Synergism of Antifungal Activity between Mitochondrial Respiration Inhibitors and Kojic Acid
Abstract
:1. Introduction
| Fungal strains | Strain characteristics | Source/Reference | |
|---|---|---|---|
| Aspergillus (Human pathogens) | |||
| A. fumigatus MYA-3626 | Aspergillosis, Reference clinical strain | ATCC a | |
| A. fumigatus AF293 | Aspergillosis, Reference clinical strain | SCVMC b | |
| A. fumigatus AF10 | Aspergillosis, Reference clinical strain | SCVMC b | |
| A. fumigatus 94-46 | Aspergillosis, Clinical isolate | SCVMC b | |
| A. fumigatus 92-245 | Aspergillosis, Clinical isolate | SCVMC b | |
| A. terreus UAB673 | Aspergillosis, Clinical isolate | CDC c | |
| A. terreus UAB680 | Aspergillosis, Clinical isolate | CDC c | |
| A. terreus UAB698 | Aspergillosis, Clinical isolate | CDC c | |
| Other filamentous fungi (Human pathogens) | |||
| Acremonium sp. CIMR 95-103 | Clinical isolate | SCVMC b | |
| Scedosporium sp. CIMR 09-246 | Clinical isolate | SCVMC b | |
| Aspergillus (Plant pathogens, etc.) | |||
| A. flavus 4212 g | Kojic acid producer, Plant pathogen, Human pathogen (aspergillosis) | NRRL d | |
| A. parasiticus 2999 | Kojic acid producer, Plant pathogen | NRRL d | |
| A. oryzae A815 | Research strain (model) | FGSC e | |
| A. niger 326 | Plant pathogen | NRRL d | |
| A. ochraceous 5175 | Plant pathogen | NRRL d | |
| A. nidulans A4 | Research strain (model) | FGSC e | |
| Penicillium (Plant pathogens, etc.) | |||
| P. expansum 974 | Plant pathogen | NRRL d | |
| P. expansum W1 | Plant pathogen | [ 26] | |
| P. expansum FR2 | Plant pathogen, Fludioxonil resistant (FLUDR) mutant derived from P. expansum W1 | [ 26] | |
| P. expansum W2 | Plant pathogen | [ 26] | |
| P. expansum FR3 | Plant pathogen, FLUDR mutant derived from P. expansum W2 | [ 26] | |
| P. chrysogenum 824 | Fleming’s penicillin-producing strain | NRRL d | |
| P. griseofulvum 2159 | Plant pathogen | NRRL d | |
| P. griseofulvum 2300 | Plant pathogen | NRRL d | |
| P. digitatum 786 | Plant pathogen | NRRL d | |
| P. italicum 983 | Plant pathogen | NRRL d | |
| P. glabrum 766 | Plant pathogen | NRRL d | |
| Yeasts | |||
| Saccharomyces cerevisiae BY4741 | Model yeast, Parental strain (Mat a his3∆1 leu2∆0 met15∆0 ura3∆0) | SGD f | |
| S. cerevisiae yap1 | Transcription factor Yap1p mutant derived from BY4741 | SGD f | |
| S. cerevisiae sod2 | Mitochondrial superoxide dismutase (Mn-SOD) mutant derived from BY4741 | SGD f | |
| S. cerevisiae sod1 | Cytosolic superoxide dismutase (Cu,Zn-SOD) mutant derived from BY4741 | SGD f | |
| S. cerevisiae glr1 | Glutathione reductase (Glr1p) mutant derived from BY4741 | SGD f | |
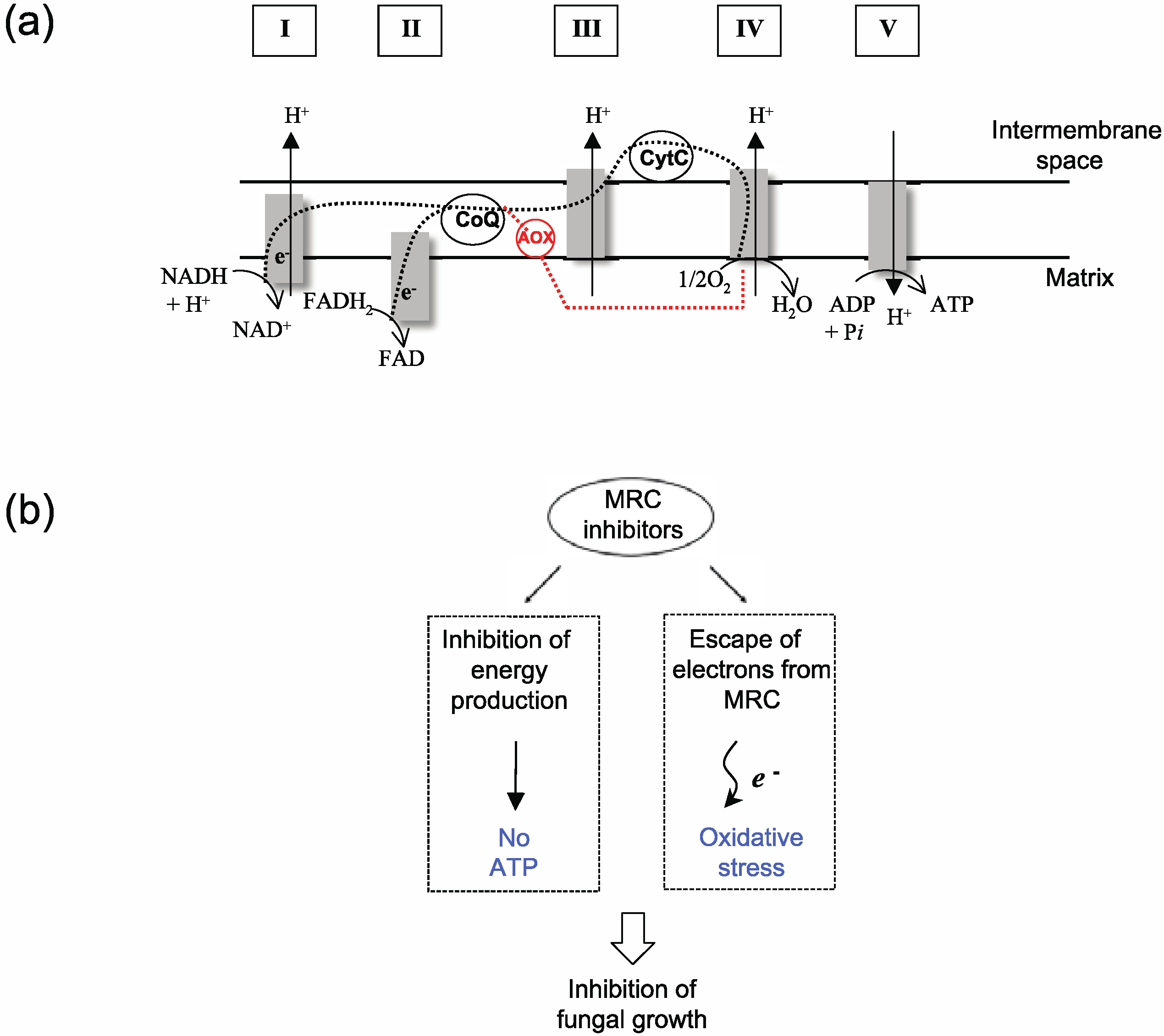
2. Results and Discussion
2.1. Enhancing Antifungal Activity of H2O2 or Complex III Inhibitors with KA Against Aspergillus or Penicillium Strains: Agar Plate Bioassay
2.1.1. Filamentous Fungi
| Strains | H2O2 | Kre-Me | PCS | AntA |
|---|---|---|---|---|
| Human pathogens | ||||
| A. fumigatus MYA-3626 | ++ | + | ++ | + |
| A. fumigatus AF293 | + b | + | ++ | + |
| A. fumigatus AF10 | ++ | + | ++ | + |
| A. fumigatus 94-46 | + | + | ++ | + |
| A. fumigatus 92-245 | + | + | ++ | + |
| A. terreus UAB673 | ++ b | + | ++ | - |
| A. terreus UAB680 | + b | + | ++ | - |
| A. terreus UAB698 | ++ b | - | - | - |
| Acremonium sp. 95-103 | ++ | ++ | n/t c | ++ |
| Scedosporium sp. 09-246 | + | ++ | n/t c | ++ |
| Penicillium strains | ||||
| P. expansum 974 | ++ | - | - | - |
| P. expansum W1 | + | - | - | - |
| P. expansum FR2 | + | - | ++ | - |
| P. expansum W2 | ++ | - | - | - |
| P. expansum FR3 | ++ | - | + | - |
| P. chrysogenum 824 | ++ | - | - | - |
| P. griseofulvum 2159 | ++ | - | - | - |
| P. griseofulvum 2300 | - | - | - | - |
| P. digitatum 786 | ++ | n/t d | n/t c | + |
| P. italicum 983 | - | + | ++ | - |
| P. glabrum 766 | - | + | + | + |
| Other Aspergillus strains | ||||
| A. flavus 4212 | - | - | - | - |
| A. parasiticus 2999 | - | - | - | - |
| A. oryzae A815 | - | - | - | - |
| A. niger 326 | - | - | - | - |
| A. ochraceous 5175 | - | - | - | - |
| A. nidulans A4 | - | + | + | - |
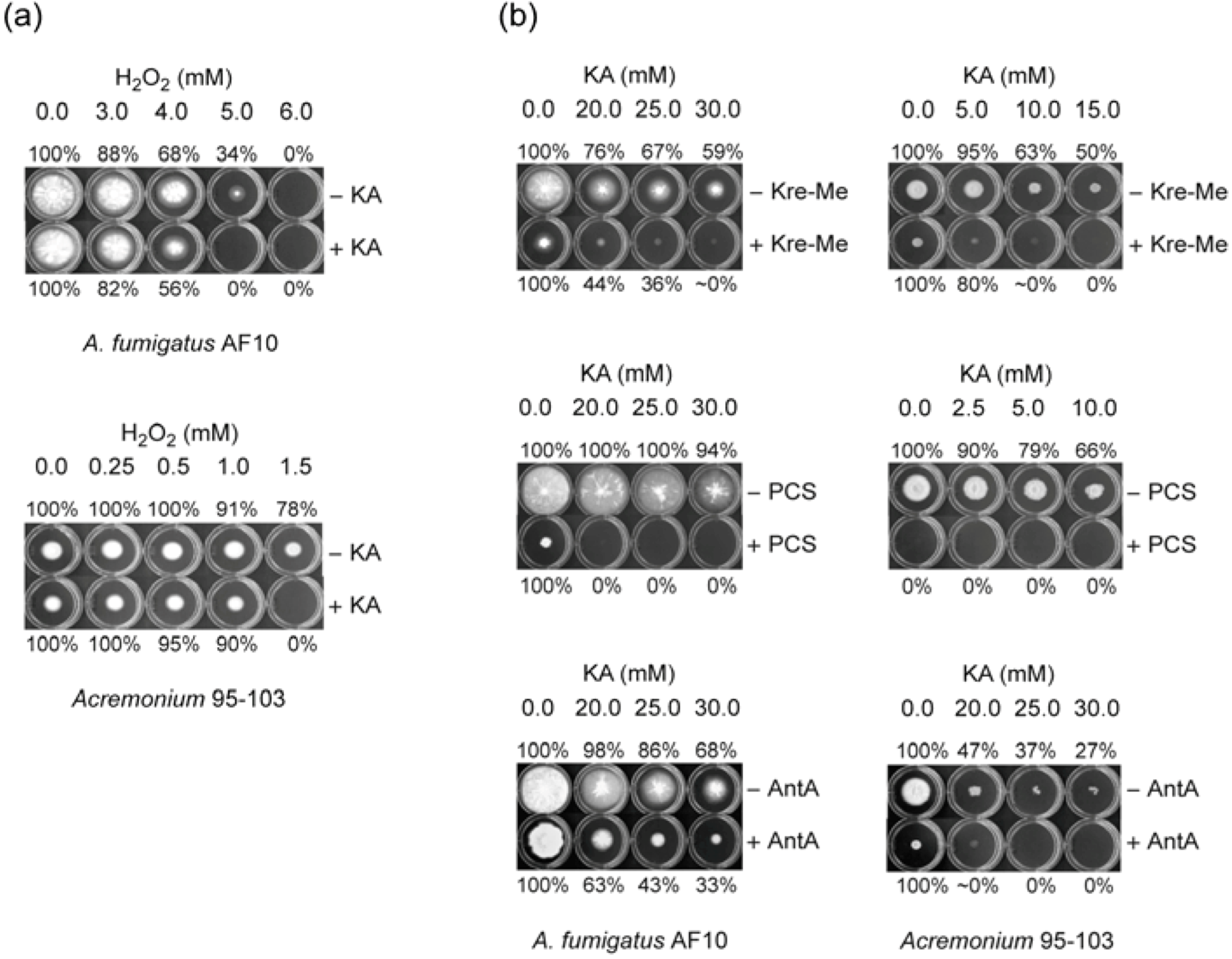
2.1.2. Antioxidant, but Not Antifungal, Effect of KA in Wild Type and Antioxidant Mutants of the Model Yeast Saccharomyces cerevisiae

2.2. Calculating Levels of Compound Interactions by Using Microtiter Plate (Microdilution) Bioassays: Human Pathogens, Penicillium Strains or A. nidulans
2.2.1. Co-Application of KA and PCS
| Strains (Human pathogens and A. nidulans) | Compounds | MIC alone | MIC combined | FICI |
| A. fumigatus AF293 | KA | 64 | 16 | 0.3 |
| PCS | >16 b | 1 | ||
| A. fumigatus MYA-3626 | KA | >64 c | 16 | 0.4 |
| PCS | >16 | 8 | ||
| A. fumigatus AF10 | KA | 64 | 16 | 0.4 |
| PCS | >16 | 4 | ||
| A. fumigatus 92-245 | KA | >64 | 16 | 0.4 |
| PCS | >16 | 8 | ||
| A. fumigatus 94-46 | KA | >64 | 16 | 0.4 |
| PCS | >16 | 8 | ||
| A. terreus UAB673 | KA | 64 | 8 | 0.1 |
| PCS | >16 | 0.5 | ||
| A. terreus UAB 680 | KA | 64 | 8 | 0.2 |
| PCS | >16 | 1 | ||
| A. nidulans A4 | KA | >64 | 32 | 0.5 |
| PCS | >16 | 8 | ||
| Acremonium sp. 95-103 | KA | 64 | 16 | 0.8 |
| PCS | 0.25 | 0.125 | ||
| Scedosporium sp. 09-246 | KA | 64 | 4 | 0.2 |
| PCS | 1 | 0.125 | ||
| Mean | KA | 89.6 | 14.8 | 0.3 |
| PCS | 25.7 | 3.9 | ||
| t-test d | KA | - | p < 0.005 | - |
| PCS | - | p < 0.005 | - | |
| Strains (Human pathogens and A. nidulans) | Compounds | MFC alone | MFC combined | FFCI |
| Acremonium sp. 95-103 | KA | >64 | 32 | 0.5 |
| PCS | 2 | 0.5 | ||
| All other strains | KA | >64 | >64 | 2 |
| PCS | >16 | >16 | ||
| Mean | KA | 128 | 118.4 | 1.9 |
| PCS | 29 | 28.9 | ||
| t-test | KA | - | p < 0.5 | - |
| PCS | - | p < 1.0 | - | |
| Strains (Penicillium strains) | Compounds | MIC alone | MIC combined | FICI |
| P. expansum FR2 | KA | >64 | 16 | 0.4 |
| PCS | 2 | 0.5 | ||
| P. expansum FR3 | KA | >64 | 32 | 0.8 |
| PCS | 2 | 1 | ||
| P. glabrum 766 | KA | >64 | 32 | 0.4 |
| PCS | >16 | 4 | ||
| P. digitatum 786 | KA | >64 | 2 | 0.5 |
| PCS | 0.25 | 0.125 | ||
| P. italicum 983 | KA | >64 | 16 | 0.3 |
| PCS | >16 | 4 | ||
| Mean | KA | 128 | 19.6 | 0.3 |
| PCS | 13.7 | 1.9 | ||
| t-test | KA | - | p < 0.005 | - |
| PCS | - | p < 0.5 | - | |
| Strains (Penicillium strains) | Compounds | MFC alone | MFC combined | FFCI |
| P. glabrum 766 | KA | >64 | 64 | 1 |
| PCS | >16 | 16 | (99.8%) | |
| P. italicum 983 | KA | >64 | 64 | 1 |
| PCS | >16 | 16 | (99.8%) | |
| All other strains | KA | >64 | > 64 | 2 |
| PCS | >16 | >16 | ||
| Mean | KA | 128 | 102.4 | 1.6 |
| PCS | 32 | 25.6 | ||
| t-test | KA | - | p < 0.5 | - |
| PCS | - | p < 0.5 | - |
2.2.2. Strains Hypersensitive to Complex III Inhibitors: Testing Acremonium, Scedosporium, P. digitatum with Kre-Me
| Strains | Compounds | MIC alone | MIC combined | FICI |
| Acremonium sp. 95-103 | KA | 64 | 8 | 0.2 |
| Kre-Me | 16 | 1 | ||
| Scedosporium sp. 09-246 | KA | 64 | 8 | 0.2 |
| Kre-Me | >16 b | 1 | ||
| P. digitatum 786 | KA | >64 c | 8 | 0.1 |
| Kre-Me | 8 | 0.5 | ||
| Mean | KA | 85.3 | 8 | 0.1 |
| Kre-Me | 18.7 | 0.8 | ||
| t-test d | KA | - | p < 0.05 | - |
| Kre-Me | - | p < 0.1 | - | |
| Strains | Compounds | MFC alone | MFC combined | FFCI |
| Acremonium sp. 95-103 | KA | >64 | 64 | 0.6 |
| Kre-Me | >16 | 2 | ||
| Scedosporium sp. 09-246 & P. digitatum 786 | KA | >64 | >64 | 2 |
| Kre-Me | >16 | >16 | ||
| Mean | KA | 128 | 106.7 | 1.5 |
| Kre-Me | 32 | 22 | ||
| t-test d | KA | - | p < 0.5 | - |
| Kre-Me | - | p < 0.5 | - |
| Fungal strains | Agents co-applied | |
|---|---|---|
| PCS (FICI, FFCI) a | Kre-Me (FICI, FFCI) a | |
| Human pathogens | ||
| A. fumigatus AF293 | (0.3, 2.0) | nt b |
| A. fumigatus MYA-3626 | (0.4, 2.0) | nt |
| A. fumigatus AF10 | (0.4, 2.0) | nt |
| A. fumigatus 92-245 | (0.4, 2.0) | nt |
| A. fumigatus 94-46 | (0.4, 2.0) | nt |
| A. terreus UAB673 | (0.1, 2.0) | nt |
| A. terreus UAB680 | (0.2, 2.0) | nt |
| Acremonium sp. 95-103 | (0.8, 0.5) | (0.2, 0.6) |
| Scedosporium sp. 09-246 | (0.2, 2.0) | (0.2, 2.0) |
| Plant pathogens | ||
| P. expansum FR2 | (0.4, 2.0) | nt |
| P. expansum FR3 | (0.8, 2.0) | nt |
| P. glabrum 766 | (0.4, 1.0) | nt |
| P. italicum 983 | (0.3, 1.0) | nt |
| P. digitatum 786 | (0.5, 2.0) | (0.1, 2.0) |
| Other Aspergillus | ||
| A. nidulans A4 | (0.5, 2.0) | nt |
3. Experimental
3.1. Fungal Strains and Culture Conditions
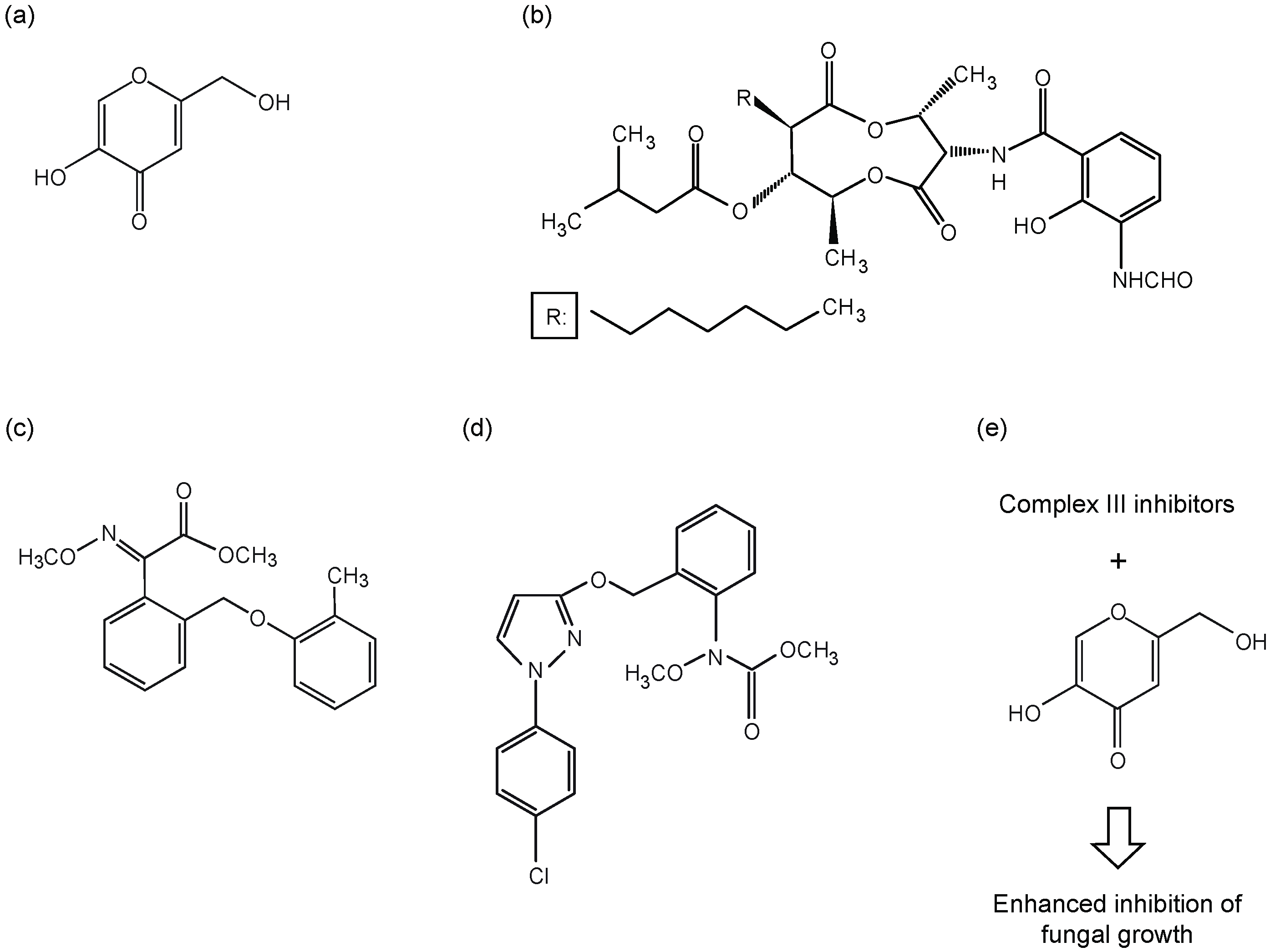
3.2. Chemicals
3.3. Antifungal Bioassay
3.3.1. Agar Plate Bioassay: Filamentous Fungi
3.3.2. Microtiter Plate (microdilution) Bioassay: Filamentous Fungi
3.3.3. Agar Plate Bioassay: S. cerevisiae
4. Conclusions
Acknowledgments
- Sample Availability: Not available.
References
- Fujita, K.; Tani, K.; Usuki, Y.; Tanaka, T.; Taniguchi, M. Growth inhibition dependent on reactive oxygen species generated by C9-UK-2A, a derivative of the antifungal antibiotic UK-2A, in Saccharomyces cerevisiae. J. Antibiot. (Tokyo) 2004, 57, 511–517. [Google Scholar] [CrossRef]
- Ruy, F.; Vercesi, A.E.; Kowaltowski, A.J. Inhibition of specific electron transport pathways leads to oxidative stress and decreased Candida albicans proliferation. J. Bioenerg. Biomembr. 2006, 38, 129–135. [Google Scholar] [CrossRef]
- Longo, V.D.; Gralla, E.B.; Valentine, J.S. Superoxide dismutase activity is essential for stationary phase survival in Saccharomyces cerevisiae. Mitochondrial production of toxic oxygen species in vivo. J. Biol. Chem. 1996, 271, 12275–12280. [Google Scholar] [CrossRef]
- Grant, C.M. Role of the glutathione/glutaredoxin and thioredoxin systems in yeast growth and response to stress conditions. Mol. Microbiol. 2001, 39, 533–541. [Google Scholar] [CrossRef]
- Costa-de-Oliveira, S.; Sampaio-Marques, B.; Barbosa, M.; Ricardo, E.; Pina-Vaz, C.; Ludovico, P.; Rodrigues, A.G. An alternative respiratory pathway on Candida krusei: Implications on susceptibility profile and oxidative stress. FEMS Yeast Res. 2012, 12, 423–429. [Google Scholar] [CrossRef] [Green Version]
- Inoue, K.; Tsurumi, T.; Ishii, H.; Park, P.; Ikeda, K. Cytological evaluation of the effect of azoxystrobin and alternative oxidase inhibitors in Botrytis cinerea. FEMS Microbiol. Lett. 2012, 326, 83–90. [Google Scholar] [CrossRef]
- McIntosh, L. Molecular biology of the alternative oxidase. Plant Physiol. 1994, 105, 781–786. [Google Scholar]
- Ostrosky-Zeichner, L.; Casadevall, A.; Galgiani, J.N.; Odds, F.C.; Rex, J.H. An insight into the antifungal pipeline: Selected new molecules and beyond. Nat. Rev. Drug Discov. 2010, 9, 719–727. [Google Scholar] [CrossRef]
- Srivastava, I.K.; Rottenberg, H.; Vaidya, A.B. Atovaquone, a broad spectrum antiparasitic drug, collapses mitochondrial membrane potential in a malarial parasit. J. Biol. Chem. 1997, 272, 3961–3966. [Google Scholar]
- MedlinePlus. Atovaquone. Available online: http://www.nlm.nih.gov/medlineplus/druginfo/meds/a693003.html/ (accessed on 12 December 2012).
- Niimi, K.; Harding, D.R.; Parshot, R.; King, A.; Lun, D.J.; Decottignies, A.; Niimi, M.; Lin, S.; Cannon, R.D.; Goffeau, A.; et al. Chemosensitization of fluconazole resistance in Saccharomyces cerevisiae and pathogenic fungi by a D-octapeptide derivative. Antimicrob. Agents Chemother. 2004, 48, 1256–1271. [Google Scholar] [CrossRef]
- Lavigne, J.P.; Brunel, J.M.; Chevalier, J.; Pages, J.M. Squalamine, an original chemosensitizer to combat antibiotic-resistant gram-negative bacteria. J. Antimicrob. Chemother. 2010, 65, 799–801. [Google Scholar] [CrossRef]
- Agarwal, A.K.; Tripathi, S.K.; Xu, T.; Jacob, M.R.; Li, X.C.; Clark, A.M. Exploring the molecular basis of antifungal synergies using genome-wide approaches. Front. Microbiol. 2012, 3, 115. [Google Scholar]
- Campbell, B.C.; Chan, K.L.; Kim, J.H. Chemosensitization as a means to augment commercial antifungal agents. Front. Microbiol. 2012, 3, 79. [Google Scholar]
- Srivastava, I.K.; Vaidya, A.B. A mechanism for the synergistic antimalarial action of atovaquone and proguanil. Antimicrob. Agents Chemother. 1999, 43, 1334–1339. [Google Scholar]
- Kim, J.H.; Chan, K.L.; Mahoney, N.; Campbell, B.C. Antifungal activity of redox-active benzaldehydes that target cellular antioxidation. Ann. Clin. Microbiol. Antimicrob. 2011, 10, 23. [Google Scholar] [CrossRef]
- Bentley, R. From miso, sake and shoyu to cosmetics: A century of science for kojic acid. Nat. Prod. Rep. 2006, 23, 1046–1062. [Google Scholar] [CrossRef]
- Chang, T.S. An updated review of tyrosinase inhibitors. Int. J. Mol. Sci. 2009, 10, 2440–2475. [Google Scholar] [CrossRef]
- Leyden, J.J.; Shergill, B.; Micali, G.; Downie, J.; Wallo, W. Natural options for the management of hyperpigmentation. J. Eur. Acad. Dermatol. Venereol. 2011, 25, 1140–1145. [Google Scholar] [CrossRef]
- Lajis, A.F.; Hamid, M.; Ariff, A.B. Depigmenting effect of kojic acid esters in hyperpigmented B16F1 melanoma cells. J. Biomed. Biotechnol. 2012, 2012, 952452. [Google Scholar]
- Niwa, Y.; Akamatsu, H. Kojic acid scavenges free radicals while potentiating leukocyte functions including free radical generation. Inflammation 1991, 15, 303–315. [Google Scholar] [CrossRef]
- Rodrigues, A.P.; Carvalho, A.S.; Santos, A.S.; Alves, C.N.; do Nascimento, J.L.; Silva, E.O. Kojic acid, a secondary metabolite from Aspergillus sp., acts as an inducer of macrophage activation. Cell Biol. Int. 2011, 35, 335–343. [Google Scholar] [CrossRef]
- Reddy, B.V.; Reddy, M.R.; Madan, C.; Kumar, K.P.; Rao, M.S. Indium(III) chloride catalyzed three-component coupling reaction: A novel synthesis of 2-substituted aryl(indolyl)kojic acid derivatives as potent antifungal and antibacterial agents. Bioorg. Med. Chem. Lett. 2010, 20, 7507–7511. [Google Scholar] [CrossRef]
- Chee, H.Y.; Lee, E.H. Fungistatic activity of kojic acid against human pathogenic fungi and inhibition of melanin production in Cryptococcus neoformans. Mycobiology 2003, 31, 248–250. [Google Scholar] [CrossRef]
- Kim, J.H.; Chang, P.K.; Chan, K.L.; Faria, N.C.; Mahoney, N.; Kim, Y.K.; Martins Mde, L.; Campbell, B.C. Enhancement of commercial antifungal agents by kojic acid. Int. J. Mol. Sci. 2012, 13, 13867–13880. [Google Scholar] [CrossRef]
- Li, H.X.; Xiao, C.L. Characterization of fludioxonil-resistant and pyrimethanil-resistant phenotypes of Penicillium expansum from apple. Phytopathology 2008, 98, 427–435. [Google Scholar] [CrossRef]
- Saccharomyces Genome Database. Available online: http://www.yeastgenome.org/ (accessed on 8 December 2012).
- Dinauer, M.C. The respiratory burst oxidase and the molecular genetics of chronic granulomatous disease. Crit. Rev. Clin. Lab. Sci. 1993, 30, 329–369. [Google Scholar] [CrossRef]
- Sokol-Anderson, M.L.; Brajtburg, J.; Medoff, G. Amphotericin B-induced oxidative damage and killing of Candida albicans. J. Infect. Dis. 1986, 154, 76–83. [Google Scholar] [CrossRef]
- Graybill, J.R.; Burgess, D.S.; Hardin, T.C. Key issues concerning fungistatic versus fungicidal drugs. Eur. J. Clin. Microbiol. Infect. Dis. 1997, 16, 42–50. [Google Scholar] [CrossRef]
- An, M.; Shen, H.; Cao, Y.; Zhang, J.; Cai, Y.; Wang, R.; Jiang, Y. Allicin enhances the oxidative damage effect of amphotericin B against Candida albicans. Int. J. Antimicrob. Agents 2009, 33, 258–263. [Google Scholar] [CrossRef]
- Gonzalez-Parraga, P.; Sanchez-Fresneda, R.; Zaragoza, O.; Arguelles, J.C. Amphotericin B induces trehalose synthesis and simultaneously activates an antioxidant enzymatic response in Candida albicans. Biochim. Biophys. Acta 2011, 1810, 777–783. [Google Scholar] [CrossRef]
- Okamoto, Y.; Aoki, S.; Mataga, I. Enhancement of amphotericin B activity against Candida albicans by superoxide radical. Mycopathologia 2004, 158, 9–15. [Google Scholar] [CrossRef]
- Fernandes, L.; Rodrigues-Pousada, C.; Struhl, K. Yap, a novel family of eight bZIP proteins in Saccharomyces cerevisiae with distinct biological functions. Mol. Cell. Biol. 1997, 17, 6982–6993. [Google Scholar]
- Lee, J.; Godon, C.; Lagniel, G.; Spector, D.; Garin, J.; Labarre, J.; Toledano, M.B. Yap1 and Skn7 control two specialized oxidative stress response regulons in yeast. J. Biol. Chem. 1999, 274, 16040–16046. [Google Scholar]
- Hughes, L.M.; Lanteri, C.A.; O’Neil, M.T.; Johnson, J.D.; Gribble, G.W.; Trumpower, B.L. Design of anti-parasitic and anti-fungal hydroxy-naphthoquinones that are less susceptible to drug resistance. Mol. Biochem. Parasitol. 2011, 177, 12–19. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute (CLSI), Reference Method for Broth Dilution Antifungal Susceptibility Testing of Filamentous Fungi: Approved Standard—Second edition. CLSI document M38-A2; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2008; Volume 28.
- Odds, F.C. Synergy, antagonism, and what the chequerboard puts between them. J. Antimicrob. Chemother. 2003, 52, 1. [Google Scholar] [CrossRef]
- Kirkman, T.W. Statistics to use. Available online: http://www.physics.csbsju.edu/stats/ (accessed on 10 December 2012).
- Vincent, J.M. Distortion of fungal hyphae in the presence of certain inhibitors. Nature 1947, 159, 850. [Google Scholar] [CrossRef]
- Kim, J.; Campbell, B.; Mahoney, N.; Chan, K.; Molyneux, R.; May, G. Chemosensitization prevents tolerance of Aspergillus fumigatus to antimycotic drugs. Biochem. Biophys. Res. Commun. 2008, 372, 266–271. [Google Scholar] [CrossRef]
- Burnett, C.L.; Bergfeld, W.F.; Belsito, D.V.; Hill, R.A.; Klaassen, C.D.; Liebler, D.C.; Marks, J.G., Jr.; Shank, R.C.; Slaga, T.J.; Snyder, P.W.; et al. Final report of the safety assessment of kojic acid as used in cosmetics. Int. J. Toxicol. 2010, 29, 244S–273S. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kim, J.H.; Campbell, B.C.; Chan, K.L.; Mahoney, N.; Haff, R.P. Synergism of Antifungal Activity between Mitochondrial Respiration Inhibitors and Kojic Acid. Molecules 2013, 18, 1564-1581. https://doi.org/10.3390/molecules18021564
Kim JH, Campbell BC, Chan KL, Mahoney N, Haff RP. Synergism of Antifungal Activity between Mitochondrial Respiration Inhibitors and Kojic Acid. Molecules. 2013; 18(2):1564-1581. https://doi.org/10.3390/molecules18021564
Chicago/Turabian StyleKim, Jong H., Bruce C. Campbell, Kathleen L. Chan, Noreen Mahoney, and Ronald P. Haff. 2013. "Synergism of Antifungal Activity between Mitochondrial Respiration Inhibitors and Kojic Acid" Molecules 18, no. 2: 1564-1581. https://doi.org/10.3390/molecules18021564






