Starch-Stabilized Iron Oxide Nanoparticles for the Photocatalytic Degradation of Methylene Blue
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
2.2. Instrumentation
2.3. Synthesis of Starch-Stabilized Iron (Fe2+/Fe3+) Oxide Nanoparticles
2.3.1. Starch–Iron (St–Fe) Nanoparticles
2.3.2. Iron–Starch (Fe–St) Nanoparticles
2.3.3. Starch-Free Iron Oxide Nanoparticles
2.4. Quantification of the Iron Content
2.5. Photo-Fenton Experiments
2.6. Spectrophotometric Detection of Hydroxyl Radicals
2.7. Colorimetric Test for Fe3+ Leaching
3. Results and Discussion
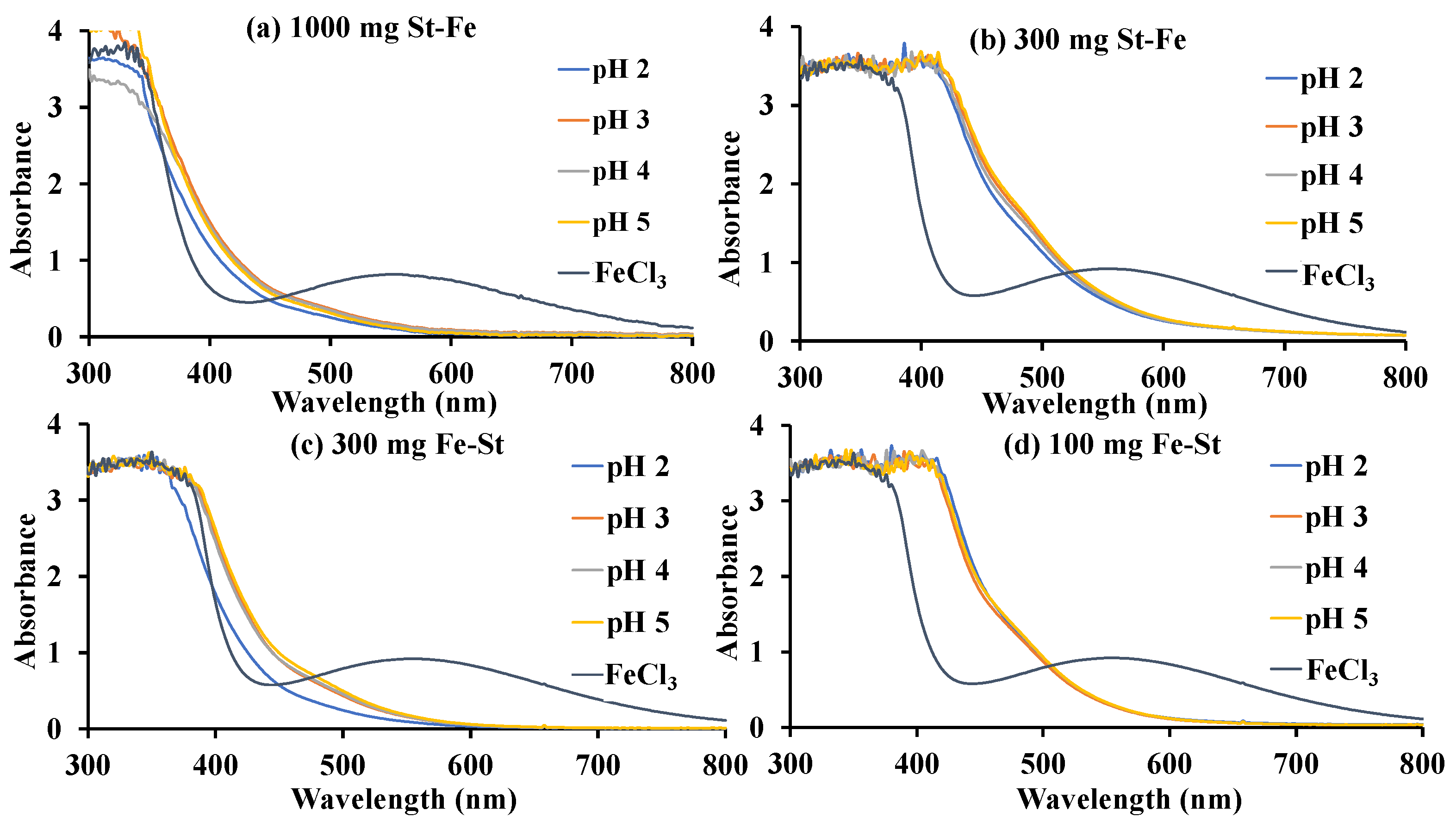
3.1. Catalyst Synthesis and Characterization
3.2. Degradation of MB with Starch-Stabilized Iron Oxide Nanoparticles
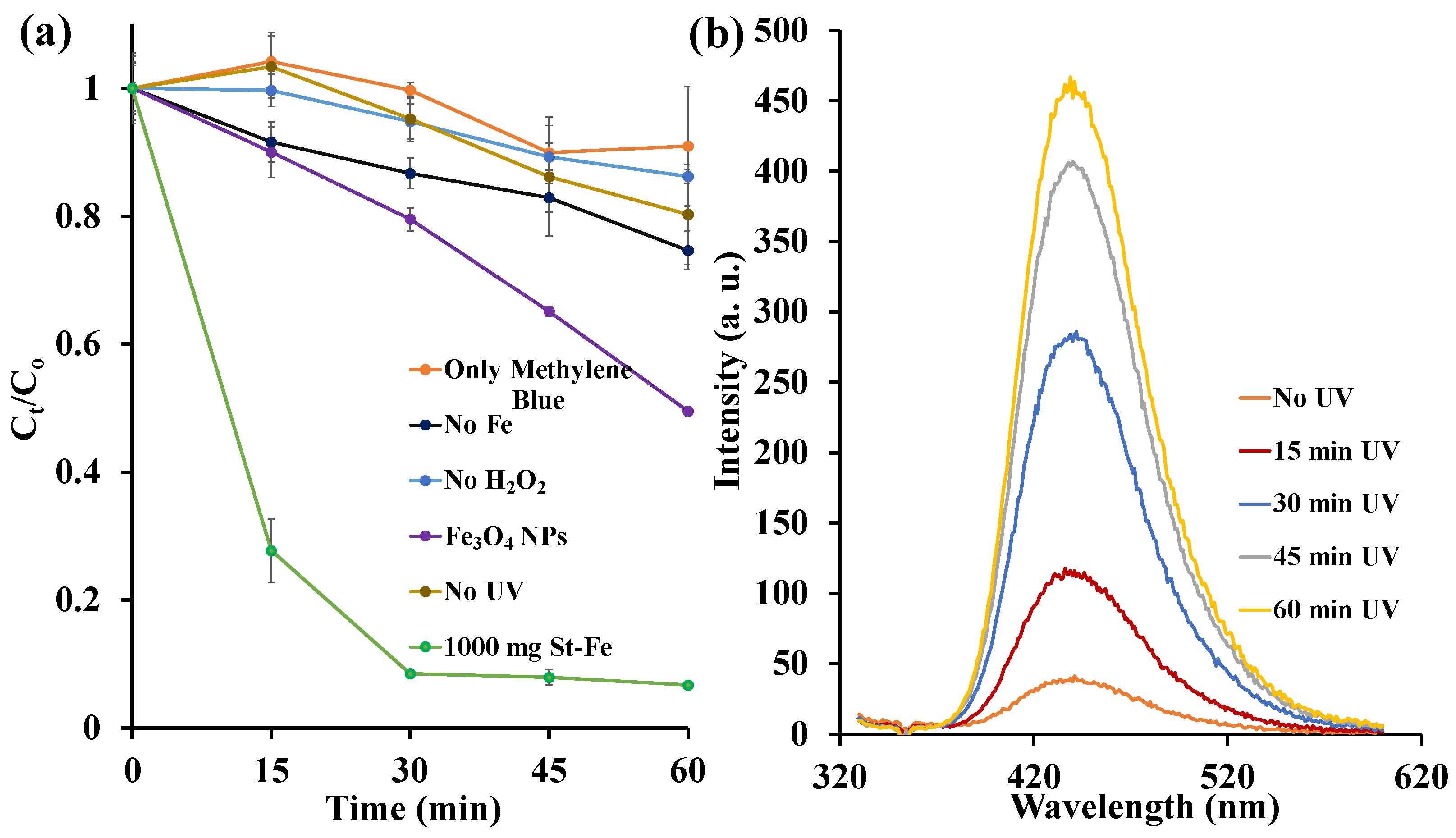
3.2.1. Control Experiments
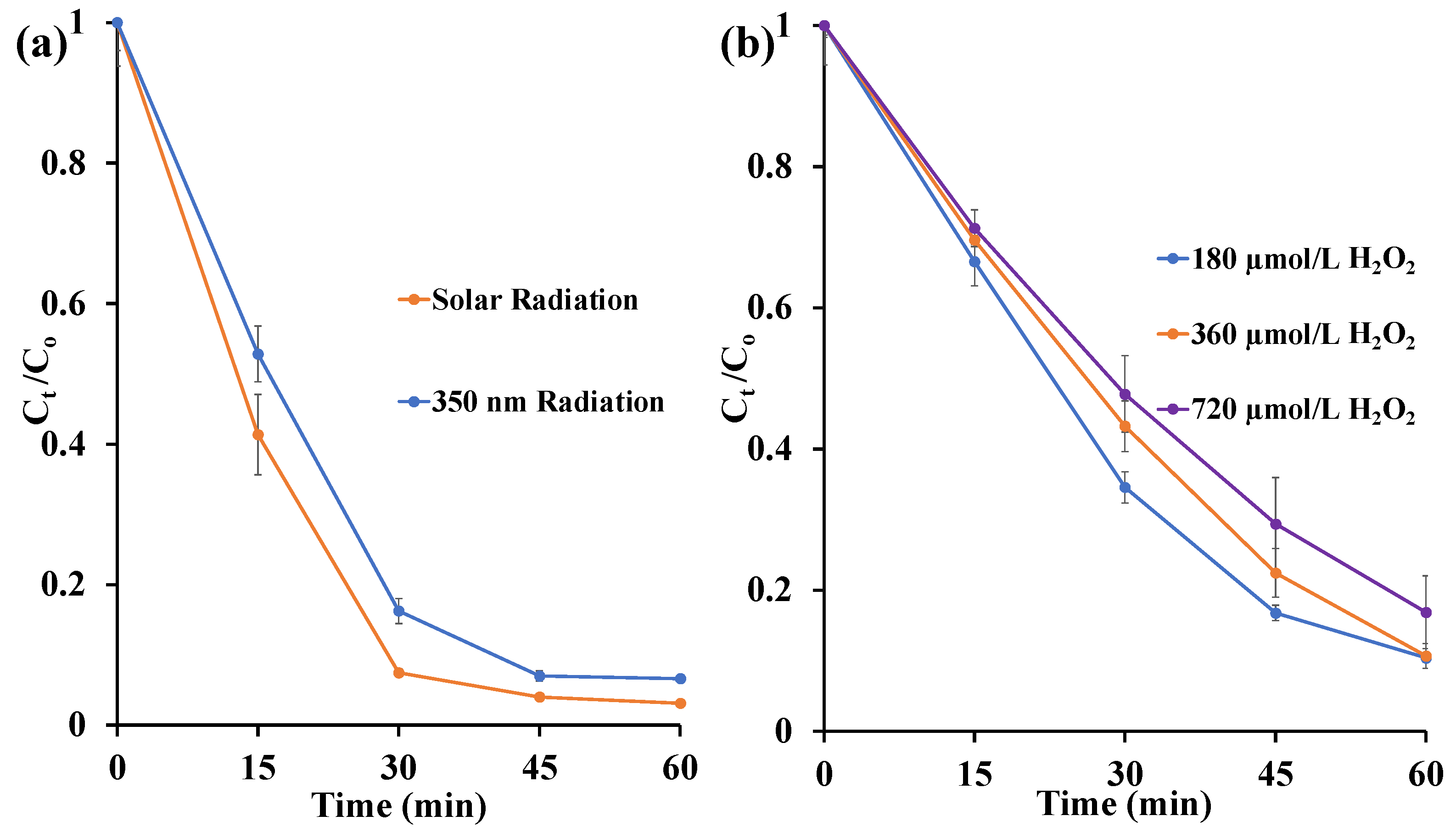
3.2.2. Influence of UV Radiation Source and H2O2 Concentration
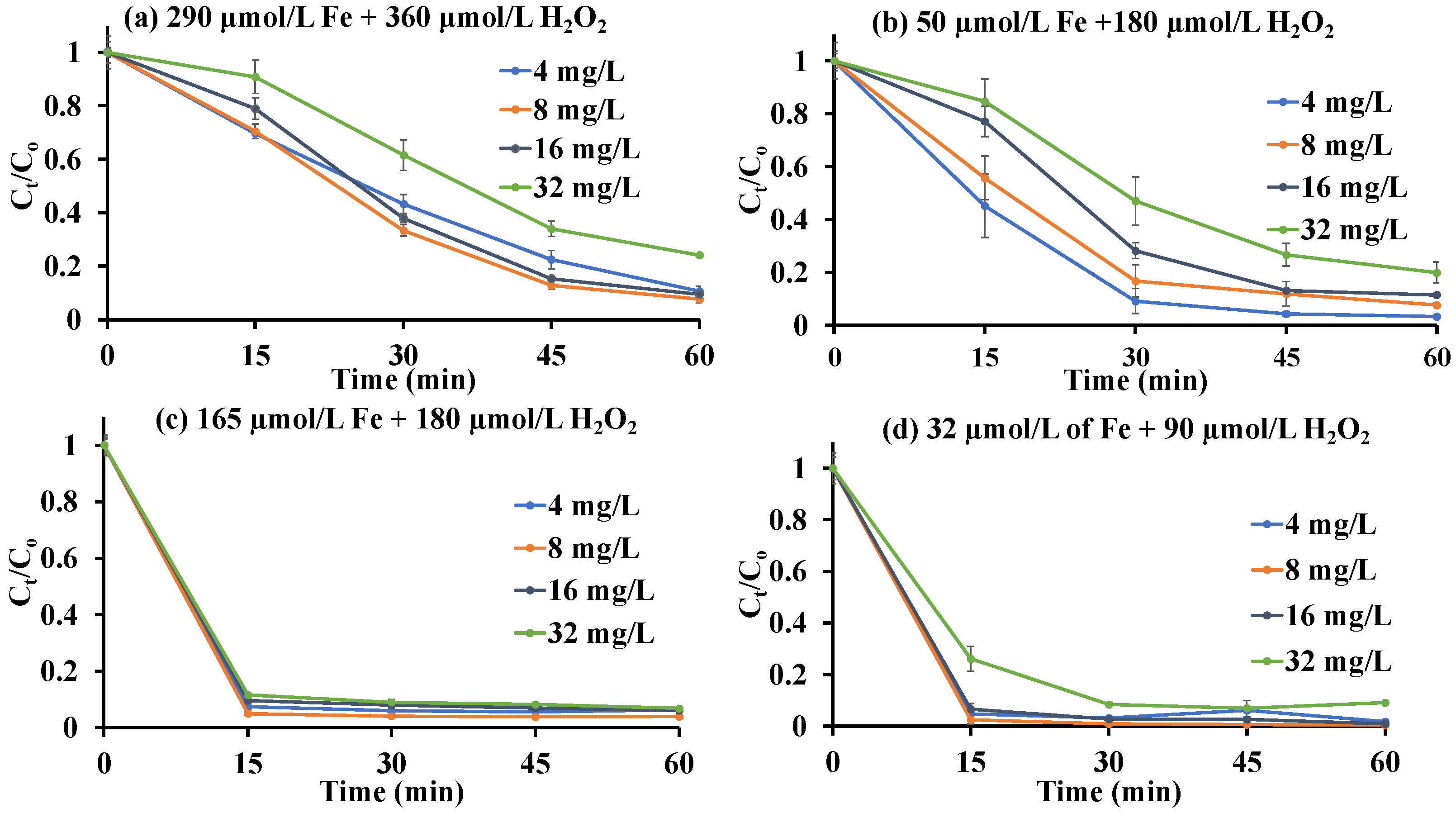
3.2.3. Optimization of the H2O2: Fe Ratio
3.2.4. Activity of the Starch-Stabilized Iron Catalysts
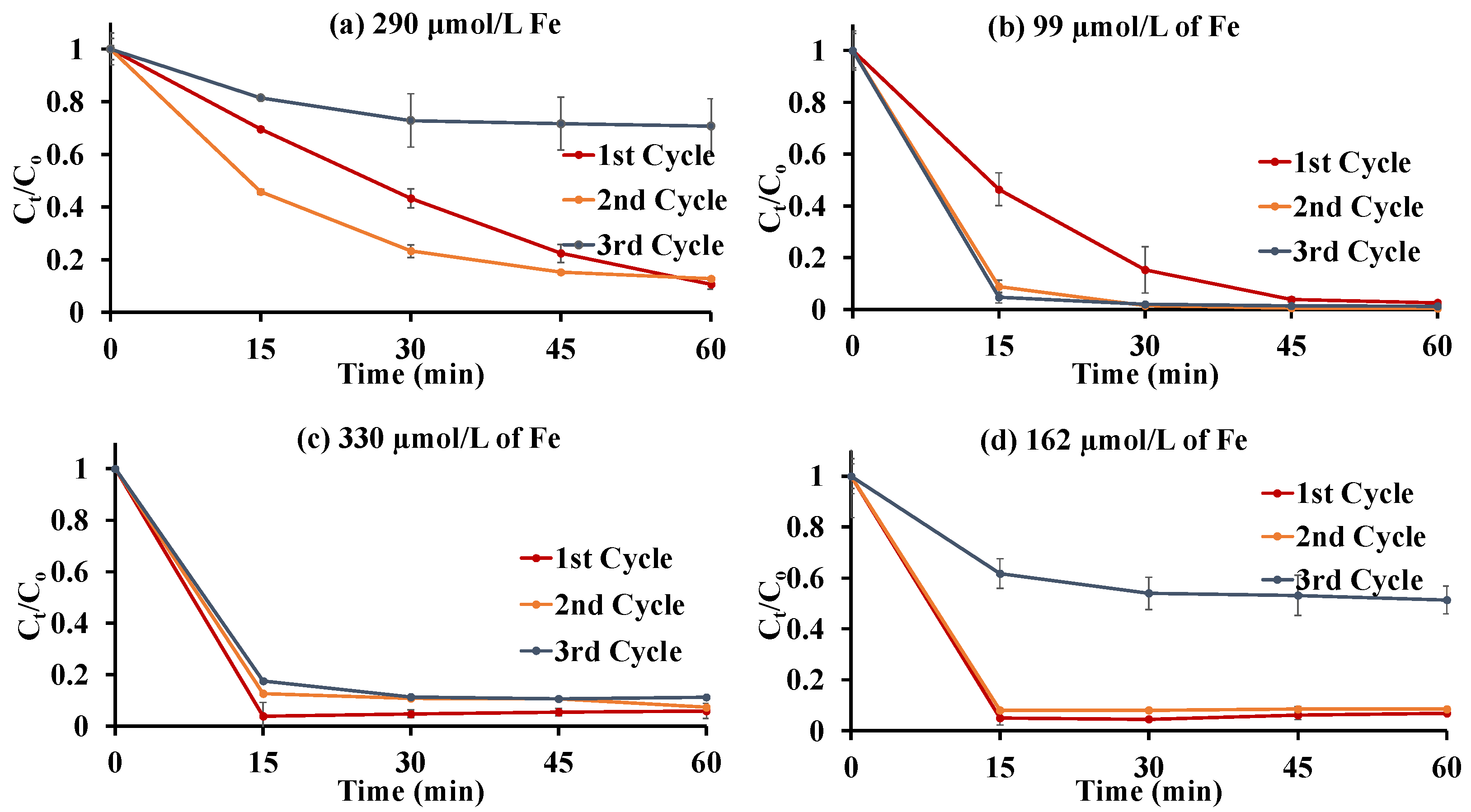
3.2.5. Multiple Additions of Methylene Blue
3.3. Total Organic Carbon (TOC) Analysis
3.4. Iron Leaching from the Starch-Stabilized NPs
3.5. Catalyst Performance Comparison with Other Reported Work
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Corradini, M.G. Synthetic Food Colors. Encycl. Food Chem. 2018, 1, 291–296. [Google Scholar]
- Arif, Z.; Sethy, N.K.; Swati; Mishra, P.K.; Verma, B. Grossly Polluting Industries and Their Effect on Water Resources in India. Chapter 3. Pollut. Water Manag. 2021, 47–65. [Google Scholar] [CrossRef]
- Punzi, M.; Mattiasson, B.; Jonstrup, M. Treatment of Synthetic Textile Wastewater by Homogeneous and Heterogeneous Photo-Fenton Oxidation. J. Photochem. Photobiol. A Chem. 2012, 248, 30–35. [Google Scholar] [CrossRef]
- Drumond Chequer, F.M.; de Oliveira, G.A.R.; Anastacio Ferraz, E.R.; Carvalho, J.; Boldrin Zanoni, M.V.; de Oliveir, D.P. Textile Dyes: Dyeing Process and Environmental Impact. In Eco-Friendly Textile Dyeing and Finishing; IntechOpen: London, UK, 2013. [Google Scholar]
- Hassaan, M.A.; Nemr, A.E. Health and Environmental Impacts of Dyes: Mini Review. Am. J. Environ. Sci. Eng. 2017, 1, 64–67. [Google Scholar]
- Sarayu, K.; Sandhya, S. Current Technologies for Biological Treatment of Textile Wastewater-A Review. Appl. Biochem. Biotechnol. 2012, 167, 645–661. [Google Scholar] [CrossRef]
- Pérez, M.; Torrades, F.; Domènech, X.; Peral, J. Fenton and Photo-Fenton Oxidation of Textile Effluents. Water Res. 2002, 36, 2703–2710. [Google Scholar] [CrossRef]
- Holkar, C.R.; Jadhav, A.J.; Pinjari, D.V.; Mahamuni, N.M.; Pandit, A.B. A Critical Review on Textile Wastewater Treatments: Possible Approaches. J. Environ. Manag. 2016, 182, 351–366. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.L.; Xu, L.J. Advanced Oxidation Processes for Wastewater Treatment: Formation of Hydroxyl Radical and Application. Crit. Rev. Environ. Sci. Technol. 2012, 42, 251–325. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhu, R.; Xi, Y.; Zhu, J.; Zhu, G.; He, H. Strategies for Enhancing the Heterogeneous Fenton Catalytic Reactivity: A Review. Appl. Catal. B Environ. 2019, 255, 117739. [Google Scholar] [CrossRef]
- Munoz, M.; de Pedro, Z.M.; Casas, J.A.; Rodriguez, J.J. Preparation of Magnetite-Based Catalysts and Their Application in Heterogeneous Fenton Oxidation—A Review. Appl. Catal. B Environ. 2015, 176–177, 249–265. [Google Scholar] [CrossRef]
- Pliego, G.; Zazo, J.A.; Garcia-Muñoz, P.; Munoz, M.; Casas, J.A.; Rodriguez, J.J. Trends in the Intensification of the Fenton Process for Wastewater Treatment: An Overview. Crit. Rev. Environ. Sci. Technol. 2015, 45, 2611–2692. [Google Scholar] [CrossRef]
- Zhang, M.; Dong, H.; Zhao, L.; Wang, D.; Meng, D. A Review on Fenton Process for Organic Wastewater Treatment Based on Optimization Perspective. Sci. Total Environ. 2019, 670, 110–121. [Google Scholar] [CrossRef] [PubMed]
- Karthikeyan, S.; Titus, A.; Gnanamani, A.; Mandal, A.B.; Sekaran, G. Treatment of Textile Wastewater by Homogeneous and Heterogeneous Fenton Oxidation Processes. Desalination 2011, 281, 438–445. [Google Scholar] [CrossRef]
- Clarizia, L.; Russo, D.; di Somma, I.; Marotta, R.; Andreozzi, R. Homogeneous Photo-Fenton Processes at near Neutral PH: A Review. Appl. Catal. B Environ. 2017, 209, 358–371. [Google Scholar] [CrossRef]
- Gligorovski, S.; Strekowski, R.; Barbati, S.; Vione, D. Environmental Implications of Hydroxyl Radicals (•OH). Chem. Rev. 2015, 115, 13051–13092. [Google Scholar] [CrossRef] [PubMed]
- Xue, W.; Huang, D.; Zeng, G.; Wan, J.; Zhang, C.; Xu, R.; Cheng, M.; Deng, R. Nanoscale Zero-Valent Iron Coated with Rhamnolipid as an Effective Stabilizer for Immobilization of Cd and Pb in River Sediments. J. Hazard. Mater. 2018, 341, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Jain, B.; Singh, A.K.; Kim, H.; Lichtfouse, E.; Sharma, V.K. Treatment of Organic Pollutants by Homogeneous and Heterogeneous Fenton Reaction Processes. Environ. Chem. Lett. 2018, 341, 947–967. [Google Scholar] [CrossRef]
- Jenita Rani, G.; Jothi Rajan, M.A.; Gnana Kumar, G. Reduced Graphene Oxide/ZnFe2O4 Nanocomposite as an Efficient Catalyst for the Photocatalytic Degradation of Methylene Blue Dye. Res. Chem. Intermed. 2017, 43, 2669–2690. [Google Scholar] [CrossRef]
- Guo, X.; Wang, K.; Xu, Y. Tartaric Acid Enhanced CuFe2O4-Catalyzed Heterogeneous Photo-Fenton-like Degradation of Methylene Blue. Mater. Sci. Eng. B Solid-State Mater. Adv. Technol. 2019, 245, 75–84. [Google Scholar] [CrossRef]
- Guo, X.; Meng, Q.; Wang, D.; Zha, F.; Tang, X.; Tian, H. S-Doped NiFe-Based Catalyst for Fast Degradation of Methylene Blue by Heterogeneous Photo-Fenton Reaction. Environ. Sci. Pollut. Res. 2021, 28, 36112–36121. [Google Scholar] [CrossRef]
- Ashraf, G.A.; Rasool, R.T.; Hassan, M.; Zhang, L. Enhanced Photo Fenton-like Activity by Effective and Stable Al–Sm M-Hexaferrite Heterogenous Catalyst Magnetically Detachable for Methylene Blue Degradation. J. Alloy Compd. 2020, 821, 153410. [Google Scholar] [CrossRef]
- Abbas Ashraf, G.; Hassan, M.; Tur Rasool, R.; Abbas, W.; Zhang, L. Mesoporous SnMgNd Substituted M-Hexaferrite Catalyzed Heterogeneous Photo-Fenton-like Activity for Degradation of Methylene Blue. J. Colloid Interface Sci. 2019, 557, 408–422. [Google Scholar] [CrossRef] [PubMed]
- Kalam, A.; Al-Sehemi, A.G.; Assiri, M.; Du, G.; Ahmad, T.; Ahmad, I.; Pannipara, M. Modified Solvothermal Synthesis of Cobalt Ferrite (CoFe2O4) Magnetic Nanoparticles Photocatalysts for Degradation of Methylene Blue with H2O2/Visible Light. Results Phys. 2018, 8, 1046–1053. [Google Scholar] [CrossRef]
- Su, L.; Xiong, Y.; Wu, M.; Duan, Z.; Zhang, Z.; Xie, W.; Zhu, D.; Luo, Y.; He, X. From Starch to Magnetic Porous Fe2O3@C: A Promising Peroxidase-Mimicking Heterogeneous Biocatalyst for Degradation of Organic Dye. Starch-Stärke 2018, 70, 1700221. [Google Scholar] [CrossRef]
- El Qada, E.N.; Allen, S.J.; Walker, G.M. Adsorption of Methylene Blue onto Activated Carbon Produced from Steam Activated Bituminous Coal: A Study of Equilibrium Adsorption Isotherm. Chem. Eng. J. 2006, 124, 103–110. [Google Scholar] [CrossRef]
- Tang, M.; Wang, D.; Hou, Y.; Buchili, P.; Sun, L. Preparation, Characterization, Bioavailability in Vitro and in Vivo of Tea Polysaccharides-Iron Complex. Eur. Food Res. Technol. 2013, 236, 341–350. [Google Scholar] [CrossRef]
- Krenkova, J.; Foret, F. Iron Oxide Nanoparticle Coating of Organic Polymer-Based Monolithic Columns for Phosphopeptide Enrichment. J. Sep. Sci. 2011, 34, 2106–2112. [Google Scholar] [CrossRef]
- Karaagac, O.; Kockar, H. Iron Oxide Nanoparticles Co-Precipitated in Air Environment: Effect of [Fe2+]/[Fe3+] Ratio. IEEE Trans. Magn. 2012, 48, 1532–1536. [Google Scholar] [CrossRef]
- Pitarresi, G.; Tripodo, G.; Cavallaro, G.; Palumbo, F.S.; Giammona, G. Inulin-Iron Complexes: A Potential Treatment of Iron Deficiency Anaemia. Eur. J. Pharm. Biopharm. 2008, 68, 267–276. [Google Scholar] [CrossRef]
- Baalousha, M.; Manciulea, A.; Cumberland, S.; Kendall, K.; Lead, J.R. Aggregation and Surface Properties of Iron Oxide Nanoparticles: Influence of pH and Natural Organic Matter. Environ. Toxicol. Chem. 2008, 27, 1875–1882. [Google Scholar] [CrossRef]
- Soshnikova, Y.M.; Roman, S.G.; Chebotareva, N.A.; Baum, O.I.; Obrezkova, M.V.; Gillis, R.B.; Harding, S.E.; Sobol, E.N.; Lunin, V.V. Starch-Modified Magnetite Nanoparticles for Impregnation into Cartilage. J. Nanopart. Res. 2013, 15, 2092. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, L. Kinetic Study of Hydroxyl Radical Formation in a Continuous Hydroxyl Generation System. RSC Adv. 2018, 8, 40632–40638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watts, R.J.; Foget, M.K.; Kong, S.H.; Teel, A.L. Hydrogen Peroxide Decomposition in Model Subsurface Systems. J. Hazard. Mater. 1999, 69, 229–243. [Google Scholar] [CrossRef]
- Mierzwa, J.C.; Rodrigues, R.; Teixeira, A.C.S.C. UV-Hydrogen Peroxide Processes. In Advanced Oxidation Processes for Wastewater Treatment: Emerging Green Chemical Technology; Academic Press: London, UK, 2018; pp. 13–48. [Google Scholar]
- Neamtu, M.; Yediler, A.; Siminiceanu, I.; Kettrup, A. Oxidation of Commercial Reactive Azo Dye Aqueous Solutions by the Photo-Fenton and Fenton-like Processes. J. Photochem. Photobiol. A Chem. 2003, 161, 87–93. [Google Scholar] [CrossRef]
- Tengrui, L.; Al-Harbawi, A.; Jun, Z.; Bo, L.M. The Effect and Its Influence Factors of the Fenton Process on the Old Landfill Leachate. J. Appl. Sci. 2007, 7, 724–727. [Google Scholar] [CrossRef]
- Parovuori, P.; Hamunen, A.; Forssell, P.; Autio, K.; Poutanen, K. Oxidation of Potato Starch by Hydrogen Peroxide. Starch-Stärke 1995, 47, 19–23. [Google Scholar] [CrossRef]
- Silver, J.; Morrison, I.E.G.; Rees, L.V.C. A Mössbauer Spectroscopic Study of Frozen Solutions of FeCl3-Phenols. Inorg. Nucl. Chem. Lett. 1979, 15, 433–436. [Google Scholar] [CrossRef]
- Zhou, G.; Fang, F.; Chen, Z.; He, Y.; Sun, H.; Shi, H. Facile Synthesis of Paper Mill Sludge-Derived Heterogeneous Catalyst for the Fenton-like Degradation of Methylene Blue. Catal. Commun. 2015, 62, 71–74. [Google Scholar] [CrossRef]
- Liu, Y.; Jin, W.; Zhao, Y.; Zhang, G.; Zhang, W. Enhanced Catalytic Degradation of Methylene Blue by α-Fe2O3/Graphene Oxide via Heterogeneous Photo-Fenton Reactions. Appl. Catal. B Environ. 2017, 206, 642–652. [Google Scholar] [CrossRef]
- Ahmed, Y.; Yaakob, Z.; Akhtar, P. Degradation and Mineralization of Methylene Blue Using a Heterogeneous Photo-Fenton Catalyst under Visible and Solar Light Irradiation. Catal. Sci. Technol. 2016, 6, 1222–1232. [Google Scholar] [CrossRef]
- Walling, S.A.; Um, W.; Corkhill, C.L.; Hyatt, N.C. Fenton and Fenton-like Wet Oxidation for Degradation and Destruction of Organic Radioactive Wastes. npj Mater. Degrad. 2021, 5, 2021. [Google Scholar] [CrossRef]


| Step | Reaction | Rate Constant (M−1 s−1) | References |
|---|---|---|---|
| Initiation | [9,10,11] | ||
| [9,10,11] | |||
| [9,11] | |||
| Propagation | [10,11] | ||
| [10] | |||
| [10] | |||
| [9,10] | |||
| - | [10] | ||
| - | [10,11] | ||
| - | [10,11] | ||
| Termination | [9,10,11,12] | ||
| [10,11] | |||
| [10,11] | |||
| [10,11] | |||
| - | [10,11] | ||
| [10,11] | |||
| [10] |
| Catalyst | Initial Weight Ratio of Iron: Citrate: Starch | Iron (% w/w) |
|---|---|---|
| 1000 mg St–Fe | ||
| 300 mg St–Fe | ||
| 300 mg Fe–St | ||
| 100 mg Fe–St |
| (a) 300 mg St–Fe 50 μmol/L Fe + 180 μmol/L H2O2 | |||
|---|---|---|---|
| Sample | Initial TOC (mg/L) | Final TOC | % Mineralized |
| Blank | 4.13 | 3.35 | - |
| 4 mg/L MB | 6.57 | 4.41 | 56.7 |
| 8 mg/L MB | 8.94 | 5.88 | 47.5 |
| 16 mg/L MB | 13.74 | 9.09 | 40.4 |
| (b) 300 mg Fe–St 33 μmol/L Fe + 45 μmol/L H2O2 | |||
| Sample | Initial TOC (mg/L) | Final TOC | % Mineralized |
| Blank | 1.41 | 0.46 | - |
| 4 mg/L MB | 3.81 | 1.66 | 50.2 |
| 8 mg/L MB | 6.22 | 3.40 | 38.9 |
| 16 mg/L MB | 11.03 | 7.75 | 24.2 |
| (c) 100 mg Fe–St 32 μmol/L Fe + 90 μmol/L H2O2 | |||
| Sample | Initial TOC (mg/L) | Final TOC (mg/L) | % Mineralized |
| Blank | 2.12 | 0.7 | - |
| 4 mg/L MB | 4.52 | 1.53 | 65.3 |
| 8 mg/L MB | 6.92 | 2.49 | 62.9 |
| 16 mg/L MB | 11.73 | 6.20 | 42.8 |
| Catalyst System | MB Conc. (mg/L) | Iron Conc. (mg/L) | H2O2 Conc. (µmol/L) | Reaction Time (min) | % Discoloured | % Mineralized |
|---|---|---|---|---|---|---|
| PMS-Fe-400 [40] | 50 | 500 | 1800 | 80 | 93.3 | 78 |
| Fe2O3/GO [41] | 40 | 100 | 1100 | 80 | 99 | 56 |
| Fe–Ni/SiO2 [42] | 40 | 85 | 3000 | 60 | 99.8 | 86.2 |
| 100 mg Fe–St | 4 | 1.8 | 90 | 60 | 99 | 65.3 |
| 100 mg Fe–St | 8 | 1.8 | 90 | 60 | 99 | 62.9 |
| 100 mg Fe–St | 32 | 1.8 | 90 | 30 | 92 | N/A |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dasgupta, N.; Nayak, M.A.; Gauthier, M. Starch-Stabilized Iron Oxide Nanoparticles for the Photocatalytic Degradation of Methylene Blue. Polysaccharides 2022, 3, 655-670. https://doi.org/10.3390/polysaccharides3030038
Dasgupta N, Nayak MA, Gauthier M. Starch-Stabilized Iron Oxide Nanoparticles for the Photocatalytic Degradation of Methylene Blue. Polysaccharides. 2022; 3(3):655-670. https://doi.org/10.3390/polysaccharides3030038
Chicago/Turabian StyleDasgupta, Natun, Milind Ajith Nayak, and Mario Gauthier. 2022. "Starch-Stabilized Iron Oxide Nanoparticles for the Photocatalytic Degradation of Methylene Blue" Polysaccharides 3, no. 3: 655-670. https://doi.org/10.3390/polysaccharides3030038






