Yeast Flocculation—Sedimentation and Flotation †
Abstract
:1. Introduction
- The yeast strains employed and their condition during pitching and throughout the wort fermentation cycle [2];
- the concentration and category of assimilable nitrogen [3];
- the spectrum of wort sugars [4];
- fermentation temperature [2];
- yeast pitching (inoculation) rate [1];
- concentration of inorganic ions [5];
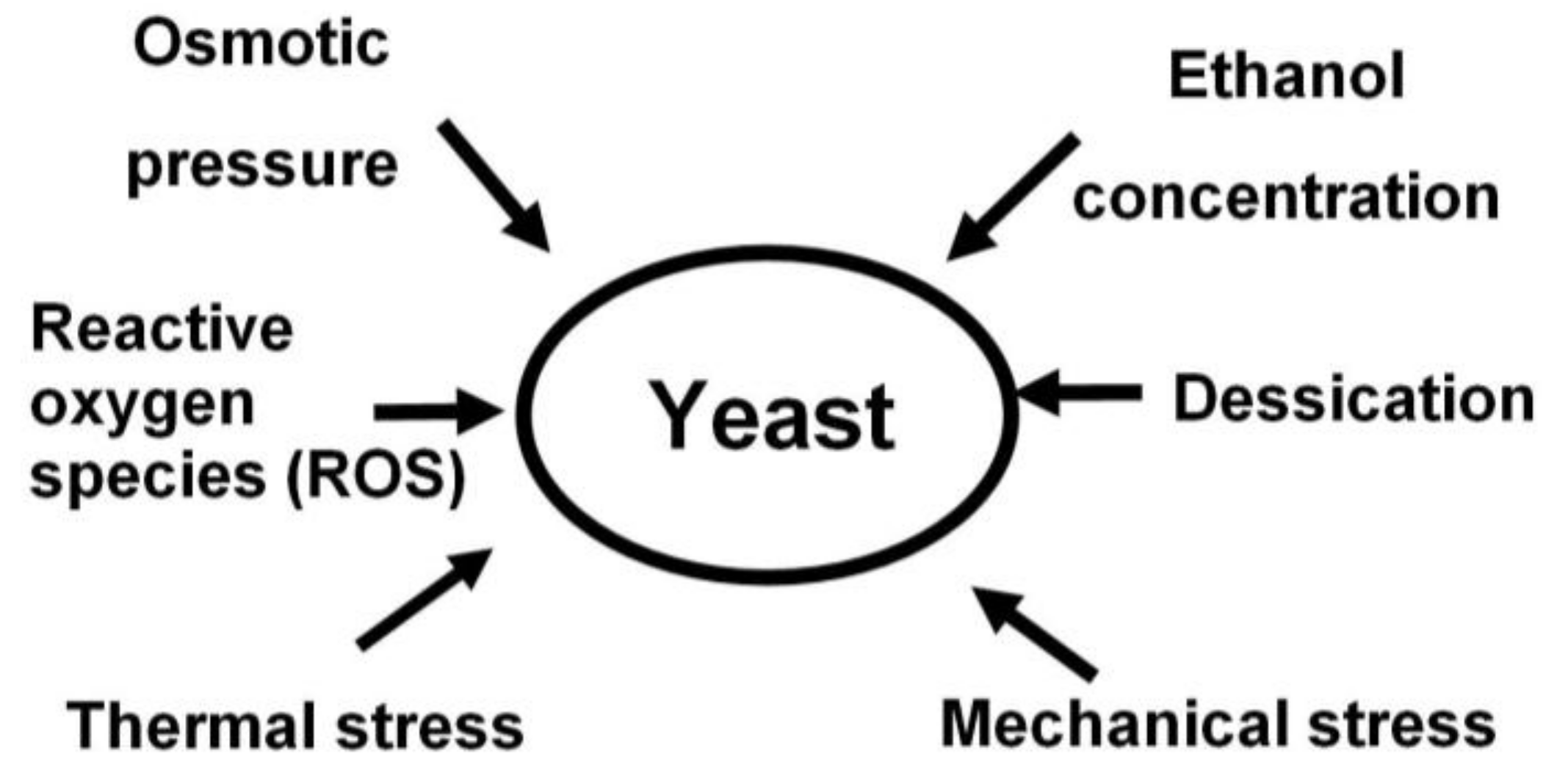
- yeast tolerance to stress factors such as: heat, osmotic pressure, temperature, ethanol, mechanical tension, desiccation, etc. [6];
- wort gravity [7];
- wort dissolved oxygen concentration at pitching and during fermentation [8];
- flocculation, adhesion, sedimentation, and cropping characteristics [9];
- culture characteristics during storage between fermentations and its subsequent re-pitching into wort [10].
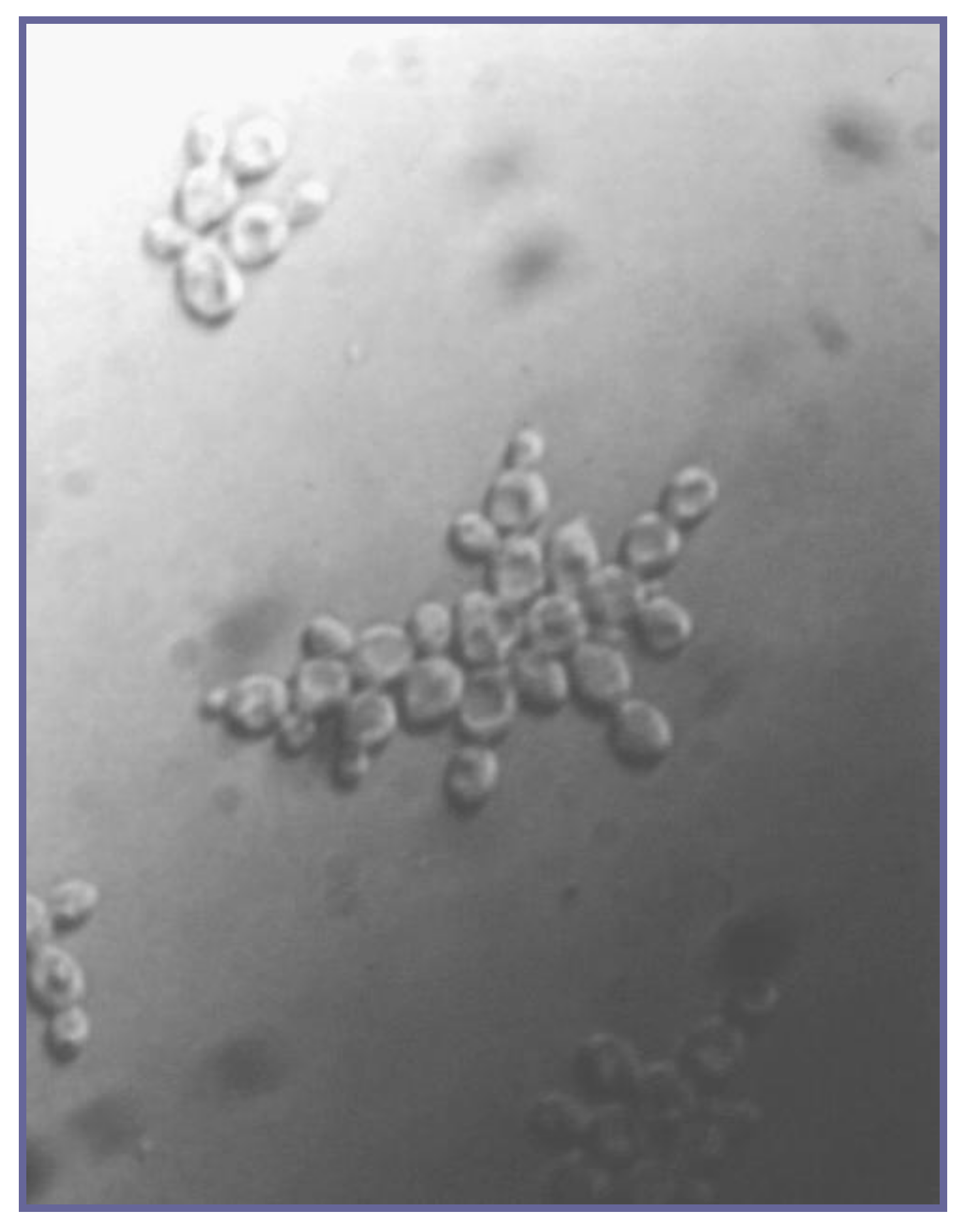
2. Yeast Flocculation
3. Measurement of Yeast Flocculation
3.1. Sedimentation Methods (For Example, the Helm Sedimentation Test)
3.2. Direct Observation of Floc Formation in the Growth/Fermentation Medium
3.3. Static Fermentation Methods
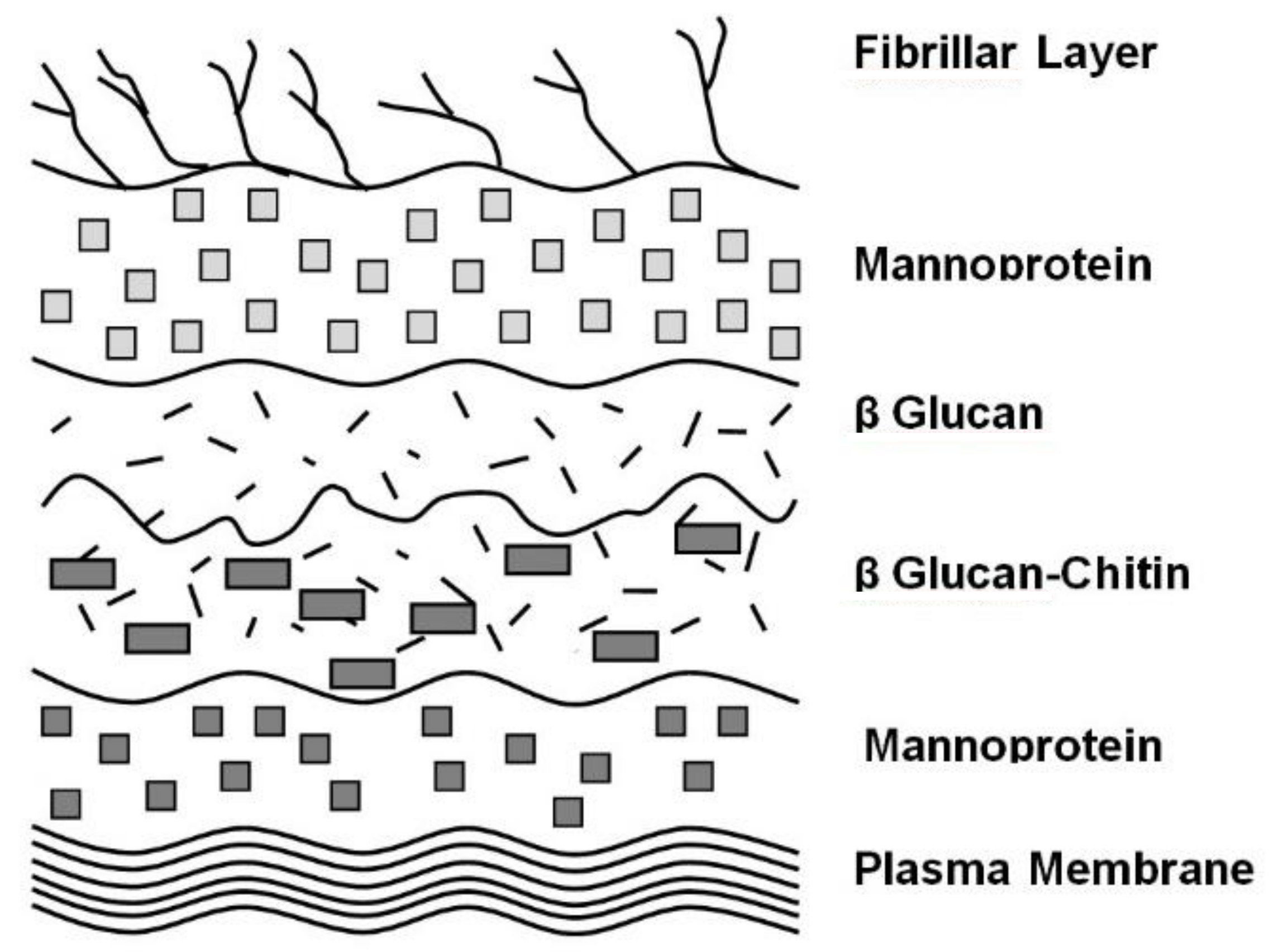
4. Cell Wall Characteristics
5. Sexual Aggregation
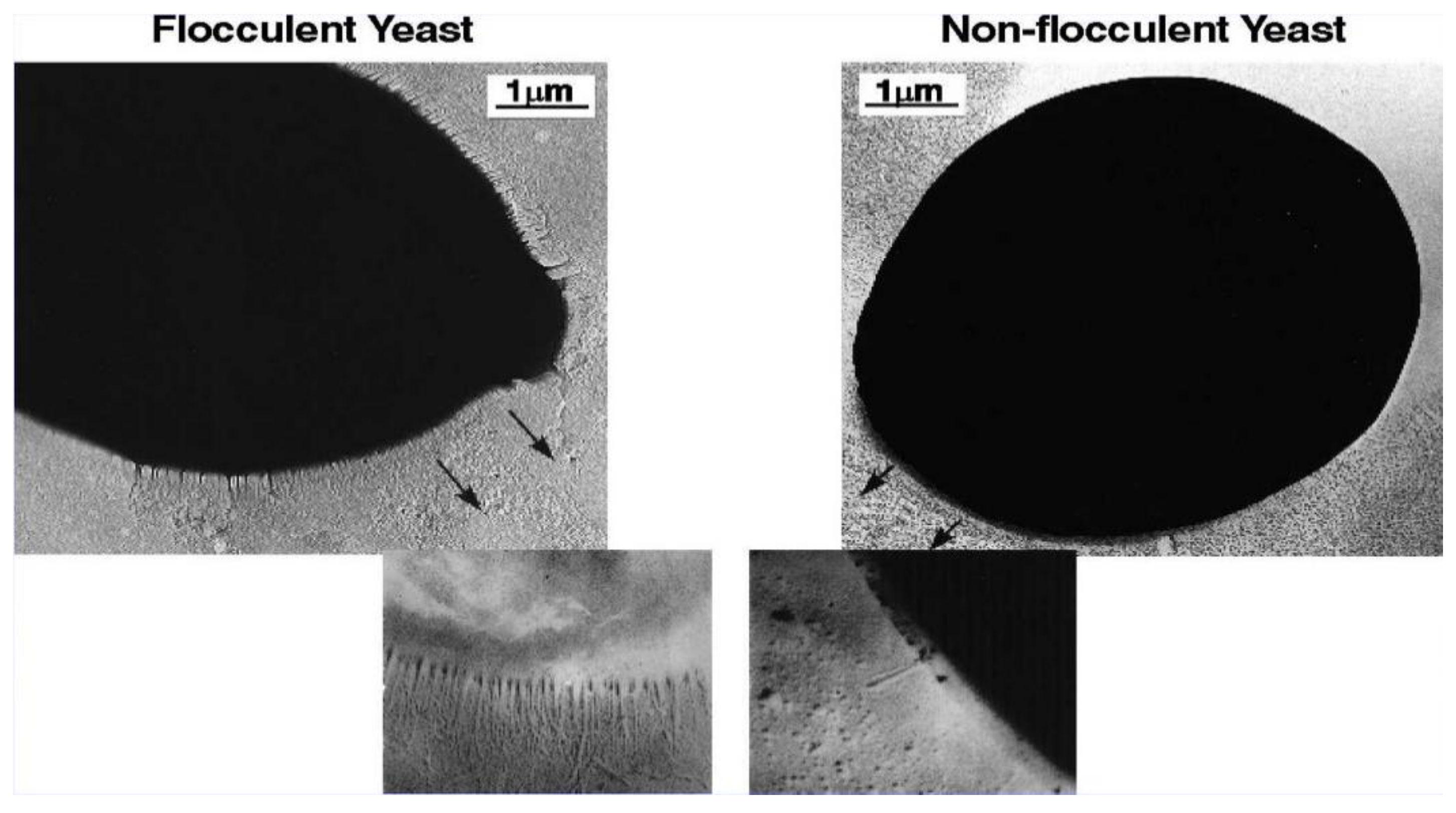
6. Yeast Cell Wall Structure and Flocculation
7. Factors Affecting Yeast Flocculation
- The carbon dioxide production rate is slow;
- wort attenuation is approaching completion—most of the fermentable sugars in wort have been removed by the yeast culture, including glucose, fructose, sucrose, maltose, and finally maltotriose [4];
- flocculation ability is high but not too high;
- yeast concentration in suspension is maximal [61];
- The way that yeast cells are packed into flocs;
- the floc size, shape, and density;
- higher gravity worts, following fermentation, results in “green” (immature) beers with a higher viscosity and density. Both these factors will retard yeast sedimentation and lead to increased osmotic pressure and ethanol prior to dilution to the fermented wort’s sales gravity and alcohol concentration.
8. Co-Flocculation
9. Genetic Control of Yeast Flocculation
10. Flocculation Gene Structure
11. The Genetic Instability of Flocculation in Brewer’s Yeast Strains
12. The Influence of Cell Surface Hydrophobicity (CSH) and Cell Surface Charge on Yeast Flocculation
13. Premature Yeast Flocculation
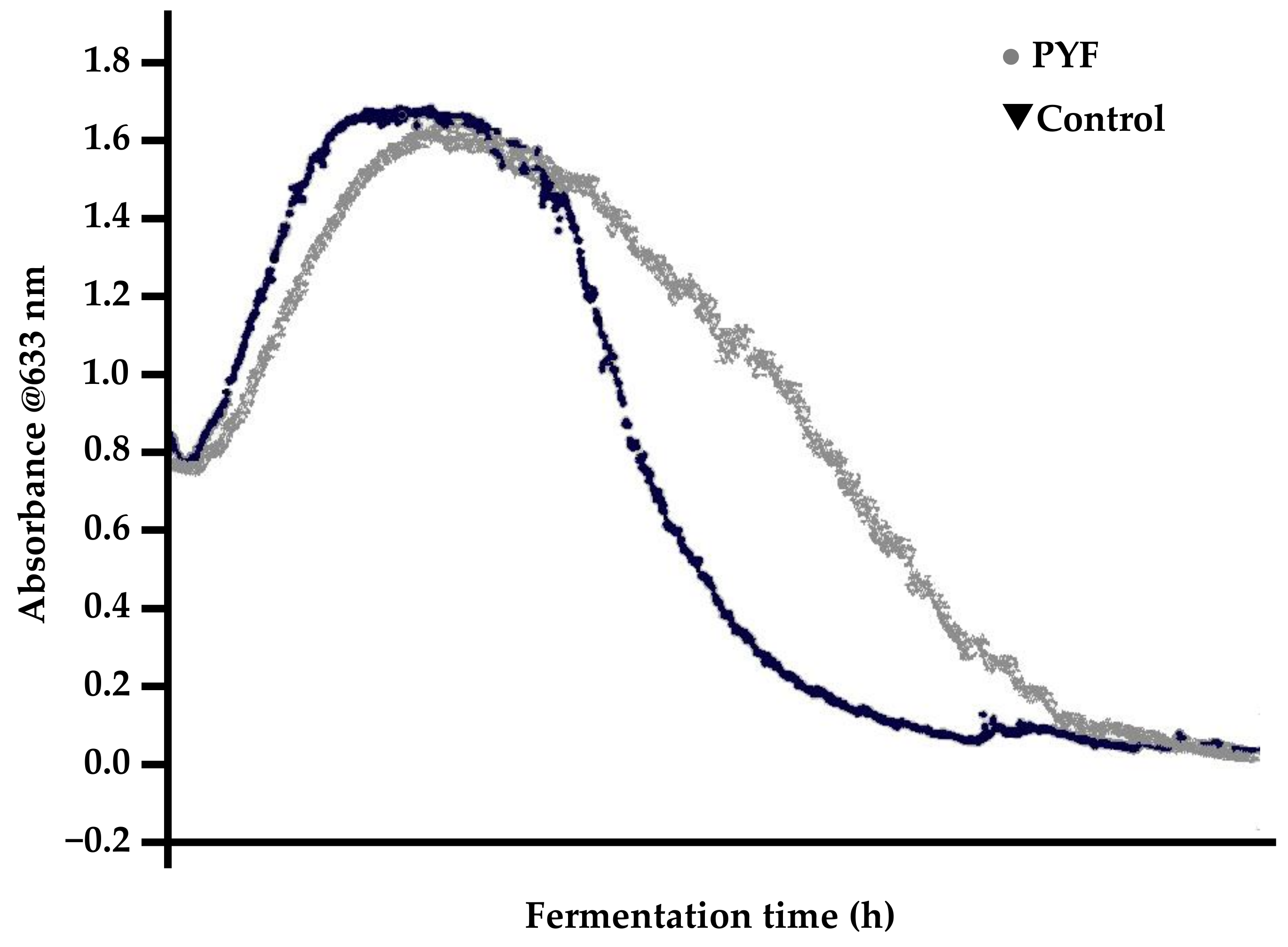
- Rapid yeast flocculation out of suspension, resulting in elevated apparent extract values relative to a “normal” malt (depending on the fermentation vessel employed);
- typically, normal fermentations exhibit parabolic yeast-in-suspension trends while PYF yeast-in-suspension curves proceed in a normal and parabolic manner to a peak and then decline in a concave fashion (Figure 14).
14. Phenotypic Effects on Flocculation
14.1. Cations
14.2. Medium pH
14.3. Temperature
14.4. Oxygen
14.5. Sugars
- Carbohydrate sources are nutrients that stimulate the loss of flocculation in a defined growth medium (for example, yeast nitrogen base (YNB)) [60];
- all metabolisable carbon sources (for example, glucose, fructose, galactose, maltose, and sucrose) induce the loss of flocculation in YNB, which ethanol does not—details to follow [4];
- the rate of sugar-induced flocculation appears to be associated with the rate of sugar metabolism;
- the rate of sugar-induced flocculation loss most likely requires energy and this process is blocked by ethanol;
- growth does not always trigger flocculation loss because cells grown in a medium containing ethanol remained flocculent;
- glucose-induced loss of flocculation requires de novo protein synthesis—cycloheximide addition (an inhibitor of protein synthesis) to glucose-growing cells impairs the loss of flocculation [133].
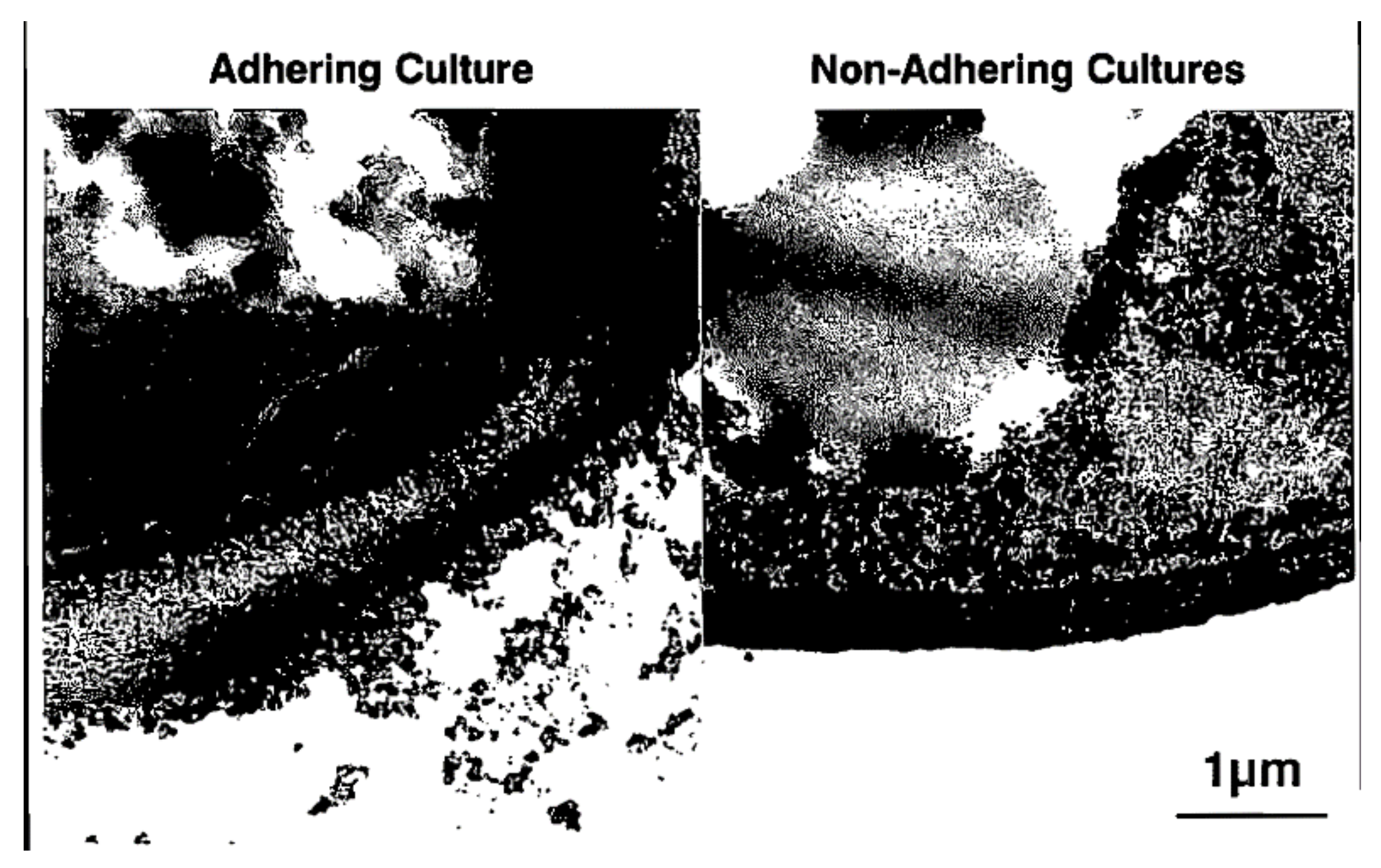
15. Adhesion and Biofilm Formation
16. Centrifuges to Crop Yeast
- Cropping of non-flocculent yeast cultures at the end of primary fermentation;
- reducing the yeast quantity from green beer before the start of secondary fermentation/maturation;
- beer recovery from cropped yeast [138];
- removal of cold break (precipitated protein, etc.) and yeast at the end of maturation;
- separation of the hot break after wort boiling.
17. Conclusions
Acknowledgments
Conflicts of Interest
References
- Stewart, G.G.; Russell, I. An Introduction to Brewing Science and Technology. Series III. Brewer’s Yeast, 2nd ed; Institute of Brewing and Distilling: London, UK, 2009. [Google Scholar]
- Stewart, G.G. Brewing and Distilling Yeasts; Springer: Cham, Switzerland, 2017; pp. 241–258. [Google Scholar]
- Lekkas, C.; Stewart, G.G.; Hill, A.; Taidi, B.; Hodgson, J. Elucidation of the role of nitrogenous wort components in wort fermentation. J. Inst. Brew. 2007, 113, 183–191. [Google Scholar] [CrossRef]
- Stewart, G.G. Studies on the uptake and metabolism of wort sugars during brewing fermentations. Tech. Quart. Master Brew. Assoc. Am. 2006, 43, 265–269. [Google Scholar] [CrossRef]
- Stewart, G.G. Biochemistry of brewing. In Biochemistry of Food; Eskin, N.A.M., Shahidi, N., Eds.; Elsevier: Oxford, UK, 2012; pp. 291–318. [Google Scholar]
- Piper, P.W.; Talreja, K.; Panaretou, B.; Moradas-Ferreira, P.; Byrne, K.; Praekelt, U.M.; Meacock, P.; Récnacq, M.; Boucherie, H. Induction of major heat-shock proteins of Saccharomyces cerevisiae, including plasma membrane Hsp30, by ethanol levels above a critical threshold. Microbiology 1994, 140, 3031–3038. [Google Scholar] [CrossRef] [PubMed]
- Stewart, G.G. Brewing Intensification; American Society for Brewing Chemists: St. Paul, MN, USA, 2014. [Google Scholar]
- Verbelen, P.J.; Saerens, S.M.; Van Mulders, S.E.; Delvaux, F.; Delvaux, F.R. The role of oxygen in yeast metabolism during high cell density brewery fermentations. Appl. Microbiol. Biotechnol. 2009, 82, 1143–1156. [Google Scholar] [CrossRef] [PubMed]
- Soares, E.V. Flocculation in Saccharomyces cerevisiae: A review. J. Appl. Microbiol. 2010, 110, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Stewart, G.G.; Hill, A.E.; Russell, I. 125th anniversary review—Developments in brewing and distilling yeast strains. J. Inst. Brew. 2013, 119, 202–220. [Google Scholar] [CrossRef]
- Stewart, G.G. The concept of nature-nurture applied to brewer’s yeast and wort fermentations. Tech. Quart. Master Brew. Assoc. Am. 2015, 52, 146–151. [Google Scholar] [CrossRef]
- Mamvura, T.A.; Paterson, A.E.; Fanucchi, D. The impact of pipe geometry variations on hygiene and success of orbital welding of brewing industry equipment. J. Inst. Brew. 2017, 123, 81–97. [Google Scholar] [CrossRef]
- Douglas, L.J. Adhesion of Candida species to epithelial surfaces. Crit. Rev. Microbiol. 1987, 15, 27–43. [Google Scholar] [CrossRef] [PubMed]
- Speers, R.A. A review of yeast flocculation. In International Brewers Symposium: Yeast Flocculation, Vitality and Viability; Master Brewers Association of the Americas: St. Paul, MN, USA, 2012. [Google Scholar]
- Miki, B.; Poon, N.H.; James, A.P.; Seligy, V.L. Possible mechanisms for flocculation interactions governed by gene FLO1 in Saccharomyces cerevisiae. J. Bacteriol. 1982, 150, 878–889. [Google Scholar] [PubMed]
- Stewart, G.G.; Russell, I. Yeast Flocculation. Brewing Science; Pollock, J.R.A., Ed.; Academic Press: London, UK, 1981; Volume 2, pp. 61–92. [Google Scholar]
- Gilliland, R.B. The flocculation characteristics of brewing yeasts during fermentation. Proc. Eur. Brew. Conv. Congr. Brighton 1951, 35–58. [Google Scholar]
- Thorne, R.S.W. Some aspects of yeast flocculation. Proc. Eur. Brew. Conv. Congr. Brighton 1951, 21–34. [Google Scholar]
- Stewart, G.G.; Russell, I.; Sills, A.M. Factors that control the utilization of wort carbohydrates by yeast. Tech. Quart. Master Brew. Assoc. Am. 1983, 20, 1–8. [Google Scholar]
- Marika, H.; Straver, J.; Kijne, J.W.; Smit, G. Cause and control of flocculation in yeast. Trends Biotechnol. 1993, 11, 228–232. [Google Scholar]
- Jin, Y.; Speers, R.A. Effect of environmental conditions on the flocculation of Saccharomyces cerevisiae. J. Am. Soc. Brew. Chem. 2000, 58, 108–116. [Google Scholar]
- Soares, E.V.; Teixeira, J.A.; Mota, M. Effect of cultural and nutritional conditions on the control of flocculation expression in Saccharomyces cerevisiae. Can. J. Microbiol. 1994, 40, 851–857. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Mulders, S.E.; Ghequire, M.; Daenen, L.; Verbelen, P.J.; Verstrepen, K.J.; Delvaux, F.R. Flocculation gene variability in industrial brewer’s yeast strains. Appl. Microbiol. Biotechnol. 2010, 88, 1321–1331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nathan, L. Improvements in the fermentation and maturation of beers. J. Inst. Brew. 1930, 36, 538–554. [Google Scholar] [CrossRef]
- Krogerus, A.; Gibson, B.R. 125th anniversary review: Diacetyl and its control during brewery fermentation. J. Inst. Brew. 2013, 119, 86–97. [Google Scholar]
- Stratford, M. Yeast flocculation—A new perspective. Adv. Microb. Physiol. 1992, 33, 1–71. [Google Scholar]
- Verstrepan, K.J.; Klis, F.M. Flocculation, adhesion and biofilm formation in yeasts. Mol. Microbiol. 2006, 60, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Helm, E.; Nohr, B.; Thorne, R.S.W. The measurement of yeast flocculation and its significance in brewing. Wall. Lab. Commun. 1953, 16, 315–326. [Google Scholar]
- Stewart, G.G.; Garrison, I. Some observations on co-flocculation in Saccharomyces cerevisiae. Am. Soc. Brew. Chem. Proc. 1972, 118–131. [Google Scholar]
- Stewart, G.G.; Russell, I. The identification, characterization, and mapping of a gene for flocculation in Saccharomyces sp. Can. J. Microbiol. 1977, 23, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Boulton, C. Yeast handling. Brew. Dig. Int. 2011, 7, 7–10. [Google Scholar]
- Calleja, G.G. Microbial Aggregation; CRC Press: Boca Raton, FL, USA, 1984. [Google Scholar]
- Day, A.W.; Poon, N.H.; Stewart, G.G. Fungal fimbriae. III. The effect of flocculation in Saccharomyces. Can. J. Microbiol. 1975, 21, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Klis, F.M.; Boorsma, A.; De Groot, P.W.J. Cell wall construction in Saccharomyces cerevisiae. Yeast 2006, 23, 185–202. [Google Scholar] [CrossRef] [PubMed]
- Claro, F.B.; Rijsbrack, K.; Soares, E.V. Flocculation onset in Saccharomyces cerevisiae: Effect of ethanol, heat and osmotic stress. J. Appl. Microbiol. 2007, 102, 693–700. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ashbee, R.; Bignell, E.M. (Eds.) Pathogenic Yeasts. The Yeast Handbook; Springer: Berlin, Germany, 2010. [Google Scholar]
- Stewart, G.G. MBAA Award of Merit Lecture. A love affair with yeast. Tech. Quart. Master Brew. Assoc. Am. 2010, 47, 4–11. [Google Scholar]
- Winge, O. On haplophase and diplophase in some Saccharomycetes. C. R. Trav. Lab. Carlsberg. Ser. Physiol. 1935, 21, 77–111. [Google Scholar]
- Gimeno, C.J.; Ljungdahl, P.O.; Styles, C.A.; Fink, G.R. Unipolar cell divisions in the yeast S. cerevisiae lead to filamentous growth; regulation by starvation and RAS. Cell 1992, 68, 1077–1090. [Google Scholar] [CrossRef]
- Zarattini, R.A.; Williams, J.W.; Ernandes, J.R.; Stewart, G.G. Bacterial-induced flocculation in selected brewing strains of Saccharomyces cerevisiae. Cerevisia Biotechnol. 1993, 18, 65–70. [Google Scholar]
- Guo, B.; Styles, C.A.; Feng, Q.; Fink, G.R. A Saccharomyces gene family involved in invasive growth, cell-cell adhesion, and mating. Proc. Nat. Acad. Sci. USA 2000, 97, 12158–12163. [Google Scholar] [CrossRef] [PubMed]
- Bilinski, C.A.; Russell, I.; Stewart, G.G. Analysis of sporulation in brewer’s yeast: Induction of tetrad formation. J. Inst. Brew. 1986, 92, 594–598. [Google Scholar] [CrossRef]
- Nishihara, H.; Toraya, T.; Fukui, S. Flocculation of cell walls of brewer’s yeast and effects of metal ions, protein-denaturants and enzyme treatments. Arch. Microbiol. 1982, 131, 112–115. [Google Scholar] [CrossRef]
- Eddy, A.A. Composite ratio of the flocculation process of top and bottom strains of Saccharomyces cerevisiae. J. Inst. Brew. 1958, 64, 143–151. [Google Scholar] [CrossRef]
- Peng, X.; Sun, J.; Iserentant, D.; Michiels, C.; Verachtert, H. Flocculation and co-flocculation of bacteria by yeasts. Appl. Microbiol. Biotechnol. 2007, 21, 31–37. [Google Scholar]
- Stratford, M. Yeast flocculation: Calcium specificity. Yeast 1992, 5, 487–496. [Google Scholar] [CrossRef]
- Stewart, G.G.; Maskell, D.L.; Speers, A. Brewing fundamentals—Fermentation. Tech. Quart. Master Brew. Assoc. Am. 2016, 53, 2–22. [Google Scholar]
- Lyons, T.P.; Hough, J.S. The role of yeast cell walls in brewing. Brew. Dig. 1970, 45, 52–60. [Google Scholar]
- Beavan, M.T.; Belki, D.; Stewart, G.G.; Rose, A.H. Changes in electrophoretic mobility and lytic enzyme activity associated with developments of flocculating ability in Saccharomyces cerevisiae. Can. J. Microbiol. 1979, 25, 888–895. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Speers, R.A. Effect of cell surface hydrophobicity, charge, and zymolectin density on the flocculation of Saccharomyces cerevisiae. J. Am. Soc. Brew. Chem. 2001, 58, 108–116. [Google Scholar]
- Smit, G.; Straver, M.H.; Lugtenberg, B.J.; Kijne, J.W. Flocculence of Saccharomyces cerevisiae cells is induced by nutrient limitation, with cell surface hydrophobicity as a major determinant. Appl. Environ. Microbiol. 1992, 58, 3709–3714. [Google Scholar] [PubMed]
- Halina, S.; Nathan, L. Lectins; Springer: Dordrecht, The Netherlands, 2007. [Google Scholar]
- Miki, B.L.A.; Poon, N.H.; Seligy, N.L. Repression and induction of flocculation interactions to Saccharomyces cerevisiae. J. Bacteriol. 1982, 150, 890–899. [Google Scholar] [PubMed]
- Goldstein, I.J.; Poretz, R.D. Isolation, physicochemical characterization and carbohydrate-binding specificity of lectins. In The Lectins; Liener, I.E., Goldstein, I.J., Eds.; Academic Press: Orlando, FL, USA, 1986; pp. 52–54. [Google Scholar]
- Stratford, M.; Assinder, S. Yeast flocculation: Flo1 and new Flo phenotypes and receptor structure. Yeast 1991, 7, 559–574. [Google Scholar] [CrossRef] [PubMed]
- Soares, E.V.; Mota, M. Flocculation onset, growth phase, and genealogical age in Saccharomyces cerevisiae. Can. J. Microbiol. 1996, 42, 539–547. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teunissen, A.W.R.H.; Steensma, H.Y. Review: The dominant flocculation genes of Saccharomyces cerevisiae constitute a new subtelomeric gene family. Yeast 1995, 11, 1001–1013. [Google Scholar] [CrossRef] [PubMed]
- Van Mulders, S.E.; Christianen, E.; Saerens, S.E.; Daenen, L.; Verbelen, P.J.; Willaert, R.; Verstrepen, K.J.; Delvaux, F.R. Phenotypic diversity of Flo protein family-mediated adhesion in Saccharomyces cerevisiae. FEMS Yeast Res. 2009, 9, 178–190. [Google Scholar] [CrossRef] [PubMed]
- Siero, C.; Reboredo, N.M.; Villa, T.G. Flocculation of industrial and laboratory strains of Saccharomyces cerevisiae. J. Ind. Microbiol. 1994, 14, 461–466. [Google Scholar] [CrossRef]
- Stewart, G.G.; Russell, I.; Garrison, I.F. Some considerations of the flocculation characteristics of ale and lager yeast strains. J. Inst. Brew. 1975, 81, 248–257. [Google Scholar] [CrossRef]
- Van Hamersveld, E.H.; van der Lans, R.G.; Luyben, K.G. Quantification of brewer’s yeast flocculation in a stirred tank: Effect of physical parameters on flocculation. Biotechnol. Bioeng. 1997, 56, 190–200. [Google Scholar] [CrossRef]
- Wilcocks, K.R.; Smart, K.A. The importance of surface charge of hydrophobicity for the flocculation of chain forming brewing yeast strains and resistance of these parameters to acid washing. FEMS Microbiol. Lett. 1995, 15, 293–297. [Google Scholar] [CrossRef]
- Speers, R.A.; Wan, Y.-Q.; Stewart, R.J. Effects of fermentation parameters and cell wall properties on yeast flocculation. J. Inst. Brew. 2006, 112, 246–254. [Google Scholar] [CrossRef]
- Jarvis, P.; Jefferson, B.; Parsons, S.A. Measuring flocstructural characteristics. Environ. Sci. Biotechnol. 2005, 4, 1–18. [Google Scholar] [CrossRef]
- Stewart, G.G.; Russell, I.; Goring, T.E. Nature-nurture anomalies—Further studies in yeast flocculation. Am. Soc. Brew. Chem. Proc. 1975, 33, 137–147. [Google Scholar]
- Axcell, B.C.; van Nierop, S.; Vundla, W. Malt induced premature yeast flocculation. Tech. Quart. Master Brew. Assoc. Am. 2000, 37, 501–504. [Google Scholar]
- Mortier, A.; Soares, E.V. Separation of yeasts by addition of flocculent cells of Saccharomyces cerevisiae. World J. Microbiol. Biotechnol. 2007, 23, 1401–1407. [Google Scholar] [CrossRef]
- Eddy, A.A.; Rudin, A.D. Part of the yeast surface apparently involved in flocculation. J. Inst. Brew. 1958, 64, 19–21. [Google Scholar] [CrossRef]
- Stewart, G.G. Seduced by yeast. J. Am. Soc. Brew. Chem. 2015, 73, 1–21. [Google Scholar]
- Gilliland, R.B. Determination of yeast viability. J. Inst. Brew. 1959, 65, 424–429. [Google Scholar] [CrossRef]
- Richards, M. The use of giant-colony morphology for the differentiation of brewing yeasts. J. Inst. Brew. 1967, 73, 162–166. [Google Scholar] [CrossRef]
- Stewart, G.G. Recent developments in the characterization of brewery yeast strains. Tech. Quart. Master Brew. Assoc. Am. 1973, 9, 183–191. [Google Scholar]
- White, F.H.; Kidney, E. The influence of yeast strain on beer spoilage bacteria. Proc. Eur. Brew. Conv. Congr. Berl.-West 1979, 801–815. [Google Scholar]
- Rossouw, D.; Bagheri, B.; Setati, E.; Bauer, F.F. Co-flocculation of yeast species, a new mechanism to govern population dynamics in microbial ecosystems. PLoS ONE 2015, 10, e0136249. [Google Scholar] [CrossRef] [PubMed]
- Fink, S.L.; Cookson, B.T. Apoptosis, pyroptosis, and necrosis: Mechanistic description of dead and dying eukaryotic cells. Infect. Immun. 2005, 73, 1907–1916. [Google Scholar] [CrossRef] [PubMed]
- Powell, C.D.; Diacetis, A.N. Long term serial repitching and the genetic and phenotypic stability of brewer’s yeast. J. Inst. Brew. 2007, 113, 67–74. [Google Scholar] [CrossRef]
- Pomper, S.; Burkholder, P.R. Studies on the biochemical genetics of yeast. Proc. Natl. Acad. Sci. USA 1949, 35, 456–464. [Google Scholar] [CrossRef] [PubMed]
- Panchal, C.J.; Russell, I.; Sills, A.M.; Stewart, G.G. Genetic manipulation of brewing and related yeast strains. Food Technol. 1984, 111, 99–106. [Google Scholar]
- Lewis, C.W.; Johnston, J.R.; Martin, P.A. The genetics of yeast flocculation. J. Inst. Brew. 1976, 82, 158–160. [Google Scholar] [CrossRef]
- Mundy, R.D.; Cormack, B. Expression of Candida glabrata adhesion following exposure to chemical preservatives. J. Infect. Dis. 2009, 199, 1891–1898. [Google Scholar] [CrossRef] [PubMed]
- Hoyer, L.L.; Green, C.B.; OH, S.H.; Zhao, X. Discovering the secrets of the Candida albicans agglutinin-like sequence (ALS) gene family—A sticky pursuit. Med. Mycol. 2008, 46, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Goffeau, A.; Barrell, B.G.; Bussey, H.; Oliver, S.G. Life with 6000 genes. Science. 1996, 274, 546, 563–567. [Google Scholar] [CrossRef] [PubMed]
- Sherman, F.; Fink, G.R.; Hicks, J.B. Methods in Yeast Genetics; Cold Spring Harbor Laboratory Press: New York, NY, USA, 1986. [Google Scholar]
- Meaden, P.G. DNA fingerprinting of brewer’s yeast. Ferment 1996, 9, 267–272. [Google Scholar]
- Soares, E.V.; Seynaeve, J. The use of succinic acid, as a pH buffer, expands the potentialities of utilisation of a chemically defined medium in Saccharomyces cerevisiae flocculation studies. Biotechnol. Lett. 2000, 22, 859–863. [Google Scholar] [CrossRef]
- Stratford, M. Induction of flocculation in brewing yeasts by change in pH value. FEMS Microbiol. Lett. 1996, 136, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Russell, I.; Stewart, G.G. Revised nomenclature of genes that control flocculation. J. Inst. Brew. 1980, 86, 120–121. [Google Scholar] [CrossRef]
- Bayly, J.C.; Douglas, L.M.; Pretorium, I.S.; Bauer, F.F.; Dranginis, A.M. Characteristics of Flo11-dependent flocculation in Saccharomyces cerevisiae. FEMS Microbiol. Lett. 2005, 5, 1151–1156. [Google Scholar]
- Bester, M.C.; Pretorius, I.S.; Bauer, F.F. The regulation of Saccharomyces cerevisiae FLO gene expression and Ca2+-dependent flocculation by Flo8p and Mss11p. Curr. Genet. 2006, 49, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Govender, P.; Domingo, J.L.; Bester, M.C.; Pretorius, I.S.; Bauer, F.F. Controlled expression of the dominant flocculation genes FLO1, FLO5 and FLO11 in Saccharomyces cerevisiae. Appl. Environ. Microbiol. 2008, 74, 6041–6052. [Google Scholar] [CrossRef] [PubMed]
- Kock, J.L.; Venter, P.; Smith, D.P.; Van Wyk, P.W.; Botes, P.J.; Coetzee, D.J.; Pohl, C.H.; Both, A.; Riedel, K.-H.; Nigam, S. A novel oxylipin-associated “ghosting” phenomenon in yeast flocculation. Antonie Van Leeuwenhoek 2000, 77, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.H.; MacGurn, J.A.; Chu, T.; Stefan, C.J.; Emr, S.D. Arrestin-related ubiquitin-ligase adaptors regulate endocytosis and protein turnover at the cell surface. Cell 2008, 135, 714–725. [Google Scholar] [CrossRef] [PubMed]
- Verstrepen, K.J.; Reynolds, T.B.; Fink, G.R. Origins of variation in the fungal cell surface. Nat. Rev. Microbiol. 2004, 2, 533–540. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Halme, A.; Bumgarner, S.; Styles, C.; Fink, G.R. Genetic and epigenetic resolution of the FLO gene family generates cell-surface variation in yeast. Cell 2004, 116, 405–415. [Google Scholar] [CrossRef]
- Dunn, B.; Sherlock, G. Reconstitution of the genome origins and evolution of the hybrid lager yeast Saccharomyces pastorianus. Genome Res. 2008, 18, 1610–1623. [Google Scholar] [CrossRef] [PubMed]
- Loney, E.R.; Inglis, P.M.; Sharp, S.; Pryde, F.E.; Kent, N.A.; Mellor, J.; Louis, E.J. Repressive and non-repressive chromatin at nature telomeres in Saccharomyces cerevisiae. Epigenet. Chromat. 2009, 2, 18–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miller, T.; Krogan, N.J.; Dover, J.; Erdjument-Bromage, H.; Tempst, P.; Johnston, M.; Greenblatt, J.F.; Shilatifard, A. COMPASS: A complex of proteins associated with a tuthorax-related SET domain protein. Proc. Natl. Acad. Sci. USA 2001, 98, 12902–12907. [Google Scholar] [CrossRef] [PubMed]
- Stewart, G.G. The Horace Brown Medal Lecture—Forty years of brewing research. J. Inst. Brew. 2009, 115, 3–29. [Google Scholar] [CrossRef]
- Bird, A. DNA methylation patterns and epigenetic memory. Genes Dev. 2002, 16, 6–21. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.; Watari, J.; Shinotusuka, K. Genetic instability of flocculation of bottom-fermenting yeast. J. Am. Soc. Brew. Chem. 2001, 59, 130–134. [Google Scholar]
- Jibiki, M.; Ishibiki, T.; Yuuki, T.; Kagami, N. Application of polymerase chain reaction to determine the flocculation properties of brewer’s lager yeast. J. Am. Soc. Brew. Chem. 2001, 59, 107–110. [Google Scholar]
- Stewart, G.G. Yeast performance and management. Brewer 1996, 82, 211–215. [Google Scholar]
- Smart, K.A.; Whisker, S. Effect of serial repitching on the fermentation properties and condition of brewing yeast. J. Am. Soc. Brew. Chem. 1996, 54, 41–44. [Google Scholar]
- Teixeira, J.M.; Teixeira, J.A.; Mota, M.; Manuela, M.; Guerra, B.; Machado, M.; Cruz, J.M.; S’Almeida, A.M. The influence of cell wall composition of a brewer’s flocculent lager yeast on sedimentation during successive industrial fermentations. Proc. Eur. Brew. Conv. Congr. Lisb. 1991, 23, 241–248. [Google Scholar]
- Wightman, P.; Quain, D.E.; Meadon, P.G. Analysis of production brewing strains of yeast by DNA fingerprinting. Lett. Appl. Microbiol. 1996, 22, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Southern, E.M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J. Mol. Biol. 1975, 98, 503–517. [Google Scholar] [CrossRef]
- Amory, D.E.; Rouxhet, P.G.; Dufour, J.P. Flocculence of brewery yeasts and their surface properties: Chemical composition, electrostatic charge and hydrophobicity. J. Inst. Brew. 1988, 94, 79–84. [Google Scholar] [CrossRef]
- Lange, C.; Nett, J.H.; Trumpower, B.L.; Hunte, C. Specific roles of protein-phospholipid interactions in the yeast cytochrome bc1 complex structure. EMBO J. 2001, 20, 6591–6600. [Google Scholar] [CrossRef] [PubMed]
- Lyons, T.P.; Hough, J.S. Flocculation of brewer’s yeast. J. Inst. Brew. 1970, 76, 564–571. [Google Scholar] [CrossRef]
- Boulton, C.; Quain, D. Brewing Yeast and Fermentation; Blackwell Science: Oxford, UK, 2001. [Google Scholar]
- Van Holle, A.; Machado, M.D.; Soares, E.V. Flocculation in ale brewing strains of Saccharomyces cerevisiae: Re-evaluation of the role of cell surface charge and hydrophobicity. Appl. Microbiol. Biotechnol. 2011, 93, 1221–1229. [Google Scholar] [CrossRef] [PubMed]
- Dengis, P.B.; Nélissen, L.R.; Rouxhet, P.G. Mechanisms of yeast flocculation, comparison of top- and bottom-fermenting strains. Appl. Environ. Microbiol. 1995, 61, 718–728. [Google Scholar] [PubMed]
- Van Lersel, M.F.M.; Meersman, E.; Arntz, M.; Robvouts, F.M.; Abee, T. Effect of environmental conditions on flocculation and immobilisation of brewer’s yeast during production of alcohol-free beer. J. Inst. Brew. 1998, 104, 131–136. [Google Scholar]
- Potter, G.; Budge, S.M.; Speers, R.A. Flocculation, cell surface hydrophobicity and 3-OH oxylipins in the SMA strain of Saccharomyces pastorianus. J. Inst. Brew. 2015, 121, 31–37. [Google Scholar] [CrossRef]
- Ishimaru, S.; Kudo, S.; Hattan, M.; Yoshida, T.; Kataoka, J. Selection of small vessels for fermentation tests in the laboratory. Rep. Res. 1967, 10, 61–65. [Google Scholar]
- Krugger, L.; Ryder, D.S.; Alcock, C.; Murray, J.P. Malt quality: Prediction of malt fermentability. Part 1. Tech. Quart. Master Brew. Assoc. Am. 1982, 19, 45–51. [Google Scholar]
- Koizumi, H.; Ogawa, T. Rapid and sensitive method to measure premature yeast flocculation activity in malt. J. Am. Soc. Brew. Chem. 2005, 63, 147–150. [Google Scholar]
- Van Nierop, S.N.E.; Rautenbach, M.; Axcell, B.C. The impact of microorganisms on barley and malt quality—A review. J. Am. Soc. Brew. Chem. 2006, 62, 69–79. [Google Scholar]
- Lake, T.C.; Speers, R.A. A Discussion of malt-induced premature yeast flocculation. Tech. Quart. Master Brew. Assoc. Am. 2008, 4, 253–262. [Google Scholar]
- Van Nierop, S.N.E.; Cameron-Clarke, A.; Axcell, B.C. Enzymatic generation of malt factors responsible for premature yeast flocculation. J. Am. Soc. Brew. Chem. 2004, 62, 108–116. [Google Scholar]
- Armstrong, K.; Bendiak, D. PYF malt: Practical brewery observations of fermentability. Tech. Quart. Master Brew. Assoc. Am. 2007, 44, 40–46. [Google Scholar] [CrossRef]
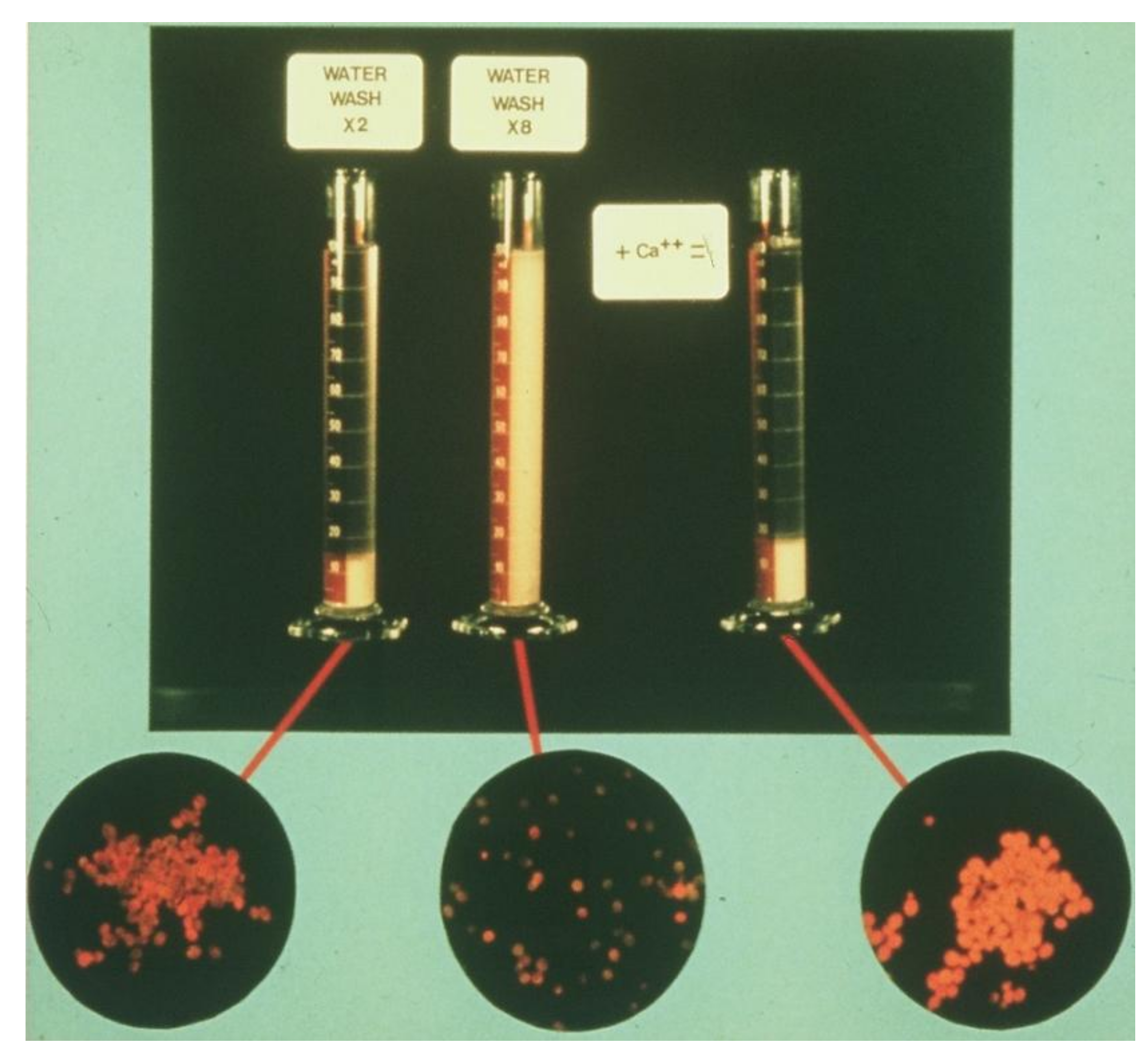
- Stewart, G.G.; Goring, T.E. Effect of some monovalent and divalent metal ions on the flocculation of brewer’s yeast strains. J. Inst. Brew. 1976, 82, 341–342. [Google Scholar] [CrossRef]
- Russell, I.; Dowhanick, T.; Raspor, P.; Stewart, G.G. Yeast flocculation—The influence of divalent ions. Proc. Eur. Brew. Conv. Congr. Zurich 1989, 529–536. [Google Scholar]
- Raspor, P.; Russell, I.; Stewart, G.G. An update of zinc ion as an effector of flocculation in brewer’s yeast strains. J. Inst. Brew. 1990, 96, 303–305. [Google Scholar] [CrossRef]
- Rees, E.M.R.; Stewart, G.G. Strain specific response of brewer’s yeast strains to zinc concentration in conventional and high gravity worts. J. Inst. Brew. 1998, 104, 255–264. [Google Scholar] [CrossRef]
- Rees, E.M.R.; Stewart, G.G. The effects of divalent ions magnesium and calcium on yeast fermentation performance in conventional (12 °P) and high (20 °P) gravity worts in both static and shaking fermentations. Proc. Eur. Brew. Conv. Congr. Maastricht 1997, 461–468. [Google Scholar]
- Gouveio, C.; Soares, E.V. Pb2+ inhibits competitively flocculation of Saccharomyces cerevisiae. J. Inst. Brew. 2004, 110, 141–145. [Google Scholar] [CrossRef]
- Stewart, G.G. Yeast mitochondria—Their influence on brewer’s yeast fermentation and medical research. Tech. Quart. Master Brew. Assoc. Am. 2004, 51, 3–11. [Google Scholar]
- Lorenz, R.T.; Parks, L.W. Involvement of heme components in sterol metabolism of Saccharomyces cerevisiae. Lipids 1991, 26, 598–603. [Google Scholar] [CrossRef] [PubMed]
- Abramova, N.; Sertil, O.; Mehta, S.; Lowry, C.V. Reciprocal regulation of anaerobic and aerobic cell wall mannoprotein gene expression in Saccharomyces cerevisiae. J. Bacteriol. 2001, 183, 2881–2887. [Google Scholar] [CrossRef] [PubMed]
- Straver, M.; Aar, P.C.V.D.; Sinit, G.; Kijne, J.W. Determinants of flocculence of brewer’s yeast during fermentation in wort. Yeast 1993, 9, 521–532. [Google Scholar] [CrossRef] [PubMed]
- Soares, E.V.; Duarte, A.A. Addition of nutrients induce a fast loss of flocculation-stirred cells of Saccharomyces cerevisiae. Biotechnol. Lett. 2002, 24, 1957–1960. [Google Scholar] [CrossRef]
- Baker, D.A.; Kirsop, B.H. Flocculation in Saccharomyces cerevisiae is influenced by wort composition and by actidione. J. Inst. Brew. 1972, 78, 454–458. [Google Scholar] [CrossRef]
- Bower, J.L.; Christensen, C.M. Disruptive technologies: Catching the wave. Harv. Bus. Rev. 1995, 73, 43–47. [Google Scholar]
- Dudbridge, M. Handbook of Lean Manufacturing; Wiley: New York, NY, USA, 2011. [Google Scholar]
- Chlup, P.H.; Bernard, D.; Stewart, G.G. Disc stack centrifuge operating parameters and their impact on yeast physiology. J. Inst. Brew. 2008, 114, 45–61. [Google Scholar] [CrossRef]
- Siebert, K.J.; Stenroose, L.E.; Reid, D.S.; Grabowski, D. Filtration difficulties resulting from damage to yeast during centrifugation. Tech. Quart. Master Brew. Assoc. Am. 1987, 24, 1–8. [Google Scholar]
- Chlup, P.H.; Stewart, G.G. Centrifuges in brewing. Tech. Quart. Master Brew. Assoc. Am. 2011, 48, 48–50. [Google Scholar] [CrossRef]
- Lewis, M.J.; Poerwantaro, W.M. Release of haze material from the cell walls of agitated yeast. J. Am. Soc. Brew. Chem. 1991, 49, 43–46. [Google Scholar]
- Stewart, G.G.; Russell, I.; Anstruther, A. (Eds.) Handbook of Brewing, 3rd ed.; Taylor & Francis Group: Abingdon, UK, 2018. [Google Scholar]
- Chlup, P.H.; Bernard, D.; Stewart, G.G. The disc stack centrifuge and its impact on yeast and beer quality. J. Am. Soc. Brew. Chem. 2007, 65, 151–155. [Google Scholar]
- Ogur, M.; St. John, R.; Nagai, S. Tetrazolium overlay technique for population studies of respiratory deficiency in yeast. Science 1957, 125, 928–929. [Google Scholar] [CrossRef] [PubMed]
- Chlup, P.H.; Conery, J.; Stewart, G.G. Detection of mannan from Saccharomyces cerevisiae. J. Am. Soc. Brew. Chem. 2007, 65, 29–37. [Google Scholar]
- Chlup, P.H.; Wang, T.; Lee, E.G.; Stewart, G.G. Assessment of the physiological status of yeast during high- and low-gravity wort fermentations determined by flow cytometry. Tech. Quart. Master Brew. Assoc. Am. 2007, 44, 286–295. [Google Scholar] [CrossRef]
- Schlee, C.; Miedl, M.; Leiper, K.A.; Stewart, G.G. The potential of confocal imaging for measuring physiological changes in brewer’s yeast. J. Inst. Brew. 2006, 112, 134–147. [Google Scholar] [CrossRef]










| Genes | Character | Sugars that Inhibit Flocculation |
|---|---|---|
| FLO1, FLO5, FLO9, FLO10 | Strong Flo1 phenotype | Only mannose |
| FL08 | Regulation of other FLO genes | Unknown |
| Lg-FLO1 | NewFlo phenotype | Mannose, glucose, sucrose, maltose, and maltotriose (not galactose) |
| FLO11 | Chain formation—pseudohyphae | No inhibition by sugars |
| FLONL, FLONS | Like NewFlo phenotype | Mannose, glucose, sucrose, maltose, maltotriose, and galactose |
| Not known | Mannose-insensitive (MI) flocculation (Ca-independent) | No inhibition by sugars |
| Yeast Culture | Flocculation Characteristic | Total Calcium Washed Off Yeast * | |
|---|---|---|---|
| Ale | S. cerevisiae | Non-flocculent | 18 |
| Non-flocculent | 19 | ||
| Flocculent | 30 | ||
| Flocculent | 42 | ||
| Lager | S. pastorianus | Non-flocculent | 12 |
| Non-flocculent | 14 | ||
| Flocculent | 20 | ||
| Flocculent | 22 | ||
| Characteristic | Before Centrifugation | After Centrifugation |
|---|---|---|
| Viability (%) | 85 | 42 |
| Extracellular pH | 4.2 | 6.0 |
| Intracellular pH | 5.8 | 5.3 |
| Damaged cells (%) | 4 | 15 |
| Glycogen (ppm) | 18 | 8 |
| Trehalose (ppm) | 22 | 6 |
| Mannan released (counts) | 400 | 1000 |
| Proteinase A (U/mL) | 3.1 | 6.2 |
| Hydrophobic polypeptides (mg/L) | 48 | 25 |
| Beer foam stability (NIBEM) | 110 | 82 |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stewart, G.G. Yeast Flocculation—Sedimentation and Flotation. Fermentation 2018, 4, 28. https://doi.org/10.3390/fermentation4020028
Stewart GG. Yeast Flocculation—Sedimentation and Flotation. Fermentation. 2018; 4(2):28. https://doi.org/10.3390/fermentation4020028
Chicago/Turabian StyleStewart, Graham G. 2018. "Yeast Flocculation—Sedimentation and Flotation" Fermentation 4, no. 2: 28. https://doi.org/10.3390/fermentation4020028