1. Introduction
The influence of dietary fats on the pathogenesis of coronary heart disease, congestive heart failure as well as vulnerability to cardiac arrhythmias and sudden cardiac death has been well documented [
1,
2]. In this regard, both the “type” and the “amount” of dietary oils and fats have been identified as important determinants [
3,
4,
5]. For example, a considerable body of supporting evidence shows that long chain (C ≥ 20)
n-3 polyunsaturated fatty acids (LC
n-3 PUFA) derived from marine sources (seafood, fish and microalgae) are particularly effective in affording cardiovascular protection [
6,
7] although more recent analyses have reported inconsistent outcomes [
8]. In similar vein, a review of recent clinical trials (2007–2013 period) showed a lack of clear benefit of fish oil supplements although high dietary intake of fish was associated with lower incidence of sudden cardiac death, congestive heart failure, myocardial infarction and stroke [
9]. Among the
n-3 PUFA, the two major LC-
n-3 PUFA are eicosapentaenoic acid (EPA, C20:5
n-3) and docosahexaenoic acid (DHA, C22:6
n-3).
A number of studies [
10,
11] have reported certain positive cardiovascular health outcomes from consumption of α-linolenic acid (ALA, C18:3
n-3), an essential
n-3 PUFA widely available from plant-based food sources including certain seed oils (e.g., flax, canola, perilla, chia, walnut,
etc.). The primary mechanism by which ALA fosters cardiovascular health benefits is usually explained in terms of it being a precursor for endogenous LC
n-3 PUFA biosynthesis of EPA and DHA, the two key fatty acid substrates for the synthesis of eicosanoid family of biological mediators. However, in humans, the conversion of ALA to EPA and DHA is inefficient, almost negligible in the case of conversion to DHA [
12,
13,
14]. This is due to the lack of an efficient elongation and desaturation process to convert ALA to EPA by any more than 5%–7% [
12]. The primary reason for this inefficiency is explained by the
n-6 PUFA linoleic acid (LA) competing with ALA at the level of the Δ6-desaturase enzyme complex [
12,
13,
14], which inserts additional double bonds to these precursor fatty acids. Accordingly, the conversion of ALA to stearidonic acid (SDA, C18:4
n-3), facilitated by this enzyme, is considered the rate-limiting step in LC
n-3 PUFA biosynthesis in vertebrates [
15].
In order to address this limited bio-conversion of ALA to EPA and beyond, there has been interest in evaluating plant oils with better conversion to EPA and DHA than ALA-rich oils. In this regard, oils containing SDA have been the subject of much interest in feeding experiments involving farm animals [
16,
17], aquaculture fish [
18], animal models [
19] and humans [
20,
21,
22]. SDA is relatively abundant in plants of the
Boraginaceae family. One of the commercially available non-GM sources of SDA is extracted from
Echium plantagineum [
23]. SDA has been found to be further metabolised
in vivo and lead to increased plasma and tissue levels of LC
n-3 PUFA both in animal models [
19] and in humans [
24]. For example, increased EPA and DPA have been observed following supplementation of humans with Echium oil (EO) [
24]. In parallel with such compositional alterations, modification of several biochemical and physiological markers for cardiovascular disease have also been observed [
25]. For example, daily supplementation with 15 g EO for four weeks lowered serum triacylglycerols in hypertriglyceridemic subjects [
24], whilst a longer feeding protocol (17 g/day EO for 8-weeks equating to 2 g/day SDA) in normal and overweight individuals was accompanied by reductions in serum cholesterol, LDL-cholesterol, oxidized-LDL, HDL-cholesterol and triacylglycerols [
25]. In contrast, a more recent randomized controlled trial [
26] of overweight and obese subjects found no change in serum triacylglycerols following EO (1.2 g/day SDA; 6-weeks). In subjects with metabolic syndrome, further to improving plasma lipid profiles, additional benefits of EO were noted by Khunt
et al. [
25] with reductions in blood pressure and plasma insulin. Interestingly, this latter study has concluded that the collective outcomes of EO on cardiovascular risk biomarkers are broader than that exerted by fish oil (1.9 g/day EPA) itself. These benefits of EO were observed in the absence of any increase in DHA in plasma and/or peripheral blood mononuclear cells.
Biochemical studies in animals, using apoB100-only LDLrKO mice, have shown that decreased lipogenic gene expression increased intravascular lipolysis and enhanced clearance of plasma very low density lipoprotein (VLDL) as potential mechanisms for the triglyceride lowering action of EO [
27,
28]. In addition to lipid lowering properties, anti-atherogenic actions of EO have also been reported [
29]. For example, both EO and FO were equally effective in reducing plasma triglycerides, total plasma cholesterol, VLDL and LDL-cholesterols and apoB lipoproteins, as reflected in the form of reduced aortic deposition of cholesterol, and surface lesion formation leading to retardation in atherogenesis [
29]. Such collective observations from both human and animal studies have led these investigators to suggest that EO may be useful as a botanical alternative to FO in reducing hypertriglyceridemia and affording athero-protection [
29,
30].
Most studies with EO have reported increased accumulation of EPA and DPA with no change in DHA in blood and cell lipids [
25] suggesting incomplete metabolism (elongation/desaturation) of the precursor fatty acids in EO. However, compositional data following EO supplementation on major organs (heart, kidney, liver) is lacking and it is unknown whether or not further conversion/incorporation of SDA and its metabolites has taken place, for example, in tissue specific manner. Moreover, several recent studies have reported EO is able to mimic biochemical measures of cardiovascular risk reduction benefits of FO [
24,
25,
27,
28]. Nevertheless, except for an indirect or secondary observation [
19] regarding blood pressure, no datum exists in relation to any direct measure(s) of a given endpoint of cardiovascular pathophysiology that may be influenced after ingesting EO. In this context, a unique characteristic of the two major LC
n-3 PUFA in fish oils—EPA and DHA—is their ability to modify ischemia-induced ventricular fibrillation and sudden cardiac death [
31]. The anti-arrhythmic actions of EPA and DHA first demonstrated in this laboratory over two decades ago in whole animal models have now been confirmed in large scale human clinical trials [
32] which has also validated the experimental model employed. To this end, despite the presence of a sound body of evidence showing increased accumulation of LC
n-3 PUFA following dietary EO (SDA), and claimed benefits on plasma lipids, its potential to facilitate any direct anti-arrhythmic benefit has not yet been evaluated. Therefore, this study was initiated with two main objectives: (A) to compare the impact of any dose-related outcomes of EO and FO on the LC
n-3 PUFA contents of membrane phospholipids in major tissues, and (B) to provide comparative data on the anti-arrhythmic potential of these two sources of
n-3 PUFA, using the well-established rat model of cardiac arrhythmia, ventricular fibrillation and sudden cardiac death [
33].
4. Discussion
The present study addressed two key questions relating to tissue fatty acid composition and cardiac arrhythmia outcomes following dietary supplementation with oils rich in
n-3 PUFA of different origin. EO contained
n-3 PUFA of C = 18 length (29% ALA and 14% of SDA) compared to FO which had a total LC
n-3 PUFA, C ≥ 20 (EPA + DHA) of >30%. Both oil types resulted in marked differences in the composition of membrane phospholipids of various tissues. The previously reported [
37] anti-arrhythmic actions of FO were reconfirmed in the present study. Whilst EO also led to an increased accumulation of LC
n-3 PUFA, mainly via EPA and DPA, the extent of protection against ischemia-induced cardiac arrhythmia and sudden cardiac death was less than that observed at comparable levels of FO supplementation.
The fatty acid data from this study suggested that ALA and SDA were virtually converted to longer chain
n-3 PUFA in heart and liver phospholipids, with no trace of 18C
n-3 PUFA left. Previous studies have also shown very efficient conversion of both ALA and SDA to EPA and DPA in the rat liver [
19]. Overall, the pattern of changes in EPA, DPA and DHA in plasma and tissue were consistent with reports from previous rat [
19] and human studies [
20]. The first salient feature of this study was that SDA supplementation did not yield nutritionally/physiologically meaningful DHA levels in plasma or tissue. In almost all published data, the magnitude of changes in tissue or plasma DHA as a consequence of supplementation with oils containing 18C
n-3 PUFA were usually negligible [
12]. On the contrary, SDA feeding has consistently shown significantly increased levels of EPA and DPA in plasma and tissues [
19]. Our data provides further evidence that in rats there is efficient conversion of SDA up to DPA, but not DHA. In this regard, these findings also mimic the observations in humans with SDA-rich oils [
20,
21,
22]. For example, James
et al. [
20] who compared the conversion of ALA, SDA to ≥20C
n-3 PUFA (EPA, DPA and DHA) reported that dietary SDA led to an increase in EPA and DPA concentrations but not DHA levels in plasma and/or erythrocytes in a double blind, parallel group design study of six weeks’ duration. The efficiency of increasing tissue EPA was 1.0, 0.3 and 0.07 for EPA, SDA and ALA, respectively. Similarly, Krul
et al. [
38] showed that the efficiency of apparent conversion of SDA to EPA in human RBC was 41%, 26% and 17% of dietary SDA for doses of 0.61, 1.89 and 5.32 g/day.
Studies using 18C
n-3 oils have shown that the efficiency of conversion of 18C
n-3 PUFA to ≥20C
n-3 PUFA declines as the dose (en%) of the dietary 18C
n-3 PUFA is increased. Gibson
et al. [
39] compared 54 different diet combinations, including various ratios of
n-6 polyunsaturated linoleic acid (LA) to ALA (
n-3 PUFA) to determine DHA synthesis from ALA in rats. They showed plasma phospholipid EPA, DPA and DHA increased rapidly within a narrow range—between 0 and 2 en% of dietary ALA, but suppressed to basal levels (2% total fatty acids) when the en% derived from total PUFA (LA + ALA) reached 3 en% and above.
Taken collectively, the fatty acid changes in plasma and tissues following SDA feeding had two main outcomes: increases in EPA and DPA and no change in the basal levels of DHA. These findings mirror what other studies have shown to be the case for plant-based C18 omega-3 oils in humans and different animal species (see review by Brenna
et al. [
12]). Furthermore, the present study has clearly shown that the patterns of changes in LC
n-3 PUFA in plasma and tissues are markedly different between EO and FO. In FO-supplemented rats, EPA and DHA generally increased with greater availability of LC
n-3 PUFA. For most parts the increased LC PUFA in plasma and tissue following supplementation with FO can be explained by way of direct incorporation of dietary EPA and DHA with some elongation and desaturation of EPA taking place. In contrast, any increase in LC
n-3 PUFA following dietary EO would be due to further metabolism of the two C18
n-3 precursor fatty acids ALA and SDA since pre-formed C20 fatty acids were absent in the diet. In EO-supplemented rats, only cardiac muscle phospholipids showed a dose-related increase in DPA. In all other cases, the changes in tissue and plasma EPA and DPA beyond the first dose (EO-1) were minimal, suggesting that the linear response phase may be between 0% and 1% dietary Echium oil.
Although the tissue levels of EPA and DHA in the FO fed rats tended to show a dose-related uptake, there exist clear tissue specific differences with regard to the extent of incorporation and further metabolism of these two fatty acids. For example, compared to DHA, the EPA content of cardiac muscle showed only a minor increase amounting to <0.5% of total phospholipid fatty acids even at the 5% oil supplementation level. This differed markedly with DHA where a clear dose-related accumulation was observed (
Table 3), and accounted for nearly 24% of cardiac membrane fatty acids at the highest dose tested (5%
w/
w). It is also evident that this increase in
n-3 PUFAs has occurred primarily at the expense of the two major
n-6 PUFA; LA (18:2
n-6) and AA (20:4
n-6). Compared to the control group, the displacement of LA by
n-3PUFA amounted to 16%, 33% and 35% at the three dose levels of FO, respectively. AA displacement amounted to 25% at the 5% supplementation level. In contrast, the LA levels in the plasma, liver and kidney had all remained unaffected despite an increased presence of dietary
n-3 PUFA. Similarly, the AA content of liver and kidney tended to show more resistance to be displaced by greater availability of dietary
n-3 PUFA. Taken collectively, these observations would lend further support to the role membrane phospholipids play in maintaining the physiological functioning of specific tissues and organs. As previous publications [
40,
41,
42,
43,
44] showed the type and amount of fatty acids in cell membranes not only influence the physical properties of the membrane bilayer (e.g., fluidity, lipid micro domains/rafts), but also modulate important biochemical functions—ion channels, transporters and enzymes. In addition, membrane and intracellular fatty acids provide substrates for the synthesis of numerous biochemical mediators including eicosanoid family of autacoids.
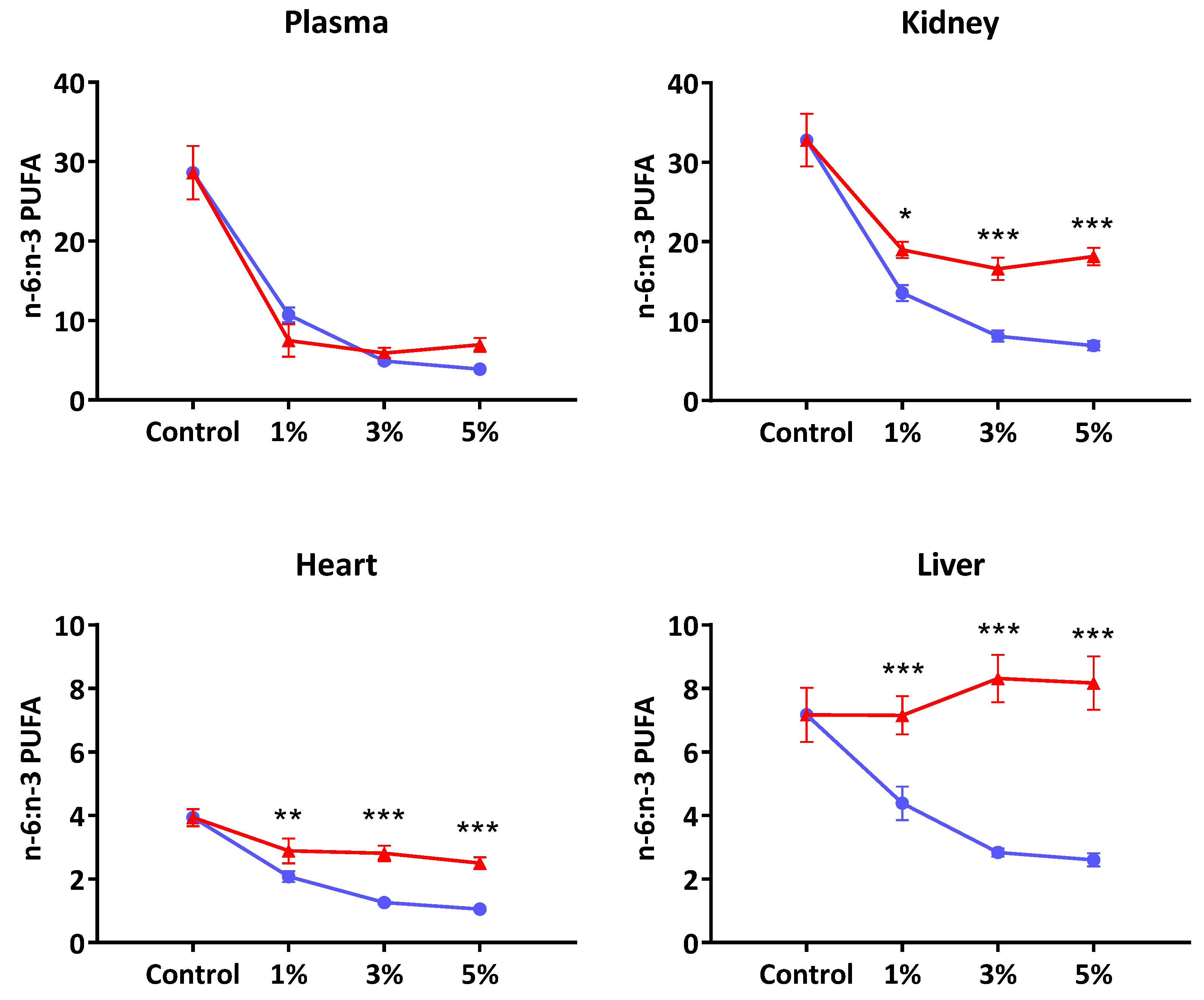
It is noted that in the three organs studied—liver, kidney and heart—the extent of perturbation of fatty acid composition by EO was much less, and reached saturation at the 1% level, compared to the changes observed following feeding FO rich in EPA and DHA that appeared to be dose-related (
Figure 1). However, the possibility exists for the relatively high presence of
n-6 PUFA (26%) as well as ALA (33%) in the EO to interfere with further metabolism of SDA which was present at a much lower level (14%). It is more likely that any substrate competition would be due to LA rather than ALA since the genetically modified SDA-soybean preparation which contained lower ALA than SDA levels [
21] also led to similar compositional changes as observed in the present study. The LA content in SDA-soybean was 31% compared to 16.6% SDA.
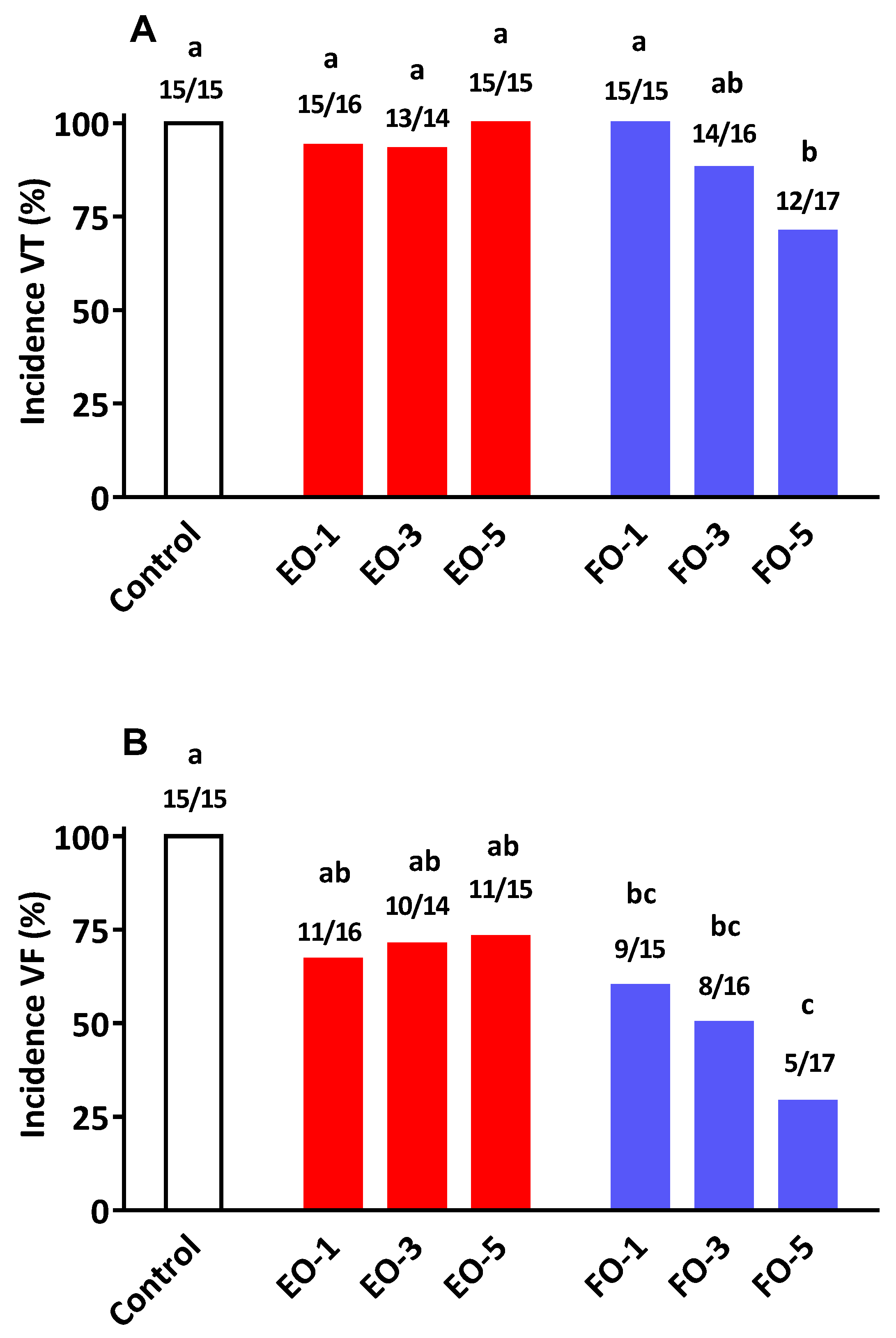
The anti-arrhythmic actions of
n-3 PUFA has been attributed to favourable changes to the heart membrane structure, favourable modulation of ion channels (e.g., Ca
2+) in cardiac tissue, and improved myocardial oxygen efficiency [
31]. The present results conclude that while there is some anti-arrhythmic action arising from EO consumption, the efficacy is not equivalent to that achieved with FO supplementation. For example, increasing the level of dietary EO neither arrested the development of VT nor its deterioration into the more serious condition of VF (
Figure 2). This differed markedly with that observed following the provision of FO, where the progression of VT to VF was reduced in a dose-related manner. At the highest supplementation level (5%
w/
w) FO not only reduced the incidence of VF (29%
vs. 73% in EO group) but such episodes once occurred lasted a much shorter period of time compared to EO and/or the control group (
Table 6). It is of interest to note that at the 3% supplementation level of EO displayed certain cardioprotective qualities (VF duration and % mortality) comparable to those found with FO. The reason(s) for this apparent protective actions of EO, only at this particular dose level, is difficult to explain since such benefits were not repeated at the higher supplementation level of 5%. Tissue fatty acid compositional data showed no clear differences between 3% and 5% oil incorporation levels. Furthermore, it is highly unlikely that a concentration–effect relationship for cardioprotection follows a bell-shape curve within the narrow band of EO feeding used in this study. Collectively, our findings in relation to cardio-protection and tissue fatty acid compositional alterations mirror previous studies where direct comparisons between SDA and EPA have been made with respect to several other biomarkers of cardiovascular health [
20,
21,
25].
The data from this study and those published by others indicate that SDA containing oils lead to elevated plasma and tissue LC n-3 PUFA. However, the magnitude of changes reported from this study and that from the literature does not provide evidence of equivalence in potency between SDA-containing oils and fish oils. In addition, potential anti-arrhythmic action of SDA oils needs to be investigated further using other animal models as well as using pure SDA or more enriched supplements to minimise the confounding effect of ALA that is present in commercial sources of SDA oils.
In summary, evaluation of the anti-arrhythmic actions of EO and FO in a rat arrhythmia model is a novel contribution from this study. We conclude that feeding Echium oil favourably changes the n-3 PUFA profiles of blood and tissues in rats, especially with respect to DPA in heart tissue. Although there are emerging evidence to suggest DPA may possess unique physiological actions including anti-platelet aggregation, pancreatic lipase inhibition and potential anti-obesity effects, its efficacy in affording direct cardio-protection has not been evaluated to date using pre-formed and more pure forms of the fatty acid. The present study provides some indirect evidence for a potential role for DPA since the changes in n-3 PUFA profile following EO were associated with some anti-arrhythmic action, although the extent of protection did not match that achieved by FO at all three dose levels studied. Furthermore, the increased DPA may play other beneficial roles unrelated to cardiac arrhythmia. This needs to be further investigated in animal models and substantiated in relevant human cohorts.