1. Introduction
The current demands for reliable electrochemical biosensing require the development of sensing (bio)platforms exhibiting large surface area to allow for the high loading of capture molecules, good biocompatibility for biological activity preservation, and excellent conductivity for electron transportation [
1].
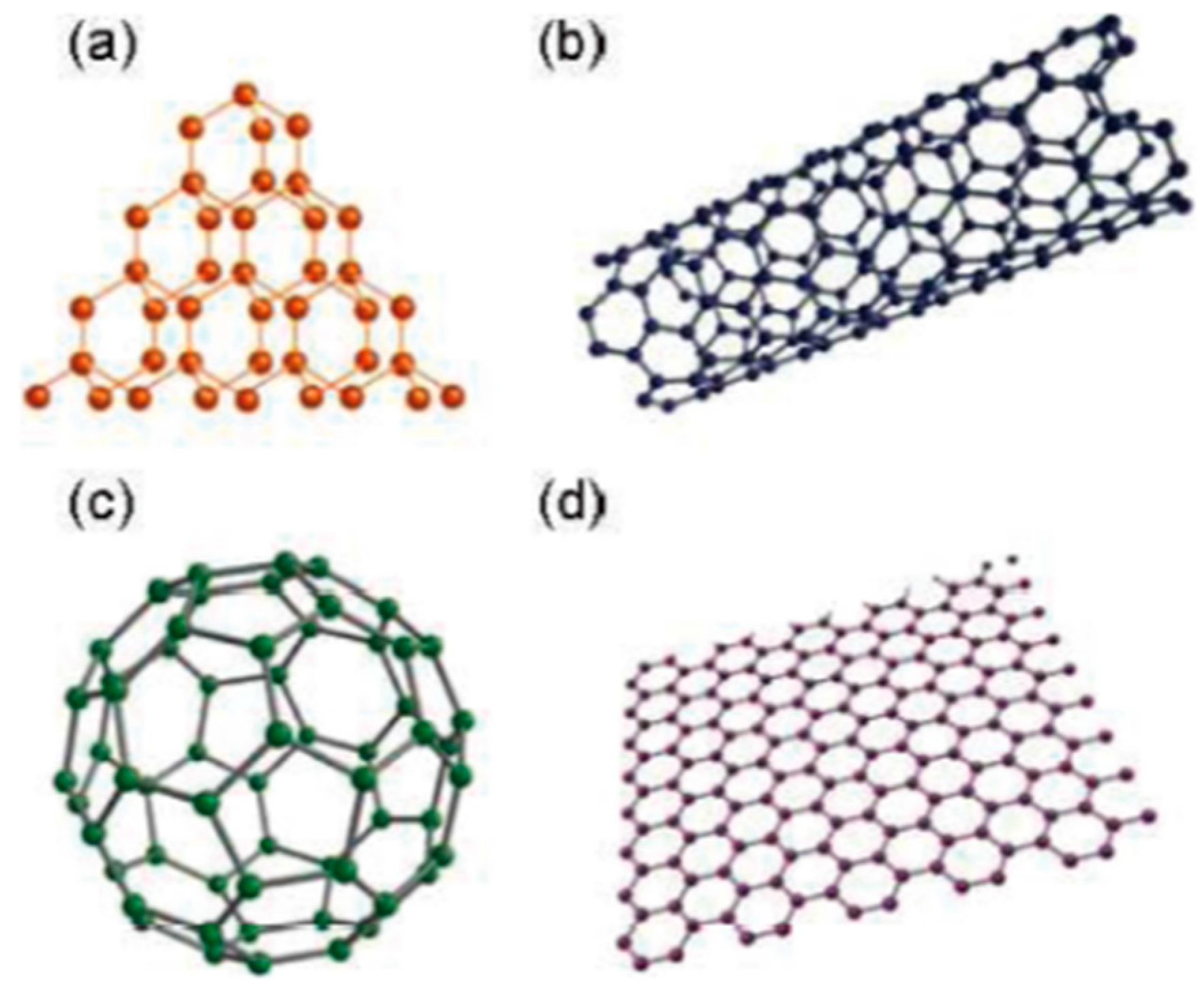
In this context, carbon nanomaterials have attracted great interest because of the structural differences in their various allotropes (e.g., graphite, fullerene, carbon nanotubes (CNT), graphene, diamond, diamond-like carbon (DLC)) (see
Figure 1) and their wide variety of structurally dependent electronic and electrochemical properties [
2]. Carbon nanomaterials cover a broad range of structures: zero-dimensional (fullerenes, diamond clusters), one-dimensional (nanotubes), two-dimensional (graphene), and three-dimensional (nanocrystalline diamond, fullerite) structures [
3]. Carbon nanomaterials such as multiwalled carbon nanotubes (MWCNTs) and fullerene (C
60) offer great versatility in terms of facile modification by functional groups, high carrier capacity incorporating both hydrophilic and hydrophobic substances, biocompatibility, relatively wide potential window, low background current, electrocatalytic capability for a variety of redox reactions, and high chemical stability [
3,
4]. This has led to their wide use in recent years for the preparation of electrochemical (bio)sensors, both as electrode modifiers and nanocarriers.
Due to their special physicochemical characteristics, fullerenes add a new dimension to the construction of highly sensitive electrochemical biosensors. Fullerenes are a very promising family of electroactive compounds with a rich electrochemistry because of the unique dimensional and electronic structure [
5] that makes them very attractive as mediators in electrochemical biosensors, allowing operation at lower potentials, thus reducing the interferences from electroactive compounds [
6].
2. Fullerenes in Electrochemical Biosensing
Fullerene (C
60), as a zero-dimensional carbon, is the most abundant representative of the fullerene family [
7]. C
60 is a truncated icosahedron made out of five and six member rings of sp
2 carbons, which plays an important role in the family of nanocarbons [
8]. C
60 has a broad range of charming properties, such as high electronic conductivity, large specific surface area, good biocompatibility, inert behavior, stable structure and good adsorption capacity towards organic molecules [
9,
10,
11]. Moreover, it is free of metallic impurities and relatively simple to implement and raise to reproducible electrocatalytic responses [
12].
Despite the wide range of interesting characteristics and properties in terms of electrical conductivity and charge transport, pure C
60 has been scarcely used as a possible electrode material. However, this hollow carbon sphere, rich in conjugated π electrons with excellent electron acceptor capability to accelerate electron transfer [
1,
8,
13], can react with amines [
14] and be successfully decorated with other materials [
6,
12,
15,
16,
17]. This explains the increasing interest in the preparation of C
60 composites with other nanomaterials to achieve new physical and chemical properties especially relevant for the application of C
60 in novel electrode materials and the fabrication of sensors and biosensors [
18]. Indeed, the combination of C
60 with nanomaterials such as nitrogen-doped carbon nanotubes (NCNTs) [
18], AuNPs [
1,
6,
15,
19], and Pt–Pd nanoparticles [
17] has been shown to be advantageous either because combining the unique properties of these two kinds of materials or the emergence of new properties leads to novel applications [
12]. Moreover, C
60 owned its inner redox activity [
15], being able to act as an electron acceptor with a dual nature of electrophilic and nucleophilic characteristics, which has led to significant interest from researchers to investigate the possibilities of using this material as a mediator in biosensing devices [
4,
15,
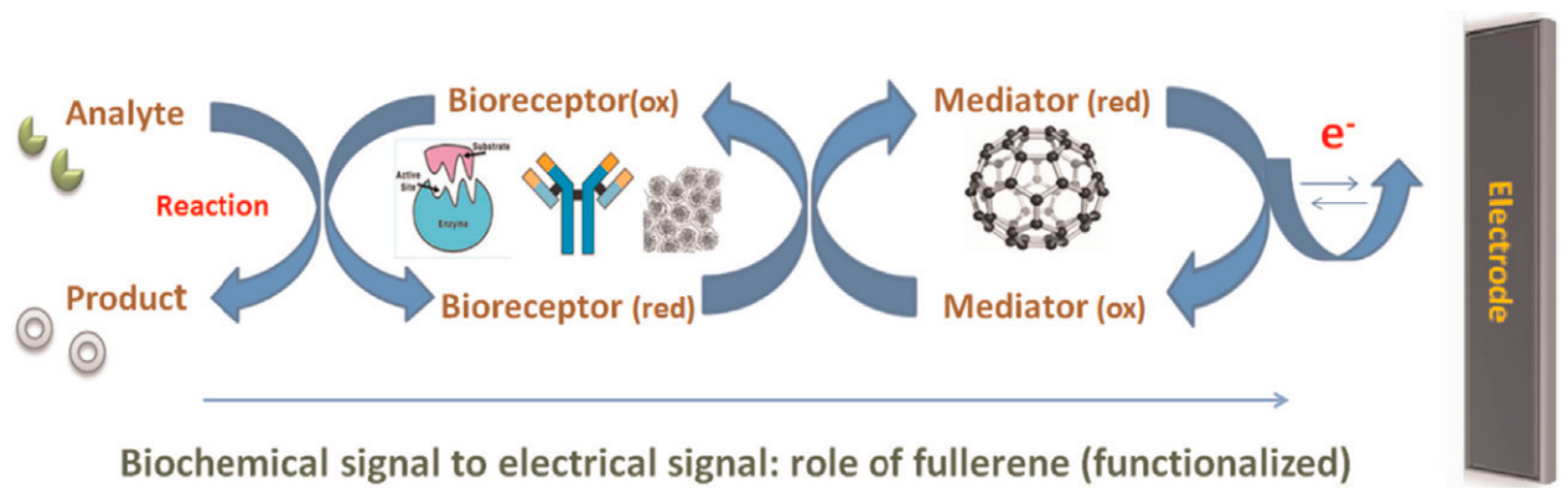
20]. Fullerene is used as a mediator between the recognition site and the electrode in electrochemical biosensors to enhance the rate of electron transfer produced due to the biocatalytic or biochemical reaction of the analyte in contact with the biological element at the recognition site (see
Figure 2) [
21]. Due to the ability of lowering the potential of electro-reduction of many different redox substrates and increasing the reaction rates, fullerenes have been used to improve the sensitivity and selectivity of the resulting biosensors [
8]. Indeed, partially reduced fullerene–C
60-modified electrodes have been demonstrated to behave as excellent working electrodes with a high electroactive surface area, excellent electronic conductivity, electrocatalytic properties and good biocompatibility [
22,
23,
24]. However, it is worth noting here that the enhanced electrocatalytic behavior observed at the reduced C
60-modified electrodes has been a controversial matter addressed by several authors. While some works support that C
60 is involved in effective electron mediation or electrocatalysis [
25], others did not find any experimental evidence of the electrocatalytic ability of C
60 when it was used as electrodes modifier [
26,
27,
28], This “apparent electrocatalytic behavior” was attributed to a re-structuration of the deposited C
60 film, which produces a porous, partially blocked electrode, and possible thin-layer effects [
29,
30].
Moreover, C
60 and their derivatives such as pristine C
60 nanoparticles are aggregated in aqueous medium and their redox activity is inhibited because of their poor solubility in water. This makes the electrochemical behavior of C
60-based nanomaterials in aqueous media unstable and irreversible. This problem can be overcome by C
60 functionalization though the covalent binding of functional groups such as carboxyl, hydroxyl, amine groups, etc. [
4,
7,
19,
21]. For example, hydroxyl fullerenes (HFs) are water soluble fullerene derivatives with a formula of C
60(OH)
n (
n = 18−24). HFs may link with a protein to form specific noncovalent complexes and protect their biological activity. They are also useful for the formation of gels, starburst polymers and composites [
31].
On the other hand, the synthesis of functionalized C
60 or their derivatives through non-covalent interactions involving supramolecular chemistry has shown to preserve the original structure and electrochemical properties of C
60 [
4].
The incorporation of fullerenes for preparing electrochemical biosensors has led to the development of a variety of fullerene functionalization methods, profiting the high surface area-to-volume ratio of these nanomaterials. Amine and carboxylic acid functional groups have been widely incorporated into fullerenes to allow them to easily react with biomolecules [
21]. From the viewpoint of fullerene chemistry, functionalized fullerene can be categorized into two basic types: exohedral, where substituents are intercalated outside the cage, and endohedral, where molecules are trapped inside the cage. It has been shown that exohedrally functionalized fullerenes bearing organic or organometallic functional groups attached to the exterior of the carbon cage are more accessible than endohedral metallofullerenes. Numerous research efforts support the notion that exohedral functionalized fullerene can be a suitable nanomaterial able to improve the sensitivity, selectivity, and reproducibility of electrochemical biosensors [
21]. Different biomolecules or organic ligands can be immobilized onto the shell of fullerenes by adsorption or covalent attachment. Since fullerenes are not harmful to biological material and due to their small size, they can locate the closest distance to the active site of biomolecules and easily accept or donate electrons to the surrounding species [
4]. Electron-releasing molecules such as amines can attack fullerene–C
60 with 60 π electrons. Therefore, biomolecules bearing NH groups are expected to bond chemically to the fullerene C
60 molecule, resulting in the formation of stable immobilized C
60-biomolecule conjugates [
4,
32]. Indeed, the functionalization of fullerene to prepare water-soluble derivatives and conjugates with enzymes (urease, glucose oxidase), proteins (hemoglobin, myoglobin), other biomolecules such as chitosan (CS), metals (e.g., Au), ferrocene (Fc), and so forth, has proved to be successfully implemented for the preparation of biosensors. This improved the sensitivity towards glucose, urea, hemoglobin, immunoglobulin, or glutathione in real samples, as well as identifying doping abuse, to analyze pharmaceutical preparations, and even to detect tumor cells after the inclusion of fullerenes in single or double walled carbon nanotubes [
4,
20,
21].
The next sections discuss briefly the main advantages offered by fullerenes and their derivatives, such as fullerene–C
60 nanoparticles and nanotubes (FNTs) [
24] in electrochemical biosensing. In addition, we overview the main strategies reported for fullerene functionalization and significant and illustrative applications in the preparation of different types of electrochemical biosensors ranging from catalytic to affinity biosensors, when the nanomaterials are used as electrode modifiers, nanocarriers, and redox nanoprobes.
3. Fullerenes in Catalytic Biosensing
The preparation of effective third-generation enzyme biosensors implies the achievement of an efficient electron transport between redox centers of proteins and electrodes. However, it is well known that achieving direct electron transfer (DET) between the redox proteins and the electrode surface is challenging because the electroactive centers of proteins usually have large and complex structures, and the active sites are buried deeply in the molecule structures [
33]. In addition, redox proteins absorbed on the electrode surface are easily irreversibly denatured [
18].
The incorporation of nanomaterials to the biosensor architecture constitutes one of the most successful approaches to overcome these challenges and develop the next generation of biosensors or nanodevices with excellent electrochemical properties [
18,
33]. The electronic, optical, and catalytic properties of many nanomaterials and their dimensions similar to those of several biomolecules provide a suitable microenvironment for proteins immobilization, maintaining their bioactivity, and at the same time facilitating electron transfer between their redox center and electrode surfaces, which have led to their intensive use for the construction of electrochemical biosensors with enhanced analytical performance. The generated hybrid systems combine the unique electronic properties of nanomaterials with the recognition and catalytic function of biomolecules, thus allowing electrically wiring redox enzymes with electrodes and reducing electron transfer distances [
6].
In the case of fullerenes, it has been shown they also provide a suitable microenvironment for the protein immobilization [
4]. Additionally, they are small enough to locate at the closest distance of the active site of the enzyme and induce the protein molecule mobility to correctly orient the redox centers to achieve a proper electron transfer. Furthermore, as commented above, they are not harmful to biological material and proteins, and can interact with enzymes through several types of immobilization strategies (entrapment, encapsulation, covalent binding, cross-linking, and adsorption) involving covalent or non-covalent bonds [
4].
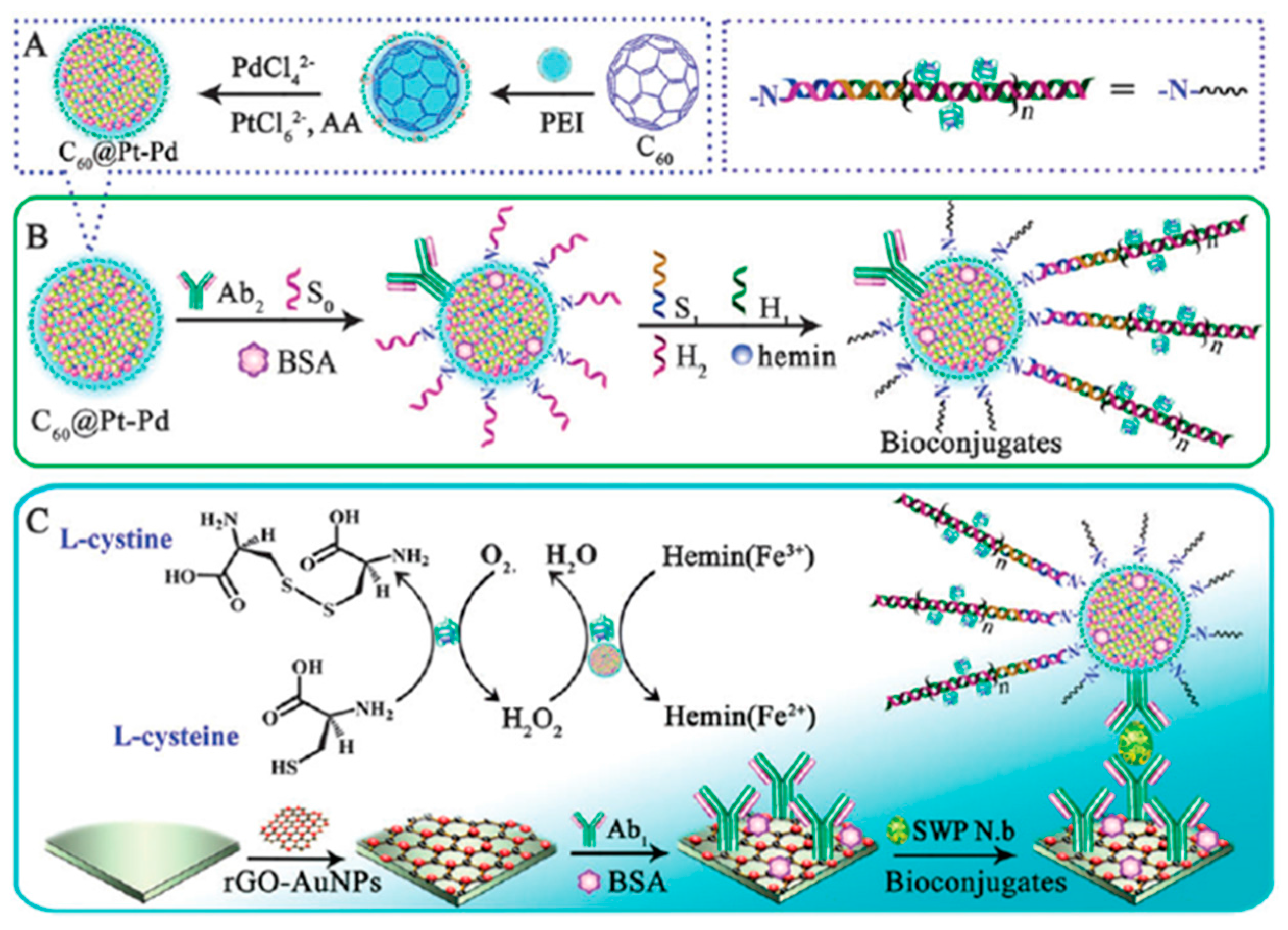
Csiszár et al. demonstrated that the electrochemistry of immobilized cyt c depended strongly on the oxidation state and porosity of the fullerene films [
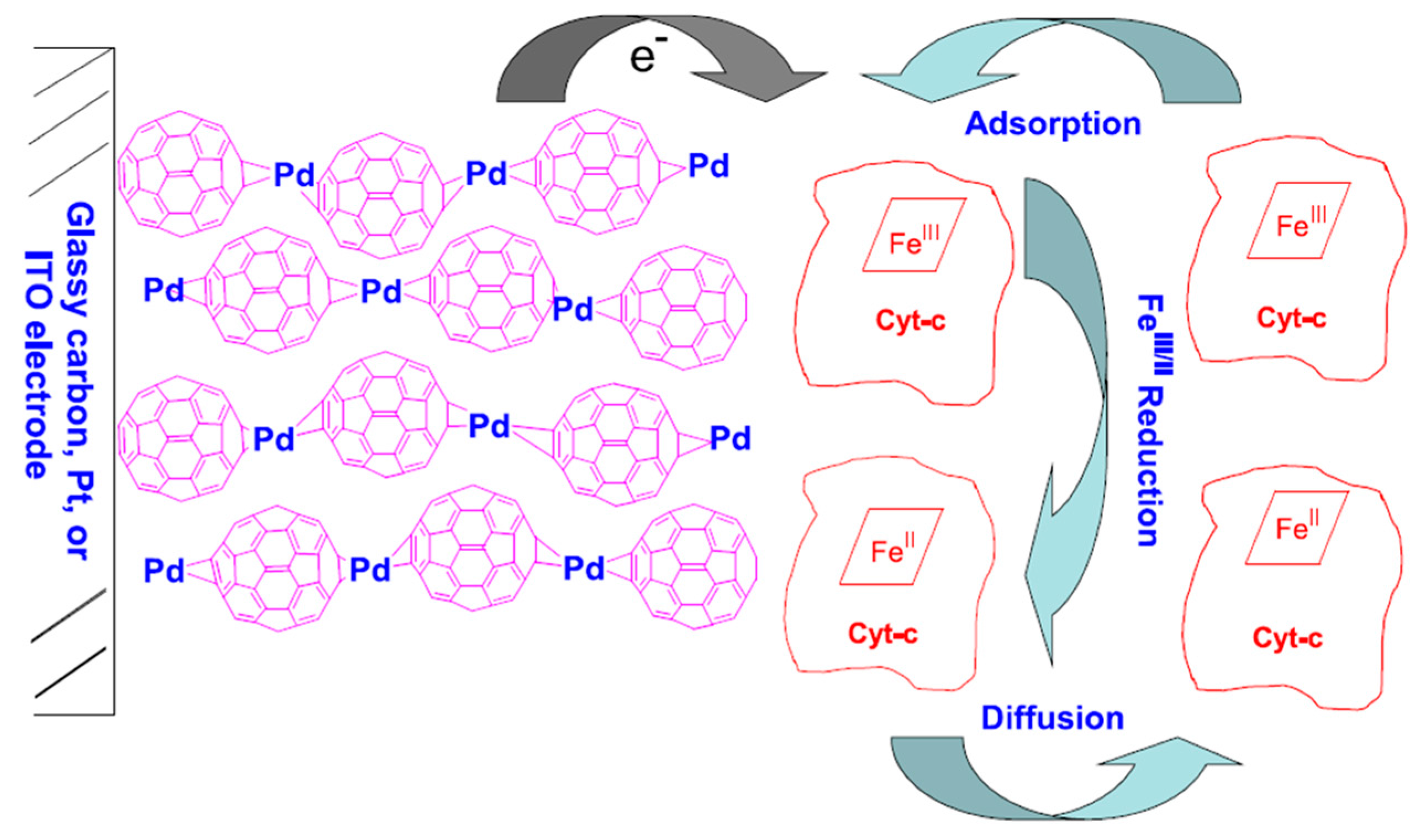
34]. It was shown that better redox reaction was accomplished on partially reduced fullerene film, which acted as a promoter, independently of the underlying electrode substrate (gold or glassy carbon). Partially reduced fullerene possesses a structure with a polar or negatively charged outside and an apolar inside which resembles biomembranes, and raised the possibility of using fullerenes as solid state modifiers to enhance the electrochemistry and stability of immobilized biomolecules compared to bare electrodes. D’Souza et al. [
35] reported the stable immobilization of cyt c on two types of fullerene film modified electrodes: one involving an electrochemically-conditioned fullerene drop-coated film and, another, an electro-polymerized fullerene-cross-linked with palladium acetate complex (C
60–Pd) film (see
Figure 3). More effective cyt c immobilization and DET was observed at the C
60–Pd polymer film-modified electrodes. Immobilization of cyt c onto the C
60–Pd polymer film-modified electrode was claimed to proceed via one or more of the following routes: (i) the electron-deficient C
60 molecules may allow the electrostatically binding to the cyt c molecules through their electron-rich aspartic-glutamic acid residue sites; (ii) the negative charges stored in the C
60 film may strongly adsorb electrostatically, via ion pairing, positively charged parts of the cyt c protein; (iii) the cyt c molecules may be trapped in the porous structure of the C
60 or C
60–Pd films.
Zhilei et al. [
13] developed an interesting third-generation glucose biosensor by the subsequent deposition of C
60, ferrocene (Fc), chitosan (CS) ionic liquid (IL) and glucose oxidase (GOx) onto a glassy carbon electrode (GCE). By using chronoamperometry at +100 mV (vs. Ag/AgCl) the GOx/C
60–Fc–CS–IL biosensor exhibited DET, a wide calibration range (1 × 10
−8–1 × 10
−5 M), an excellent limit of detection (LOD) of 3 × 10
−9 M and long-term stability of 30 weeks due to the favorable microenvironment provided by the CS–IL network. This biosensor demonstrated accurate determination of glucose in blood serum samples. The excellent performance of this biosensor was attributed both to the electrocatalytic activity of C
60 and Fc, which accelerate the electrochemical reaction, and to the network of CS–IL, which provided a favorable microenvironment to keep the enzyme bioactivity, and the electron conduction pathways for GOx through Fc and C
60.
Another glucose electrochemical biosensor was reported by Lin et al., involving the synthesis of a fullerene C
60–glucose oxidase conjugate (C
60–GOx) and its electrodeposition on a GCE previously modified with a mixed-valence cluster of cobalt(II) hexacyanoferrate [
32]. The C
60–GOx/cobalt(II) hexacyanoferrate-modified electrode exhibited efficient electrocatalytic activity toward the liberated H
2O
2 and allowed cathodic detection of glucose at 0.0 mV (vs. Ag/AgCl reference electrode). This biosensor showed a long life (≥100 days), could be repeatedly reused for a long time, showed good selectivity, linear response up to 8 mM glucose, and a LOD of 1.6 × 10
−6 M. Interestingly in this biosensor the presence of ascorbic acid almost did not affect the response obtained for glucose.
Sheng et al. [
18] achieved direct electrochemistry of hemoglobin (Hb) immobilized by a fullerene–C
60–NCNTs/chitosan composite matrix. The C
60 filled within NCNT could electronically dope the NCNT, then acting as electron mediator to facilitate the electrochemical reaction of the protein and achieving the fast electron transfer between Hb and the electrode surface. It was suggested that the C
60–NCNTs/CHIT composite film may mimic some physiological process. Moreover, this electrochemical platform catalyzed the reduction of H
2O
2, kept 95% of the initial sensitivity after 2 months of storage, and provided a wide linearity range (2.0–225.0 μM) and a low LOD (1 μM). This approach can be easily extended to other redox enzymes or proteins and find application for the design of biofuel cells and for the determination of H
2O
2 in biological and pharmaceutical samples.
Saeedfar et al. [
9] developed a potentiometric urea biosensor based on the use of a C
60–urease bioconjugate prepared by covalent immobilization of urease onto COOH-modified C
60 deposited on a screen-printed electrode (SPE) coated with a good adhesion acrylic membrane entrapping a hydrogen ionophore. The biosensor exhibited a linear response from 2.31 × 10
−3 to 8.28 × 10
−5 M urea and a stability of up to 140 days. This great stability was attributed to the prevented leaching of the enzyme by the acrylic membrane.
Gao et al. [
31] developed a sensitive and selective glucose biosensor showing the direct electrochemistry of GOx by immobilizing GOx–HFs nano-complexes on a GCE protected with a chitosan (Chit) membrane. Amperometric detection at −0.35 V (vs. Ag/AgCl) provided a response for glucose ranging from 0.05 to 30 mM (with 0.05–1.0 mM and 3–10 mM) and provided a LOD of 5 ± 1 μM. The presented results demonstrated that Chit/HFs preserved the conformational structure of the enzyme providing the resultant biosensor with high sensitivity and selectivity.
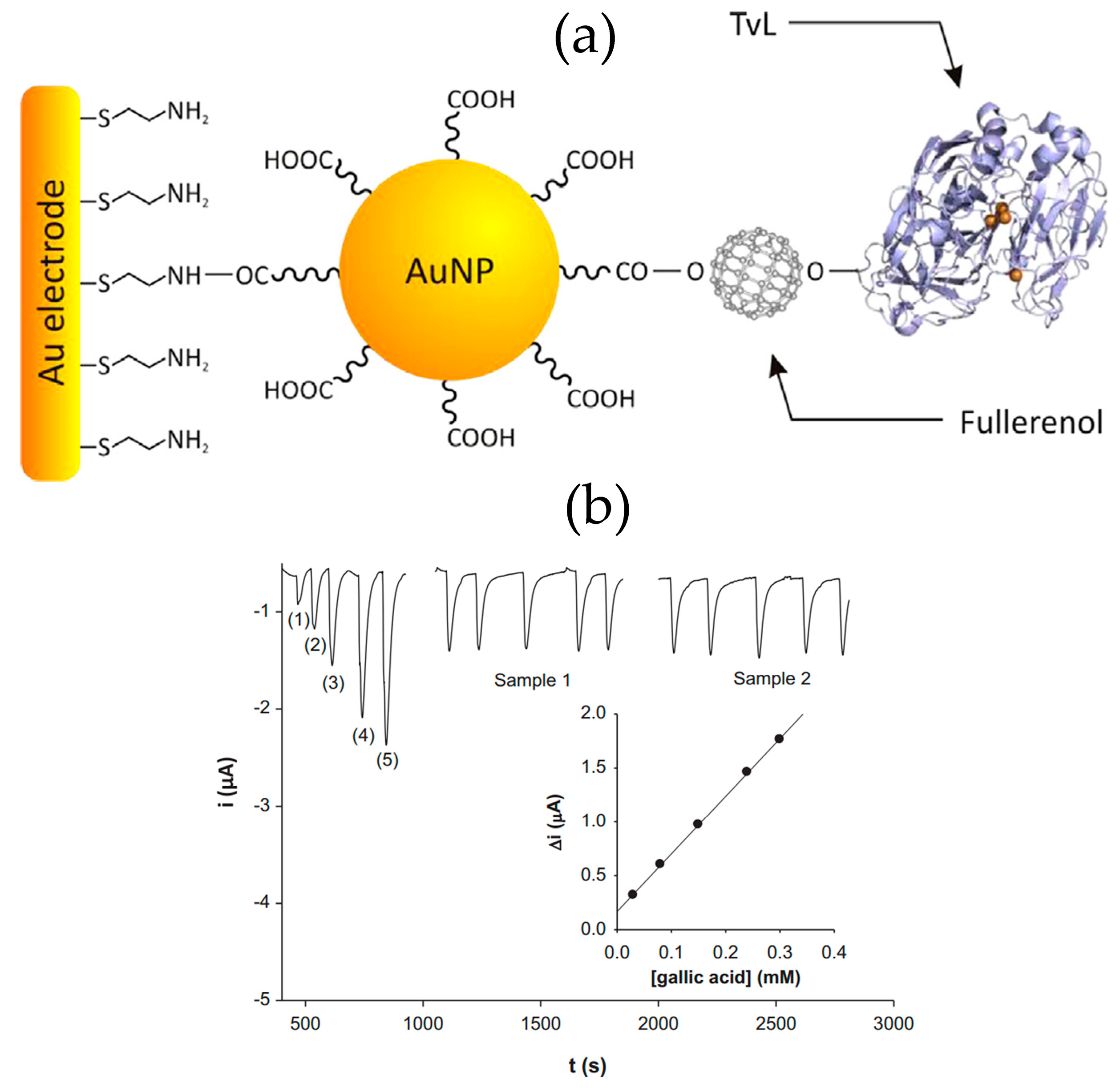
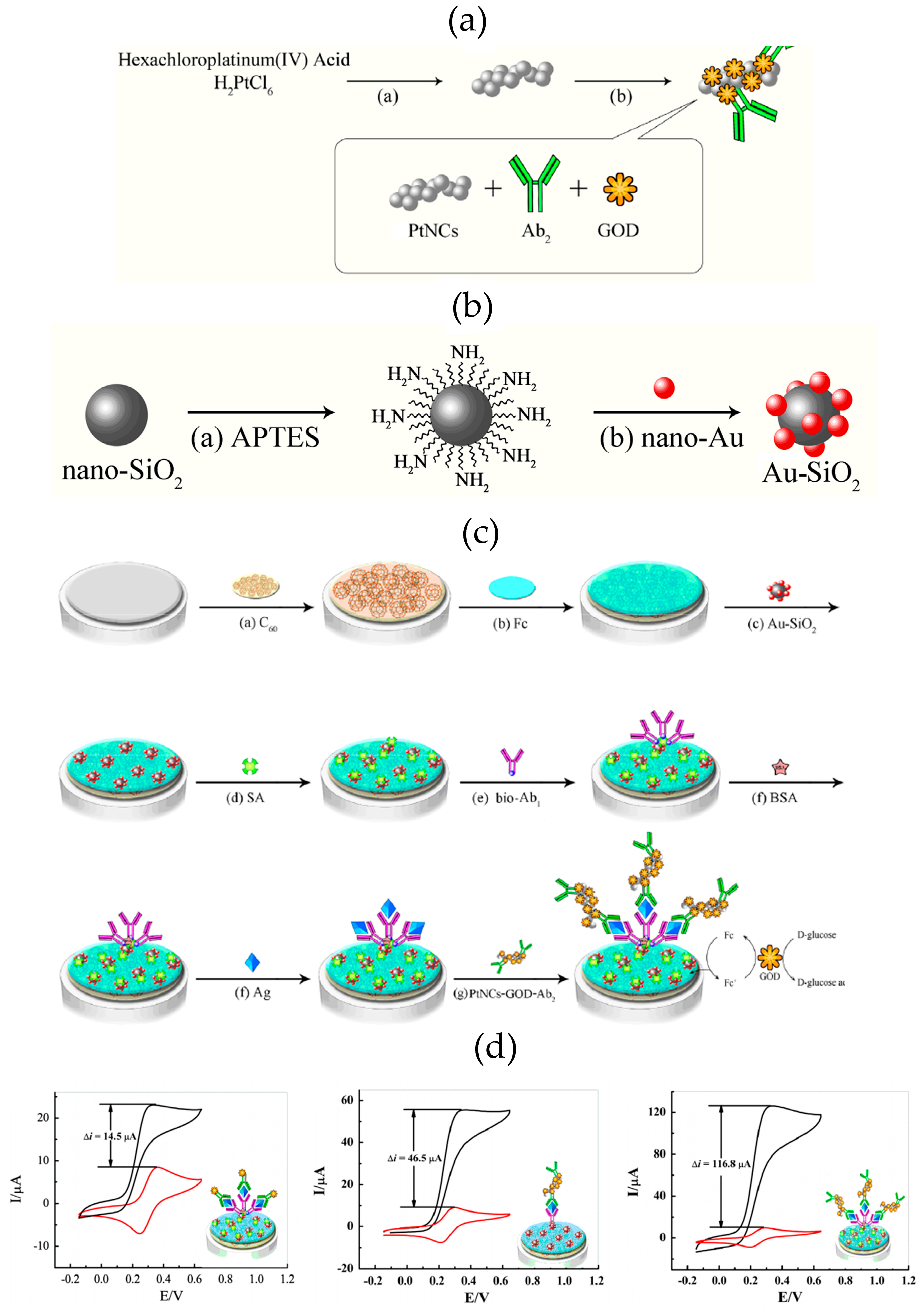
A nanostructured laccase biosensor was developed by Lanzellotto et al. [
6] by exploiting the beneficial features of poly hydroxy-fullerenes (fullerenols) and AuNPs (see
Figure 4a). The biosensor construction involved the immobilization of functionalized AuNPs on an Au/SPE modified with a cysteamine self-assembled monolayer. Subsequently, fullerenols were linked onto the AuNP-modified electrode and the
Trametes versicolor laccase enzyme (TvL) was immobilized. Using gallic acid as standard, and chronoamperometric detection (E
app = −0.100 V vs. Ag/AgCl), the biosensor exhibited DET, a linear range from 0.03 to 0.30 mM, an LOD of 0.006 mM, and provided reliable results in the analysis of commercial wines (
Figure 4b). Recently Tortolini et al. [
33] used this laccase biosensor as an amperometric detector in a flow injection analysis method for the determination of polyphenols in wine samples. These biosensing platforms, based on the coupling of two different nanostructured materials (gold nanoparticles and fullerenols), displayed interesting electrochemical features in terms of good stability and reproducibility, an increased amount of electroactive protein immobilized and enhanced electron transfer between the redox center of the protein and the electrode surface.
Barberis et al. performed the simultaneous amperometric detection of ascorbic acid (AA) and antioxidant capacity through the measurement of phenolic compounds in fruit juices using fullerenes–C
60/C
70 or nanotube-modified graphite sensor-biosensor systems, and ascorbate oxidase [
10]. Results showed that the combination of fullerene and ascorbate oxidase resulted in the complete AA shielding and in the highest selecting capacity toward AA, while nanotubes only increased sensitivity without the ability to discriminate between the AA and the phenols contribution to the antioxidant capacity. These results led authors to hypothesize that fullerenes absorb more enzyme during dips, and can therefore oxidize more AA before it reaches the transducer surface. Although these biosensors systems were much cheaper compared with traditional methods, they are limited by the fragility of graphite rods and the laborious preparation protocol.
5. General Conclusions, Main Challenges and Future Directions
Nanostructured materials have been significantly used in recent years to create a new generation of electrochemical biosensors with improved analytical performance, miniaturization ability, and reduced response time, which are features of great interest in fields such as clinical diagnosis. Among different nanomaterials, carbon nanomaterials and particularly fullerene–C60 and some of their derivatives (FC60NPs and FNTs) have been shown to hold potential promise for this purpose due to their unique electrochemical and physicochemical properties, together with biocompatibility characteristics.
This article gives an overview of the key aspects of the electrochemical biosensors reported so far involving the use of fullerenes and derivatives as electrode modifiers, nanocarriers and redox nanoprobes, and in connection to various kinds of biomolecules (enzymes, antibodies and oligonucleotides). The prospects of effectively modifying fullerene-based nanomaterials to ramp up the sensitivity and functionality of the resulting biodevices are also discussed together with challenges to be faced and future directions.
Results reported in this field demonstrate that electrodes modified with partially reduced fullerene–C
60 show exceptional properties such as large electroactive surface area able to immobilize large amounts of bioreceptors, the promotion of the biomolecules electrochemistry, excellent electronic conductivity, and good biocompatibility [
4]. From the discussed works, it is deduced that fullerene–C
60 and derivatives not only provide a suitable immobilization platform for DNA and antibodies, but have also the ability to induce a proper orientation in redox-active proteins, allowing for the direct electron transfer of enzymes and other proteins such as cyt c [
35], GOx [
13,
31], Hb [
18] and laccase [
6,
33]. Fullerene nanomaterials can act also as efficient nanocarriers due to the high surface area and good biocompatibility in the preparation of electrochemical biosensors with enhanced sensitivity [
17]. Moreover, these materials have been used as redox nanoprobes to prepare electrochemical affinity biosensors through the synthesis of hydrophilic C
60-based nanomaterials and exploiting the inner redox activity of C
60 [
15]. While C
60 and derivatives have been used as electrode modifiers in catalytic biosensors, in electrochemical affinity biosensors they have been employed as electrode modifiers [
1,
5,
6,
9,
10,
11,
12,
13,
18,
24,
31,
32,
33,
34,
35,
36,
37,
38], nanocarriers [
15,
17,
19] and redox nanoprobes [
15]. Although most of the work has been done in developing catalytic biosensors and immunosensors, there is no doubt that fullerenes can be used as an active material in biosensing devices for the determination of biomolecules of a genetic nature. Indeed, the illustrative examples selected clearly show that electrochemical biosensors involving fullerene nanomaterials can be designed for the determination of a wide variety of target analytes such as glucose, H
2O
2, poplyphenols and AA, neurotransmitters such as DA, epinenephrine and norepinephrine, mycotoxins (DON), proteins (TNF-α, HSP70, PBGF-BB, EPO and SWP N.b), bacteria (
E. coli) and oligonucleotides (dsDNA).
However, despite the great progress that has occurred in recent years and the bright prospects for this field, the function of fullerenes is still under investigation. In fact, in order to exploit their potentiality to the fullest, it is essential to discover promising advantages of their use in the construction of biosensing devices other than enhancing the electron transfer rate. Another major issue that requires attention is the examination of how conveniently this nanomaterial can be incorporated into a biosensor and how robustly it can deliver its performance under different experimental conditions to detect and measure different types of biomolecules, in comparison to biosensors prepared so far with other nanomaterials for similar purposes. Although the lack of systematic comparative studies of fullerene- and other nanomaterials-based electrochemical biosensing devices does not allow us to discuss this aspect exhaustively, in general the popularity C
60 has somewhat diminished in recent years with the rise of CNTs and graphene, more scalable and practical carbon nanomaterials. However, the uniform size and shape as well as its availability for chemical modification makes C
60 widely used at this time for other types of applications such as therapeutic purposes taking advantage of its anti-human immunodeficiency virus (HIV) activity by targeting important HIV enzymes [
40].
Although biosensors constructed with fullerene offer high sensitivity, selectivity and good reproducibility for electrochemical biosensing, the cost of this nanomaterial and of the agents and tools required for its functionalization is an important limitation to be considered for their application in point-of-care (POC) diagnosis where cost-effective and easy-to-handle configuration are demanded. However, it is expected that the advantages already demonstrated and those that may arise in future research will compensate for these limitations. Fullerene-based biosensors may achieve the wide acceptance of other nanomaterials-based biosensors and play an important role also in POC diagnosis. A mandatory prior step to this stage is to demonstrate the robustness and convenient behavior of the fullerene-involved biosensors in challenging biological samples and not just in buffered solutions and doped clinical samples, as occurred in most of the reported works. Another major challenge requiring proper attention is to explore the possibilities for multiplexing. It is striking that given the current growing interest in performing multidetermination, to date there is not any electrochemical biosensor using fullerenes used for the determination of more than one analyte. Moreover, the range of applications is still limited, and further investigation in new functionalization routes, together with the exploration of other fullerene–C60 analogues for the construction of electrochemical biosensors, is envisaged to expand the range of applications as well as to improve their performance in terms of sensitivity and stability. On the other hand, due to the biocompatibility of fullerene–C60 and derivatives, additional works should be also focused on functionalization with other types of bioreceptors such as microorganisms, cells or their organelles. The integration of the fullerene-involved biosensors in automated and miniaturized systems to achieve electrochemical POC systems is another key challenge.
In summary, despite the further efforts required to address these important issues, the rapid development and attention in the research of functionalizing fullerene and cutting-edge biosensor technology over the past few years emphasizes the possibility that, in the near future, fullerene-based biosensors will appear as effective devices in electrochemical biosensing and be applicable commercially for POC diagnosis.