14N NMR Spectroscopy Study of Binding Interaction between Sodium Azide and Hydrated Fullerene
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental
C60HyFn production, Characterization and Preparation of C60FWS
2.2. Titration
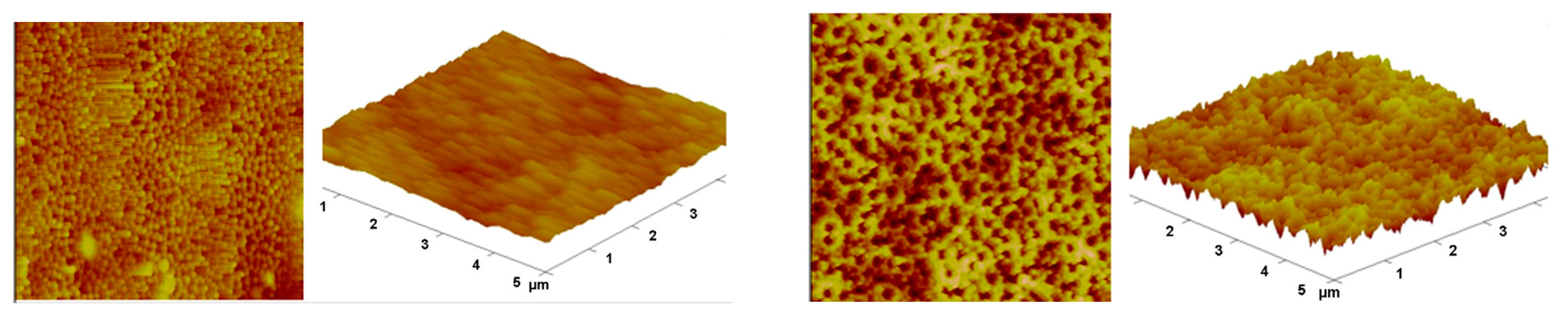
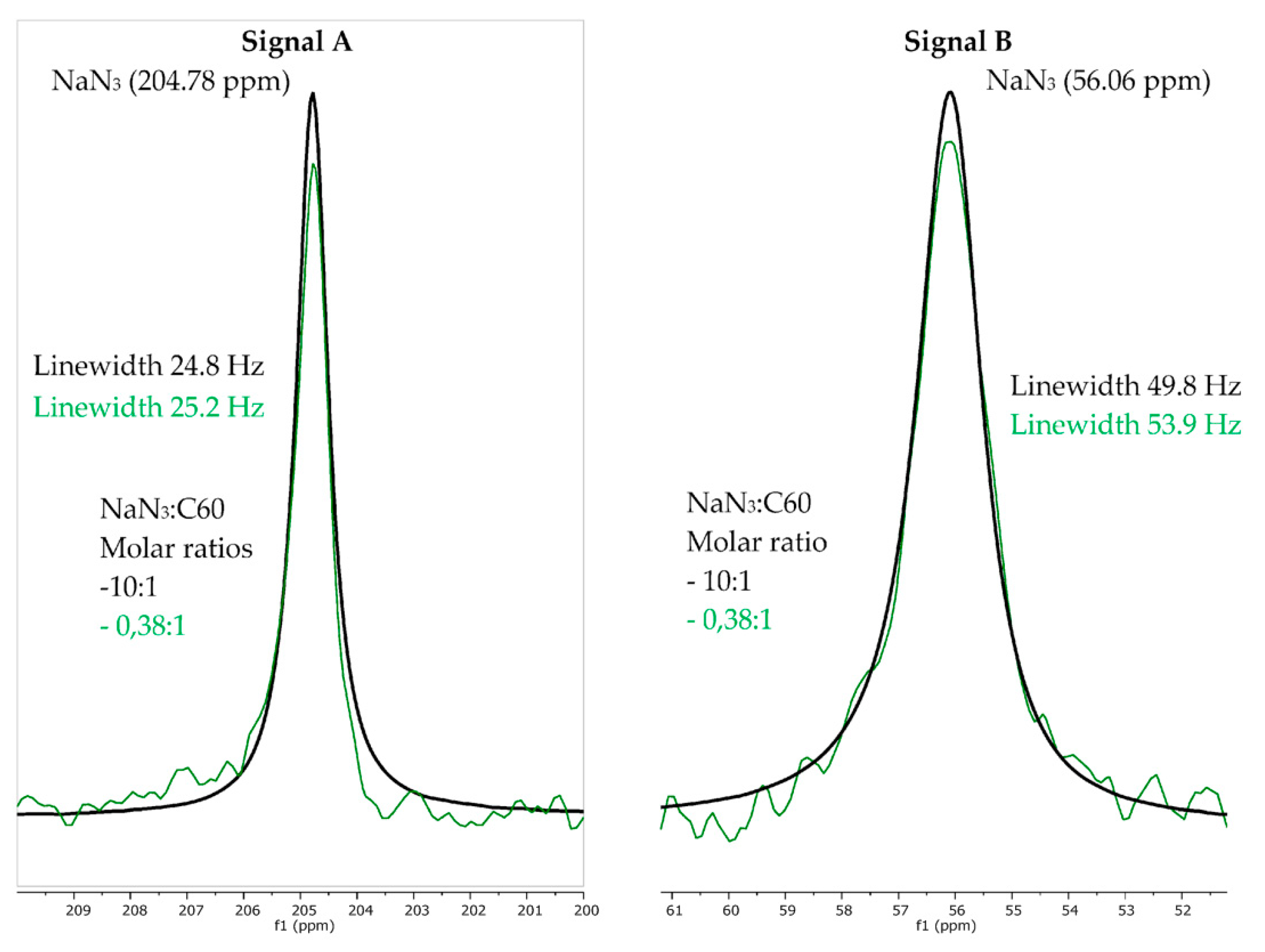
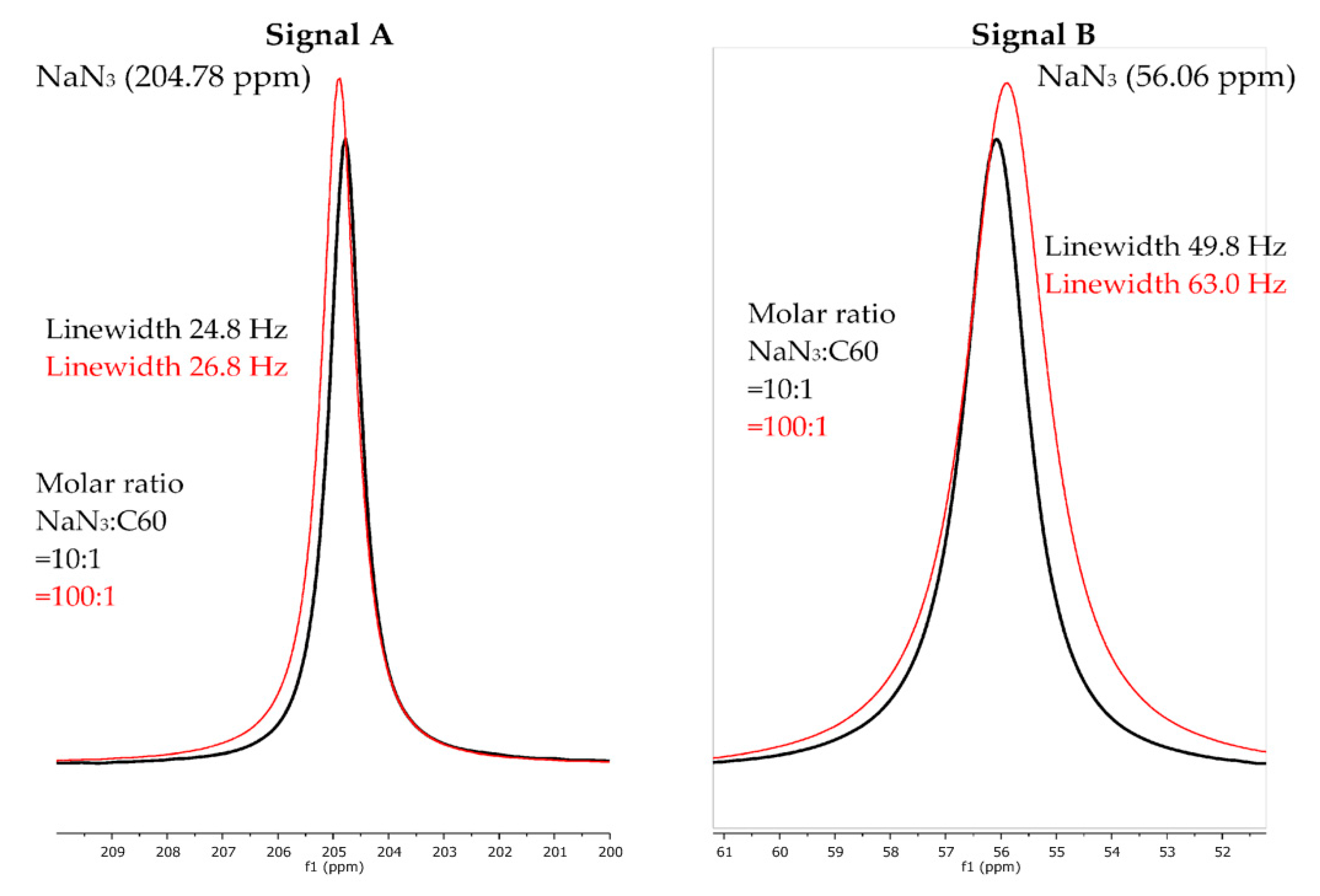
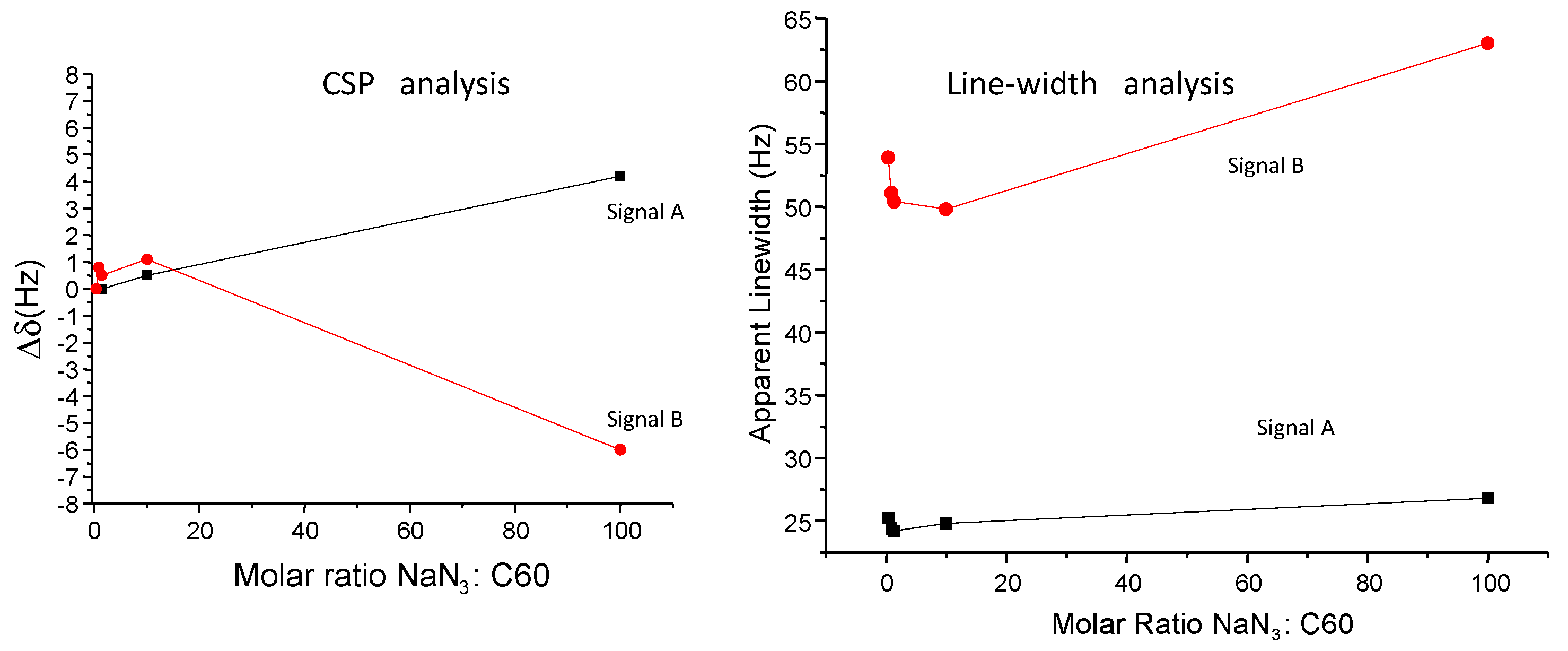
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Petrović, M.; Gonzalez, S.; Barceló, D. Analysis and removal of emerging contaminants in wastewater and drinking water. TrAC Trends Anal. Chem. 2003, 22, 685–696. [Google Scholar] [CrossRef]
- Focazio, M.J.; Kolpin, D.W.; Barnes, K.K.; Furlong, E.T.; Meyer, M.T.; Zaugg, S.D.; Thurman, M.E. A national reconnaissance for pharmaceuticals and other organic wastewater contaminants in the United States—II) Untreated drinking water sources. Sci. Total Environ. 2008, 402, 201–216. [Google Scholar] [CrossRef] [PubMed]
- Buser, H.R.; Poiger, T.; Müller, M.D. Occurrence and environmental behavior of the chiral pharmaceutical drug ibuprofen in surface waters and in wastewater. Environ. Sci. Technol. 1999, 33, 2529–2535. [Google Scholar] [CrossRef]
- Heberer, T. Occurrence, fate, and removal of pharmaceutical residues in the aquatic environment: A review of recent research data. Toxicol. Lett. 2002, 131, 5–17. [Google Scholar] [CrossRef]
- Ternes, T.A. Occurrence of drugs in German sewage treatment plants and rivers. Water Res. 1998, 32, 3245–3260. [Google Scholar] [CrossRef]
- Metcalfe, C.D.; Koenig, B.G.; Bennie, D.T.; Servos, M.; Ternes, T.A.; Hirsch, R. Occurrence of neutral and acidic drugs in the effluents of Canadian sewage treatment plants. Environ. Toxicol. Chem. 2003, 22, 2872–2880. [Google Scholar] [CrossRef] [PubMed]
- Castiglioni, S.; Bagnati, R.; Fanelli, R.; Pomati, F.; Calamari, D.; Zuccato, E. Removal of pharmaceuticals in sewage treatment plants in Italy. Environ. Sci. Technol. 2006, 40, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Joss, A.; Keller, E.; Alder, A.C.; Göbel, A.; McArdell, C.S.; Ternes, T.; Siegrist, H. Removal of pharmaceuticals and fragrances in biological wastewater treatment. Water Res. 2005, 39, 3139–3152. [Google Scholar] [CrossRef] [PubMed]
- Giger, W.; Alder, A.C.; Golet, E.M.; Kohler, H.P.E.; McArdell, C.S.; Molnar, E.; Suter, M.J.F. Occurrence and fate of antibiotics as trace contaminants in wastewaters, sewage sludges, and surface waters. CHIMIA Int. J. Chem. 2003, 57, 485–491. [Google Scholar] [CrossRef]
- McKeen, S. (Ed.) High Production Volume (HPV) Chemicals. In The Organization for Economic Co-Operation and Development; Environment Directorate: Paris, France, 2010. [Google Scholar]
- Rodríguez-Kábana, R.; Robertson, D.G. Nematicidal and herbicidal properties of potassium azide. Nematropica 2000, 30, 146–147. [Google Scholar]
- Wells, S. Granular Forms of Sodium and Potassium Azide as a Nematicide for Established Turfgrasses. Ph.D. Thesis, Auburn University, Auburn, AL, USA, 2009. [Google Scholar]
- Betterton, E.A. Environmental fate of sodium azide derived from automobile airbags. Crit. Rev. Environ. Sci. Technol. 2003, 33, 423–458. [Google Scholar] [CrossRef]
- Haase, J.; Banert, K. Large-scale preparation and usage of azides. In Organic Azides: Syntheses and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Use of Dendrimers to Enhance Selective Separation of Nanofiltration and Reverse Osmosis Membranes. Available online: https://www.usbr.gov/research/dwpr/reportpdfs/report140.pdf (accessed on 1 February 2017).
- Velizarov, S.; Crespo, J.G.; Reis, M.A. Removal of inorganic anions from drinking water supplies by membrane bio/processes. Rev. Environ. Sci. Bio/Technol. 2004, 3, 361–380. [Google Scholar] [CrossRef]
- Park, H.; Chang, I.; Lee, K. Membranes and Module Modification. In Principles of Membrane Bioreactors for Wastewater Treatment; CRC press Taylor & Francis Group: New York, NY, USA, 2015; pp. 281–282. [Google Scholar]
- Liu, Z.H.; Ogejo, J.A.; Pruden, A.; Knowlton, K.F. Occurrence, fate and removal of synthetic oral contraceptives (SOCs) in the natural environment: A review. Sci. Total Environ. 2011, 409, 5149–5161. [Google Scholar] [CrossRef] [PubMed]
- Jassby, D.; Chae, S.R.; Hendren, Z.; Wiesner, M. Membrane filtration of fullerene nanoparticle suspensions: Effects of derivatization, pressure, electrolyte species and concentration. J. Colloid Interface Sci. 2010, 346, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Chae, S.R.; Therezien, M.; Budarz, J.F.; Wessel, L.; Lin, S.; Xiao, Y.; Wiesner, M.R. Comparison of the photosensitivity and bacterial toxicity of spherical and tubular fullerenes of variable aggregate size. J. Nanopart. Res. 2011, 13, 5121–5127. [Google Scholar] [CrossRef]
- Jassby, D.; Chae, S.R.; Hendren, Z.; Wiesner, M.R. Membrane filtration of fullerene nanoparticle suspensions: Effects of derivatization, pressure and electrolyte concentration. J. Colloid Interface Sci. 2010, 346, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Chae, S.R.; Hotze, E.M.; Wiesner, M.R. Possible Applications of Fullerene Nanomaterials in Water Treatment and Reuse. In Nanotechnology Applications for Clean Water; William Andrew Publishing: Norwich, NY, USA, 2008. [Google Scholar]
- Chae, S.R.; Wang, S.; Hendren, Z.D.; Wiesner, M.R.; Watanabe, Y.; Gunsch, C.K. Effects of fullerene nanoparticles on Escherichia coli K12 respiratory activity in aqueous suspension and potential use for membrane biofouling control. J. Membr. Sci. 2009, 329, 68–74. [Google Scholar] [CrossRef]
- Jones, O.A.H.; Voulvoulis, N.; Lester, J.N. Human pharmaceuticals in wastewater treatment processes. Crit. Rev. Environ. Sci. Technol. 2005, 35, 401–427. [Google Scholar] [CrossRef]
- Buseck, P.S.; Tsipursky, R.; Hettich, R. Fullerenes from the geological environment. Science 1992, 257, 215–217. [Google Scholar] [CrossRef] [PubMed]
- Krasnovyd, S.V.; Konchits, A.A.; Shanina, B.D.; Valakh, M.Y.; Yanchuk, I.B.; Yukhymchuk, V.O.; Skoryk, M.A. Local structure and paramagnetic properties of the nanostructured carbonaceous material shungite. Nanoscale Res. Lett. 2015, 10, 78. [Google Scholar] [CrossRef] [PubMed]
- Buseck, P.R. Geological fullerenes: Review and analysis. Earth Planet. Sci. Lett. 2002, 203, 781–792. [Google Scholar] [CrossRef]
- Augustyniak-Jabłokow, M.A.; Yablokov, Y.V.; Andrzejewski, B.; Kempiński, W.; Łoś, S.; Tadyszak, K.; Zhikharev, V.A. EPR and magnetism of the nanostructured natural carbonaceous material shungite. Phys. Chem. Miner. 2010, 37, 237–247. [Google Scholar] [CrossRef]
- Andrievsky, G.V.; Kosevich, M.V.; Vovk, M.; Shelkovsky, V.S.; Vashchenko, L.A. On the production of an aqueous colloidal solution of fullerenes. J. Chem. Soc. Chem. Commun. 1995, 12, 1281–1282. [Google Scholar] [CrossRef]
- Chachibaia, T.; Pastor, M. The State-Of-The-Art chemical analytical method for detection of sodium azide by 14N NMR spectroscopy. J. Nano Stud. 2015, 11, 8–15. [Google Scholar]
- Krebs, F. Determination of the Biological Harmful Effects of Substances Endangering Water Quality with the Assimilation-Depletion Test (A-D Test). Deutsche Gewässerkundliche Mitteilungen 1991, 35, 161–170. [Google Scholar]
- O’Neill, M.J. (Ed.) Sodium Azide, The Merck Index, an Encyclopedia of Hemicals, Drugs, and Biologicals, 14th ed.; RSC Publishing: Cambridge, UK, 2006; pp. 1478–1479. [Google Scholar]
- Akhmetov, A.R.; Tuktarov, A.R.; Dzhemilev, U.M.; Yarullin, I.R.; Gabidullina, L.A. First example of the interaction of fullerene C60 with hydrazoic acid. Rus. Chem. Bull. 2011, 60, 1885–1887. [Google Scholar] [CrossRef]
- Osuna Oliveras, S. Theoretical Studies of the Exohedral Reactivity of Fullerene Compounds. Ph.D. Thesis, Universitat de Girona, Girona, Spain, 2010. [Google Scholar]



© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chachibaia, T.; Martin Pastor, M. 14N NMR Spectroscopy Study of Binding Interaction between Sodium Azide and Hydrated Fullerene. C 2017, 3, 13. https://doi.org/10.3390/c3020013
Chachibaia T, Martin Pastor M. 14N NMR Spectroscopy Study of Binding Interaction between Sodium Azide and Hydrated Fullerene. C. 2017; 3(2):13. https://doi.org/10.3390/c3020013
Chicago/Turabian StyleChachibaia, Tamar, and Manuel Martin Pastor. 2017. "14N NMR Spectroscopy Study of Binding Interaction between Sodium Azide and Hydrated Fullerene" C 3, no. 2: 13. https://doi.org/10.3390/c3020013





