Chemistry of the M (M=Fe, Ca, Ba)-Se-H2O Systems at 25 °C
Abstract
:1. Introduction
2. Chemistry of the M (M=Fe, Ca, Ba)-Se-H2O Systems at 25 °C
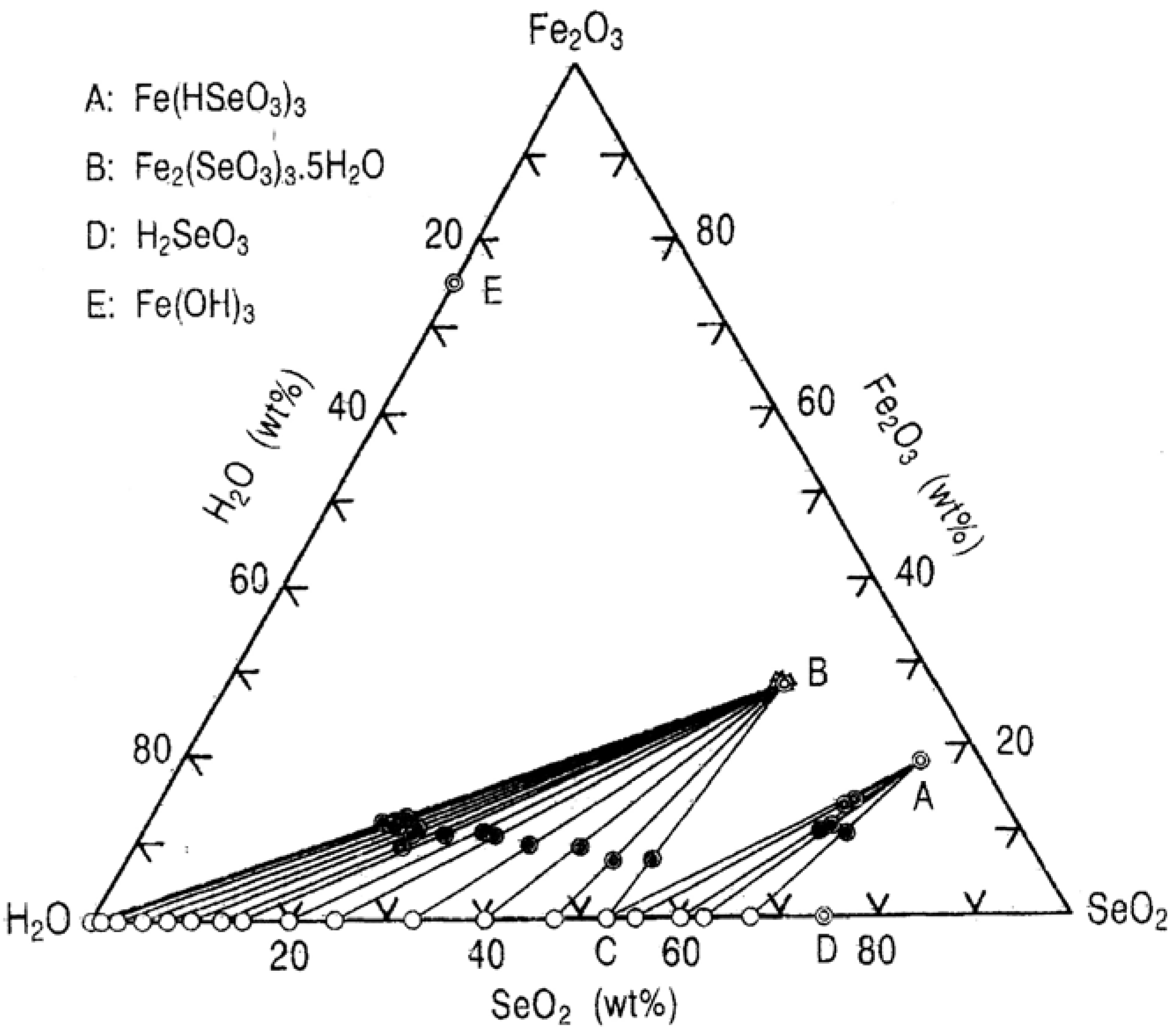
2.1. Equilibrium in the Fe(III)-Se(IV)-H2O System [12]
2.2. Equilibrium in the Fe(III)-Se(VI)-H2O System
2.3. Equilibrium in the Ca-Se(IV)-H2O and Ca-Se(VI)-H2O Systems [13]
2.4. Equilibrium in the Ba-Se(IV)-H2O and Ba-Se(VI)-H2O Systems [14]
3. Conditional Free Energies of Formation for Calcium Selenites and Selenates, and Barium Selenites and Selenates
- ΔfGφ298.15 CaSeO3∙H2O = −284.2 kcal/mol (−1189.1 kJ/mol)
- ΔfGφ298.15 Ca2SeO3(OH)2∙2H2O = −556.9 kcal/mol (−2330.1 kJ/mol).
- ΔfGφ298.15 CaSeO4∙2H2O = −352.7 kcal/mol (−1475.6 kJ/mol)
- ΔfGφ298.15 Ca2SeO4(OH)2 = −453.1 kcal/mol (−1895.9 kJ/mol)
- ΔfGφ298.15 BaSeO3= −230.6 kcal/mol (−964.7 kJ/mol).
- ΔfGφ298.15 BaSeO4= −249.0 kcal/mol (−1041.8 kJ/mol).
4. Removal of Selenium from Industrial Waste Water
- Removal of selenium(IV) as ferric selenite and sulfate ion as calcium sulfate
- Removal of selenium(VI) as barium selenate and sulfate ion as barium sulfate
- Removal of selenium(VI) by reduction-precipitation of selenium(VI) to elemental selenium with ferrous hydroxide or by adsorption of selenium(VI) on the polyamine-type weakly basic ion exchange resin (Eporasu K-6).
4.1. Removal of Selenium with Ferric Species [15]
4.2. Removal of Selenium as Barium Selenate [15]
4.3. Removal of Selenium(VI) by Reduction-Precipitation of Selenium(vi) to Elemental Selenium with Ferrous Hydroxide [15]
4.4. Removal of Selenium(iv,vi) from Solution Containing Sulfuric Acid by Three-Stage Process Using by Reduction-Precipitation of Selenium(vi) to Elemental Selenium with Ferrous Hydroxide in the Third Stage [15]
5. Adsorption of Selenium(VI) on Polyamine – Type Weakly Basic Ion Exchange Resin [16]
5.1. Adsorption of Selenium(VI) in Batch Experiments
5.2. Adsorption of Se(VI) in Column Experiments
5.3. Removal of Selenium(IV,VI) from Solution Containing Sulfuric Acid by Three-Stage Process Using by Adsorption of Selenium(VI) on the Polyamine-Type Weakly Basic Ion Exchange Resin (Eporasu K-6) in the Third Stage
6. Conclusions
References
- Koren, D.W.; Gould, W.D.; Lortie, L. Selenium Removal from Waste Water. In Water Processing Recycling in Mining and Metallurgical Industries; CIM: Edmonton, Alberta, Canada, 1992; pp. 171–182. [Google Scholar]
- Kapoor, A.; Tanjore, S.; Viraraghavan, T. Removal of Selenium from Water and Wastewater. Int. J. Environ. Stud. 1995, 49, 137–147. [Google Scholar] [CrossRef]
- Mirza, A.H.; Ramachandran, V. Removal of Arsenic and Selenium from Wastewaters - A Review. In Second International Symposium on Extraction and Processing for the Treatment and Minimization of Wastes, Scottsdale, Arizona, USA, October 27–30, 1996; Ramachandran, V., Nesbitt, C.C., Eds.; TMS: Warrendale, PA, USA, 1996; pp. 563–582. [Google Scholar]
- Hata, R.; Nishimura, N.; Umetsu, Y. Removal of Selenium from Copper Anode Slimes and Waste Water. Ryusan to kogyo 1997, 50, 155–163. [Google Scholar]
- Twidwell, L.G.; McCloskey, J.; Miranda, P.; Gale, M. Technologies and Potential Technologies for Removing Selenium from Process and Wastewater. Proceedings REWAS’99-Global Symposium on Recycling, Waste Treatment and Clean Technology, San Sebastian, Spain, September 5–9, 1999; Gaballah, I., Hager, J., Solozaral, R., Eds.; TMS: Warrendale, PA, USA, 1999; pp. 1645–1656. [Google Scholar]
- Simizu, S.; Hasegawa, N.; Yoshioka, A.; Iwahori, S. Elimination of Selenium in Effluent from Precious Metals Refinery. In Second International Symposium on Extraction and Processing for the Treatment and Minimization of Wastes, Scottsdale, Arizona, USA, October 27–30, 1996; Ramachandran, V., Nesbitt, C.C., Eds.; TMS: Warrendale, PA, USA, 1996; pp. 205–214. [Google Scholar]
- Altringer, P.B.; Larsen, D.M.; Gardner, K.R. Bench Scale Process Development of Selenium Removal from Waste Water Using Facultative Bacteria. International Symposium on Biohydrometallurgy, Jackson Hole, Wyoming, USA, August 13–18, 1989; Salley, J., McCready, R.G.L., Wilchlacz, P.L., Eds.; pp. 643–657. [Google Scholar]
- Lortie, L.; Gould, W.D.; Tajan, S.; McCready, R.G.L.; Cheng, K. Reduction of Selenate and Selenite to Elemental Selenium by a Pseudomonas stutzeri isolate. Appl. Environ. Microbiol. 1992, 58, 4042–4044. [Google Scholar] [PubMed]
- Barnes, J.M.; McNew, E.B.; Polman, J.K.; McCune, J.H.; Torma, A.E. Selanate Reduction by Pseudomonas-Stutzeri. In Mineral Bioprocessing, Santa Barbara, California, USA, June 16–12, 1991; Smith, R.W., Misra, M., Eds.; TMS: Warrendale, PA, USA, 1991; pp. 367–375. [Google Scholar]
- Oremland, R.S.; Hollibaugh, J.T.; Maest, A.S.; Presse, T.S.; Miller, L.G.; Culbertson, C.W. Selanete Reduction to Elemental Selenium by Anaerobic Bacteria in Sediments and Culture: Biogeochemical Significance of a Novel Sulfate-Independent Respiration. App. Environ. Microbiol. 1989, 55, 2333–2343. [Google Scholar]
- Fujita, M.; Ike, M.; Nishimoto, S.; Takahashi, K.; Kashiwa, M. Isolation and Characterization of Novel Selenate-Reducing Bacterium, Bacillus sp. SF-1. J. Ferment. Bioeng. 1997, 83, 517–522. [Google Scholar] [CrossRef]
- Nishimura, T.; Hata, R.; Umetsu, Y. Phase Equilibria in the Fe2O3-SeO2-H2O System. Hydrometallurgy 2005, 79, 110–120. [Google Scholar] [CrossRef]
- Nishimura, T.; Hata, R. Chemistry of the Ca-Se(IV)-H2O and Ca-Se(VI)-H2O Systems at 25 °C. Hydrometallurgy 2007, 89, 346–356. [Google Scholar] [CrossRef]
- Hata, R.; Nishimura, T.; Umetsu, Y. Solubility and Stability Regions of Barium Selenites and Barium Selenates in Aqueous Solution at 25 °C. Can. Met. Quart. 2004, 43, 57–65. [Google Scholar] [CrossRef]
- Nishimura, T.; Hata, R.; Umetsu, Y. Removal of Selenium from Industrial Waste Water. In Minor Elements 2000: Processing and Environmental Aspects of As, Sb, Se, Te, and Bi; Young, C., Ed.; SEM: Littleton, CO, USA, 2000; pp. 355–362. [Google Scholar]
- Nishimura, T.; Hashimoto, H.; Nakayama, M. Removal of Selenium(VI) from Aqueous Solution with Polyamine-type Weakly Basic Ion Exchange Resin. Separ. Sci. Technol. 2007, 42, 3155–3167. [Google Scholar] [CrossRef]
- Murphy, A.P. Removal of Selenate from Water by Chemical Reduction. Ind. Eng. Chem. Res. 1988, 27, 187–191. [Google Scholar] [CrossRef]
- Hamada, S.; Ishikawa, Y.; Shirai, T. Composition and Stability of Fe(III)-Se(IV) Complex. Nippon Kagaku Zasshi 1965, 86, 74–78. [Google Scholar] [CrossRef]
- Lien, R.H.; Dinsdale, B.E.; Gardner, K.R.; Altringer, P.B. Chemical and Biological Cyanide Destruction and Selenium Removal from Precious Metals Tailings Pond Water. In EPD Congress’90, Anaheim, California, USA, February 19-22, 1990; Gaskell, D.R., Ed.; TMS: Warrendale, PA, USA, 1990; pp. 323–339. [Google Scholar]
Sample Availability: Samples of the compounds are available from authors. |


















| Total Se | Se(IV) | Se(VI) | Fe(III) | Ca |
|---|---|---|---|---|
| 754 | 11 | 743 | 1.4 | 595 |
| BaCl2 added(mol/L) | Total Se(mg/L) | Se(IV)(mg/L) | Se(VI)(mg/L) | Ba(mg/L) |
|---|---|---|---|---|
| 0.03 | 414 | 4.3 | 410 | < 0.02 |
| 0.035 | 102 | 0.35 | 102 | 43 |
| 0.035 | 51 | 0.27 | 51 | 106 |
| 0.04 | 8.9 | 0.18 | 8.7 | 608 |
| 0.045 | 4.7 | 0.05 | 4.7 | 1150 |
| 0.05 | 3.4 | < 0.05 | 3.4 | 1860 |
| 0.075 | 1.7 | < 0.05 | 1.7 | 5180 |
| Condition | Total Se | Se(IV) | Se(VI) | Ba | Fe |
|---|---|---|---|---|---|
| Feed | 4.7 | 0.05 | 4.7 | 1150 | < 0.02 |
| Fe(II)/Se mole ratio = 50 & T = 25 °C | 0.05 | < 0.05 | 0.05 | 657 | < 0.02 |
| Fe(II)/Se mole ratio = 20 & T = 70 °C | 0.06 | < 0.05 | 0.06 | 631 | < 0.02 |
| Feed | 51 | 0.27 | 51 | 106 | < 0.02 |
| Fe(II)/Se mole ratio = 50 & T = 25 °C | 0.1 | < 0.05 | 0.1 | < 0.02 | 3.8 |
| Fe(II)/Se mole ratio = 20 & T = 70 °C | 0.05 | < 0.05 | 0.05 | < 0.02 | < 0.02 |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nishimura, T.; Hata, R.; Hasegawa, F. Chemistry of the M (M=Fe, Ca, Ba)-Se-H2O Systems at 25 °C. Molecules 2009, 14, 3567-3588. https://doi.org/10.3390/molecules14093567
Nishimura T, Hata R, Hasegawa F. Chemistry of the M (M=Fe, Ca, Ba)-Se-H2O Systems at 25 °C. Molecules. 2009; 14(9):3567-3588. https://doi.org/10.3390/molecules14093567
Chicago/Turabian StyleNishimura, Tadahisa, Ryosuke Hata, and Fumihiko Hasegawa. 2009. "Chemistry of the M (M=Fe, Ca, Ba)-Se-H2O Systems at 25 °C" Molecules 14, no. 9: 3567-3588. https://doi.org/10.3390/molecules14093567




