BB’-2H), 7.85 (1H, s, C-H), 9.07 (1H, s, E-H), 9.68 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 175.82, 159.98, 143.54, 136.74, 131.76, 129.45, 129.17, 126.81, 126.11, 117.28, 104.64, 100.57, 88.64, 81.23, 52.78, 44.60, 37.73, 36.62, 34.90, 31.20, 26.35, 24.92, 24.74, 20.60, 13.20.
4-[(10S)-Dihydroartemisinin-10-oxy]benzaldehyde N4-(4-ethoxyphenyl)thiosemicarbazone (10c). Yield 31.80%, m.p. 114–116 °C. MS (ESI) m/z: 580 (M-H); 1H-NMR (300 MHz, CDCl3) δ: 0.96 (3H, d, J = 6.0 Hz, 16-CH3), 1.02 (3H, d, J = 7.2 Hz, 15-CH3), 1.44 (3H, s, 14-CH3), 2.33–2.44 (1H, m, 4-H), 2.81–2.86 (1H, m, 9-H), 4.05 (2H, q, J = 6.9 Hz, 4’-OCH2CH3), 5.46 (1H, s, 12-H), 5.55 (1H, d, J = 3.3 Hz, 10-H), 7.14 (2H, d, J = 8.7 Hz, AA’-2H), 7.60 (2H, d, J = 8.7 Hz, BB’-2H), 7.91 (1H, s, C-H), 9.02 (1H, s, E-H), 10.25 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 176.53, 159.79, 158.31, 143.24, 131.07, 129.34, 127.22, 117.22, 114.34, 104.62, 100.57, 88.62, 81.25, 55.77, 52.79, 44.62, 37.72, 36.63, 34.90, 31.21, 26.35, 24.92, 24.73, 20.60, 19.33, 13.20.
4-[(10S)-Dihydroartemisinin-10-oxy]benzaldehyde N4-(2,5-dimethylphenyl)thiosemicarbazone (10d). Yield 38.00%, m.p. 120–122 °C. MS (ESI) m/z: 566 (M+H); 1H-NMR (300 MHz, CDCl3) δ: 0.96 (3H, d, J = 5.7 Hz, 16-CH3), 1.02 (3H, d, J = 7.2 Hz, 15-CH3), 1.44 (3H, s, 14-CH3), 2.32 (3H, s, 2’-CH3), 2.37 (3H, s, 5’-CH3), 2.81–2.86 (1H, m, 9-H), 5.46 (1H, s, 12-H), 5.55 (1H, d, J = 3.3 Hz, 10-H), 7.14 (2H, d, J = 8.7 Hz, AA’-2H), 7.60 (2H, d, J = 8.7 Hz, BB’-2H), 7.90 (1H, s, C-H), 8.93 (1H, s, E-H), 10.13 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 176.91, 159.74, 143.29, 137.45, 134.48, 134.14, 131.69, 129.28, 127.64, 127.47, 127.29, 117.23, 104.62, 100.59, 88.62, 81.24, 52.79, 44.63, 37.72, 36.64, 34.92, 31.21, 26.36, 24.92, 24.74, 21.42, 20.61, 18.28, 13.21.
4-[(10S)-Dihydroartemisinin-10-oxy]benzaldehyde N4-(3,5-dimethylphenyl)thiosemicarbazone (10e). Yield 16.19%, m.p. 125–127 °C. MS (ESI) m/z: 566 (M+H); 1H-NMR (CDCl3) δ: 0.97 (3H, d, J = 5.7 Hz, 16-CH3), 1.03 (3H, d, J = 7.2 Hz, 15-CH3), 1.44 (3H, s, 14-CH3), 2.35 (6H, s, 3’-CH3, 5’-CH3), 2.81–2.86 (1H, m, 9-H), 5.46 (1H, s, 12-H), 5.56 (1H, d, J = 3.3 Hz, 10-H), 7.16 (2H, d, J = 8.7 Hz, AA’-2H), 7.61 (2H, d, J = 8.7 Hz, BB’-2H), 7.80 (1H, s, C-H), 9.08 (1H, s, E-H), 9.38 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 176.56, 159.77, 143.10, 137.43, 135.80, 135.15, 130.18, 129.32, 127.19, 126.46, 122.87, 117.23, 104.62, 100.58, 88.61, 81.24, 52.80, 44.62, 37.72, 36.64, 34.91, 31.21, 27.21, 26.35, 24.92, 24.73, 20.60, 19.68, 13.20.
4-[(10S)-Dihydroartemisinin-10-oxy]benzaldehyde N4-(3-chloro-2-methylphenyl)thiosemicarbazone (10f). Yield 26.44%, m.p. 150–152 °C. MS (ESI) m/z: 584 (M-H); 1H-NMR (CDCl3) δ: 0.97 (3H, d, J = 6.0 Hz, 16-CH3), 1.03 (3H, d, J = 7.2 Hz, 15-CH3), 1.44 (3H, s, 14-CH3), 2.39 (3H, s, 2’-CH3), 2.81–2.86 (1H, m, 9-H), 5.46 (1H, s, 12-H), 5.56 (1H, d, J = 3.3 Hz, 10-H), 7.15 (2H, d, J = 8.7 Hz, AA’-2H), 7.61 (2H, d, J = 8.7 Hz, BB’-2H), 7.89 (1H, s, C-H), 8.95 (1H, s, E-H), 10.00 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 177.06, 159.74, 143.29, 138.08, 136.65, 133.88, 129.28, 127.32, 126.04, 117.22, 104.62, 100.58, 88.62, 81.25, 52.79, 44.62, 37.72, 36.64, 34.91, 31.21, 26.36, 24.93, 24.74, 20.84, 20.62, 14.33, 13.22.
4-[(10S)-Dihydroartemisinin-10-oxy]benzaldehyde N4-(2-fluorophenyl)semicarbazone (10g). Yield 72.28%, m.p. 175–177 °C. MS (ESI) m/z: 538 (M-H); 1H-NMR (CDCl3) δ: 0.97 (3H, d, J = 6.0 Hz, 16-CH3), 1.04 (3H, d, J = 7.5 Hz, 15-CH3), 1.45 (3H, s, 14-CH3), 2.34–2.45 (1H, m, 4-H), 2.81–2.86 (1H, m, 9-H), 5.48 (1H, s, 12-H), 5.56 (1H, d, J = 3.3 Hz, 10-H), 7.16 (2H, d, J = 8.7 Hz, AA’-2H), 7.61 (2H, d, J = 8.7 Hz, BB’-2H), 7.77 (1H, s, C-H), 8.49 (1H, s, E-H), 8.98 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 159.21, 154.37, 141.72, 136.54, 130.66, 128.57, 128.05, 128.00, 127.17, 124.02, 121.44, 117.25, 104.60, 100.69, 88.63, 81.26, 52.82, 44.66, 37.73, 36.65, 34.93, 31.25, 26.39, 24.94, 24.75, 20.63, 13.25.
4-[(10S)-Dihydroartemisinin-10-oxy]benzaldehyde N4-(4-fluorophenyl)semicarbazone (10h). Yield 44.63%, m.p. 124–126 °C. MS (ESI) m/z: 538 (M-H); 1H-NMR (CDCl3) δ: 0.97 (3H, d, J = 6.0 Hz, 16-CH3), 1.04 (3H, d, J = 7.5 Hz, 15-CH3), 1.45 (3H, s, 14-CH3), 2.34-2.45 (1H, m, 4-H), 2.82–2.87 (1H, m, 9-H), 5.48 (1H, s, 12-H), 5.56 (1H, d, J = 3.0 Hz, 10-H), 7.16 (2H, d, J = 8.7 Hz, AA’-2H), 7.59 (2H, d, J = 8.7 Hz, BB’-2H), 7.74 (1H, s, C-H), 8.08 (1H, s, E-H), 8.76 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 159.27, 154.12, 142.40, 136.95, 129.28, 128.78, 127.72, 121.17, 117.20, 104.61, 100.63, 88.62, 81.26, 52.81, 44.64, 37.73, 36.64, 34.92, 31.24, 26.37, 24.94, 24.74, 20.61, 13.23.
4-[(10S)-Dihydroartemisinin-10-oxy]benzaldehyde N4-(4-ethoxyphenyl)semicarbazone (10i). Yield 52.15%, m.p. 158–159 °C. MS (ESI) m/z: 564 (M-H); 1H-NMR (CDCl3) δ: 0.97 (3H, d, J = 5.7 Hz, 16-CH3), 1.04 (3H, d, J = 7.2 Hz, 15-CH3), 1.42 (3H, t, J = 7.2 Hz, 4’-OCH2CH3), 1.45 (3H, s, 14-CH3), 2.34–2.44 (1H, m, 4-H), 2.81–2.86 (1H, m, 9-H), 4.03 (2H, q, J = 6.9 Hz, 4’-OCH2CH3), 5.48 (1H, s, 12-H), 5.55 (1H, d, J = 3.3 Hz, 10-H), 7.15 (2H, d, J = 8.7 Hz, AA’-2H), 7.58 (2H, d, J = 8.7 Hz, BB’-2H), 7.73 (1H, s, C-H), 7.97 (1H, s, E-H), 8.81 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 159.18, 156.34, 154.57, 141.83, 131.32, 128.66, 128.03, 122.24, 117.15, 114.53, 104.58, 100.64, 88.60, 81.27, 55.82, 52.81, 44.66, 37.72, 36.65, 34.93, 31.25, 26.37, 24.93, 24.74, 20.61, 19.50, 13.23.
4-[(10S)-Dihydroartemisinin-10-oxy]benzaldehyde N4-(2,5-dimethylphenyl)semicarbazone (10j). Yield 80.05%, m.p. 172–173 °C. MS (ESI) m/z: 548 (M-H); 1H-NMR (CDCl3) δ: 0.97 (3H, d, J = 5.7 Hz, 16-CH3), 1.03 (3H, d, J = 7.2 Hz, 15-CH3), 1.45 (3H, s, 14-CH3), 2.33 (3H, s, 2’-CH3), 2.36 (3H, s, 5’-CH3), 2.81–2.86 (1H, m, 9-H), 5.48 (1H, s, 12-H), 5.55 (1H, d, J = 3.3 Hz, 10-H), 7.16 (2H, d, J = 8.7 Hz, AA’-2H), 7.57 (2H, d, J = 8.7 Hz, BB’-2H), 7.76 (1H, s, C-H), 8.12 (1H, s, E-H), 8.95 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 159.15, 154.62, 141.61, 133.83, 131.36, 128.57, 127.63, 122.14, 117.22, 104.59, 100.70, 88.62, 81.26, 52.83, 44.67, 37.72, 36.65, 34.95, 31.25, 26.38, 24.74, 21.12, 20.62, 18.34, 13.24.
4-[(10S)-Dihydroartemisinin-10-oxy]benzaldehyde N4-(3,5-dimethylphenyl)semicarbazone (10k). Yield 84.60%, m.p. 169–171 °C. MS (ESI) m/z: 548 (M-H); 1H-NMR (CDCl3) δ: 0.97 (3H, d, J = 6.0 Hz, 16-CH3), 1.04 (3H, d, J = 7.2 Hz, 15-CH3), 1.45 (3H, s, 14-CH3), 2.33 (6H, s, 3’-CH3, 5’-CH3), 2.81–2.86 (1H, m, 9-H), 5.48 (1H, s, 12-H), 5.56 (1H, d, J = 3.3 Hz, 10-H), 7.16 (2H, d, J = 8.7 Hz, AA’-2H), 7.59 (2H, d, J = 8.7 Hz, BB’-2H), 7.76 (1H, s, C-H), 8.05 (1H, s, E-H), 9.01 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 159.19, 154.33, 141.79, 137.44, 135.94, 132.01, 130.30, 128.67, 128.04, 121.58, 117.71, 117.17, 104.59, 100.67, 88.60, 81.27, 52.83, 44.67, 37.73, 36.66, 34.94, 31.26, 26.37, 24.94, 24.75, 20.61, 20.24, 19.03, 13.23.
4-[(10S)-Dihydroartemisinin-10-oxy]benzaldehyde N4-(3-chloro-2-methylphenyl)semicarbazone (10l). Yield 92.97%, m.p. 177–179 °C. MS (ESI) m/z: 568 (M-H); 1H-NMR (CDCl3) δ: 0.97 (3H, d, J = 5.7 Hz, 16-CH3), 1.03 (3H, d, J = 7.5 Hz, 15-CH3), 1.45 (3H, s, 14-CH3), 2.81–2.86 (1H, m, 9-H), 5.48 (1H, s, 12-H), 5.56 (1H, d, J = 3.3 Hz, 10-H), 7.57 (2H, d, J = 8.7 Hz, BB’-2H), 7.76 (1H, s, C-H), 8.20 (1H, s, E-H), 8.98 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 159.15, 154.79, 141.71, 137.49, 136.14, 128.56, 128.24, 126.43, 126.24, 120.86, 117.21, 104.59, 100.69, 88.62, 81.26, 52.83, 44.67, 37.73, 36.66, 34.94, 31.25, 26.38, 24.94, 24.74, 21.03, 20.62, 13.73, 13.24.
3.6. General Procedure for the Synthesis of 4-[(10S)dihydroartemisinin-10-oxycarbonyl]benzaldehyde N4-(substitued phenyl)semicarbazones 12a-f
To a CH2Cl2 solution (100 mL) of dihydroartemisinin (2a, 11.36 g, 40 mmol) and 4-formylbenzoic acid (7.21 g, 48 mmol), DCC (9.90 g, 48 mmol) and DMAP (1.47 g, 12 mmol) were added at 0–5 °C for 1 h. After 1 d reaction at room temperature, the mixture was filtered and filtrate was concentrated in vacuo. The residue was purified on silica gel column with petroleum ether/ethyl acetate (6:1) to afford 11. Yield 40.46 %, m.p. 81–82 °C. MS (ESI) m/z: 439 (M+Na); 1H-NMR (CDCl3) δ: 0.94 (3H, d, J = 7.2 Hz, 16-CH3), 0.99 (3H, d, J = 5.7 Hz, 15-CH3), 1.43 (3H, s, 14-CH3), 2.35–2.45 (1H, m, 4-H), 2.74–2.81 1H, m, 9-H), 5.54 (1H, s, 12-H), 6.03 (1H, d, J = 9.6 Hz, 10-H), 7.97 (2H, d, J = 8.1 Hz, AA’-2H), 8.28 (2H, d, J = 8.1 Hz, BB’-2H), 10.12 (1H, s, C-H); 13C-NMR (75 MHz, CDCl3) δ: 190.89, 168.31, 162.30, 133.17, 117.52, 104.61, 100.73, 88.56, 81.13, 52.69, 44.62, 37.70, 36.63, 34.89, 31.10, 26.34, 24.72, 23.67, 20.60, 13.11. Equimolar quantities of 11 (2 mmol) and 7a-f (2 mmol) was dissolved in EtOH (20 mL) catalyzed by glacial acetic acid (3 drops) and stirred for 2 h at room temperature. The precipitate was filtered and purified on silica gel column with CH2Cl2/CH3OH (80:1) to afford 12a-f.
4-[(10S)-Dihydroartemisinin-10-oxycarbonyl]benzaldehyde N4-(2-fluorophenyl)semicarbazone (12a). Yield 17.62%, m.p. 145–147 °C. MS (ESI) m/z: 590 (M+Na); 1H-NMR (CDCl3) δ: 0.94 (3H, d, J = 7.2 Hz, 16-CH3), 0.99 (3H, d, J = 5.7 Hz, 15-CH3), 1.44 (3H, s, 14-CH3), 2.35–2.46 (1H, m, 4-H), 2.74–2.81 (1H, m, 9-H), 5.55 (1H, s, 12-H), 6.02 (1H, d, J = 9.9 Hz, 10-H), 7.72 (2H, d, J = 8.4 Hz, AA’-2H), 7.89 (1H, s, C-H), 8.14 (2H, d, J = 8.4 Hz, BB’-2H), 8.20 (1H, s, E-H), 9.46 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 166.32, 158.65, 154.63, 142.01, 136.62, 130.66, 128.63, 128.14, 128.03, 127.22, 124.03, 121.35, 117.33, 104.62, 100.70, 88.45, 81.27, 52.80, 44.51, 37.67, 36.73, 34.89, 31.20, 36.37, 24.90, 24.71, 20.59, 13.12.
4-[(10S)-Dihydroartemisinin-10-oxycarbonyl]benzaldehyde N4-(4-fluorophenyl)semicarbazone (12b). Yield 81.92%, m.p. 139–141 °C. MS (ESI) m/z: 590 (M+Na); 1H-NMR (CDCl3) δ: 0.93 (3H, d, J = 7.2 Hz, 16-CH3), 0.99 (3H, d, J = 5.7 Hz, 15-CH3), 1.43 (3H, s, 14-CH3), 2.35–2.45 (1H, m, 4-H), 2.73–2.81 (1H, m, 9-H), 5.55 (1H, s, 12-H), 6.02 (1H, d, J = 9.9 Hz, 10-H), 7.71 (2H, d, J = 8.4 Hz, AA’-2H), 7.91 (1H, s, C-H), 8.12 (2H, d, J = 8.1 Hz, BB’-2H), 8.13 (1H, s, E-H), 9.99 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 166.02, 159.32, 154.34, 142.36, 136.89, 129.33, 128.78, 128.91, 121.20, 117.15, 104.61, 100.60, 88.63, 81.32, 52.77, 44.56, 37.73, 36.55, 34.90, 31.22, 26.41, 24.91, 24.70, 20.62, 13.16.
4-[(10S)-Dihydroartemisinin-10-oxycarbonyl]benzaldehyde N4-(4-ethoxyphenyl)semicarbazone (12c). Yield 42.11%, m.p. 152–153 °C. MS (ESI) m/z: 616 (M+Na); 1H-NMR (CDCl3) δ: 0.93 (3H, d, J = 7.2 Hz, 16-CH3), 0.98 (3H, d, J = 5.1 Hz, 15-CH3), 1.43 (3H, s, 14-CH3), 2.35–2.45 (1H, m, 4-H), 2.73–2.80 (1H, m, 9-H), 4.04 (2H, q, J = 6.9 Hz, 4’-OCH2CH3), 5.54 (1H, s, 12-H), 6.01 (1H, d, J = 9.6 Hz, 10-H), 7.70 (2H, d, J = 8.1 Hz, AA’-2H), 7.90 (1H, s, C-H), 8.03 (1H, s, E-H), 8.12 (2H, d, J = 8.1 Hz, BB’-2H), 10.11 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 166.87, 159.52, 154.62, 142.78, 131.31, 128.70, 128.03, 122.17, 117.22, 114.51, 104.59, 100.60, 88.56, 81.32, 55.83, 52.79, 44.65, 37.71, 36.63, 34.90, 31.21, 26.36, 24.90, 24.72, 20.60, 19.45, 13.21.
4-[(10S)-Dihydroartemisinin-10-oxycarbonyl]benzaldehyde N4-(2,5-dimethylphenyl)semicarbazone (12d). Yield 85.69%, m.p. 155–156 °C. MS (ESI) m/z: 600 (M+Na); 1H-NMR (CDCl3) δ: 0.94 (3H, d, J = 7.2 Hz, 16-CH3), 0.99 (3H, d, J = 5.7 Hz, 15-CH3), 1.43 (3H, s, 14-CH3), 2.73–2.80 (1H, m, 9-H), 5.54 (1H, s, 12-H), 6.02 (1H, d, J = 9.6 Hz, 10-H), 7.67 (2H, d, J = 8.4 Hz, AA’-2H), 7.89 (1H, s, C-H), 8.11 (1H, s, E-H), 8.13 (2H, d, J = 8.4 Hz, BB’-2H), 9.68 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 167.14, 159.02, 154.61, 141.70, 134.23, 131.41, 128.48, 127.55, 121.11, 117.20, 104.62, 100.60, 88.56, 81.31, 52.78, 44.72, 37.66, 36.69, 34.90, 31.24, 26.42, 24.67, 21.10, 18.33, 13.21.
4-[(10S)-Dihydroartemisinin-10-oxycarbonyl]benzaldehyde N4-(3,5-dimethylphenyl)semicarbazone (12e). Yield 63.18%, m.p. 154–155 °C. MS (ESI) m/z: 600 (M+Na); 1H-NMR (CDCl3) δ: 0.93 (3H, d, J = 7.2 Hz, 16-CH3), 0.98 (3H, d, J = 5.7 Hz, 15-CH3), 1.43 (3H, s, 14-CH3), 2.73–2.80 (1H, m, 9-H), 5.54 (1H, s, 12-H), 6.01 (1H, d, J = 9.9 Hz, 10-H), 7.68 (2H, d, J = 8.4 Hz, AA’-2H), 7.93 (1H, s, C-H), 8.08 (1H, s, E-H), 8.11 (2H, d, J = 8.4 Hz, BB’-2H), 10.06 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 167.71, 159.22, 154.31, 141.77, 134.90, 132.01, 130.86, 128.66, 128.02, 121.56, 117.71, 117.20, 104.61, 100.68, 88.61, 81.32, 52.80, 44.73, 37.67, 36.70, 34.86, 31.20, 26.43, 24.91, 24.73, 20.60, 20.16, 19.21, 13.21.
4-[(10S)-Dihydroartemisinin-10-oxycarbonyl]benzaldehyde N4-(3-chloro-2-methylphenyl)semicarbaz-one (12f). Yield 63.54%, m.p. 173–174 °C. MS (ESI) m/z: 620 (M+Na); 1H-NMR (CDCl3) δ: 0.94 (3H, d, J = 6.9 Hz, 16-CH3), 0.98 (3H, d, J = 5.7 Hz, 15-CH3), 1.43 (3H, s, 14-CH3), 2.73–2.80 (1H, m, 9-H), 5.54 (1H, s, 12-H), 6.01 (1H, d, J = 9.9 Hz, 10-H), 7.67 (2H, d, J = 8.4 Hz, AA’-2H), 7.92 (1H, s, C-H), 8.12 (2H, d, J = 8.1 Hz, BB’-2H), 8.21 (1H, s, E-H), 10.19 (1H, s, F-H); 13C-NMR (75 MHz, CDCl3) δ: 166.32, 159.14, 154.81, 141.68, 137.11, 136.10, 128.61, 128.15, 126.42, 126.21, 120.94, 117.20, 104.56, 100.72, 88.61, 81.26, 52.80, 44.66, 37.70, 36.74, 34.92, 31.31, 26.42, 24.89, 24.70, 21.01, 20.64, 13.71, 13.20.