1. Introduction
Of the 20 natural proteinogenic amino acids, 19 are chiral, and being readily commercially available, they are potentially useful educts for enantiospecific synthesis. Typically the nitrogen atom needs to be protected to allow further manipulations of the carboxylic acid center. Common nitrogen protecting groups include removable alkyl groups such as benzyl and substituted benzyl groups, or more frequently carbamates such as tert-butyl, benzyl or fluorenylmethyl carbamates (Boc, Cbz and Fmoc, respectively). Common nitrogen protecting groups do not fare well in guarding the enantiomeric purity of α-chiral amino carbonyl compounds, because they fail to shield the acidic α-proton from removal. Surprisingly little attention has been paid to the fact that most of the chemistry emanating from these valuable chiral pool educts often leads to the destruction of significant amounts of the chiral information and even under carefully controlled conditions some erosion of enantiopurity is observed. The 9-phenylfluoren-9-yl (Pf) group has proven to be an outstanding choice for protecting the α-center of the amino acid derivatives against epimerization and some 250 papers and patents have been published on the chemistry of the Pf group acting as a protecting group on nitrogen. Rather than offering a comprehensive review, we have selected representative references to highlight the pros and cons of the chemistry based on Pf protection.
2. The Phenylfluorenyl Group
2.1. Generalities
Structurally the phenylfluorenyl (Pf) group resembles the trityl protecting group. Trityl groups were first used to protect amines instead of
t-butyl and phenylmethyl carbamates (Fmoc, Boc, Cbz) in order to protect the α-center, and
N-trityl amino compounds were observed to retain their enantiopurity with >90% ee [
1]. However, the resulting tritylamines are acid labile, thus rendering them of limited use, and alternative protecting groups of similar size were needed. The Pf group, known to be solvolytically >6,000 times more acid stable than trityl [
2] entered the picture in the 1980s [
3].
The strength of the Pf group lies in its sterically demanding size that protects the α-center. It has been calculated that in an
N-Pf α-amino acid derivative, the steric bulk of the Pf group forces the compound to adopt a conformation in which the dihedral angle between the carbonyl group and the α-hydrogen is ~ 0° or 180° [
4]. In cyclic compounds the Pf group drives the α-hydrogen into an equatorial position [
5]. (
Figure 1 and
Figure 2)
Figure 1.
Conformational analysis of cyclic and acyclic compounds.
Figure 1.
Conformational analysis of cyclic and acyclic compounds.
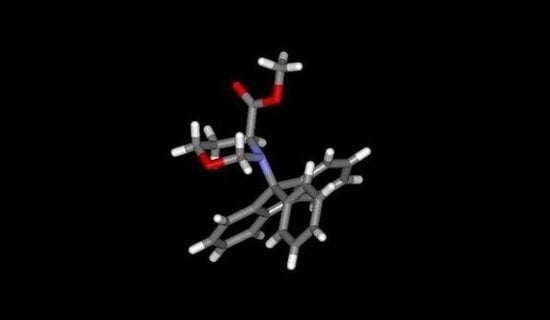
Figure 2.
Crystal structure of
N-Pf alanine
tert-butyl ester [
6].
Figure 2.
Crystal structure of
N-Pf alanine
tert-butyl ester [
6].
Not only the steric bulk of the Pf group, but also stereoelectronics may play a role in its ability to protect the α-center. For example in the case of esters, this could be further explained as follows: the most favorable conformation for deprotonation is one in which the α-hydrogen-carbon bond lies in a plane orthogonal to the plane of the carbonyl system, allowing for maximum orbital overlap as the α-carbon rehybridizes from sp3 to sp2. However, the α-proton and the carbonyl group in N-Pf-protected esters are nearly coplanar. Thus the rate of α-deprotonation would be stereoelectronically retarded in any ester for which such a conformation is highly favored.
According to crystal structure analyses the α-ester group and the fluorenyl ring are in close contact. This is strongly supported by the fact that the proton NMR shows a methyl ester resonance at 2.92 ppm (an unusually upfield chemical shift). So, achieving a stereoelectronically favorable conformation during deprotonation would require the ester group to rotate into the region of space occupied by the Pf group, resulting in allylic strain in the enolate-like transition state. This has been observed in the context with the alkylation of
N-Pf-
L-glutamate [
7].
The racemization of
N-Pf-compounds could be considered very unlikely after further investigating the stability in basic conditions. In strongly basic conditions the Pf anion can act as a leaving group giving rise to an imine (
Scheme 1). This elimination occurs faster/easier than the inversion and reprotonation of the anion [
5].
Scheme 1.
The decomposition of N-Pf amino aldehyde in basic conditions.
Scheme 1.
The decomposition of N-Pf amino aldehyde in basic conditions.
2.2. Phenylfluorenylation
Phenylfluorenylation was originally carried out by modifying and applying the existing method for making
N-trityl compounds [
3], which is still the most widely used. This traditional method utilizes 9-bromo-9-phenylfluorene (PfBr,
1). The protection is based on improving Br’s role as a leaving group with halogenophilic lead nitrate. The amino acid can be used free as such, when it has to be temporarily protected with TMSCl. It is also possible to use the corresponding methyl ester HCl salt
2. (
Scheme 2) Phenylfluorenylation is slow, taking several days, but the reaction is robust and the yields are generally good [
8].
Scheme 2.
Traditional phenylfluorenylation [
8].
Scheme 2.
Traditional phenylfluorenylation [
8].
2.3. Cleavage of Pf from nitrogen
The Pf group is usually removed by hydrogenolysis [
9] and solvolysis under strongly acidic conditions [
5]. It can also be removed by lithium or sodium in ammonia solution [
10], by TMSOTf in the presence of triethyl silane [
11] or by iodine in methanol [
12].
2.4. Phenylfluorenyl cation
Phenyfluorenyl cations have been made from fluorenol with strong acids such as sulphonic or triflic acid. In solution the cations are deep red and with an absorption maximum at λ
max = 494 nm. Quenching the cation with methanol forms Pf methyl ether as the main product [
13].
3. The Phenylfluorenyl Group in Synthesis
The Pf group has been used as a protecting group for nitrogen atoms exclusively in amino acids. It maintains the configurational integrity of the α-amino compounds during different kinds of C-C forming reactions. Enolization and alkylation [
14], Wittig and aldol reactions [
15], as well as Grignard addition [
14] of
N-Pf-amino carbonyl compounds are examples of reactions where the enantiomeric purity is preserved. Actually this applies to all examples presented in this review, although it is not mentioned in the form of ee’s (>99.9%) later on.
3.1. Amino aldehydes
Carbamate protected amino aldehydes are versatile intermediates in the synthesis of polyfunctional amino acids, peptide analogues, sphingolipids and aminosugars. They are configurationally unstable and racemize, even upon rapid chromatography on silica gel [
16]. The enantiomeric purity is usually not maintained in nucleophilic additions. Trityl-protected amino aldehydes are less prone to racemization than carbamate protected ones, but they are extremely acid labile, even under mildly acidic conditions.
N-Pf-Amino aldehydes are configurationally stable when subjected to both additions and flash chromatography [
5].
N-Pf-Amino aldehydes can be prepared from amino acids by reducing the ester to the alcohol with LiAlH
4 and then oxidizing it to aldehyde by Swern oxidation [
17]. Another option is to form an isoxazolidide and reduce it to the aldehyde with LiAlH
4 [
5]. Preparation of aldehydes is outlined in
Scheme 3.
Scheme 3.
Preparation of amino aldehydes.
Scheme 3.
Preparation of amino aldehydes.
Grignard, Wittig and aldol additions of
N-Pf-amino aldehydes are outlined in
Scheme 4. It is worth noticing that these reactions are not diastereoselective [
5].
Scheme 4.
Grignard, Wittig and aldol addition of N-Pf-amino aldehydes.
Scheme 4.
Grignard, Wittig and aldol addition of N-Pf-amino aldehydes.
3.2. Amino ketones
N-Pf-Amino ketones can be prepared by nucleophilic addition either to a carboxylic ester [
18], an aldehyde [
14], an oxazolidinone [
19] or an isoxazolidide [
10]. However, the most widely used routes, presumably because of their good yields compared to others, are via an aldehyde or an oxazolidinone. A secondary alcohol is formed in the addition to an aldehyde and it needs to be further oxidized (
Scheme 5).
Scheme 5.
Ketone formation by nucleophilic addition to aldehyde [
14].
Scheme 5.
Ketone formation by nucleophilic addition to aldehyde [
14].
The advantage of proceeding through the oxazolidinone is the temporary full protection of nitrogen. The ketone can be formed by treatment with an alkyl lithium, for example. The corresponding Grignard reaction leads to
N-alkylated product, presumably through an iminium ion intermediate [
19](
Scheme 6).
Scheme 6.
A method for preparation of N-Pf amino ketones through an oxazolidinone intermediate.
Scheme 6.
A method for preparation of N-Pf amino ketones through an oxazolidinone intermediate.
4. Enolization and Alkylation
Enolization and alkylation are important reactions in synthesis, especially if they can be performed regioselectively. α-Amino ketones possess two enolizable positions (
Figure 3), often leading to poor selectivity in their alkylations. Amide- and carbamate protected aminoketones are easily alkylated at the α-carbon, which is often undesired. Their regioselective enolization in the α’-carbon is possible by kinetic control using LDA as the base.
Figure 3.
Easily enolizable positions in amino ketones.
Figure 3.
Easily enolizable positions in amino ketones.
Pf directs the regioselectivity of enolization and alkylation in both cyclic and acyclic compounds. The Pf protected amino ketones are deprotonated and alkylated solely at α’-carbon. This feature of the Pf group has been most widely utilized. Enolization is commonly performed with KHMDS as a base and alkyl halides as electrophiles [
14].
4.1. Stereoselectivity of alkylation
In general, the diastereoselectivity in alkylations of
N-Pf-amino compounds is poor. The main product in the alkylation in
Scheme 7 is
syn, with up to 5:1 selectivity. It should be noted that the diastereoselectivity is dependent on the base, enolate cation and electrophile [
14]. In spite of its size, the Pf group does not contribute to the selectivity.
Scheme 7.
Regioselectivity of amino ketones is due to the base.
Scheme 7.
Regioselectivity of amino ketones is due to the base.
The
syn-selectivity of this alkylation can be explained by two transition state models. In the chelation model the nitrogen atom and the enolate oxygen atom are coplanar because of the electrostatic interaction with the K
+-cation. In the other model the amino and methyl groups are
gauche to the enolate oxygen. The electrophile approaches from the less hindered face, which forms the
syn isomer in the chelation model and
anti isomer in the non-chelated model. The chelated model is assumed to be more stable, because the steric interactions (allylic A
1,3-strain) between the α- and vinyl protons and methyl and oxygen are small. (
Scheme 8)
Scheme 8.
Chelation model [
14].
Scheme 8.
Chelation model [
14].
The influence of electrophiles in diastereoselectivity is presented in
Table 1.
Table 1.
Diastereoselectivity in regioselective alkylation.
Table 1.
Diastereoselectivity in regioselective alkylation.
| Alkyl halide | Time / h | Yielda / % | dr 16 (a:b) |
|---|
| MeI | 3 | 94 | 2.2:1 |
| BnI | 6 | 80 | 5:1 |
| Allyl bromide | 6 | 77 | 5:1 |
| Methyl α-bromopropanoate | 8,5 | 38 | 2.2:1 |
4.2. Alkylation of nitrogen and synthesis of pipecolates
The alkylation of nitrogen is often unwanted, but it can be utilized in preparing pipecolates from
N-Pf-aspartate (
Scheme 9) [
3,
20]. However, the electrophile must be particularly reactive, such as 3-bromochloropropane (BCP) [
21], for
N-alkylation to succeed.
Scheme 9.
N- and C-alkylation of L-aspartate methyl ester.
Scheme 9.
N- and C-alkylation of L-aspartate methyl ester.
The pipecolate structure formed in the ring forming reaction is rigid, so it can be alkylated diastereoselectively. The axial benzyl ester shields the upper side of the piperidine ring, which makes the electrophile approach from the lower side (
Scheme 10).
Scheme 10.
Alkylation of pipecolates [
3].
Scheme 10.
Alkylation of pipecolates [
3].
4.3. Aids in alkylation
In some cases the alkylation may need a cosolvent, like DMPU, DMEU or HMPA, to break the enolate complex. For example the alkylation of serine derived oxazolidinone and oxazolidide by lithium and potassium enolates in THF has been helped to go forward with DMEU (
Scheme 11). KHMDS yields both mono- and di-alkylated product. LiHMDS gives mainly monoalkylated product [
10].
Scheme 11.
Alkylation of oxazolidinone and oxazolidide.
Scheme 11.
Alkylation of oxazolidinone and oxazolidide.
5. Alkylation of L-Aspartate and L-Glutamate
Enolization and alkylation of
L-aspartate at the β-carbon is a robust and a straightforward reaction [
22]. In this particular case, the Pf group has been reported to protect both the α- and β-centers from racemization. Diastereoselectivity of the alkylation is not good and cannot be explained unambiguously. An almost 1:1 mixture is generally formed, but excellent selectivities can also be achieved. For dialkylation, the Pf protecting group has recently been used as a directing group in the synthesis of polytheonamide B [
23].
5.1. Stereoselectivity of alkylation of L-aspartate
Improving the stereoselectivity of alkylation of
N-Pf-aspartate ester enolates has been attempted by fully protecting the nitrogen. The highest stereoselectivity (50:1
syn:anti/
anti:syn) has been achieved by diprotecting the nitrogen with Pf and Bn [
24]. In that case the selectivity is determined so that KHMDS gives dominantly the
anti- while LiHMDS gives the
syn-product. The impact. of the electrophiles on selectivity are presented in
Table 2.
Table 2.
Alkylation of N-Pf-Bn-D-aspartate.
Table 2.
Alkylation of N-Pf-Bn-D-aspartate.
![Molecules 15 06512 i001]() |
|---|
| Electrophile | KHMDS, -23 °C | LiHMDS, -23 °C | LiHMDS, -78 °C |
|---|
| 26 (a:b) | 26 (a:b) | 26 (a:b) |
|---|
| Allyl iodide | 1:10 | 23:1 | - |
| MeI | 2:1 | >50:1 | >50:1 |
| BnBr | >1:50 | >50:1 | >50:1 |
It has been shown by trapping the enolate formed in the reaction with TMSCl that potassium and lithium bases form different enolates. Potassium base forms a
Z-enolate and a cyclic chelated transition state. The
anti product arises when the electrophile approaches from the less hindered face opposite to the Pf group. Lithium enolate has a
E-geometry so it can not chelate. The electrophile approaches from the less hindered face and the product is
syn (
Figure 4) [
24].
Figure 4.
Silyl ketene acetals of potassium and lithium enolates, and the geometries that affect the stereoselectivity of alkylation.
Figure 4.
Silyl ketene acetals of potassium and lithium enolates, and the geometries that affect the stereoselectivity of alkylation.
5.2. Hydroxylation and amination of L-aspartate
Hydroxylation (
Table 3) and amination of N-Pf-aspartate enolates have shown that stereoselectivity is dependent on interactions of many factors. The stereogenic center is affected by the enolate geometry, counter cation, reaction conditions, cosolvent, electrophile and the ionization degree of the amino group [
25].
Table 3.
Hydroxylation of L-aspartate methyl ester.
Table 3.
Hydroxylation of L-aspartate methyl ester.
![Molecules 15 06512 i002]() |
|---|
| 18a | Base (mol %) | Cosolvent | 27 (a:b) | Yield 27 (%)a |
|---|
| 1 | KHMDS (180) | - | 3:1 | 90 |
| 2 | LHMDS (300) | - | 1:8 | 65 |
| 3 | LDA (300) | - | 2:1 | 45 |
| 4 | LTMP (180) | - | 1:2 | 22 |
| 5 | LHMDS (300) | DMPU | 8:1 | 92 |
| 6 | LHMDS (300) | HMPA | 11:1 | 74 |
| 7 | LHMDS (300) | DME | 1:2.5 | 70 |
| 8 | LHMDS (300) | TMEDA | 1:8 | 80 |
| 9 | LHMDS (300) | PMDET | 1:5 | 75 |
| 10 | KHMDS (180) | 18-crown-6 | - | |
| 11 | LHMDS (300) | 12-crown-4 | - | |
| 12 | n-BuLi (100) / LHMDS (300) | - | 1:20 | 60 |
| 13 | n-BuLi (100) / LHMDS (300) | HMPA | 2:1 | 95 |
The use of Davis’ reagent as the hydroxylating agent gives a 1:1 mixture of diastereomers under all conditions examined.
N-Pf-Aspartate
tert-butyl ester and free acid react with poor selectivity. Enolates generated with LiHMDS do not react at all with MoOPH [
25].
The selectivity has been explained by mechanistic studies. The enolate is presumably in equilibrium with an open and a chelated form, in which the Pf group is in an equatorial position. Cosolvents like DMPU and HMPA that coordinate strongly to metals prefer the open form, as well as K
+ as the countercation. Poorly coordinating ligands like HMDS, THF or
n-BuLi as the base, prefer the chelated form. Hard, neutral cosolvents such as HMPA and DMPU form stronger complexes with Li
+ than neutral softer bases such as DME, TMEDA or PMDET [
25] (
Figure 5). An electrophile such as MoOPH that does not complex with the enolate metal cation, approaches from the less hindered face. Ligands that make too strong complexes with the counter cation of the enolate form a naked enolate that does not react.
Figure 5.
Transition state models for enolates.
Figure 5.
Transition state models for enolates.
The stereoselectivity of amination is poor, at least when DTBAD or DBAB are used. With these reagents in the conditions described in
Table 3, a 1:1 mixture of diastereomers is formed in nearly every case. However, the diastereomers can be easily separated by crystallization, and epimerized to the other isomer. Entry 6 (
Table 3) with DTBAD as the electrophile is an exception, where diastereoselectivity is as high as 30:1 (
anti:
syn). In all cases the major isomer in amination is
anti [
25].
Scheme 12.
Amination of N-Pf-aspartate dimethyl ester.
Scheme 12.
Amination of N-Pf-aspartate dimethyl ester.
5.3. Substituted prolines from L-glutamate
The regioselective enolization and alkylation of
L-glutamic acid have been used in enantiospecific synthesis of 4-substituted prolines (
Scheme 13). The
anti:
syn stereoselectivity in this alkylation varies from 1:2 to 1:3 [
26].
Scheme 13.
Synthesis of 4-substituted prolines.
Scheme 13.
Synthesis of 4-substituted prolines.
The stereo center in γ-methyl glutamate
30 can be epimerized to increase the proportion of the major
syn isomer, but the diastereomeric ratio of the
anti and
syn isomers could not be inverted. The effect of base in the epimerization is presented in
Table 4.
Table 4.
Epimerization of γ-methyl glutamate.
Table 4.
Epimerization of γ-methyl glutamate.
![Molecules 15 06512 i003]() |
|---|
| Base | Temperature / °C | Time / h | 30 (a:b) |
|---|
| KHMDS | -78 | 0.7 | 1:3 |
| LDA | -78 | 0.7 | 1:2.3 |
| BMDAa | -78 | 2 | 1:6.5 |
| NaOMe | 20 | 20 | 1:2.7 |
5.3.1. Alkylation of hydroxyproline
N-Pf-Hydroxyproline can be oxidized to corresponding ketone (
Scheme 14), that can be further regioselectively enolized and alkylated. The stereoselectivity is again due to both the base and the electrophile [
27] and the effects of bases and electrophiles on the stereoselectivity of the alkylation are presented in
Table 5.
Scheme 14.
Oxidation of hydroxyproline 32.
Scheme 14.
Oxidation of hydroxyproline 32.
Table 5.
Alkylation of
N-Pf-proline ketone and its stereoselectivity [
27].
Table 5.
Alkylation of N-Pf-proline ketone and its stereoselectivity [27].
![Molecules 15 06512 i004]() |
|---|
| 34 | Base (Mol %) | RX (mol %) | Yield 35 (%) (b:a) | Yield 36 (%) |
|---|
| 1 | NaHMDS (140) | MeI (500) | 65 (3:1) | 14 |
| 2 | KHMDS (200) | MeO2CCH2Br (220) | 81 (1:2) | - |
| 3 | NaHMDS (140) | H2C=CHCH2I (500) | 54 (>6:1) | 5 |
| 4 | KHMDS (280) | PhCH2Br (300) | 24 (1:1) | 2 |
| 5 | KHMDS (400) | MeI (1000) | - | 82 |
| 6 | KHMDS (400) | EtO2CCH2I (1000) | - | 52 |
| 7 | KHMDS (400) | H2C=CHCH2I (1000) | - | 69 |
| 8 | KHMDS (400) | PhCH2Br (670) | - | 29 |
N-Pf-Proline ketone
34a can be hydroxylated regio- and stereoselectively into alcohol
37 and finally into all
cis-dihydroxyproline
38 [
28] (
Scheme 15).
Scheme 15.
Synthesis of N-Pf-dihydroxyproline.
Scheme 15.
Synthesis of N-Pf-dihydroxyproline.
The above-presented regioselective enolization of
N-Pf-proline ketone can also be utilized in the synthesis of 4-substituted-3,4-didehydroprolines [
29]. Here the ketone is transformed into the corresponding enol triflate, which is then coupled to a suitable substituent (
Table 6).
Similar enolisation protocols have been utilised in the synthesis of kainic acid derivatives [
30,
31].
Table 6.
Synthesis of 4-substituted-3,4-didehydroprolines.
Table 6.
Synthesis of 4-substituted-3,4-didehydroprolines.
![Molecules 15 06512 i005]() |
|---|
| 39 | R | | Conditions | Yield 40 (%) |
|---|
| 1 | CO2H | CO/KOAc | DMF, rt, 66 h | 87 |
| 2 | CO2Me | CO/MeOH | DMF, reflux, 2 h | 81 |
| 3 | Ph | PhSnMe3 | THF, reflux, 16 h | 60 |
| 4 | Me | SnMe4 | THF, reflux, 48 h | 5-50 |
| 5 | ![Molecules 15 06512 i006]() | Bu3SnCH=CHSnBu3 | THF, reflux, 48 h | 40 |
6. Reduction and Oxidation of N-Pf-amino Compounds
Ketones, triple bonds and esters present in N-Pf-amino compounds can be reduced to alcohols, alkenes and aldehydes without removing the Pf protection. However, it should be remembered that the Pf group is readily hydrogenolyzed in Pd/C-hydrogenation and it is not stable under acidic ester hydrolysis. It is often possible to perform the hydrogenolysis of the Pf group and protection with Boc in the same step, effectively changing the protecting group to Boc in one operation.
6.1. Hydride reductions
In the hydride reduction of the carbonyl in
N-Pf-dimethyl proline ketone, the stereochemistry of the product is due to reducing agent. LiAlH
4 and NaBH
4 lead to different products, although with poor diastereoselectivity [
27].
Scheme 16.
Selectivity in the reduction of substituted prolines.
Scheme 16.
Selectivity in the reduction of substituted prolines.
N-Pf-
D-Tyrosine was used in the synthesis of (+)-anisomycin [
32]. The pyrrolidine structure in the target molecule was formed by using both hydride and palladium-catalyzed reductions in addition to Swern oxidation. No epimerization was observed and the Pf stayed intact (
Scheme 17).
Scheme 17.
Synthesis of (+)-anisomycin.
Scheme 17.
Synthesis of (+)-anisomycin.
The main product in the Grignard reaction is
syn, which can be explained by chelation controlled Cram-model (
Figure 6). The steric bulk of the Pf group might be the cause of
syn-selectivity (
syn:
anti 10:1). In the transition state the nucleophile attacks from the less hindered face opposite to the Pf amine.
Figure 6.
The transition state of syn-selectivity in Grignard-reaction.
Figure 6.
The transition state of syn-selectivity in Grignard-reaction.
The pyrrolidine ring is formed from the alcohol derived acetates by iodination of the double bond in compound
45. The stereoselectivity of the iodination is without a doubt due to the steric hindrance posed by the Pf group. An epi-iodinium ring is formed on the less hindered side of the double bond regardless of the spatial arrangement of the acetate (
Scheme 18).
Scheme 18.
Proposed mechanism for the stereoselective iodoamination.
Scheme 18.
Proposed mechanism for the stereoselective iodoamination.
Anisomycins have been made by a related reaction sequence, in which the same allyl alcohol intermediate is utilized [
32]. In the previous synthesis the stereochemistry of the product (
syn) was determined in the addition step to aldehyde
9, but in this case it can be directed to form either
anti or
syn. The addition is carried out by Gringard reaction with vinyl magnesium bromide, in which a diastereomeric mixture of alcohols
45 is formed. The alcohol mixture is oxidized to allylic ketone
48 by Swern oxidation. The formed ketone can be reduced stereoselectively. Results concerning the relation between the conditions and the stereochemistry of the product are presented in
Table 7.
As can be seen, the best stereo- and regioselectivity is attained with (S)-BINAL that favours the anti-product. By Felkin-Ahn-model predicted syn-product is formed with BH3.(CH3)2S. According to the polar Felkin-Ahn-model the most electron withdrawing group is placed where the largest substituent is according to classical FA-model.
Table 7.
Effect of the conditions and reagents in the stereoselectivity of the reduction.
Table 7.
Effect of the conditions and reagents in the stereoselectivity of the reduction.
![Molecules 15 06512 i007]() |
|---|
| 49 | Reagent | Conditions | 45 (a:b) | Yield 45 (%) |
|---|
| 1 | BH3-(CH3)2S | Toluene, -78 °C | 1:9 | 91 |
| 2 | (S)-BINAL | THF, -78 °C | >95:1 | 89 |
| 3 | (R)-BINAL | THF, -78 °C | 1:6 | 82 |
| 4 | NaBH4 | THF, -10 °C | | a |
| 5 | DIBAL-H | THF, -78 °C | | a |
| 6 | L-Selectride | THF, -78 °C | 1:5 | 45 |
| 7 | KS-Selectride | THF, -78 °C | 1:6 | 38 |
| 8 | Red-Al | THF, -40 °C | 1:8 | 20 |
The main product is formed when the nucleophile approaches the C-H bond in a Bürgi-Dunitz angle from the less hindered side. This decreases the energy of the transition state (
Figure 7). Simple hydride reducing agents such as NaBH
4 and DIBAL-H favor 1,4-reduction instead of 1,2-reduction [
32].
Figure 7.
Comparison of Felkin-Ahn model in selective reduction.
Figure 7.
Comparison of Felkin-Ahn model in selective reduction.
6.2. Reductive amination
Reductive amination was the key step in the synthesis of indolizidinone amino acids [
33]. The Pf group was hydrogenolyzed from the Claisen condensation product
50, originally formed by regioselective enolization of
N-Pf-glutamic acid
29. The hydrogenolysis was followed by ring closure to
51 and further cyclization to lactams
52a,b. The diastereoselectivity of the reductive amination is due to the pressure of hydrogen gas and the acid used (
Table 8).
Table 8.
The effect of hydrogen pressure and the acid on the diastereomeric ration in reductive amination.
Table 8.
The effect of hydrogen pressure and the acid on the diastereomeric ration in reductive amination.
![Molecules 15 06512 i008]() |
|---|
| 51 | H2 pressure/atm | Acid | Yield 52a (%) | Yield 52b (%) | dr (46a:46b) |
|---|
| 1 | 1 | TFA | 41 | 22 | 65:35 |
| 2 | 6 | TFA | 52 | 10 | 84:16 |
| 3 | 11 | TFA | 54 | 6 | 90:10 |
| 4 | 1 | 6 M HCl | 45 | 22 | 67:33 |
| 5 | 6 | 6 M HCl | 64 | 8 | 89:11 |
| 6 | 11 | 6 M HCl | 66 | 3 | 96:4 |
6.3. Dihydroxylation
In general, dihydroxylation of allylic amines is not stereoselective, on which the Pf group has no impact. The dihydroxylation of
N-Pf-amino alkene
53 with KMnO
4 or OsO
4 forms a mixture of diastereomers [
4].
Scheme 19.
Dihydroxylation of N-Pf-amino alkene.
Scheme 19.
Dihydroxylation of N-Pf-amino alkene.
With the Sharpless DHQ- and DHQD-ligands the diastereoselectivity is improved only to 2:1 and 1:2.6 (
55a:b). If a similar alkene
56 is subjected to conjugate addition, a mixture of different products is formed dependent on the temperature and other conditions. Considering the structures of the products, one can note that C-C-bond forming reaction is slow and the Pf protected nitrogen is reactive towards intramolecular electrophiles [
4].
Scheme 20.
Conjugate addition and the products.
Scheme 20.
Conjugate addition and the products.
9. Phenylfluorenyl in Total Synthesis
Many enantiospecific syntheses have been possible thanks to the regioselective enolization and alkylation of
N-Pf-amino compounds. This reaction sequence has been applied in the total syntheses of dipeptides [
44], (+)-pilocarpine [
45], (-)-kainic acid [
46] and pipecolates such as (+)-vincamines [
20], for example. Also achiral compounds, such as prodigiosin [
47] have been synthesized using the Pf-protecting group.
Variously functionalized L-asparagine derivatives have been used as starting materials in particularly many cases. It is common that alkylation is followed by ester hydrolysis, which has often turned out to be problematic. For example, Pf is cleaved by the usual acidic ester hydrolysis. For this reason Pf is typically used only in the early steps to direct enolization and to avoid racemization. After the critical phase it is changed to a more familiar protecting group such as Boc.
9.1. (+)-Vincamine
The synthesis of the pipecolate structure
22b is one of the first examples in which Pf has been used as a protecting group [
3]. Regioselective enolization was unsuccessful with carbamate protection, therefore trityl protection was investigated. However, trityl amines were not usable because of their acid lability, and that is why Pf-protection was tried. The pipecolate structure has been used in the total syntheses of apovincamine [
3] and later vincamine [
20].
(+)-Vincamine (
92), an alkaloid that has an influence on brain’s blood circulation, is used in nootropic memory enhancing purposes. There are several routes to racemic vincamine, and it can be synthesized enantiopure from
L-asparagine [
20]. The details of the synthesis of pipecolates was presented in
Section 4.2 (synthesis of piperidine and alkylation of pipecolate) (
Scheme 32).
Scheme 32.
The synthesis of (+)-vincamine.
Scheme 32.
The synthesis of (+)-vincamine.
9.2. (+)-Pilocarpine
Pilocarpine (
97), an alkaloid isolated from tropical American
Pilocarpus shrubs, is a muscarinic receptor agonist. It is used as a parasympathomamimetic compound towards glaucoma and xerostomia. There are several syntheses for racemic pilocarpine, and it can be made enantiopure from
L-asparagine [
45].
N-Pf-Protected asparagine
18a is alkylated, converted from amino acid to bromo acid to finally form an α,β-disubstituted succinate
98 and further the product.
Scheme 33.
The synthesis of (+)-pilocarpine.
Scheme 33.
The synthesis of (+)-pilocarpine.
9.3. (-)-Kainic acid
Kainic acid (
102) is a marine natural product belonging to the group of kainoids, glutamate analogs, that exhibits a wide range of biological activities. Kainoids are believed to have an influence on nervous system and they have been discovered to be useful also as insecticides. In this synthesis of kainic acid an inexpensive
trans-hydroxy-
L-proline is used as the starting material. The key step is diastereoselective alkylation of a keto derivative of
N-Pf-hydroxyproline
33, which is followed by stereoselective reduction of the keto function. In this synthesis, Pf is used solely in these steps and then replaced with a benzyloxycarbonyl (Cbz) in order to facilitate the upcoming cuprate substitution, which ultimately leads to the desired product [
46].
Scheme 34.
The synthesis of (-)-kainic acid.
Scheme 34.
The synthesis of (-)-kainic acid.
9.4. γ-Lactams from dipeptides
Lactams are conformational constraints in peptide skeletons and are effective structural tools for probing the active conformation of bioactive peptides. In the same way, altering their structure gives more information about how the conformation affects on biological activity.
Scheme 35.
Synthesis of γ-lactam dipeptides.
Scheme 35.
Synthesis of γ-lactam dipeptides.
Different kinds of dipeptides leading to γ-lactams have been made enantiopure from
N-Pf-
L-asparagine. Synthesis exploits the well-known feature of Pf of preventing enolization at the α-carbon of this diester
29 that enables regioselective alkylation. It also protects the α-
tert-butyl ester from reduction. Pf-protection is, however, changed after it had served its above-mentioned function because it does not tolerate the acidic conditions needed to cleave
tert-butyl ester [
44].
10. Phenylfluorenyl Derivatives
Modifications of the core structure of Pf, and how they might effect on the characteristics of Pf as a protecting group, have not been studied. Different halogen-, alkyl- or aryl-substituted fluorenyl compounds have been made and examined in different contexts [
48,
49,
50], but using Pf-related structures as protecting groups for nitrogen is very rare. The only known Pf-derivative used in synthesis is
p-bromo phenylfluorenyl (BrPf) (
Figure 14), that has been made to make it easier to remove Pf under mildly acidic conditions [
51].
Figure 14.
p-Bromophenylfluorenyl.
Figure 14.
p-Bromophenylfluorenyl.
The removal of BrPf has two phases. First, BrPf is activated by palladium-catalyzed amination between the aryl bromine and morpholine. The
p-aminophenylfluorenyl compound that is formed forms a more stable cation than Pf-cation, and it can be removed under mildly acidic conditions. It can also be removed selectively in the presence of
tert-butyl esters and carbamates. This works also the other way around, so that
tert-butyl esters and carbamates can be removed selectively in the presence of BrPf (
Scheme 36) [
51].
Scheme 36.
Palladium-catalyzed cross-coupling in the removal of BrPf.
Scheme 36.
Palladium-catalyzed cross-coupling in the removal of BrPf.
11. Phenylfluorenyl on Solid Support
Solid phase organic synthesis is a useful tool for making large libraries of molecules. The substrate is temporarily bound to a polymeric resin, which is removed at the end of the reaction sequence. The intermediate resin bound products can be filtered from the solutions and there is no need for a separate purification. Reactions are usually driven to completion by using excess reagents. Slowness is the most common disadvantage of heterogenic reactions [
52].
Synthetic strategies based on solid supported reagents are also becoming more common in the synthesis of small molecules. There is a so called linker between the polymer and the first building block, to which the molecule is attached and by the help of which the polymer can be removed. There exist several different linkers to amines, but they are usually sensitive to strong bases, acids or conditions in organometallic reactions, which constricts their use. Trityl linkers have been used to attach amines, carboxylic acids and alcohols.
Pf as a protecting group for α-amino carbonyls has proven its utility in liquid phase synthesis. Its possibilities in solid phase synthesis have been examined [
53,
54,
55] and it can act as a linker in the syntheses of alkaloids, heterocycles and amino acids. Structurally stable Pf-protected amino compounds on solid support are a valuable source in the building of chiral amino compounds libraries. Pf-linker can be made from BrPf, for example (
Scheme 37).
Scheme 37.
Pf-linker from BrPf.
Scheme 37.
Pf-linker from BrPf.
12. Conclusions
The phenylfluorenyl group has been proven to reliably protect the α-center in amino carbonyl compounds against epimerisation. It is especially useful in regioselective reactions such as enolization and alkylation. Despiet its steric bulk, it does not seem to have a significant influence on the diastereoselectivity of these or any other reactions. Very little is actually known about the Pf groups real steric and/or electronic effects or possibilities in synthesis. Much remains to be explored, especially to learn more about the characteristics of its derivatives in terms of stability, diastereoselectivity and the protection of the amine itself. At this point the Pf group is, though, unique in its way to allow a chemist to efficiently protect the usually stereochemically vulnerable α-center of α-amino acids and ketones against epimerization.