2.1. Experimental Setup
The experiment was conducted at Cornell University’s Kenneth Post Laboratory greenhouse complex in Ithaca, New York at a latitude of approximately 42° N and a longitude of 76° W. Trial 1 occurred between 24 February and 21 March 2016, and Trial 2 occurred between 8 March and 4 April 2016. All environmental parameters were monitored by an Argus system (Argus Control Systems Ltd., Surrey, BC, Canada), in which data samples were collected every two seconds, averaged every 2 min, and logged. The experiment was conducted in a middle section of a multi-sectioned glass greenhouse with dimensions 9 m × 10 m × 7 m high to the ridge, oriented east-west. The greenhouse was kept at a constant temperature of 24 °C, maintained by two identical forced air water-to-air heat exchangers on opposite ends of the greenhouse, rated at 115,000 kJ/h.
A daily light integral (DLI) was maintained at 17 mol/m2·day of photosynthetically active radiation (PAR) by a combination of natural and supplemental light. Day length increased over the course of the experiment’s post-seedling stage from 11:43 h to 12:55 h. Since we maintained the same DLI over the entire experiment, we assumed there was no effect from the day length change. Supplemental light was provided by an array of 20 high-pressure sodium (HPS) lights (General Electric, 400 watt clear S51/O, Mogul Base rated ED18 HSP, LU 400/H/ECO). Lights were arrayed to ensure that each plant received the same amount of light. A LiCor quantum sensor (LI-190R, LI-COR Biosciences, Lincoln, NE, USA) was used to quantify lighting at the plant level. CO2 was not controlled or measured, but was assumed to be ambient. Humidity was not controlled but monitored and left at ambient conditions, typically between 50% and 60% during the daytime period. Root zone temperature was not controlled, but was within 1.9 °C of the average room temperature of 24 °C. Each trial took place over 27 days with the last 13 of these days in the bucket environment. All plants were grown in buckets, described in more detail below.
All seeds for all trials were germinated in an ebb and flow seeding bench and then were transplanted after 14 days to the bucket system for days 15 to 27. Seedlings at day 14 were randomly selected from the available seedlings after discarding atypical (including large or small) plants.
2.2. Treatments
Two trials of 10 replicates of four treatments were performed. The four treatments are shown in
Table 1. Each treatment will be referenced by their respective acronym throughout the paper. The H5 treatment of conventional hydroponic at pH 5.8 was considered the control treatment, and was used as a basis for comparisons with other treatments.
Nutrient concentrations used for the four treatments at the beginning of the trial and then at the replacement of solution every two days are given in
Table 2. H5 was a standard Sonneveld and Straver nutrient solution for lettuce [
8], modified to half concentration, adjusted to pH 5.8, and without silicon. H7 was the same nutrient solution as H5, adjusted to pH 7.0. Two hundred liters of H5 and H7 solution were prepared at once using reverse osmosis (RO) water and concentrated stock solutions. The modified solutions were created following details provided in our previous publications [
7,
9]. The starting nutrient conditions for our four treatments were assumed to be the same as H5, H7, and A7 reported by Vandam et al. [
10], since they used the same fish system, nutrient solution stocks, and protocols, and were analyzed within one month of when we started this experiment. A7 and SA7 nutrient elemental concentrations would be similar, since the same water from the fish system was used for both. We did not investigate whether the autoclaving process had any impact on the compounds present due to heat transformation.
Chelated iron (Fe-DTPA, brand name Sprint 330, BASF Corporation, Research Triangle Park, Durham, NC, USA) was included for all treatments at 11 mg/L (10% of the material is active as elemental iron or 1.1 mg/L). A7 received the Fe-DTPA immediately after its removal from the main aquaponic system, while SA7 received Fe-DTPA after sterilization. The Fe values in
Table 2 are for total Fe present, which would be exclusively from the chelated additive for H5 and H7, while the higher Fe value for A7 included the Fe in the chelated material, as well as Fe from other compounds in the fish water and residual non-chelated Fe from previous experiments. The chelated form of Fe from the fish system water was assumed to be minimal, since the chelated product breaks down fairly rapidly to other Fe compounds due to UV radiation from sunlight, as was the case for our experimental setup. Humic and fulvic acids can act as natural chelators and are the byproduct of decomposition [
11], and thus were likely present in our system. Selectivity for elements and the concentration of these natural chelators was not evaluated in this experiment.
Nutrient solutions in each bucket were changed every two days to restore nutrient conditions to their initial known values. Given the frequent change to known conditions and that nutrient solutions are designed to provide surplus nutrients, we considered our data collection every two days to be sufficient to characterize the nutrient conditions that the plants were grown under. Lettuce plants were harvested after 27 days of growth to ensure that elements would not be depleted between solution changes, e.g., our plants at 27 days were only 15% to 20% of a commercial harvest size of 150 g. From a mass balance perspective, we calculated that macro elements in solution declined by only 2% to 4% from their initial values.
The H5 and H7 solutions were adjusted daily to the targeted pH five days in advance of plant placement using KOH for base and HNO
3 for acid corrections. While H5 did not require any corrections after the first day, H7 experienced precipitation from the elevated pH that required daily adjustments for a period (discussed in our previous papers [
7,
9]). The five-day adjustment period for the nutrient solutions was to ensure that the majority of any pH-related precipitation occurred before we used the nutrient solution for the plants and that the pH in the stock container remained relatively constant once the experiment was initiated.
A7 used unsterilized water from a 1400-L recirculating aquaculture system growing approximately 52 adult koi (
Cyprinus carpio) being fed 90 g per day of a commercial koi pelleted feed. Koi feed (Max Growth Diet floating extruded pellet) was purchased from Blackwater Creek Farms (Eustis, FL, USA,
www.koisale.com). All food was used within six months of the mill date to ensure nutritional quality; details of the fish system and feeding protocols are given in our earlier papers [
7,
9]). SA7 solution was obtained from the same system as A7 and then autoclaved following an established liquid autoclaving protocol [
12]. Our sterilization protocol heated the nutrient solutions to approximately 120 °C and 1.36 atmospheres of gage pressure. Autoclave tape was applied to ensure that the liquid reached the necessary sterilization temperature. After autoclaving, the nutrient solutions were allowed to cool to room temperature before use.
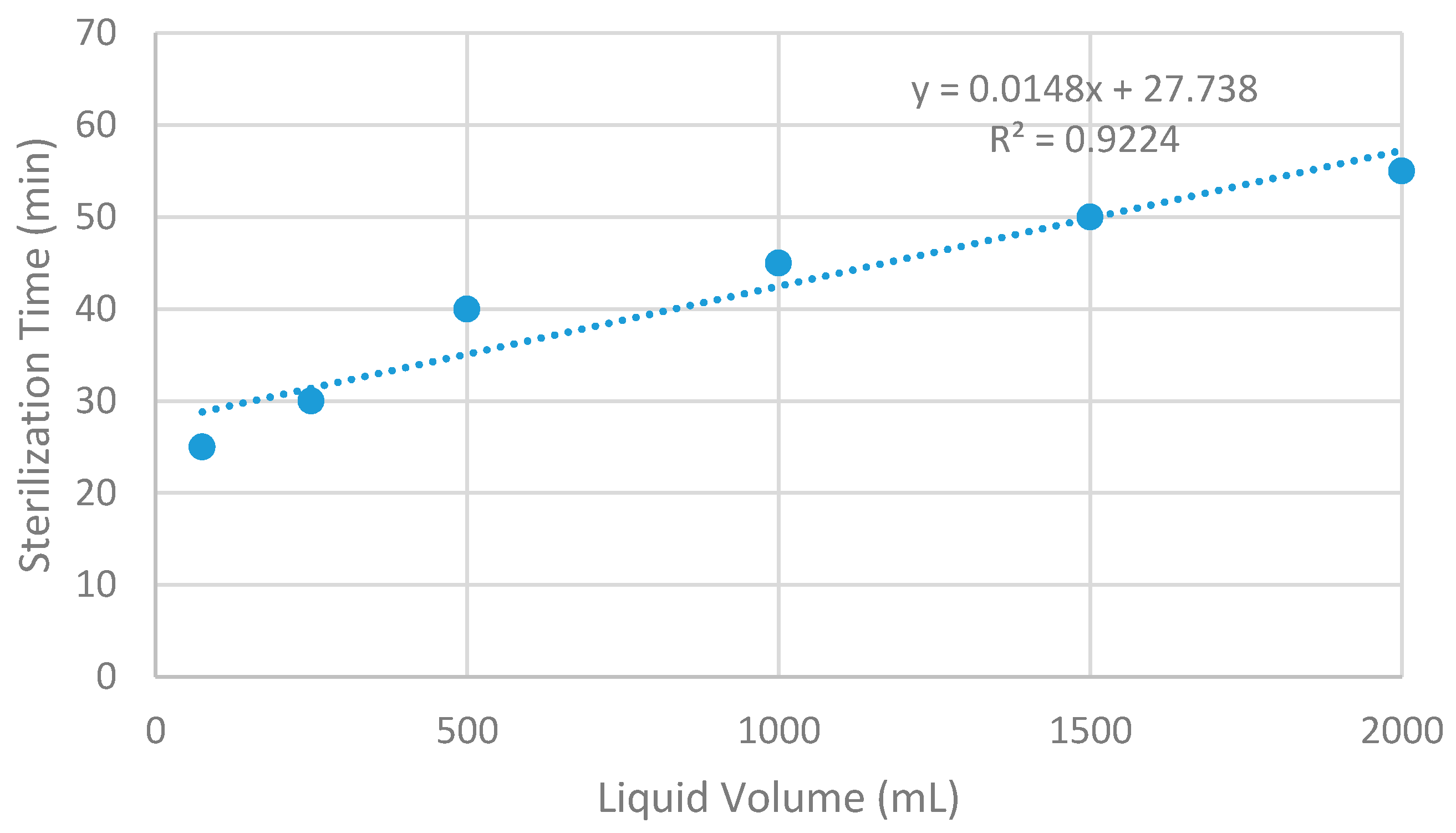
Based upon the data from the referenced manual [
12], the regression shown in
Figure 1 was developed and used to prescribe the minimum autoclaving time-period for sterilization; we rounded this time up to 90 min to be confident of solution sterilization. To verify that autoclave sterilization was effective in reducing microorganism populations, an adenosine triphosphate (ATP)-based monitoring system (novaLUM System manufactured by Charm Sciences, Lawrence, MA, USA) was utilized to determine the relative living cells in the solution via Relative Light Units (RLU). RLUs are a comparative measure of ATP present in a solution after cell lysing. A high RLU indicates that the solution contains a large amount of ATP, meaning it contains a large amount of living cells [
13]. Verification data is reported in
Section 3.
2.3. Experimental Protocol
The bucket system consisted of 40 plastic buckets, each filled near to the brim with 4 L of one of the four treatments (
Figure 2). Treatments and buckets were placed randomly within the growing area. Each bucket contained an air stone placed in the nutrient solution that constantly aerated the nutrient solution from common air pumps. Each bucket was covered with a 200-mm diameter lid containing three 25-mm diameter holes drilled equidistant from each other. Buckets and lids were painted silver to reduce chelation degradation and limit algal growth by preventing sunlight from reaching the nutrient solution.
Flats of rockwool (Grodan AO25/40, 25 mm; Grodan, Roermond, The Netherlands) were prepared by soaking them in RO water for 10–20 min and then flushing each flat with RO water for 5 min. Flats were then soaked for an additional 10–20 min using the ½ Sonneveld and Straver solution adjusted to a pH of 4.5 to 5.0, following which flats were flushed with 6 to 7 L of nutrient solution that had been adjusted to a pH of 5.8 (control pH for H5). We then did a final flushing using RO water for 7 min and considered the flats ready for seed placement.
Un-pelleted lettuce seeds (Lactuca sativa cv. Flandria) purchased from Rijk-Zwaan (De Lier, The Netherlands) were planted in the rockwool cubes 14 days prior to the desired transplant date and placed in the mentioned greenhouse space within the DLI maintained light array. The day of seeding is the reference date, day 1. Standard perforated 1020 trays of seeds in rockwool were covered with an opaque cover for the first 24 h, after which the cover was replaced with a clear germination cover. This cover was removed after the fourth day, leaving the seedlings exposed. Seedlings were placed in an ebb and flood bench that supplied nutrient solution at pH 5.8 four times a day (7 a.m., 11 a.m., 3 p.m., and 7 p.m.). Individual cubes were separated from the pre-cut rockwool slab and seedlings that were considered typical (not large, smaller, or abnormal) were then transplanted to the bucket system on Day 14. One cube was placed in each of the three lid holes. Roots were submerged in nutrient solution when the cubes were placed in the lids. New buckets were used in Trial 1 (cleaned and disinfected before use) and then cleaned thoroughly before reuse in Trial 2.
Once placed into the buckets, the nutrient solutions were emptied every two days (days 16, 18, 20, 22, 24, and 26) and replaced with fresh nutrient solution for each treatment. Plants were removed at day 27, compared to normal harvests occurring at day 35 (150 g plants). We harvested at the earlier date to minimize the changes occurring in natural day length and before the late stage exponential growth of the lettuce plant. Also, our experience was that we would see any effects of nutrient deficiencies or environmental variables well before plants reach a typical market size at day 35.
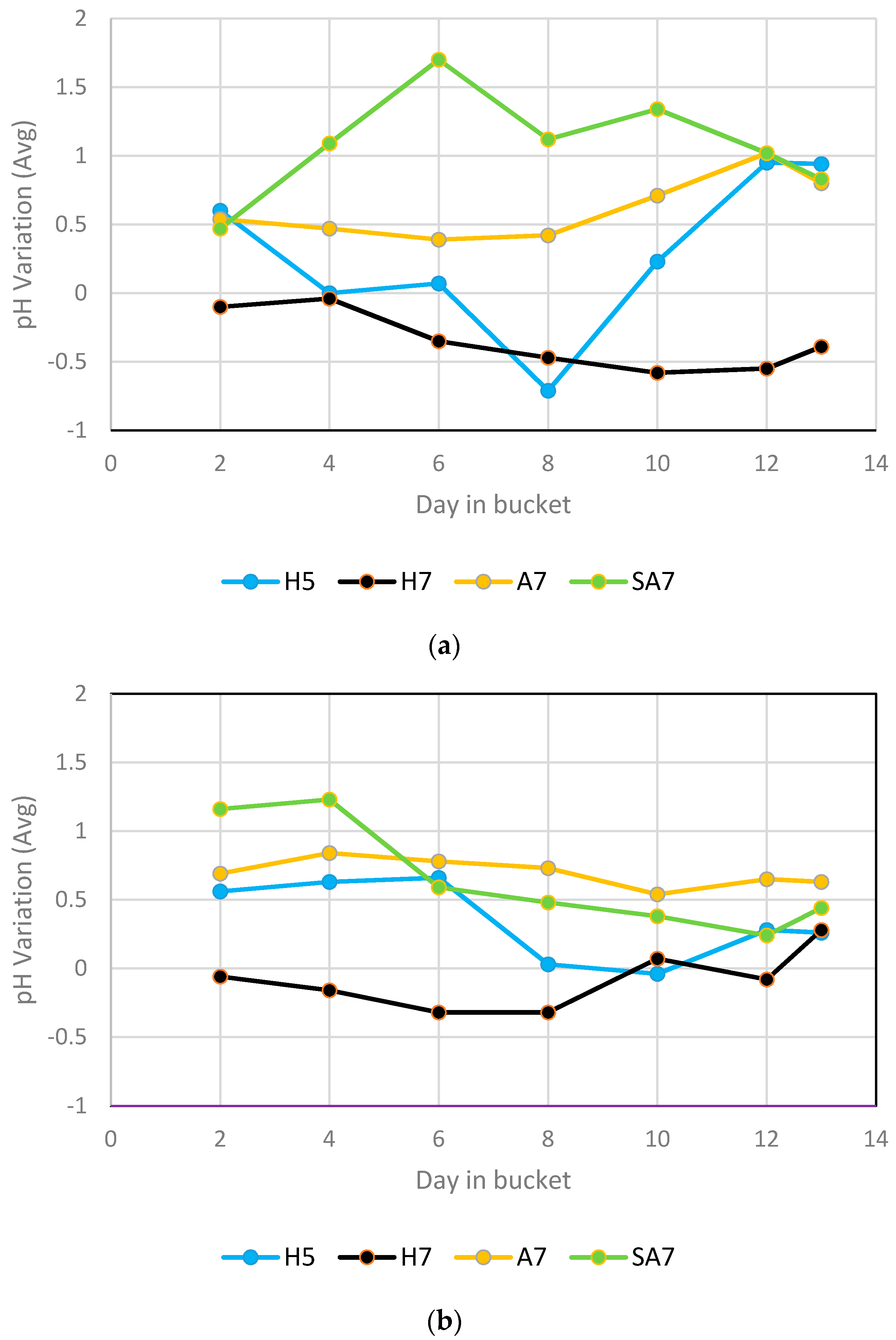
Frequent replacement of nutrient solution, in addition to the factors mentioned in the treatments in
Section 2.2, were intended to maintain pH and nutrient availability at near target values. The pH variations of each solution immediately before replacing the nutrient solution with fresh replacement at the target pH value are shown in
Figure 3.
2.4. Measurements
The pH, electrical conductivity (EC), and root zone temperature were measured prior to the addition of new nutrient solution to each bucket. All treatment pH values were adjusted to their target values (5.80 ± 0.02 for H5, 7.00 ± 0.02 for H7, A7, and SA7) before the addition of nutrient solutions to individual buckets. Old nutrient solution from each treatment was collected into a barrel and mixed thoroughly. After mixing, the pH, EC, and root zone temperature were measured and recorded for each of the treatments. Change in nutrient concentrations was not measured and was assumed to be minimal due to the small head size at harvest, circa 16% of a commercial head target size of 150 g.
Nutrient solution data were recorded for the final time on day 27. Shoots were cut level with the rockwool cube and individual fresh weights were measured and recorded. Each shoot was individually bagged and placed in a 70 °C drying oven for approximately 72 h. After this time-period, each shoot’s dry weight was measured and recorded, from which the dry weight (DW) to fresh weight (FW) ratio (DW/FW) or dry matter percentage was determined.