Synthesis, Characterization and Biological Activities of Cu(II), Co(II), Mn(II), Fe(II), and UO2(VI) Complexes with a New Schiff Base Hydrazone: O-Hydroxyacetophenone-7-chloro-4-quinoline Hydrazone
Abstract
:1. Introduction
2. Results and Discussion
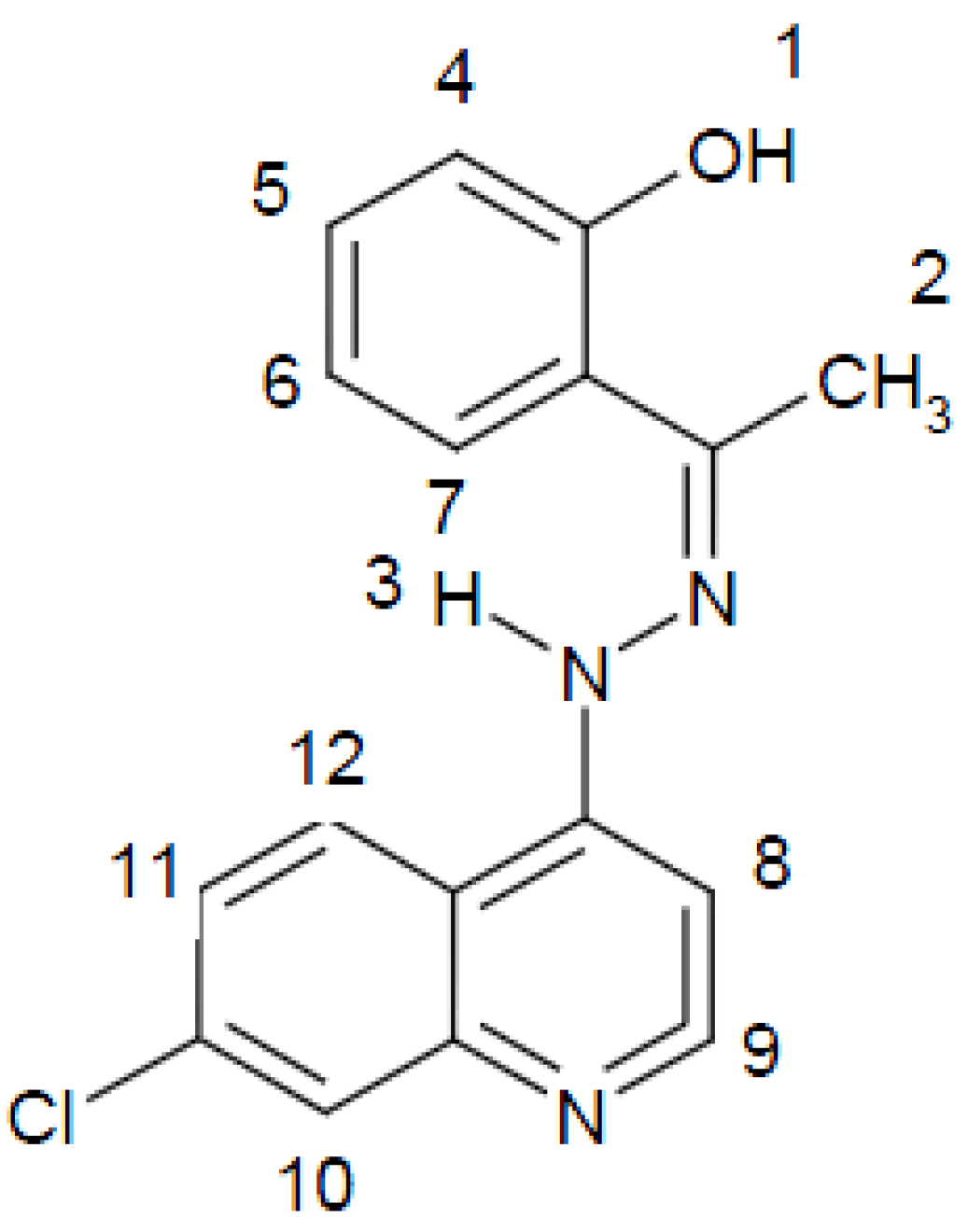
2.1. Characterization of the Ligand
| Compound | F.W. | Color | Yield(%) | M.P. (°C) | Elemental analysis, Found / (Calcd) % | Solubility | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| C | H | N | CL | M | ||||||
| C17H14N3OCl (HL) | 311.5 | Deep Orange | 66 | 225 | 65.37 (65.49) | 4.39 (4.49) | 13.48 (13.48) | 11.30 (11.4) | -- | Soluble in most common organic solvent |
| [(HL)2Cu].EtOH (1) C36H32N6O3Cl2Cu | 731 | Green | 33 | 287 | 59.00 (59.10) | 4.27 (4.38) | 11.29 (11.49) | 9.51 (9.71) | 8.66 (8.76) | Soluble in acetone and insoluble in methanol and ethanol |
| [Ni (HL)2(OH2)2] 2EtOH (2) C28H42N6O6Cl2Ni | 808 | Pale Green | 25 | 220 | 56.24 (56.44) | 5.13 (5.20) | 10.40 (10.40) | 8.79 (8.79) | 7.30 (7.30) | Soluble in most common organic solvent |
| [(L)2Ni2(OH2)6]a (3) C34H36N6O8Cl2Ni2 | 845 | Yellowish Green | 47 | 250 (decomp) | 48.15 (48.28) | 4.07 (4.26) | 9.75 (9.94) | 8.08 (8.28) | 13.67 (13.96) | Soluble in acetone and insoluble in methanol and ethanol |
| [(L)2Co2(OH2)6]a (4) C34H36N6O8Cl2Co2 | 845 | Brown | 34 | 260 (decomp) | 48.00 (48.18) | 4.50 (4.28) | 10.12 (9.99) | 8.07 (8.26) | 13.79 (13.98) | Soluble in most common organic solvent except diethyl ether |
| [(L)2Mn(OH2)6]a (5) C34H36N6O8Cl2Mn2 | 837 | Deep brown | 48 | 225 | 49.00 (48.75) | 4.50 (4.30) | 9.10 (10.04) | 8.27 (8.36) | 12.97 (13.14) | Soluble in acetone and insoluble in methanol and ethanol and ether |
| [(HL)2UO2(EtOH)] (6) C36H32N6O5Cl2U | 937 | Red | 75 | 225 (decomp) | 46.31 (46.10) | 3.51 (3.42) | 9.21 (8.96) | 7.73 (7.58) | 25.20 (25.40) | Soluble in most common organic solvent except diethylether |
| [(L)2Fe2Cl2(OH2)4] 2EtOHa (7) C38H44N6O8Cl4Fe2 | 966 | Reddish brown | 55 | 250 (decomp) | 47.35 (47.20) | 4.26 (4.55) | 8.58 (8.70) | 14.52 (14.70) | 11.45 (11.59) | Soluble in most common organic solvent except diethylether |
| Chemical shift, δTMS (ppm) | Assignment a |
|---|---|
| 14.5 | [s, 1H] (1) |
| 2.4 | [s, 3H] (2) |
| 11.7 | [s, 1H] (3) |
| 7.9–8.3 | [m, 9H, Ar–H and quinoline–H] |
| Compound | ν(C=N) | ν(N-H) | ν(N-N) | ν(M-N) | ν(M-O) | ν(OH), H2O or alcohol | Other bands |
|---|---|---|---|---|---|---|---|
| HL | 1524 s | 3300 m | 1140s | --- | --- | 3530m, br (νOH-phenolic) | 1278 (δ OH-phenolic) |
| [(HL)2Cu] EtOH (1) | 1520 s | 3215 m | 1136 s | 420 w | 540 m | 3426 m, br (νOH-alcohol | --- |
| [(HL)2Ni(OH 2)2] 2EtOH (2) | 1510 m | 3200 s | 1137 w | 425 w | 520 m | 3440 m, br (νOH-coordinated water over-lapped with (νOH- alcohol | --- |
| [(L)2Ni2(OH2)6] (3) | 1514 sh | --- | 1125 w | 410 w | 515 m | 3436 m, br (νOH-coordinated water | --- |
| [(L)2Co2(OH)2)6] (4) | 1527 sh | --- | 1136 S | 445W | 520W | 3438 m, br (νOH-coordinated water | --- |
| [(L)2Mn(OH2)6] (5) | 1507 m | --- | 1137 s | 465 w | 520 m | 3439 m, br (νOH-coordinated water | --- |
| [(HL)2UO2(EtOH)] (6) | 1505 | 3214 sh | 1134 s | 460 w | 540 w | 3435 m, br (νOH-coordinated water over-lapped with (νOH-coordinated alcohol | 901 s ν3(O=U=O |
| [(L)2Fe2Cl2(OH2)4] 2EtOH (7) | 1515 s | --- | 1138 W | 460 W | 515 W | 3435 m, br (νOH-coordinated water overlapped with (νOH- alcohol | --- |
| Compound | μeff.b B.M. | μcomble c found (expected d)B.M. | *π→π, n→π* and charge transfer transitions | d→dTransition(cm−1) | EC f |
|---|---|---|---|---|---|
| HL | --- | --- | 48067, 44642, 36231, 26176,24752 | --- | --- |
| [(HL)2Cu] EtOH (1) | 1.807 | 1.807 | 36765 ,28612, 22396 | 15850 | 2.4 |
| [HL)2Ni(OH 2)2] 2EtOH (2) | 2.85 | 2.85 | 37037, 28653, 22371 | 15850 | 2.0 |
| [(L)2Ni2(OH2)6] (3) | --- | 4.44 (5.71) | 37437, 28670,22300 | 1518.45, 11957 | 2.6 |
| [(L)2Co2(OH)2)6] (4) | --- | 7.12 1(10.4) | 370307, 28600, 22271 | 15250, 15390 | 2.4 |
| [(L)2Mn(OH2)6] (5) | --- | 5.39 (8.40) | 37085, 28500, 22471 | 21331.9, 11733.33 | 3.6 |
| [(HL)2UO2(EtOH)] (6) | --- | --- | 37537, 27853, 22871 | 22500, 19040 | 1.5 |
| [(L)2Fe2Cl2(OH2)4] 2EtOH (7) | --- | 7.49 (10.80) | 37137, 27653, 22571 | 14331, 13850 | 68 |
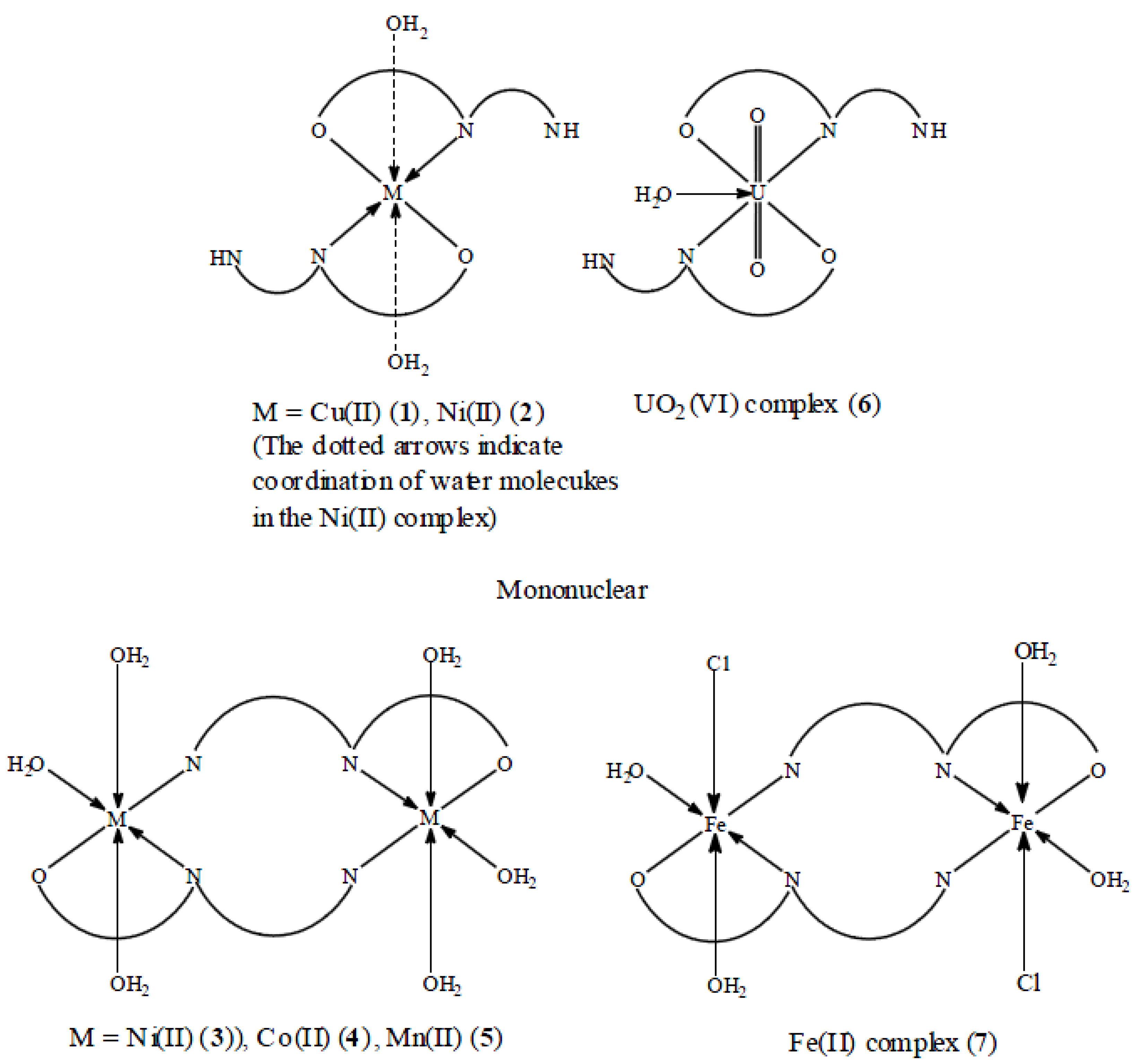
2.2. Metal Complexes
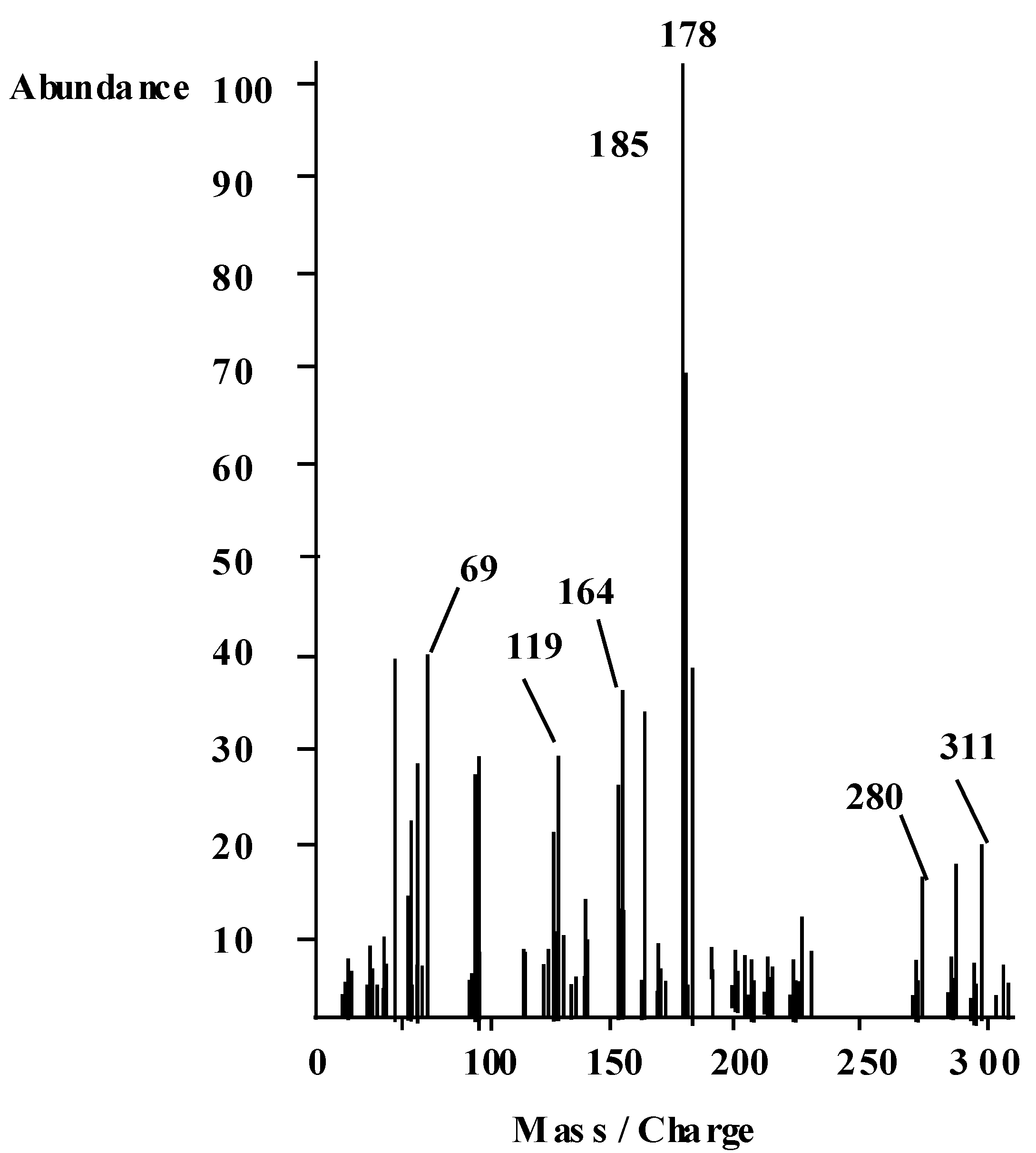
2.3. IR Spectra of the Metal Complexes
2.4. Magnetic Moments and Electronic Spectral Data of the Metal Complexes
2.5. Molar Conductance of the Metal Complexes
2.6. 1H-NMR Spectrum of the Uranyl Complex
- (1). The disappearance of the signal due to the phenolic OH group is attributed to its involvement in coordinating the uranylcation, While the signal due to the NH group was broad and appeared at δ = 12.4 ppm compared to that of the ligand which appeared at, δ =11.7 ppm, i.e., shifted to low-field.
- (2). The signals due to the aromatic ring showed fine structure and appear as four separate signals at δ = 7.14, 7.53 and 7.6 ppm.
- (3). The NH group which did not take part in coordinating the uranylcation disappeared on deuteration.
- (4). The CH3 group signal remained unchanged as observed in the parent ligand.
2.7. Antimicrobial Activity and Minimum Inhibitory Concentration (MIC)
| Compound | Conc.% | Micro-organism | ||
|---|---|---|---|---|
| S. aureus ATCC * 6538MIC *** 25% | E. coli ATCC 8739 MIC 25% | C. albicans ATCC 10231MIC 25% | ||
| o-Hydroxyacetophenone-7-chloro-4-quinoline hydrazine (HL) | 1 ** | |||
| 25 | +ve | −ve | +ve | |
| 50 | +ve | −ve | +ve | |
| 100 | +ve | +ve | +ve | |
| [(HL)2Cu] EtOH (1) | 1 | +ve | +ve | +ve |
| 25 | +ve | +ve | +ve | |
| 50 | +ve | +ve | −ve | |
| 100 | −ve | −ve | −ve | |
| [HL)2Ni(OH2)2] 2EtOH (2) | 1 | +ve | −ve | −ve |
| 25 | +ve | +ve | +ve | |
| 50 | +ve | +ve | +ve | |
| 100 | −ve | −ve | −ve | |
| [(L)2Ni2(OH2)6] (3) | 1 ** | |||
| 25 | −ve | +ve | −ve | |
| 50 | −ve | −ve | −ve | |
| 100 | −ve | −ve | −ve | |
| [(L)2Co2(OH2)6] (4) | 1 | +ve | +ve | +ve |
| 25 | −ve | +ve | −ve | |
| 50 | −ve | −ve | −ve | |
| 100 | −ve | −ve | −ve | |
| [(L)2Mn(OH2)6] (5) | 1 ** | |||
| 25 | −ve | +ve | −ve | |
| 50 | −ve | −ve | −ve | |
| 100 | −ve | −ve | −ve | |
| [(HL)2UO2(EtOH)] (6) | 1 | +ve | +ve | +ve |
| 25 | −ve | +ve | −ve | |
| 50 | −ve | +ve | −ve | |
| 100 | −ve | −ve | −ve | |
| [(L)2Fe2Cl2(OH2)4].2EtOH (7) | 1 | +ve | +ve | +ve |
| 25 | −ve | +ve | −ve | |
| 50 | −ve | −ve | −ve | |
| 100 | −ve | −ve | −ve | |
| 7-Chloro-4-hydrazinoquinoline | 1 | +ve | +ve | +ve |
| 25 | −ve | −ve | −ve | |
| 50 | −ve | −ve | −ve | |
| 100 | −ve | −ve | −ve | |
3. Experimental
3.1. General
3.2. Synthesis of the Schiff Base Hydrazone Ligand (HL)
3.2.1. Synthesis of 7-Chloro-4-hydrazinoquinoline
3.2.2. Synthesis of the ligand HL
3.3. Synthesis of the Metal Complexes
3.3.1. Cu(II) Complex, [(HL)2Cu] EtOH
3.3.2. Ni(II) Complexes
3.3.2.1. In the Absence of LiOH, [(HL)2Ni(OH2)2] 2EtOH
3.3.2.2. In the Presence of LiOH, [(L)2Ni(OH2)6]
3.3.3. Co(II) Complex, [[(L)2CO2(OH2)6]
3.3.4. Mn(II)Complex, [(L)2Mn2(OH2)6]
3.3.5. UO2(VI) Complex, [(HL)2UO2(EtOH)]
3.3.6. Fe(III) Complex, [(L)2Fe2Cl2(OH)4] EtOH
3.4. Synthesis of Samples for Microbiological Analysis
3.5. Physical Measurements and Analysis
3.6. Pharmacology
4. Conclusions
- Samples Availability: Samples are available from the author.
References and Notes
- Chohan, Z.H.; Sheazi, S.K.A. Synthesis and characterization of some Co(II), Cu(II) and Ni(II) complexes with nicolinylhydrazine derivatives and their biological role of metals and anions (SO42−, NO3−, C2O42− and CH3CO2−) on the antibacterial properties. Synth. React. Inorg. Met.-Org. Chem. 1999, 29, 105–118. [Google Scholar] [CrossRef]
- Jayabalakrishnan, C.; Natarajan, K. Synthesis, characterization and biological activities of ruthenium(II) carhonyl complexes containing bifunctional tridentate Schiff bases. Synth. React. Inorg. Met.-Org. Chem. 2011, 983–995. [Google Scholar]
- Jeeworth, T.; Wah, H.L.; Bhowon, K.; Ghoorhoo, M.G.; Babooram, D. Synthesis and antibacterial/catalytic properties of schiff bases and Schiff base metal complexes derived from 2,3-diaminopyridine. Synth. Read. Inorg. Met.-Org. Chem. 2000, 30, 1023–1038. [Google Scholar] [CrossRef]
- Dharmaraj, N.; Viswanalhamurthi, P.; Natarajan, K. Ruthenium(Il) complexes containing bidentate Schiff bases and their antifungal activity. Trans. Met. Chem. 2000, 26, 105–109. [Google Scholar]
- Colins, C.H.; Lyne, P.M. Microhiul Melfwds; University Park Press: Baltimore, MD, USA, 1970; p. 422. [Google Scholar]
- Savanini, L.; Chiasserini, L.; Gaeta, A.; Pellerano, C. Synthesis, anti-tubercular, evaluation of 4-quinolylhydrazones. Bioorg. Med. Chem. 2002, 10, 2193–2198. [Google Scholar] [CrossRef]
- Sankar, R.; Vijayalakshmi, S.; Rajagopan, S.; Kaliyappan, T. Synthesis, spectral, thermal, and chelation potentials of polymeric hydrazone based on 2,4-dihydroxy benzophenone. J. Appl. Polym. Sci. 2010, 117, 2146–2152. [Google Scholar]
- Albertini, R.; Pinelli, S.; Lunghi, P. Synthesis, structural characterization, biological activity of helicinthiosemicarbazone monohydrate and a copper(II) complex of salicyaldehyde thiosemicarbazone. Inorg. Chim. Acta 1999, 286, 134–141. [Google Scholar] [CrossRef]
- Al-Shaalan, N. Antimicrobial activity and spectral, magnetic and thermal studies of some transition metal complexes of a Schiff base hydrazone containing a quinoline moiety. Molecules 2007, 12, 1080–1091. [Google Scholar] [CrossRef]
- Elo, H.; Sunila, I.; Lumme, P. Studies on the acute toxicity of antineoplastic metal chelate, trans-bis (salicyaldoximato) coppery(II) in rats. Inorg.Chim. Acta 1987, 136, 61–63. [Google Scholar] [CrossRef]
- Ali, M.; Kabir, M.H.; Nazimuddin, M.; Majumder, S.M.; Tarafder, M.T.; Akhair, M. Synthesis, characterization and antiiungal properties of some four-coordinate nickel(II) and four- and five-coordinate copper(II) complexes containing tridentate thiosemicarbazones and heterocyclic bases. Indian J. Chem. 1988, 27A, 1064–1067. [Google Scholar]
- Kakimoto, S.; Yashamoto, K. Studies on antitubercular compounds. X. Condensation products of aldehydes and acid hydrazides of pyridine group. Pharm. Bull. 1956, 4, 4. [Google Scholar]
- Agarwal, R.K.; Singh, L.; Sharma, D.K.; Singh, R. Synthesis, spectral and thermal investigations of some oxovanadium(IV) complexes of isonicotinic acid hydrazide. Turk. J. Chem. 2005, 29, 309–310. [Google Scholar]
- Hill, H.A.; Zarb-Adami, N. Some nickel(II) complexes of n-hydroxyethyl and n-hydroxypropylsalicyaldimines. J. Inorg. Nucl. Chem. 1975, 37, 2443–2447. [Google Scholar] [CrossRef]
- Salib, K.A.; Iskak, M.F.; El-Behairy, M.; Abd El-Halim, H.F. Synthesis, spectroscopic studies of a novel Schiff base Derived from o-acetoacetylphenol and ethylamine and its metal complexes. Synth. React. Inorg. Met.-Org. Chem. 2003, 33, 1667–1687. [Google Scholar] [CrossRef]
- El-Behery, M.; El-Twigry, H. Synthesis, magnetic, s pectral, and antimicrobial studies of Cu(II), Ni(II) Co(II), Fe(III), and UO2(II) complexes of a new Schiff base hydrazone derived from 7-chloro-4-hydrazinoquinoline. Spectrochim. Acta Part A 2007, 66, 28–36. [Google Scholar] [CrossRef]
- Williams, D.H.; Fleming, I. Spectroscopic Methods in Organic Chemistry; McGraw-Hill: New York, NY, USA, 1966; p. 94. [Google Scholar]
- Silverstein, R.M.; Bassler, G.C.; Morrill, T.C. Spectrometric Identification of Organic Compounds; John Wiley & Sons: New York, NY, USA, 1991. [Google Scholar]
- Dyer, J.R. Applications of Absorption Spectroscopy of Organic Compounds; Prentice Hall: London, UK, 1965; p. 15. [Google Scholar]
- Ueno, K.; Martel, A.E. Ultraviolet and visible absorption spectra of metal chelates of bisacetylacetoneethylenediimine and related compounds. J. Phys. Chem. 1957, 61, 257–261. [Google Scholar] [CrossRef]
- Bailer, J.C.; Emeleus, H.J.; Nyholm, R.; Trotman-Dickinson, A.F. Comprehensive Inorganic Chemistry; Pergamon Press: Oxford, UK, 1975; p. 517, 1153, 1088, 1048. [Google Scholar]
- Casellato, U.; Fregona, D.; Sitran, S.; Tamburini, S.; Vigato, P.A. Preparation and properties of mono, homo- and heterobinuclear complexes with a new heptadentate Schiff base ligand. Inorg. Chim. Acta 1984, 95, 306–316. [Google Scholar]
- Figgs, B.N. Introduction to Ligand Field; Wiley: New York, NY, USA, 1966. [Google Scholar]
- Lever, A.B.P. Inorganic Electronic Spectroscopy, 2nd ed; Elsevier: Amsterdam, The Netherlands, 1984. [Google Scholar]
- Gruber, S.J.; Harris, C.M.; Sinn, E. Metal complexes as ligands. IV. Bi-and tri-nuclear complexes derived from metal complexes of tetradentate salicylaldimines. J. Inorg. Nucl. Chem. 1968, 30, 1805–1830. [Google Scholar] [CrossRef]
- Baldwin, M.E. Sulphitobis(ethylenediamine)cobalt(III) complexes. J. Chem. Soc. 1961, 3123–3128. [Google Scholar] [CrossRef]
- William, H.; Stephen, V. Theory and Application of Microbiological Assay; Academic Press: San Diego, CA, USA, 1989; p. 320. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Al-Shaalan, N.H. Synthesis, Characterization and Biological Activities of Cu(II), Co(II), Mn(II), Fe(II), and UO2(VI) Complexes with a New Schiff Base Hydrazone: O-Hydroxyacetophenone-7-chloro-4-quinoline Hydrazone. Molecules 2011, 16, 8629-8645. https://doi.org/10.3390/molecules16108629
Al-Shaalan NH. Synthesis, Characterization and Biological Activities of Cu(II), Co(II), Mn(II), Fe(II), and UO2(VI) Complexes with a New Schiff Base Hydrazone: O-Hydroxyacetophenone-7-chloro-4-quinoline Hydrazone. Molecules. 2011; 16(10):8629-8645. https://doi.org/10.3390/molecules16108629
Chicago/Turabian StyleAl-Shaalan, Nora H. 2011. "Synthesis, Characterization and Biological Activities of Cu(II), Co(II), Mn(II), Fe(II), and UO2(VI) Complexes with a New Schiff Base Hydrazone: O-Hydroxyacetophenone-7-chloro-4-quinoline Hydrazone" Molecules 16, no. 10: 8629-8645. https://doi.org/10.3390/molecules16108629




