The Effect of Cell Immobilization by Calcium Alginate on Bacterially Induced Calcium Carbonate Precipitation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Microorganism and Growth Medium
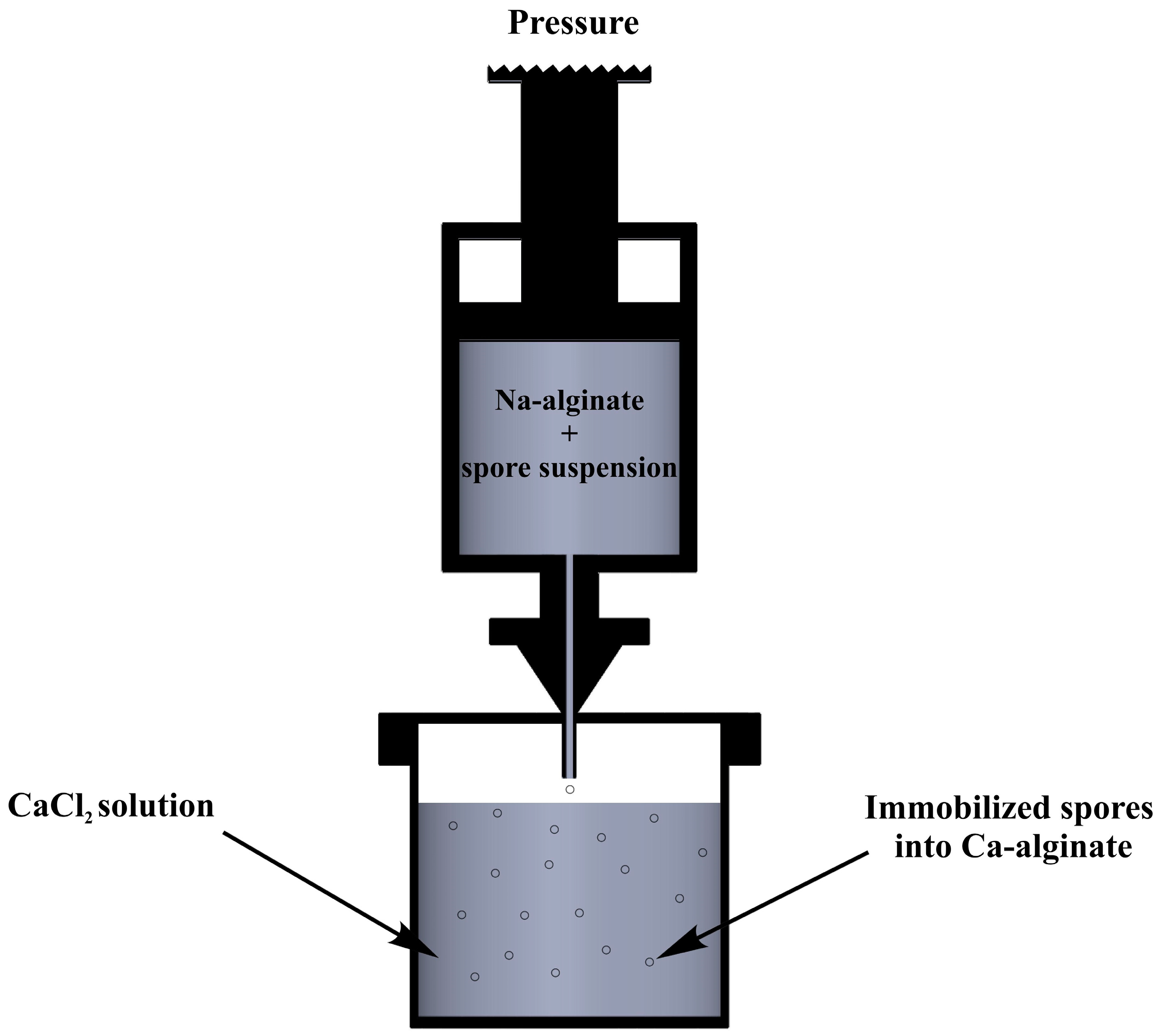
2.3. Synthesis of Calcium Alginate and Immobilization Process
2.4. Experimental Design and Fermentation Procedure
2.5. CaCO3 Extraction
2.6. Crystal Characterization and Morphological Observation
3. Results and Discussion
3.1. Immobilization of Bacterial Cell into Ca-Alginate
3.2. Bacterially Induced CaCO3 Precipitation
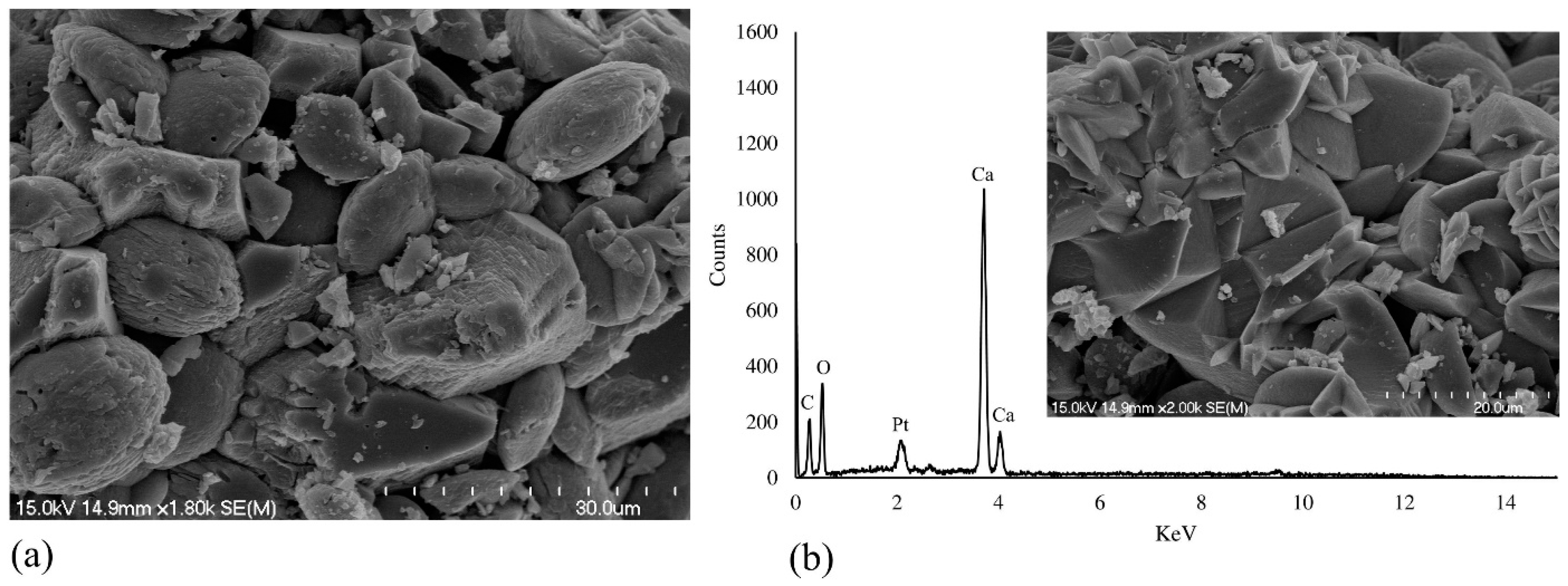
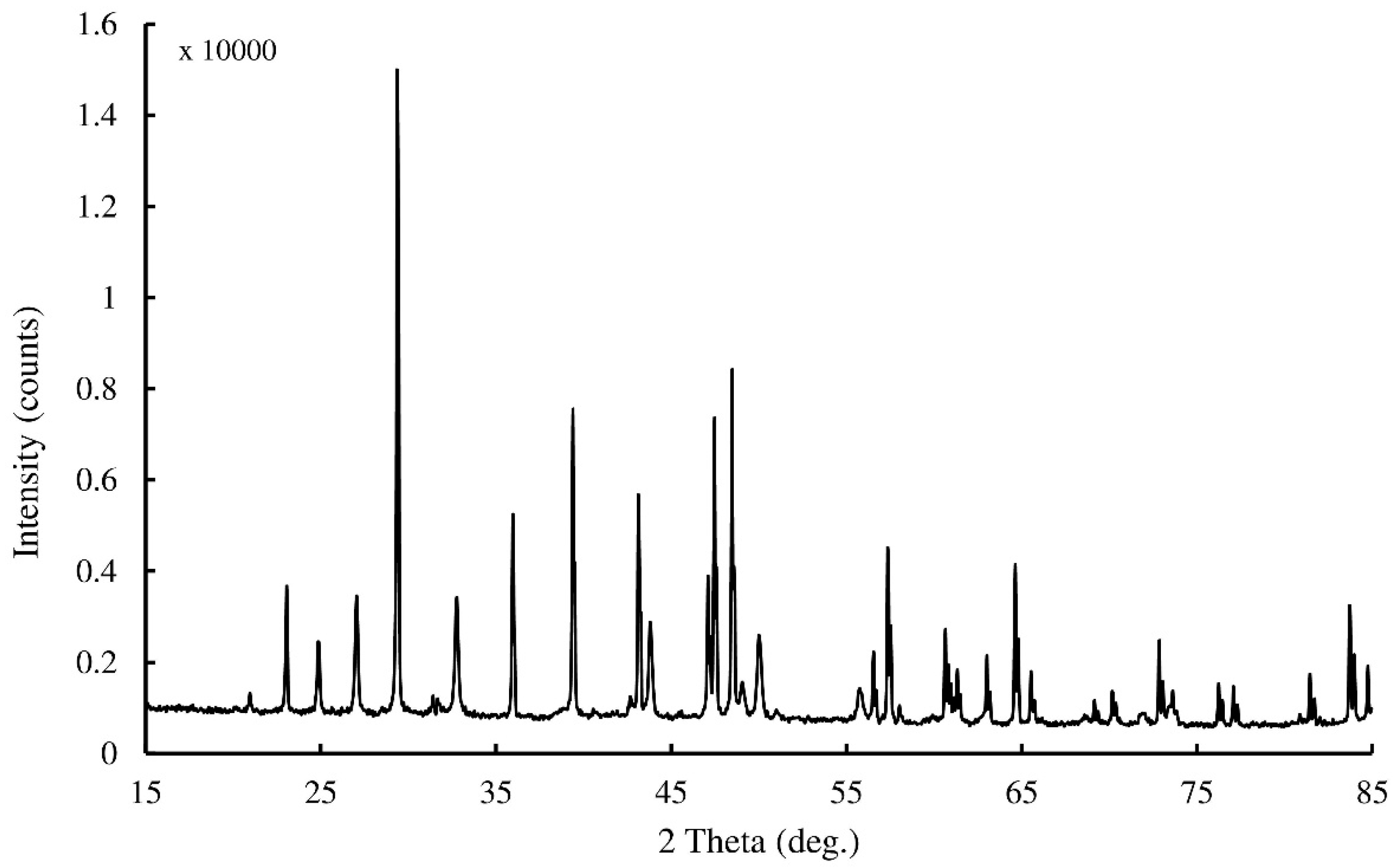
3.3. Morphological Observation and Crystal Characterization
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Van Paassen, L.A.; Daza, C.M.; Staal, M.; Sorokin, D.Y.; van der Zon, W.; van Loosdrecht, M.C.M. Potential soil reinforcement by biological denitrification. Ecol. Eng. 2010, 36, 168–175. [Google Scholar] [CrossRef]
- Whiffin, V.S.; van Paassen, L.A.; Harkes, M.P. Microbial carbonate precipitation as a soil improvement technique. Geomicrobiol. J. 2007, 24, 417–423. [Google Scholar] [CrossRef]
- Burbank, M.B.; Weaver, T.J.; Green, T.L.; Williams, B.; Crawford, R.L. Precipitation of calcite by indigenous microorganisms to strengthen liquefiable soils. Geomicrobiol. J. 2011, 28, 301–312. [Google Scholar] [CrossRef]
- Cheng, L.; Cord-Ruwisch, R. Upscaling effects of soil improvement by microbially induced calcite precipitation by surface percolation. Geomicrobiol. J. 2014, 31, 396–406. [Google Scholar] [CrossRef]
- Warren, L.A.; Maurice, P.A.; Parmar, N.; Ferris, G.F. Microbially mediated calcium carbonate precipitation: Implications for interpreting calcite precipitation and for solid-phase capture of inorganic contaminants. Geomicrobiol. J. 2001, 18, 93–115. [Google Scholar]
- Dhami, N.K.; Reddy, M.S.; Mukherjee, A. Biomineralization of calcium carbonates and their engineered applications: A review. Front. Microbiol. 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Tiano, P.; Biagiotti, L.; Mastromei, G. Bacterial bio-mediated calcite precipitation for monumental stones conservation: Methods of evaluation. J. Microbiol. Methods 1999, 36, 139–145. [Google Scholar] [CrossRef]
- Okyay, T.O.; Rodrigues, D.F. Biotic and abiotic effects on CO2 sequestration during microbially-induced calcium carbonate precipitation. FEMS Microbiol. Ecol. 2015, 91. [Google Scholar] [CrossRef] [PubMed]
- De Muynck, W.; Leuridan, S.; Van Loo, D.; Verbeken, K.; Cnudde, V.; De Belie, N.; Verstraete, W. Influence of pore structure on the effectiveness of a biogenic carbonate surface treatment for limestone conservation. Appl. Environ. Microbiol. 2011, 77, 6808–6820. [Google Scholar] [CrossRef] [PubMed]
- Dhami, N.K.; Reddy, M.S.; Mukherjee, A. Improvement in strength properties of ash bricks by bacterial calcite. Ecol. Eng. 2012, 39, 31–35. [Google Scholar] [CrossRef]
- Sierra-Beltran, M.G.; Jonkers, H.M.; Schlangen, E. Characterization of sustainable bio-based mortar for concrete repair. Constr. Build. Mater. 2014, 67, 344–352. [Google Scholar] [CrossRef]
- Van Tittelboom, K.; De Belie, N.; De Muynck, W.; Verstraete, W. Use of bacteria to repair cracks in concrete. Cem. Concr. Res. 2010, 40, 157–166. [Google Scholar] [CrossRef]
- Seifan, M.; Samani, A.K.; Berenjian, A. Bioconcrete: Next generation of self-healing concrete. Appl. Microbiol. Biotechnol. 2016, 100, 2591–2602. [Google Scholar] [CrossRef] [PubMed]
- Seifan, M.; Samani, A.K.; Burgess, J.J.; Berenjian, A. The effectiveness of microbial crack treatment in self healing concrete. In High Value Processing Technologies; Berenjian, A., Jafarizadeh-Malmiri, H., Song, Y., Eds.; Nova Science Publishers Inc.: New York, NY, USA, 2016; pp. 97–124. [Google Scholar]
- Seifan, M.; Samani, A.K.; Berenjian, A. New insights into the role of pH and aeration in the bacterial production of calcium carbonate (CaCO3). Appl. Microbiol. Biotechnol. 2017, 101, 3131–3142. [Google Scholar] [CrossRef] [PubMed]
- Jonkers, H.M.; Thijssen, A.; Muyzer, G.; Copuroglu, O.; Schlangen, E. Application of bacteria as self-healing agent for the development of sustainable concrete. Ecol. Eng. 2010, 36, 230–235. [Google Scholar] [CrossRef]
- Stevenson, A.; Cray, J.A.; Williams, J.P.; Santos, R.; Sahay, R.; Neuenkirchen, N.; McClure, C.D.; Grant, I.R.; Houghton, J.D.; Quinn, J.P. Is there a common water-activity limit for the three domains of life? ISME J. 2015, 9, 1333–1351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stevenson, A.; Burkhardt, J.; Cockell, C.S.; Cray, J.A.; Dijksterhuis, J.; Fox-Powell, M.; Kee, T.P.; Kminek, G.; McGenity, T.J.; Timmis, K.N. Multiplication of microbes below 0.690 water activity: Implications for terrestrial and extraterrestrial life. Environ. Microbiol. 2015, 17, 257–277. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, A.; Hallsworth, J.E. Water and temperature relations of soil actinobacteria. Environ. Microbiol. Rep. 2014, 6, 744–755. [Google Scholar] [CrossRef] [PubMed]
- Cray, J.A.; Connor, M.C.; Stevenson, A.; Houghton, J.D.; Rangel, D.E.; Cooke, L.R.; Hallsworth, J.E. Biocontrol agents promote growth of potato pathogens, depending on environmental conditions. Microb. Biotechnol. 2016, 9, 330–354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seifan, M.; Samani, A.K.; Berenjian, A. Induced calcium carbonate precipitation using Bacillus species. Appl. Microbiol. Biotechnol. 2016, 100, 9895–9906. [Google Scholar] [CrossRef] [PubMed]
- Kourkoutas, Y.; Bekatorou, A.; Banat, I.M.; Marchant, R.; Koutinas, A.A. Immobilization technologies and support materials suitable in alcohol beverages production: A review. Food Microbiol. 2004, 21, 377–397. [Google Scholar] [CrossRef]
- Bell, A.N.; Magill, E.; Hallsworth, J.E.; Timson, D.J. Effects of alcohols and compatible solutes on the activity of β-galactosidase. Appl. Biochem. Biotechnol. 2013, 169, 786–794. [Google Scholar] [CrossRef] [PubMed]
- Cray, J.A.; Stevenson, A.; Ball, P.; Bankar, S.B.; Eleutherio, E.C.; Ezeji, T.C.; Singhal, R.S.; Thevelein, J.M.; Timson, D.J.; Hallsworth, J.E. Chaotropicity: A key factor in product tolerance of biofuel-producing microorganisms. Curr. Opin. Biotechnol. 2015, 33, 228–259. [Google Scholar] [CrossRef] [PubMed]
- Bezerra, M.A.; Santelli, R.E.; Oliveira, E.P.; Villar, L.S.; Escaleira, L.A. Response surface methodology (rsm) as a tool for optimization in analytical chemistry. Talanta 2008, 76, 965–977. [Google Scholar] [CrossRef] [PubMed]

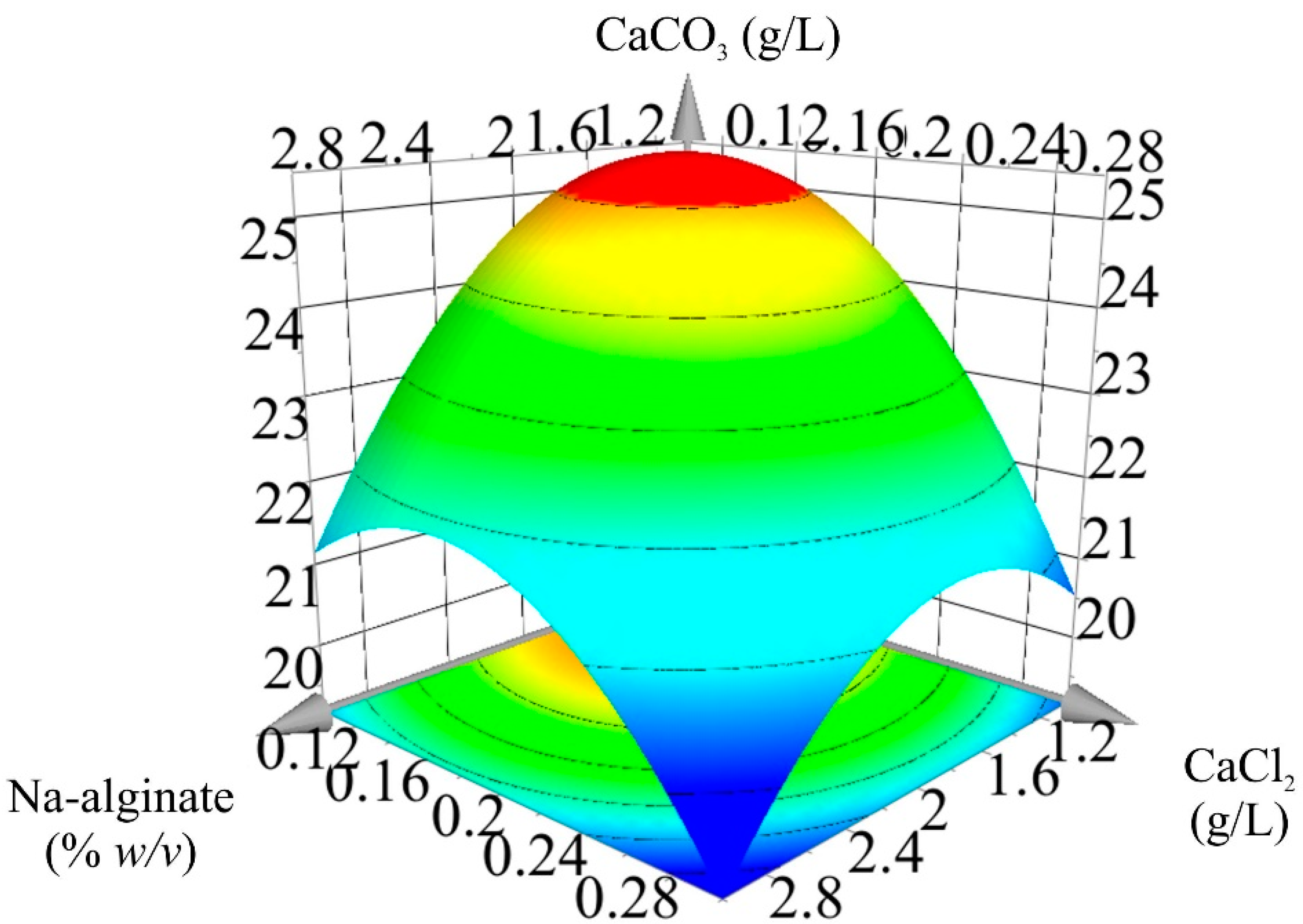
| Experimental Run | Na-Alginate (w/v) | CaCl2 (M) | CaCO3 (g/L) |
|---|---|---|---|
| 1 | 1 | 0.1 | 24.93 |
| 2 | 3 | 0.1 | 21.32 |
| 3 | 1 | 0.3 | 20.17 |
| 4 | 3 | 0.3 | 19.27 |
| 5 | 1 | 0.2 | 24.99 |
| 6 | 3 | 0.2 | 21.37 |
| 7 | 2 | 0.1 | 24.69 |
| 8 | 2 | 0.3 | 21.52 |
| 9 | 2 | 0.2 | 24.59 |
| 10 | 2 | 0.2 | 23.96 |
| 11 | 2 | 0.2 | 24.62 |
| Terms | Coefficient | Std. Err. * | p-Value |
|---|---|---|---|
| Constant | 24.493 | 0.236 | 0.000 |
| X1 | −1.355 | 0.187 | 0.000 |
| X2 | −1.663 | 0.187 | 0.000 |
| X12 | −1.459 | 0.289 | 0.004 |
| X22 | −1.537 | 0.289 | 0.003 |
| X1·X2 | 0.676 | 0.230 | 0.032 |
| Source of Variation | DF * | SS * | MS (Variance) * | F-Value | p-Value | SD * |
|---|---|---|---|---|---|---|
| Total | 11 | 5793.834 | 526.712 | - | - | - |
| Constant | 1 | 5747.827 | 5747.827 | - | - | - |
| Total corrected | 10 | 46.007 | 4.601 | - | - | 2.145 |
| Regression | 5 | 44.949 | 8.990 | 42.47 | 0.000 | 2.998 |
| Residual | 5 | 1.058 | 0.212 | - | - | 0.460 |
| Lack of Fit | 3 | 0.780 | 0.260 | 1.868 | 0.367 | 0.510 |
| Pure error | 2 | 0.278 | 0.139 | - | - | 0.373 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seifan, M.; Samani, A.K.; Hewitt, S.; Berenjian, A. The Effect of Cell Immobilization by Calcium Alginate on Bacterially Induced Calcium Carbonate Precipitation. Fermentation 2017, 3, 57. https://doi.org/10.3390/fermentation3040057
Seifan M, Samani AK, Hewitt S, Berenjian A. The Effect of Cell Immobilization by Calcium Alginate on Bacterially Induced Calcium Carbonate Precipitation. Fermentation. 2017; 3(4):57. https://doi.org/10.3390/fermentation3040057
Chicago/Turabian StyleSeifan, Mostafa, Ali Khajeh Samani, Shaun Hewitt, and Aydin Berenjian. 2017. "The Effect of Cell Immobilization by Calcium Alginate on Bacterially Induced Calcium Carbonate Precipitation" Fermentation 3, no. 4: 57. https://doi.org/10.3390/fermentation3040057







