Porous Diatomaceous Earth/Nano-Zinc Oxide Composites: Preparation and Antimicrobial Applications
Abstract
:1. Introduction
2. Experimental Section
2.1. Chemicals and Materials
2.2. Instrumentation
2.3. Experimental
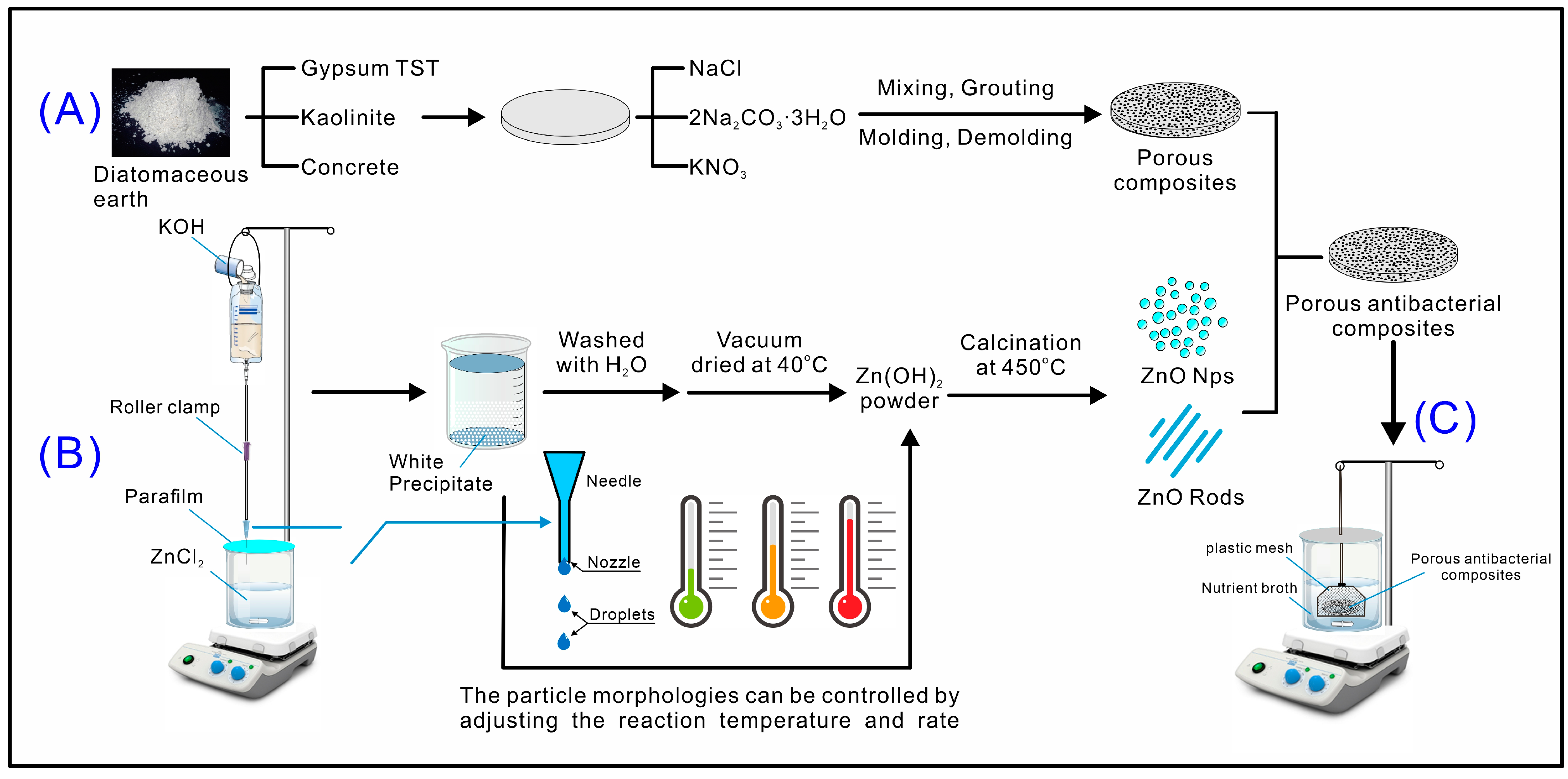
2.3.1. Diatomaceous Earth Mold
2.3.2. Direct Precipitation Synthesis of ZnO Powder
2.4. Surface Area Measurement and BET Particle Size Calculation of ZnO Powder
2.5. Antibacterial Effect of the Porous DE/Nano-ZnO Composites
3. Results and Discussion
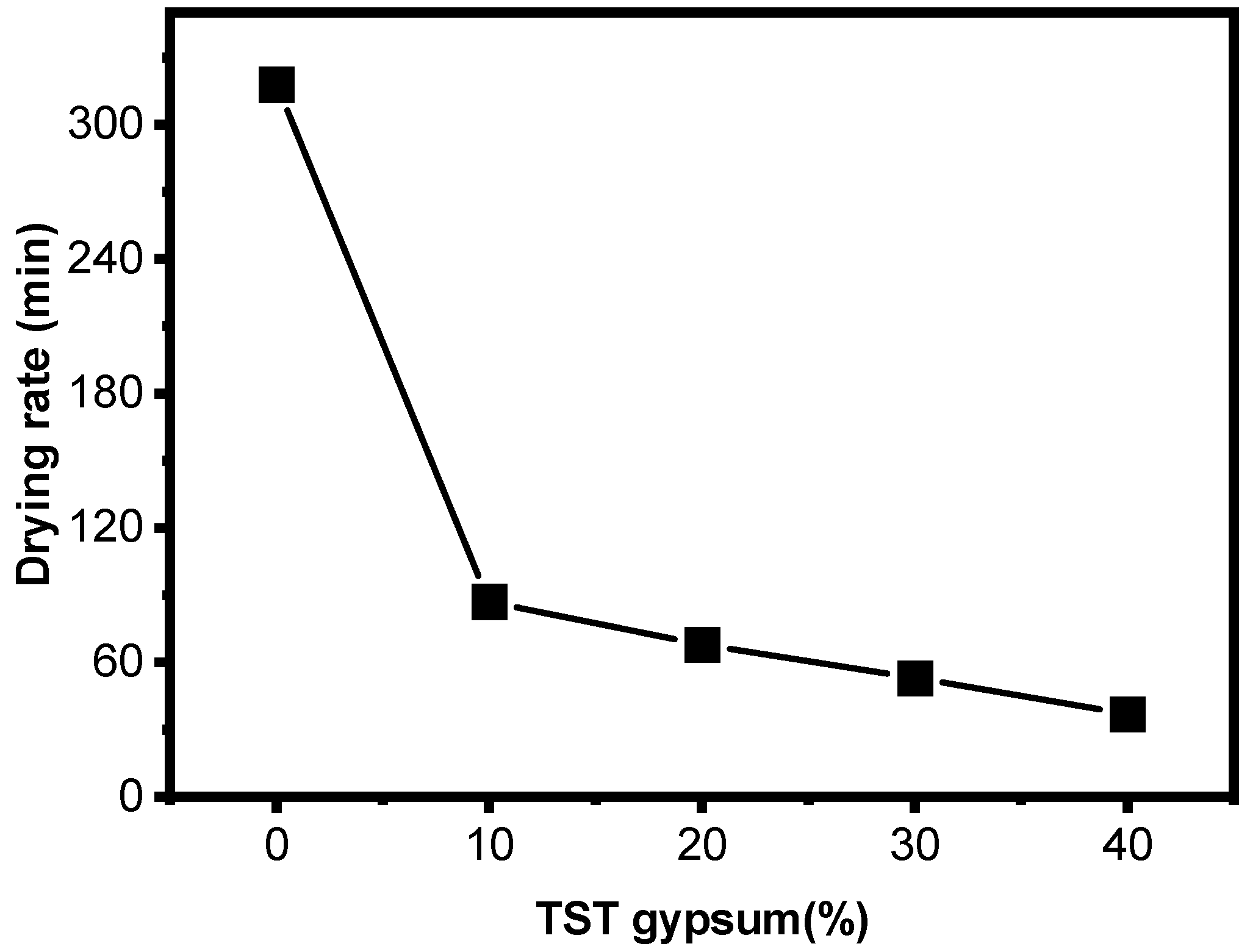
3.1. Comparison of Adding Inorganic Materials to Diatomaceous Earth for Mold Making
3.2. Comparison of Porosity of Diatomaceous Earth Composites
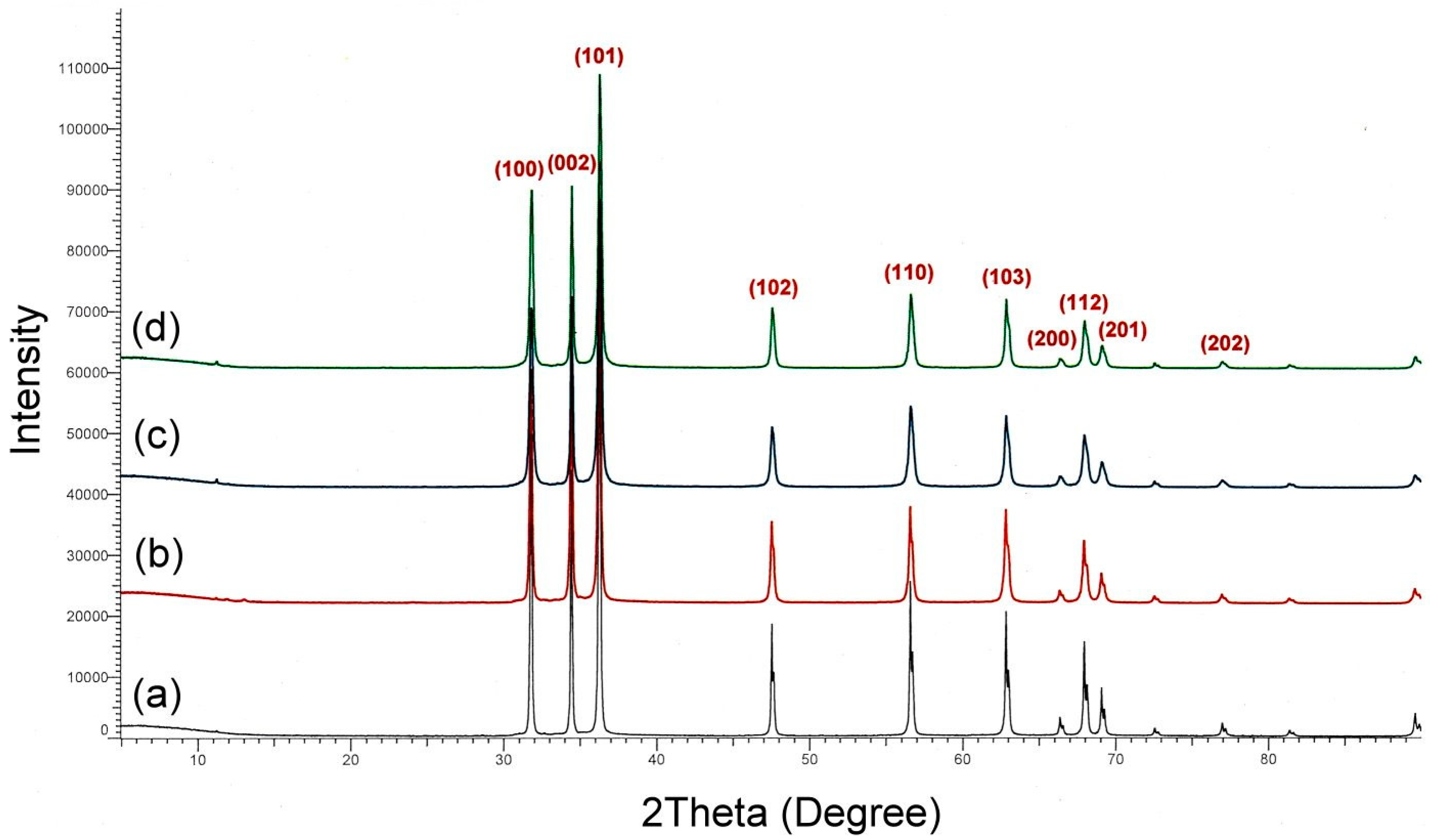
3.3. Preparation of High-Surface Area ZnO Powder
3.4. Comparison of Antibacterial Properties of Porous Diatomaceous Earth/ZnO Composites
3.4.1. Comparison of Antibacterial Properties with Different Amounts of ZnO
3.4.2. Fluorescence Analysis of the Antimicrobial Experiment on DE Composites with Added ZnO
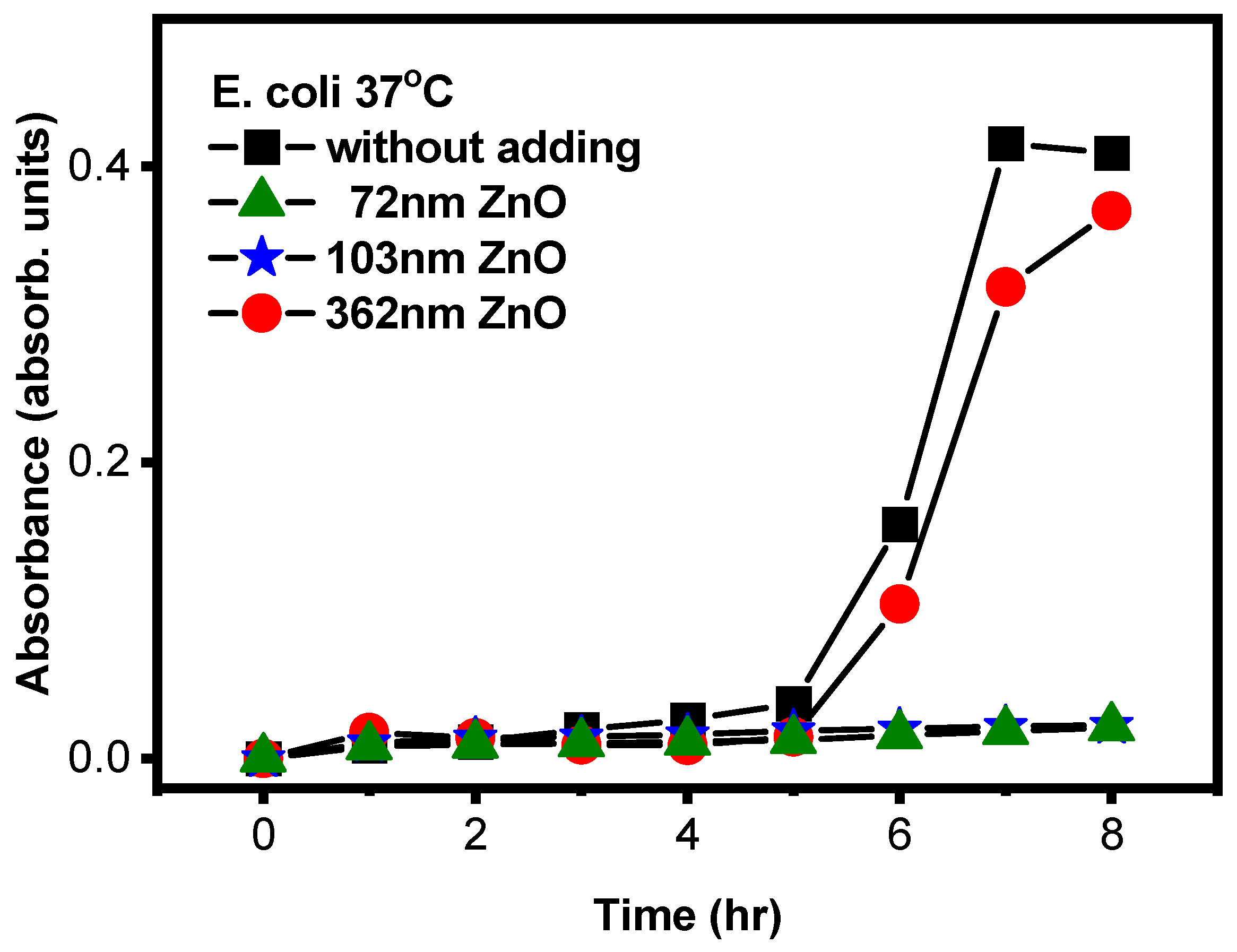
3.4.3. Comparison of Antibacterial Properties of ZnO with Different Particle Sizes
3.5. Practical Application of Porous Diatomaceous Earth/Nano-ZnO Composites
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mete, Z. Benefication of Kütahya-Alayunt diatomite ores. Akdeniz Univ. Eng. Fac. J. 1988, 1, 184–201. [Google Scholar]
- Hadjar, H.; Hamdi, B.; Jaber, M.; Brendle, J.; Kessaissia, Z.; Balard, H.; Donnet, J.B. Elaboration and characterisation of new mesoporous materials from diatomite and charcoal. Microporous Mesoporous Mater. 2008, 107, 219–226. [Google Scholar] [CrossRef]
- Zheng, R.; Gao, H.; Guan, J.; Ren, Z.; Tian, J. Characteristics of cationic red X-GRL adsorption by diatomite tailings. J. Wuhan Univ. Technol. 2017, 32, 1038–1047. [Google Scholar] [CrossRef]
- Sun, Z.; Yang, X.; Zhang, G.; Zheng, S.; Frost, R.L. A novel method for purification of low grade diatomite powders in centrifugal fields. Int. J. Miner. Process. 2013, 125, 18–26. [Google Scholar] [CrossRef]
- Zheng, R.; Ren, Z.; Gao, H.; Zhang, A.; Bian, Z. Effects of calcination on silica phase transition in diatomite. J. Alloys Compd. 2018, 757, 364–371. [Google Scholar] [CrossRef]
- Yılmaz, B.; Ediz, N. The use of raw and calcined diatomite in cement production. Cem. Concr. Compos. 2008, 30, 202–211. [Google Scholar] [CrossRef]
- Al-Ghouti, M.A.; Khraisheh, M.A.M.; Allen, S.J.; Ahmad, M.N. The removal of dyes from textile wastewater: A study of the physical characteristics and adsorption mechanisms of diatomaceous earth. J. Environ. Manag. 2003, 69, 229–238. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, J.; Yang, W.; Wu, J.; Chen, R. Adsorption properties for urokinase on local diatomite surface. Appl. Surf. Sci. 2003, 206, 20–28. [Google Scholar] [CrossRef]
- Martinovic, S.; Vlahovic, M.; Boljanac, T.; Pavlovic, L. Preparation of filter aids based on diatomites. Int. J. Miner. Process. 2006, 80, 255–260. [Google Scholar] [CrossRef]
- Tsai, W.-T.; Lai, C.-W.; Kuo, J. Characterization and adsorption properties of diatomaceous earth modified by hydrofluoric acid etching. J. Colloid Interface Sci. 2006, 297, 749–754. [Google Scholar] [CrossRef]
- Yusan, S.; Gok, C.; Erenturk, S.; Aytas, S. Adsorptive removal of thorium (IV) using calcined and flux calcined diatomite from Turkey: Evaluation of equilibrium, kinetic and thermodynamic data. Appl. Clay Sci. 2012, 67–68, 106–116. [Google Scholar] [CrossRef]
- Karaman, S.; Karaipekli, A.; Sarı, A.; Biçer, A. Polyethylene glycol (PEG)/diatomite composite as a novel shape-stabilized phase change material for thermal energy storage. Sol. Energy Mater. Sol. C 2011, 95, 1647–1653. [Google Scholar] [CrossRef]
- Xu, B.; Li, Z. Paraffin/diatomite composite phase change material incorporated cement-based composite for thermal energy storage. Appl. Energy 2013, 105, 229–237. [Google Scholar] [CrossRef]
- Li, X.Y.; Sanjayan, J.G.; Wilson, J.L. Fabrication and stability of form-stable diatomite/paraffin phase change material composites. Energy Build. 2014, 76, 284–294. [Google Scholar] [CrossRef]
- Sarı, A.; Bicer, A. Thermal energy storage properties and thermal reliability of some fatty acid esters/building material composites as novel form-stable PCMs. Sol. Energy Mater. Sol. C 2012, 101, 114–122. [Google Scholar] [CrossRef]
- Nomura, T.; Okinaka, N.; Akiyama, T. Impregnation of porous material with phase change material for thermal energy storage. Mater. Chem. Phys. 2009, 115, 846–850. [Google Scholar] [CrossRef]
- Qian, T.; Li, J.; Min, X.; Deng, Y.; Guan, W.; Ning, L. Diatomite: A Promising Natural Candidate as Carrier Material for Low, Middle and High Temperature Phase Change Material. Energy Convers. Manag. 2015, 98, 34–45. [Google Scholar] [CrossRef]
- Moezzi, A.; McDonagh, A.M.; Cortie, M.B. Zinc oxide particles: Synthesis, properties and applications. Chem. Eng. J. 2012, 185–186, 1–22. [Google Scholar] [CrossRef]
- Chauhan, R.; Kumar, A.; Tripathi, R.; Kumar, A. Advancing of Zinc Oxide Nanoparticles for Cosmetic Applications. In Handbook of Consumer Nanoproducts; Bhushan, B., Ed.; Springer: Cham, Switzerland, 2022; pp. 1057–1072. [Google Scholar]
- Kim, K.-B.; Kim, Y.W.; Lim, S.K.; Roh, T.H.; Bang, D.Y.; Choi, S.M.; Lim, D.S.; Kim, Y.J.; Baek, S.-H.; Kim, M.-K.; et al. Risk assessment of zinc oxide, a cosmetic ingredient used as a UV filter of sunscreens. J. Toxicol. Environ. Health Part B 2017, 20, 155–182. [Google Scholar] [CrossRef]
- Taghizadeh, S.-M.; Lal, N.; Ebrahiminezhad, A.; Moeini, F.; Seifan, M.; Ghasemi, Y.; Berenjian, A. Green and Economic Fabrication of Zinc Oxide (ZnO) Nanorods as a Broadband UV Blocker and Antimicrobial Agent. Nanomaterials 2020, 10, 530. [Google Scholar] [CrossRef]
- Zhao, X.; Xin, Z.; He, Q.; Wang, S.; Han, X.; Fu, Z.; Xu, X. Recent Progress in ZnO-Based Nanostructures for Photocatalytic Antimicrobial in Water Treatment: A Review. Appl. Sci. 2022, 12, 7910. [Google Scholar]
- Abdul Mutalib, A.A.; Jaafar, N.F. ZnO photocatalysts applications in abating the organic pollutant contamination: A mini review. Total Environ. Res. Themes 2022, 3–4, 100013. [Google Scholar] [CrossRef]
- Sun, T.; Qiu, J.; Liang, C. Controllable fabrication and photocatalytic activity of ZnO nanobelt arrays. J. Phys. Chem. C 2008, 112, 715–721. [Google Scholar] [CrossRef]
- Thauer, E.; Zakharova, G.S.; Andreikov, E.I.; Adam, V.; Wegener, S.A.; Nölke, J.-H.; Singer, L.; Ottmann, A.; Asyuda, A.; Zharnikov, M.; et al. Novel synthesis and electrochemical investigations of ZnO/C composites for lithium-ion batteries. J. Mater. Sci. 2021, 56, 13227–13242. [Google Scholar] [CrossRef]
- Chen, H.-C.; Lyu, Y.-R.; Fang, A.; Lee, G.-J.; Karuppasamy, L.; Wu, J.J.; Lin, C.-K.; Anandan, S.; Chen, C.-Y. The Design of ZnO Nanorod Arrays Coated with MnOx for High Electrochemical Stability of a Pseudocapacitor Electrode. Nanomaterials 2020, 10, 475. [Google Scholar] [CrossRef] [PubMed]
- Schlur, L.; Carton, A.; Lévêque, P.; Guillon, D.; Pourroy, G. Optimization of a new ZnO nanorods hydrothermal synthesis method for solid state dye sensitized solar cells applications. J. Phys. Chem. C 2013, 117, 2993–3001. [Google Scholar] [CrossRef]
- Son, D.Y.; Im, J.H.; Kim, H.S.; Park, N.G. 11% Efficient perovskite solar cell based on ZnO nanorods: An effective charge collection system. J. Phys. Chem. C 2014, 118, 16567–16573. [Google Scholar] [CrossRef]
- Wang, J.X.; Sul, X.W.; Yang, Y.; Huang, H.; Lee, Y.C.; Tan, O.K.; Vayssieres, L. Hydrothermally grown oriented ZnO nanorod arrays for gas sensing applications. Nanotechnology 2006, 17, 4995–4998. [Google Scholar] [CrossRef]
- Wang, H.T.; Kang, B.S.; Ren, F.; Tien, L.C.; Sadik, P.W.; Norton, D.P.; Pearton, S.; Lin, J. Hydrogen-selective sensing at room temperature with ZnO nanorod. Appl. Phys. Lett. 2005, 86, 243503. [Google Scholar] [CrossRef]
- Kilinc, N.; Cakmak, O.; Kosemen, A.; Ermek, E.; Ozturk, S.; Yerli, Y.; Ozturk, Z.Z.; Urey, H. Fabrication of 1D ZnO nanostructures on MEMS cantilever for VOC sensor application. Sens. Actuators B Chem. 2014, 202, 357–364. [Google Scholar] [CrossRef]
- Murali, M.; Kalegowda, N.; Gowtham, H.G.; Ansari, M.A.; Alomary, M.N.; Alghamdi, S.; Shilpa, N.; Singh, S.B.; Thriveni, M.C.; Aiyaz, M.; et al. Plant-Mediated Zinc Oxide Nanoparticles: Advances in the New Millennium towards Understanding Their Therapeutic Role in Biomedical Applications. Pharmaceutics 2021, 13, 1662. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; He, L.; Mustapha, A.; Li, H.; Hu, Z.Q.; Lin, M. Antibacterial activities of zinc oxide nanoparticles against Escherichia coli O157:H7. J. Appl. Microbiol. 2009, 107, 1193–1201. [Google Scholar] [CrossRef] [PubMed]
- Babayevska, N.; Przysiecka, Ł.; Iatsunskyi, I.; Nowaczyk, G.; Jarek, M.; Janiszewska, E.; Jurga, S. ZnO size and shape effect on antibacterial activity and cytotoxicity profile. Sci. Rep. 2022, 12, 8148. [Google Scholar] [CrossRef] [PubMed]
- Sirelkhatim, A.; Mahmud, S.; Seeni, A.; Kaus, N.H.M.; Ann, L.C.; Bakhori, S.K.M.; Hasan, H.; Mohamad, D. Review on Zinc Oxide Nanoparticles: Antibacterial Activity and Toxicity Mechanism. Nano-Micro Lett. 2015, 7, 219–242. [Google Scholar] [CrossRef]
- Moradpoor, H.; Safaei, M.; Mozaffari, H.R.; Sharifi, R.; Imani, M.M.; Golshahe, A.; Bashardoust, N. An overview of recent progress in dental applications of zinc oxide nanoparticles. RSC Adv. 2021, 11, 21189. [Google Scholar] [CrossRef]
- Dayakar, T.; Venkateswara Rao, K.; Bikshalu, K.; Rajendar, V.; Park, S.-H. Novel synthesis and structural analysis of zinc oxide nanoparticles for the non enzymatic glucose biosensor. Mater. Sci. Eng. C 2017, 75, 1472–1479. [Google Scholar] [CrossRef]
- Elumalai, K.; Velmurugan, S. Green synthesis, characterization and antimicrobial activities of Zinc Oxide nanoparticles from the leaf extract of Azadirachta indica (L.). Appl. Surf. Sci. 2015, 345, 329–336. [Google Scholar] [CrossRef]
- Dobrucka, R.; Długaszewska, J. Biosynthesis and antibacterial activity of Zno nanoparticles using Trifolium Pratense flower extract. J. Nanosci. Nanotechnol. 2016, 23, 517–523. [Google Scholar] [CrossRef]
- Li, M.; Zhu, L.; Lin, D. Toxicity of ZnO Nanoparticles to Escherichia coli: Mechanism and the Influence of Medium Components. Environ. Sci. Technol. 2011, 45, 1977–1983. [Google Scholar] [CrossRef]
- Liu, H.; Qiao, Z.; Jang, Y.O.; Kim, M.G.; Zou, Q.; Lee, H.J.; Koo, B.; Kim, S.-H.; Yun, K.; Kim, H.-S.; et al. Diatomaceous earth/zinc oxide micro-composite assisted antibiotics in fungal Therapy. Nano Converg. 2021, 8, 32. [Google Scholar] [CrossRef]
- Kairyte, K.; Kadys, A.; Luksiene, Z. Antibacterial and antifungal activity of photoactivated ZnO nanoparticles in suspension. J. Photochem. Photobiol. B 2013, 128, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Studart, A.R.; Gonzenbach, U.T.; Tervoort, E.; Gauckler, L.J. Processing Routes to Macroporous Ceramics: A Review. J. Am. Ceram. Soc. 2006, 89, 1771–1789. [Google Scholar] [CrossRef]
- Kaliaguine, S. Porous Materials: Processing and Applications; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Li, Y.; Chen, C.; Wang, X. Porous Materials: Design, Synthesis, and Applications. Sci. China Chem. 2017, 60, 1143–1153. [Google Scholar]
- Deville, S. Freeze-Casting of Porous Materials: Review of Current Achievements and Issues. Adv. Eng. Mater. 2008, 10, 155–169. [Google Scholar] [CrossRef]
- Cao, G.Z. Nanostructures and Nanomaterials: Synthesis, Properties, and Applications; World Scientific: Singapore, 2004. [Google Scholar]
- Tas, A.C. Synthesis of Biomimetic Ca-Hydroxyapatite Powders at 37 °C in Synthetic Body Fluids. Biomaterials 2000, 21, 1429–1438. [Google Scholar]
- Sing, K.S. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Jones, N.; Ray, B.; Ranjit, K.T.; Manna, A.C. Antibacterial activity of ZnO nanoparticle suspensions on a broad spectrum of microorganisms. FEMS Microbiol. Lett. 2008, 279, 71–76. [Google Scholar] [CrossRef]
- Romadhan, M.F.; Suyatma, N.E.; Taqi, F.M. Synthesis of ZnO Nanoparticles by Precipitation Method with Their Antibacterial Effect. Indones. J. Chem. 2016, 16, 117–123. [Google Scholar] [CrossRef]
- Mahmood, N.B.; Saeed, F.R.; Gbashi, K.R.; Mahmood, U.S. Synthesis and characterization of zinc oxide nanoparticles via oxalate co-precipitation method. Mater. Lett. X 2022, 13, 100126. [Google Scholar] [CrossRef]
- Purwaningsih, S.Y.; Pratapa, S.; Triwikantoro; Darminto. Nano-sized ZnO Powders Prepared by Co-precipitation Method with Various pH. AIP Conf. Proc. 2016, 1725, 020063. [Google Scholar]
- Hwang, J.-J.; Wu, C.-Y.; Hung, Y.-H.; Li, M.-X.; Luo, K.-H.; Jia, H.-W.; Balitaan, J.N.I.; Lin, S.-R.; Yeh, J.-M. Biomimetic PMMA Coating Surface and its Application on Inhibition of Bacterial Attachment and Anti-biofilm Performance. Surf. Interfaces 2023, 36, 102548. [Google Scholar] [CrossRef]







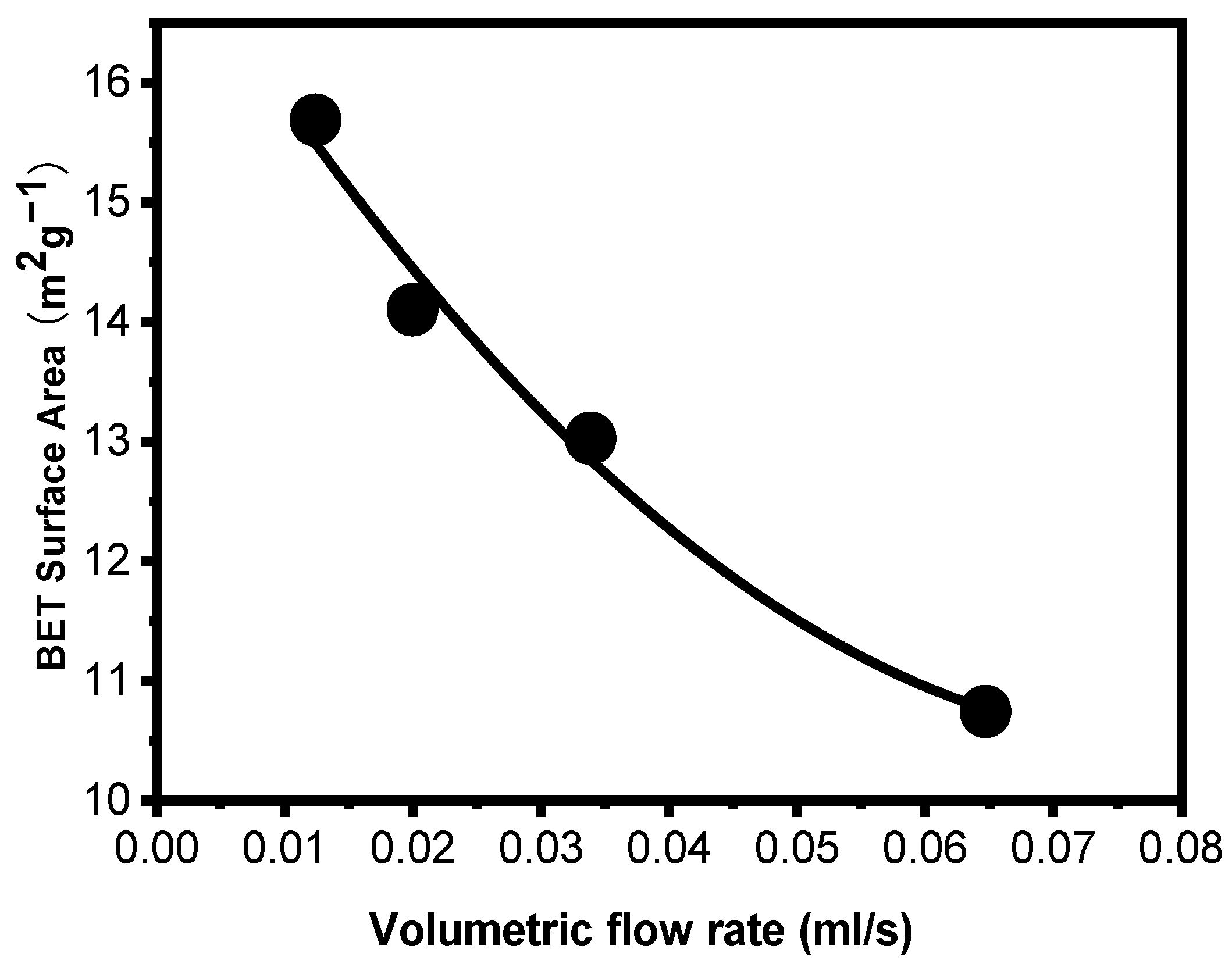


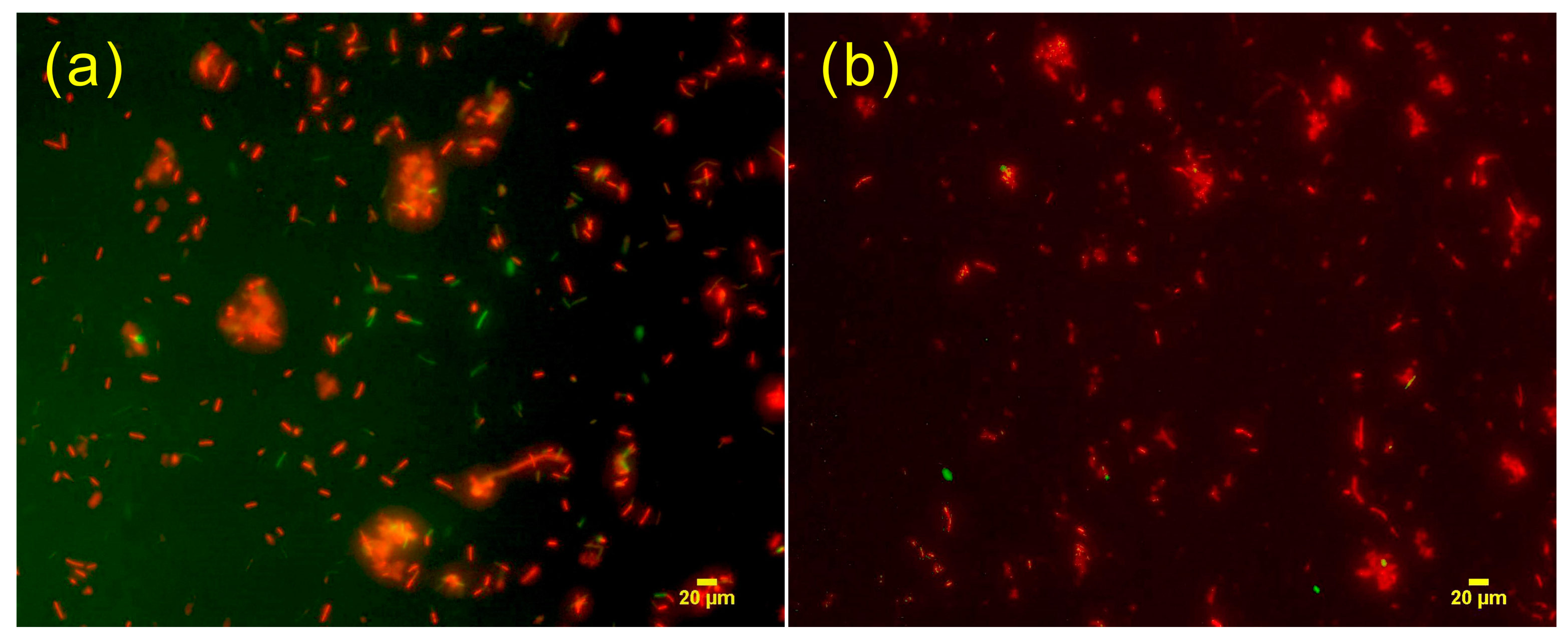

| Drop rate (drop/s) | 0.5 | 1 | 2 | 3 |
| Weight (g) | 7.2449 | 7.2809 | 7.3123 | 7.1168 |
| Yield (%) | 89.03 | 89.47 | 89.85 | 87.45 |
| BET surface area (m2/g) | 2.2691 | 2.4754 | 1.9543 | 2.1196 |
| dBET (nm) | 472 | 432 | 548 | 505 |
| Temperature (℃) | 25 | 40 | 60 | 80 |
| Yield (%) | 89.85 | 94.84 | 89.38 | 92.01 |
| BET surface area (m2/g) | 2.9568 | 10.3577 | 14.8765 | 11.3191 |
| Pore volume (cm3/g) | 0.001457 | 0.005221 | 0.007403 | 0.005644 |
| dBET (nm) | 362 | 103 | 72 | 143 |
| ZnO (%) | 0 | 1 | 5 | 10 |
| Absorption (OD600nm) | 0.8323 | 0.6674 | 0.2860 | 0.1191 |
| Antibacterial rate (%) | − | 19.81 | 65.64 | 85.69 |
| ZnO dBET (nm) | 0 | 72 | 103 | 362 |
| Absorption (OD600nm) | 0.4091 | 0.0209 | 0.0224 | 0.3701 |
| Antibacterial rate (%) | - | 94.89 | 94.52 | 9.53 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chung, C.-C.; Hwang, J.-J. Porous Diatomaceous Earth/Nano-Zinc Oxide Composites: Preparation and Antimicrobial Applications. J. Compos. Sci. 2023, 7, 204. https://doi.org/10.3390/jcs7050204
Chung C-C, Hwang J-J. Porous Diatomaceous Earth/Nano-Zinc Oxide Composites: Preparation and Antimicrobial Applications. Journal of Composites Science. 2023; 7(5):204. https://doi.org/10.3390/jcs7050204
Chicago/Turabian StyleChung, Chin-Chun, and Jiunn-Jer Hwang. 2023. "Porous Diatomaceous Earth/Nano-Zinc Oxide Composites: Preparation and Antimicrobial Applications" Journal of Composites Science 7, no. 5: 204. https://doi.org/10.3390/jcs7050204
APA StyleChung, C.-C., & Hwang, J.-J. (2023). Porous Diatomaceous Earth/Nano-Zinc Oxide Composites: Preparation and Antimicrobial Applications. Journal of Composites Science, 7(5), 204. https://doi.org/10.3390/jcs7050204






