Role of UDP-Glucuronosyltransferase 1A1 in the Metabolism and Pharmacokinetics of Silymarin Flavonolignans in Patients with HCV and NAFLD
Abstract
:1. Introduction
2. Results
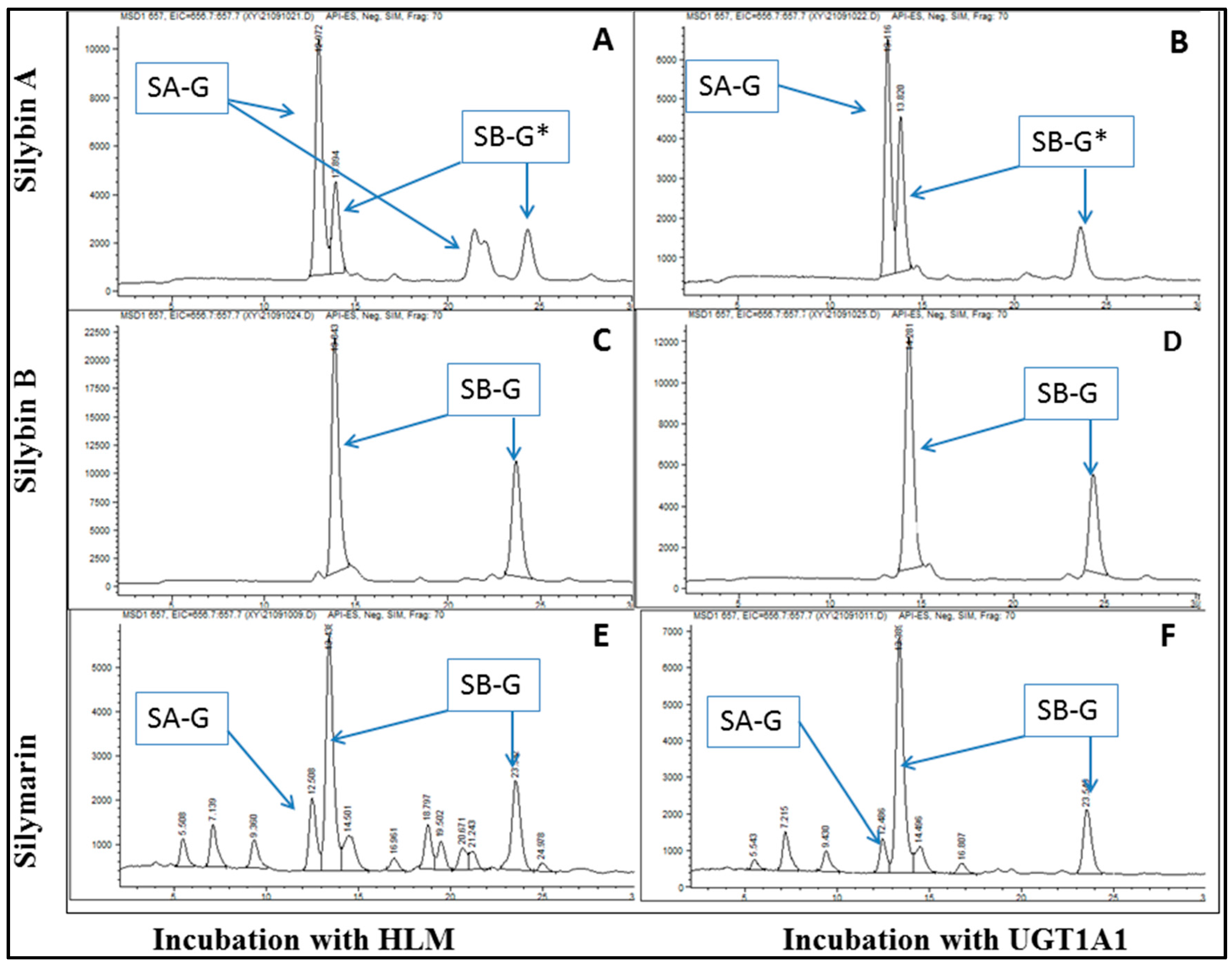
2.1. Enzyme Kinetics of Silymarin Glucuronidation in Recombinant Human UGT1A1 by Substrate Depletion Assay
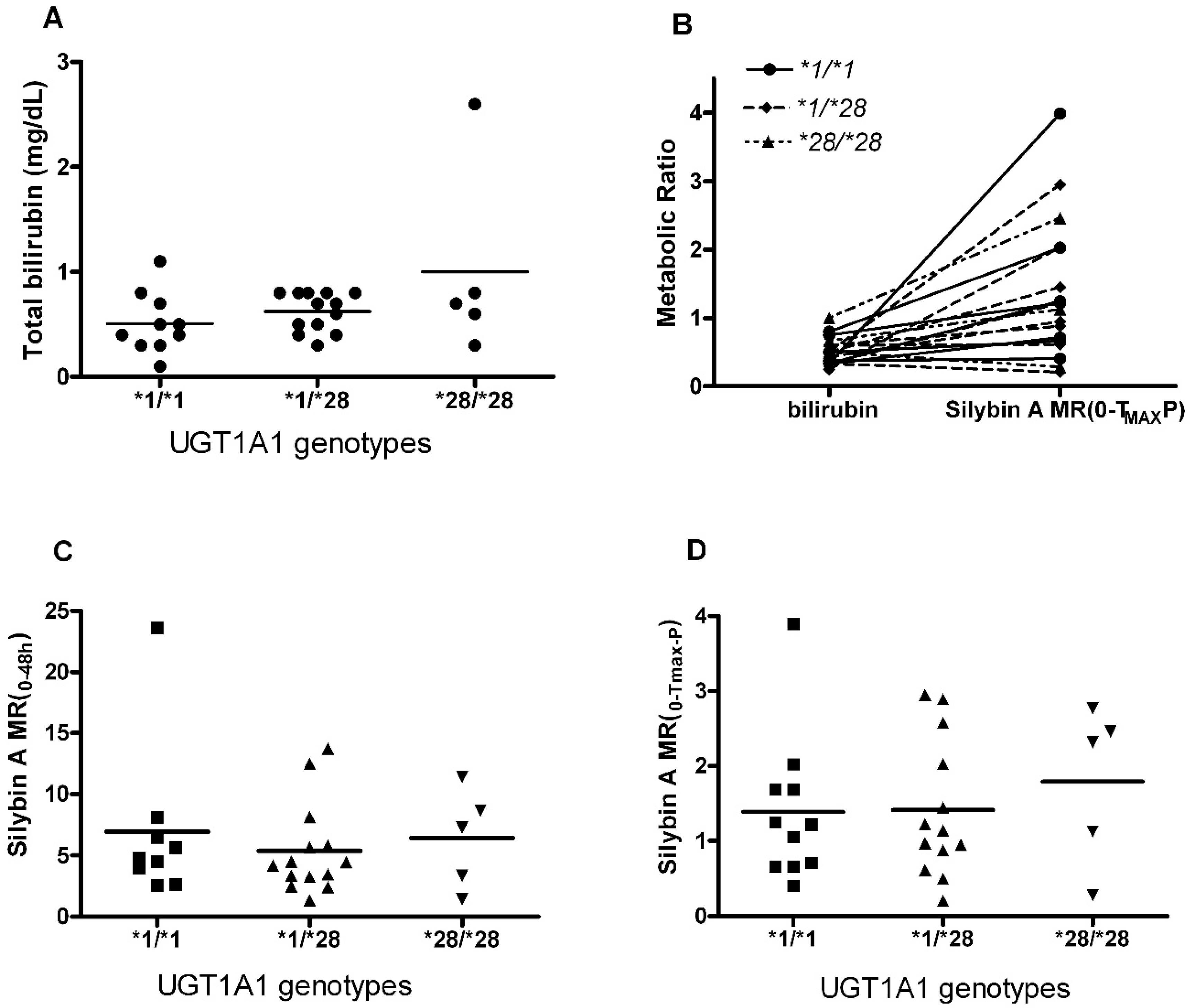
2.2. Influence UGT1A1*28 Polymorphism on SA and SB Plasma Exposures
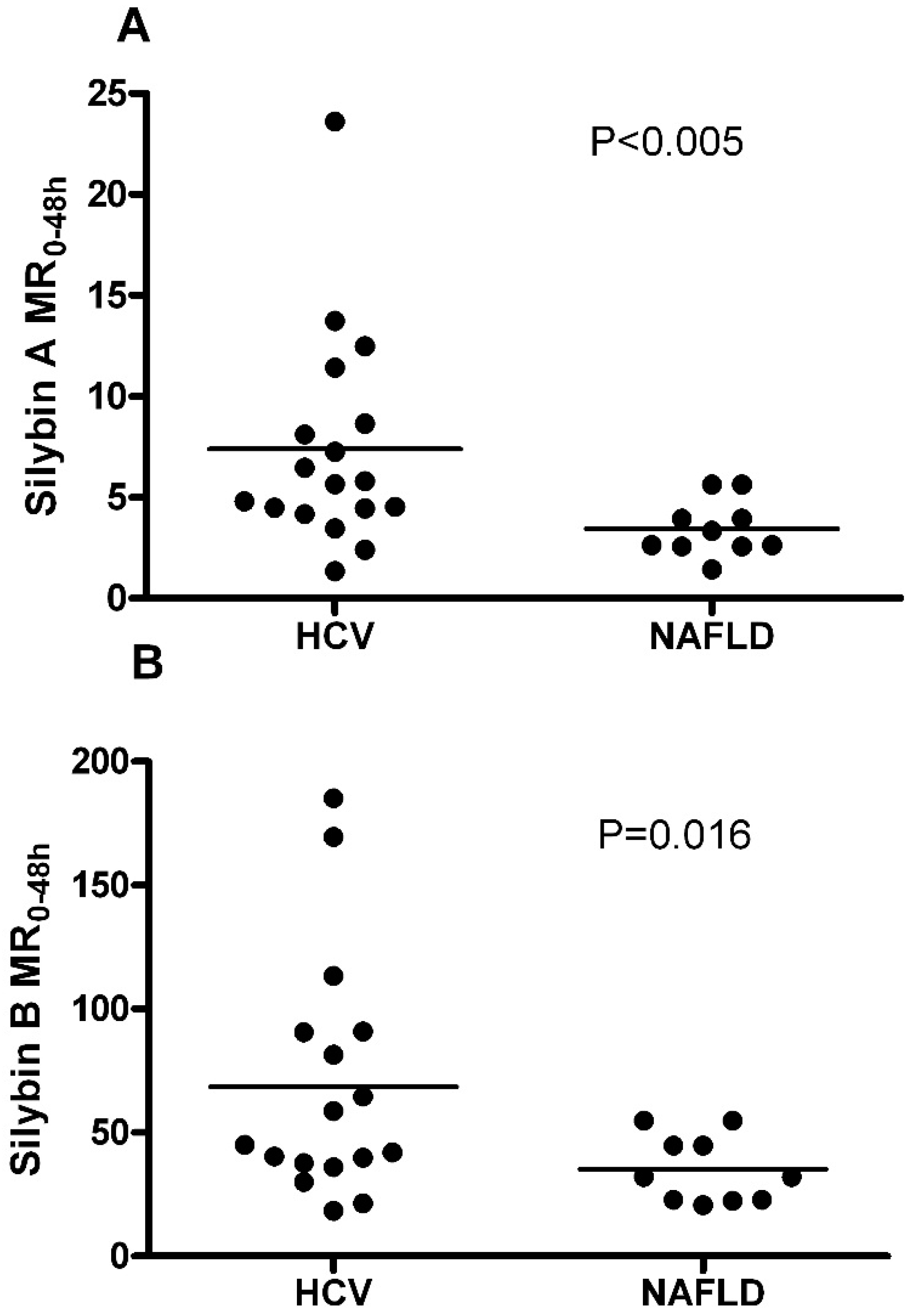
2.3. Effect of Liver Disease Type on Metabolic Ratios for SA and SB
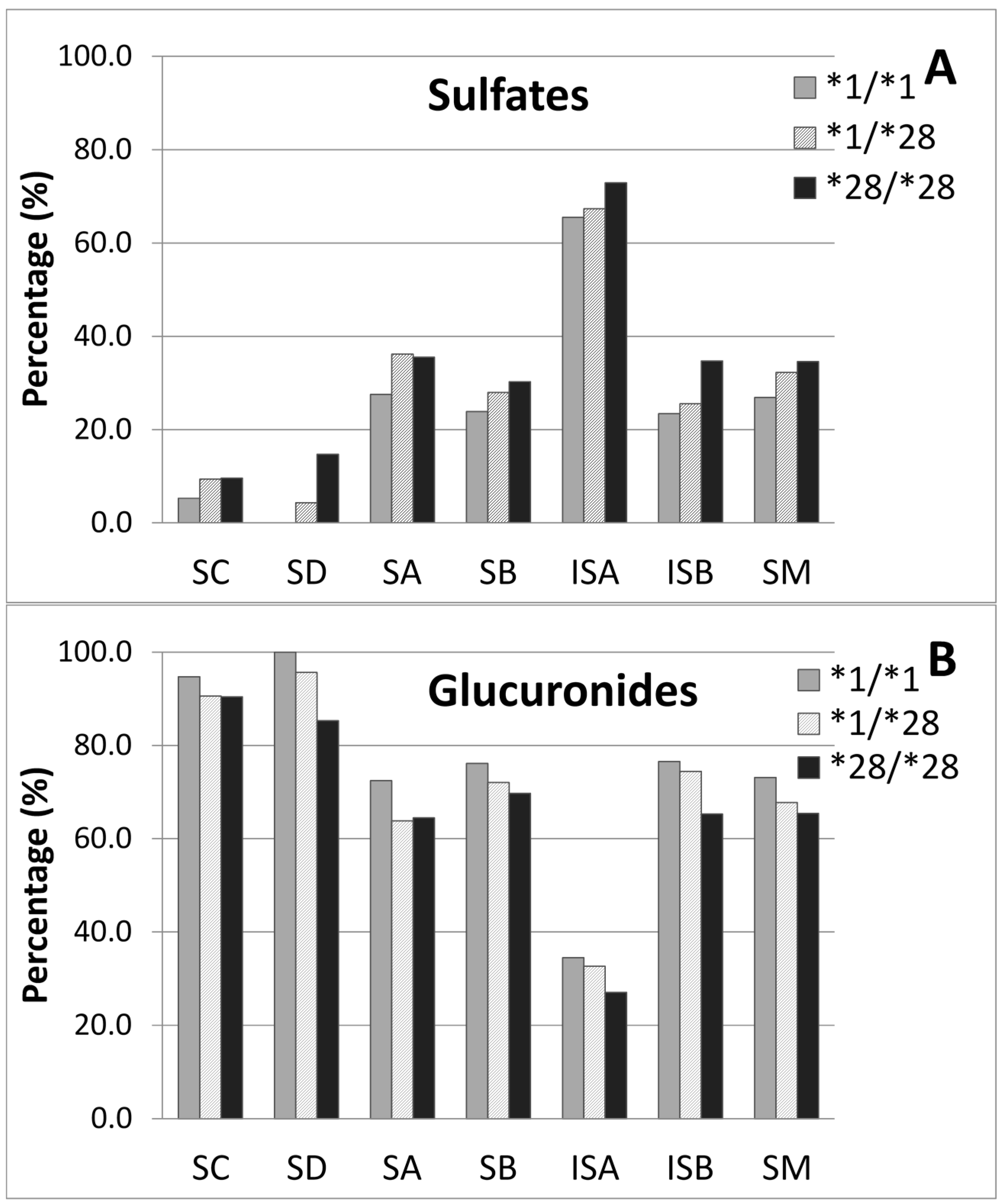
2.4. Metabolites Profile of Silymarin and UGT1A1*28 Effects
3. Materials and Methods
3.1. Subjects and Study Design
3.2. Chemicals and Reagents
3.3. LC/MS Conditions
3.4. UGT Genotyping
3.5. UGT1A1 Substrate Depletion Assay
3.6. Biosynthesis of Glucuronides for Each Silymarin Flavonolignan
3.7. Quantification of Total Glucuronide and Sulfate Concentrations in Pooled Plasma Samples
3.8. Pharmacokinetics Analysis
3.9. Statistical Analysis
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
References
- Flora, K.; Hahn, M.; Rosen, H.; Benner, K. Milk thistle (Silybum marianum) for the therapy of liver disease. Am. J. Gastroenterol. 1998, 93, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.C.; Graf, T.N.; Sparacino, C.M.; Wani, M.C.; Wall, M.E. Complete isolation and characterization of silybins and isosilybins from milk thistle (Silybum marianum). Org. Biomol. Chem. 2003, 1, 1684–1689. [Google Scholar] [CrossRef] [PubMed]
- Seeff, L.B.; Curto, T.M.; Szabo, G.; Everson, G.T.; Bonkovsky, H.L.; Dienstag, J.L.; Shiffman, M.L.; Lindsay, K.L.; Lok, A.S.; Di Bisceglie, A.M.; et al. Herbal product use by persons enrolled in the hepatitis C Antiviral Long-Term Treatment Against Cirrhosis (HALT-C) Trial. Hepatology 2008, 47, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Abenavoli, L.; Capasso, R.; Milic, N.; Capasso, F. Milk thistle in liver diseases: Past, present, future. Phytother. Res. 2010, 24, 1423–1432. [Google Scholar] [CrossRef] [PubMed]
- Ahmed-Belkacem, A.; Ahnou, N.; Barbotte, L.; Wychowski, C.; Pallier, C.; Brillet, R.; Pohl, R.T.; Pawlotsky, J.M. Silibinin and related compounds are direct inhibitors of hepatitis C virus RNA-dependent RNA polymerase. Gastroenterology 2010, 138, 1112–1122. [Google Scholar] [CrossRef] [PubMed]
- Polyak, S.J.; Morishima, C.; Lohmann, V.; Pal, S.; Lee, D.Y.; Liu, Y.; Graf, T.N.; Oberlies, N.H. Identification of hepatoprotective flavonolignans from silymarin. Proc. Natl. Acad. Sci. USA 2010, 107, 5995–5999. [Google Scholar] [CrossRef] [PubMed]
- Rutter, K.; Scherzer, T.M.; Beinhardt, S.; Kerschner, H.; Stättermayer, A.F.; Hofer, H.; Popow-Kraupp, T.; Steindl-Munda, P.; Ferenci, P. Intravenous silibinin as ‘rescue treatment’ for on-treatment non-responders to pegylated interferon/ribavirin combination therapy. Antivir. Ther. 2011, 16, 1327–1333. [Google Scholar] [CrossRef] [PubMed]
- György, I.; Antus, S.; Blázovics, A.; Földiák, G. Substituent effects in the free radical reactions of silybin: Radiation-induced oxidation of the flavonoid at neutral pH. Int. J. Radiat. Biol. 1992, 61, 603–609. [Google Scholar] [CrossRef] [PubMed]
- Day, C.P.; James, O.F. Steatohepatitis: A tale of two “hits”? Gastroenterology 1998, 114, 842–845. [Google Scholar] [CrossRef]
- Chalasani, N.; Deeg, M.A.; Crabb, D.W. Systemic levels of lipid peroxidation and its metabolic and dietary correlates in patients with nonalcoholic steatohepatitis. Am. J. Gastroenterol. 2004, 99, 1497–1502. [Google Scholar] [CrossRef] [PubMed]
- Koruk, M.; Taysi, S.; Savas, M.C.; Yilmaz, O.; Akcay, F.; Karakok, M. Oxidative stress and enzymatic antioxidant status in patients with nonalcoholic steatohepatitis. Ann. Clin. Lab. Sci. 2004, 34, 57–62. [Google Scholar] [PubMed]
- Jain, S.K.; Pemberton, P.W.; Smith, A.; McMahon, R.F.; Burrows, P.C.; Aboutwerat, A.; Warnes, T.W. Oxidative stress in chronic hepatitis C: Not just a feature of late stage disease. J. Hepatol. 2002, 36, 805–811. [Google Scholar] [CrossRef]
- Bahmani, M.; Shirzad, H.; Rafieian, S.; Rafieian-Kopaei, M. Silybum marianum: Beyond Hepatoprotection. J. Evid. Based Complement. Altern. Med. 2015, 20, 292–301. [Google Scholar] [CrossRef] [PubMed]
- Fried, M.W.; Navarro, V.J.; Afdhal, N.; Belle, S.H.; Wahed, A.S.; Hawke, R.L.; Doo, E.; Meyers, C.M.; Reddy, K.R. Silymarin in NASH and C Hepatitis (SyNCH) Study Group. Effect of silymarin (milk thistle) on liver disease in patients with chronic hepatitis C unsuccessfully treated with interferon therapy: A randomized controlled trial. JAMA 2012, 308, 274–282. [Google Scholar] [CrossRef] [PubMed]
- Hawke, R.L.; Schrieber, S.J.; Soule, T.A.; Wen, Z.; Smith, P.C.; Reddy, K.R.; Wahed, A.S.; Belle, S.H.; Afdhal, N.H.; Navarro, V.J.; et al. SyNCH Trial Group. Silymarin ascending multiple oral dosing phase I study in noncirrhotic patients with chronic hepatitis C. J. Clin. Pharmacol. 2010, 50, 434–449. [Google Scholar] [CrossRef] [PubMed]
- Javed, S.; Kojli, K.; Ali, M. Reassessing bioavailability of silymarin. Altern. Med. Rev. 2011, 16, 239–249. [Google Scholar] [PubMed]
- Jančová, P.; Siller, M.; Anzenbacherová, E.; Křen, V.; Anzenbacher, P.; Simánek, V. Evidence for differences in regioselective and stereoselective glucuronidation of silybin diastereomers from milk thistle (Silybum marianum) by human UDP-glucuronosyltransferases. Xenobiotica 2011, 41, 743–751. [Google Scholar] [CrossRef] [PubMed]
- Miranda, S.R.; Lee, J.K.; Wen, Z.; Brouwer, K.L.R.; Smith, P.C.; Hawke, R.L. Disposition of silymarin flavonolignans in isolated perfused gunn rat livers: Role of phase II metabolism and UGT1A1. Drug Metab. Rev. 2008, 40, 316. [Google Scholar]
- Guillemette, C. Pharmacogenomics of human UDP-glucuronosyltransferase enzymes. Pharmacogenom. J. 2003, 3, 136–158. [Google Scholar] [CrossRef] [PubMed]
- Iyer, L.; King, C.D.; Whitington, P.F.; Green, M.D.; Roy, S.K.; Tephly, T.R.; Coffman, B.L.; Ratain, M.J. Genetic predisposition to the metabolism of irinotecan (CPT-11). Role of uridine diphosphate glucuronosyltransferase isoform 1A1 in the glucuronidation of its active metabolite (SN-38) in human liver microsomes. J. Clin. Investig. 1998, 101, 847–854. [Google Scholar] [CrossRef] [PubMed]
- Gil, J.; Sasiadek, M. Gilbert syndrome: The UGT1A1*28 promoter polymorphism as a biomarker of multifactorial diseases and drug metabolism. Biomark. Med. 2012, 6, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, Y.; Minani, H. An overview of the recent progress in irinotecan pharmacogenetics. Pharmacogenomics 2010, 11, 391–406. [Google Scholar] [CrossRef] [PubMed]
- Hoskins, J.M.; Goldberg, R.M.; Qu, P.; Ibrahim, J.G.; McLeod, H.L. UGT1A1*28 genotype and irinotecan-induced neutropenia: Dose matters. J. Natl. Cancer Inst. 2007, 99, 1290–1295. [Google Scholar] [CrossRef] [PubMed]
- Wenning, L.A.; Petry, A.S.; Kost, J.T.; Jin, B.; Breidinger, S.A.; DeLepeleire, I.; Carlini, E.J.; Young, S.; Rushmore, T.; Wagner, F.; et al. Pharmacokinetics of raltegravir in individuals with UGT1A1 polymorphisms. Clin. Pharmacol. Ther. 2009, 85, 623–627. [Google Scholar] [CrossRef] [PubMed]
- Feher, J.; Deak, G.; Muzes, G.; Lang, I.; Niederland, V.; Nekam, K.; Karteszi, M. Liver-protective action of silymarin therapy in chronic alcoholic liver diseases. Orv. Hetil. 1989, 130, 2723–2727. [Google Scholar] [PubMed]
- Pares, A.; Planas, R.; Torres, M.; Caballeria, J.; Viver, J.M.; Acero, D.; Panes, J.; Rigau, J.; Santos, J.; Rodes, J. Effects of silymarin in alcoholic patients with cirrhosis of the liver: Results of a controlled, double-blind, randomized and multicenter trial. J. Hepatol. 1998, 28, 615–621. [Google Scholar] [CrossRef]
- Tănăsescu, C.; Petrea, S.; Băldescu, R.; Macarie, E.; Chiriloiu, C.; Purice, S. Use of the romanian product silimarina in the treatment of chronic liver diseases. Med. Interne 1988, 26, 311–322. [Google Scholar] [PubMed]
- Lampe, J.W.; Bigler, J.; Horner, N.K.; Potter, J.D. UDP-glucuronosyltransferase (UGT1A1*28 and UGT1A6*2) polymorphisms in Caucasians and Asians: Relationships to serum bilirubin concentrations. Pharmacogenetics 1999, 9, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Schrieber, S.J.; Wen, Z.; Vourvahis, M.; Smith, P.C.; Fried, M.W.; Kashuba, A.D.; Hawke, R.L. The pharmacokinetics of silymarin is altered in patients with hepatitis C virus and nonalcoholic fatty liver disease and correlates with plasma caspase-3/7 activity. Drug Metab. Dispos. 2008, 36, 1909–1916. [Google Scholar] [CrossRef] [PubMed]
- Wen, Z.; Dumas, T.E.; Schrieber, S.J.; Hawke, R.L.; Fried, M.W.; Smith, P.C. Pharmacokinetics and metabolic profile of free, conjugated, and total silymarin flavonolignans in human plasma after oral administration of milk thistle extract. Drug Metab. Dispos. 2008, 36, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Iyer, L.; Das, S.; Janisch, L.; Wen, M.; Ramírez, J.; Karrison, T.; Fleming, G.F.; Vokes, E.E.; Schilsky, R.L.; Ratain, M.J. UGT1A1*28 polymorphism as a determinant of irinotecan disposition and toxicity. Pharmacogenom. J. 2002, 2, 43–47. [Google Scholar] [CrossRef]
- Congiu, M.; Mashford, M.; Slavin, J.L.; Desmond, P.V. UDP glucuronosyltransferase mRNA levels in human liver disease. Drug Metab. Dispos. 2002, 30, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.H.; Lou, H.X.; Ren, D.M.; Sun, L.R.; Ma, B.; Ji, M. Stereoselective metabolism of silybin diastereoisomers in the glucuronidation process. J. Pharm. Biomed. Anal. 2004, 34, 1071–1078. [Google Scholar] [CrossRef] [PubMed]
- Kren, V.; Ulrichova, J.; Kosina, P.; Stevenson, D.; Sedmera, P.; Prikrylová, V.; Halada, P.; Simánek, V. Chemoenzymatic preparation of silybin beta-glucuronides and their biological evaluation. Drug Metab. Dispos. 2000, 28, 1513–1517. [Google Scholar] [PubMed]
- Wang, S.W.; Kulkarni, K.H.; Tang, L.; Wang, J.R.; Yin, T.; Daidoji, T.; Yokota, H.; Hu, M. Disposition of flavonoids via enteric recycling: UDP-glucuronosyltransferase (UGT) 1As deficiency in Gunn rats is compensated by increases in UGT2Bs activities. J. Pharmacol. Exp. Ther. 2009, 329, 1023–1031. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.Y.; Hwang, H.; Yousaf, A.M.; Kim, D.W.; Shin, Y.J.; Bae, O.N.; Kim, Y.I.; Kim, J.O.; Yong, C.S.; Choi, H.G. Silymarin-loaded solid nanoparticles provide excellent hepatic protection: Physicochemical characterization and in vivo evaluation. Int. J. Nanomed. 2013, 8, 3333–3343. [Google Scholar]
- Kidd, P.; Head, K. A review of the bioavailability and clinical efficacy of milk thistle phytosome: A silybin-phosphatidylcholine complex (Siliphos). Altern. Med. Rev. 2005, 10, 193–203. [Google Scholar] [PubMed]
- Sample Availability: Not Available.

| Flavonolignan | Km (μM) | Vmax (pmol/mg/min) | Clint (μL/mg/min) |
|---|---|---|---|
| Silybin A | 23.7 ± 17.7 | 2610 ± 1500 | 110.0 |
| Silybin B | 8.7 ± 0.8 | 5920 ± 4550 | 691.0 |
| Silydianin | 9.0 ± 0.8 | 12,740 ± 3710 | 1411.0 |
| Demographics and Laboratory Values | UGT1A1 Genotype | ||
|---|---|---|---|
| *1/*1 | *1/*28 | *28/*28 | |
| Number of subjects | 10 | 14 | 5 |
| Male:Female | 3:7 | 8:6 | 4:1 |
| HCV:NAFLD | 6:4 | 10:4 | 3:2 |
| White:Black:Hispanic | 9:1:0 | 13:0:1 | 1:4:0 |
| Age (year) a | 51.4 (27.8, 60.6) | 50.5 (43.0, 57.2) | 48.7 (43.1, 55.9) |
| BMI (kg/m2) a | 28.9 (21.1, 42.4) | 29.5 (21.5, 36.6) | 34.8 (25.7, 39.2) |
| Total Bilirubin (mg/dL) a | 0.45 (0.1, 1.1) | 0.65 (0.3, 0.8) | 0.7 (0.3, 2.6) |
| Direct Bilirubin (mg/dL) a | 0.29 (0.1, 0.8) | 0.26 (0.1, 0.6) | 0.47 (0.3, 0.6) |
| ALT (U/L) a | 88.5 (48, 164) | 94 (58, 322) | 79 (52, 158) |
| Platelet Count (cells/mm3) a | 228.5 (139, 339) | 194.5 (162, 319) | 254 (108, 327) |
| Flavonolignan | UGT1A1*1/*1 (n = 10) | UGT1A1*1/*28 (n = 14) | UGT1A1*28/*28 (n = 5) | p-Value |
|---|---|---|---|---|
| Median (25th, 75th) | Median (25th, 75th) | Median (25th, 75th) | *1/*1 vs. *1/*28 vs. *28/*28 | |
| Silybin A | 0.17 (0.13, 0.2) | 0.19 (0.15, 0.23) | 0.12 (0.1, 0.23) | 0.78 |
| Silybin B | 0.02 (0.02, 0.03) | 0.02 (0.01, 0.03) | 0.02 (0.02, 0.04) | 0.76 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xie, Y.; Miranda, S.R.; Hoskins, J.M.; Hawke, R.L. Role of UDP-Glucuronosyltransferase 1A1 in the Metabolism and Pharmacokinetics of Silymarin Flavonolignans in Patients with HCV and NAFLD. Molecules 2017, 22, 142. https://doi.org/10.3390/molecules22010142
Xie Y, Miranda SR, Hoskins JM, Hawke RL. Role of UDP-Glucuronosyltransferase 1A1 in the Metabolism and Pharmacokinetics of Silymarin Flavonolignans in Patients with HCV and NAFLD. Molecules. 2017; 22(1):142. https://doi.org/10.3390/molecules22010142
Chicago/Turabian StyleXie, Ying, Sonia R. Miranda, Janelle M. Hoskins, and Roy L. Hawke. 2017. "Role of UDP-Glucuronosyltransferase 1A1 in the Metabolism and Pharmacokinetics of Silymarin Flavonolignans in Patients with HCV and NAFLD" Molecules 22, no. 1: 142. https://doi.org/10.3390/molecules22010142





