The Mediatorless Electroanalytical Sensing of Sulfide Utilizing Unmodified Graphitic Electrode Materials
Abstract
:1. Introduction
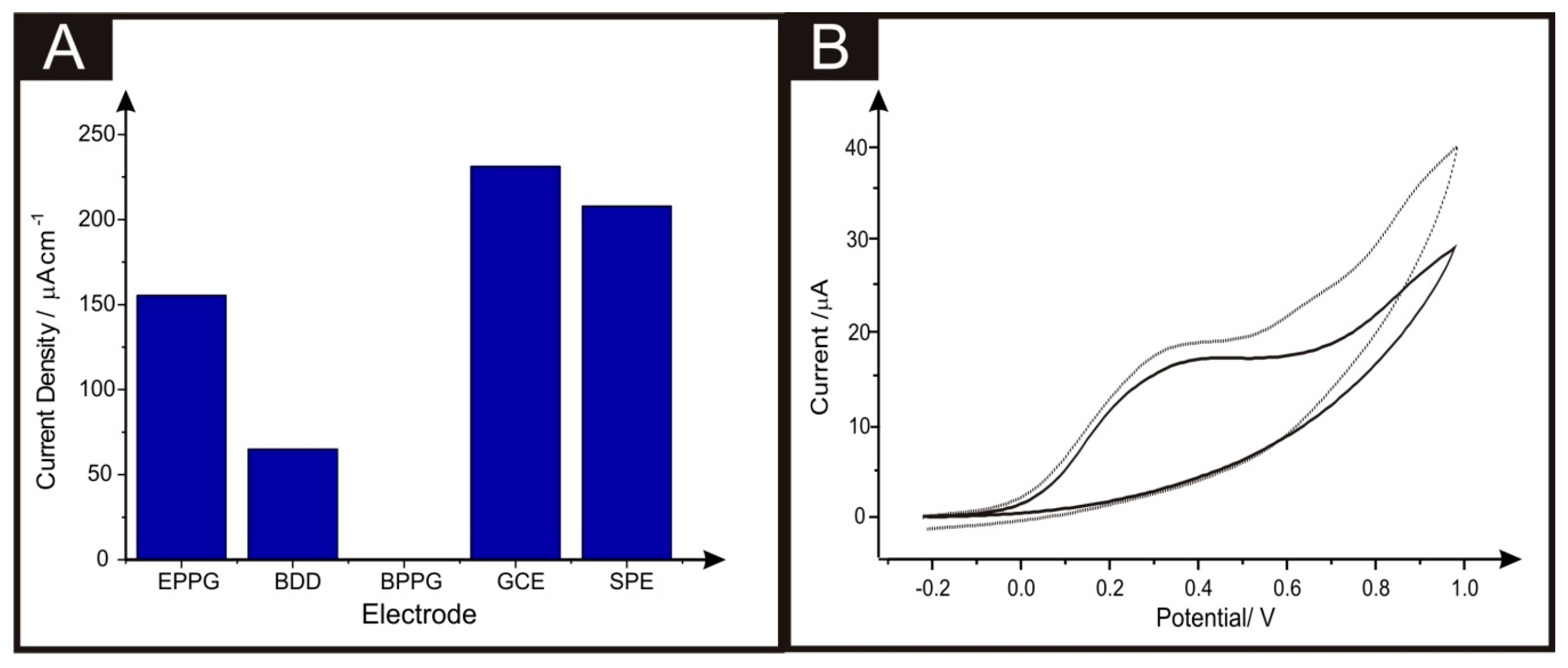
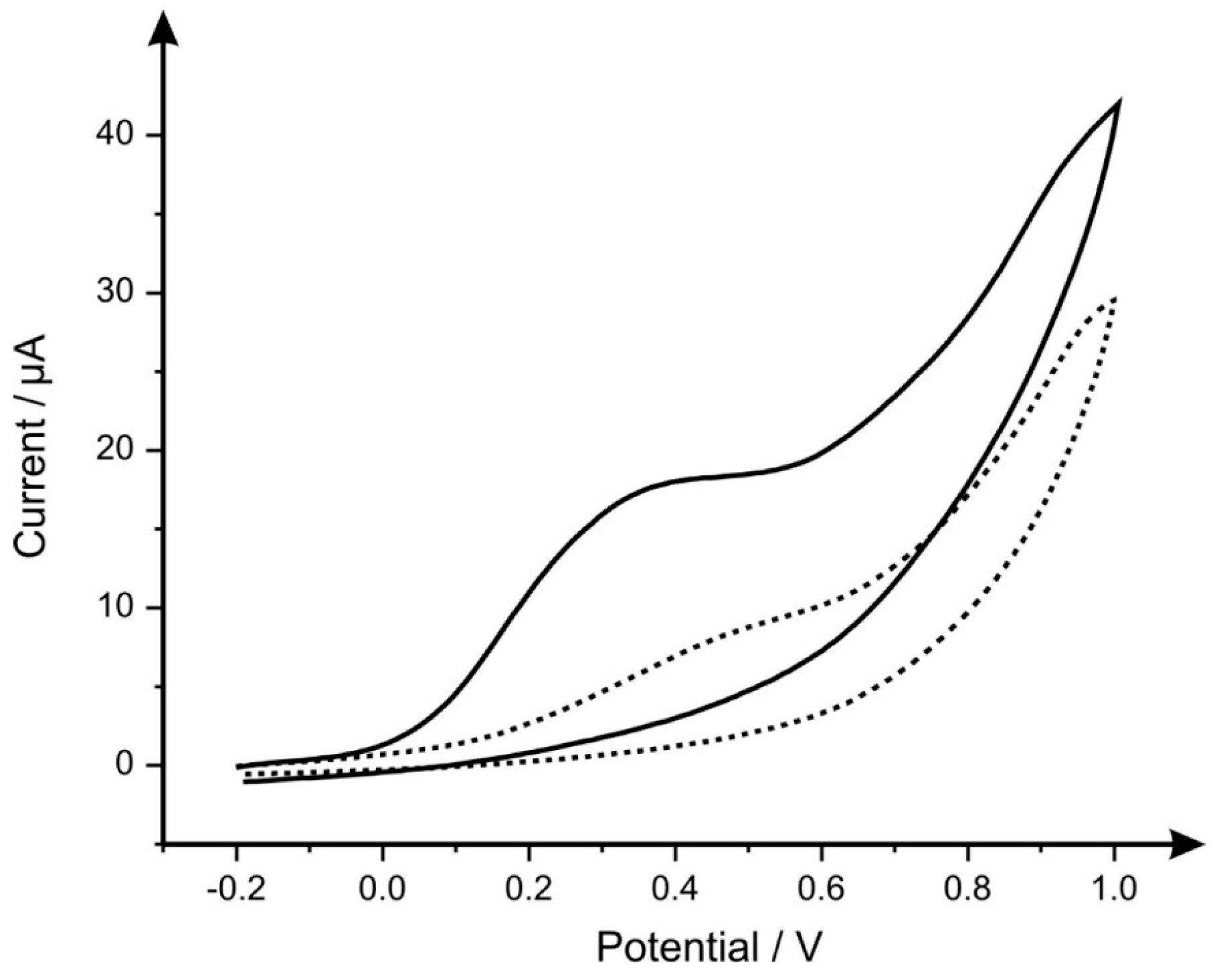
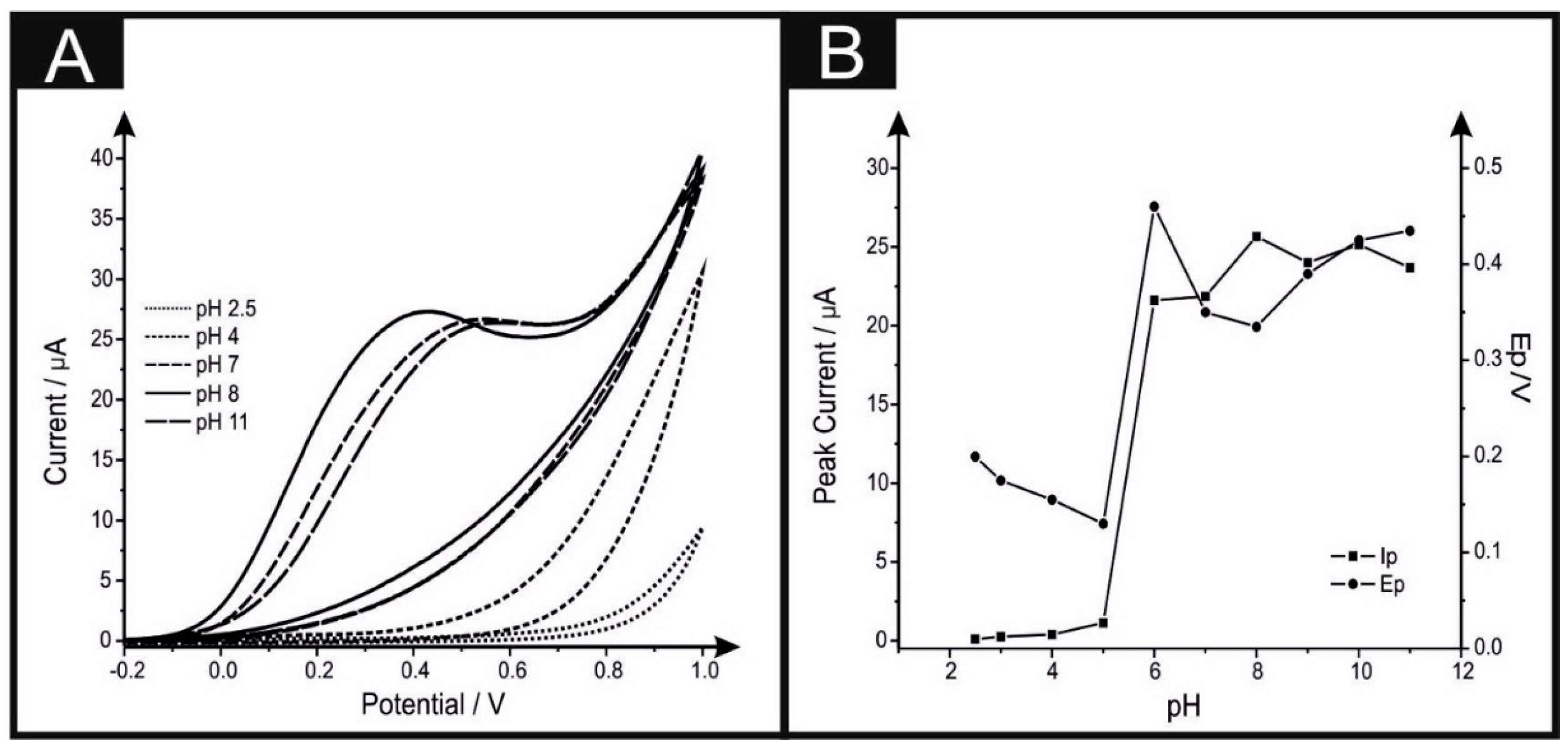
2. Results and Discussion
3. Materials and Methods
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ertek, B.; Vu, D.L.; Cervenka, L.; Dilgin, Y. Flow injection amperometric detection of sulfide using using a prussian blue modified glassy carbon electrode. Anal. Sci. 2012, 28, 1075–1080. [Google Scholar] [CrossRef] [PubMed]
- Radford-Knoery, J.; Cutter, G.A. Determination of carbonyl sulfide and hydrogen sulfide species in natural waters using specialized collection procedures and gas chromatography with flame photometric detection. Anal. Chem. 1993, 65, 976–982. [Google Scholar] [CrossRef]
- Colon, M.; Iglesias, M.; Hidalgo, M.; Todoli, J.L. Sulfide and sulfate determination in water samples by means of hydrogen sulfide generation-inductively coupled plasma-atomic emission spectrometry. J. Anal. At. Spectrom. 2008, 23, 416–418. [Google Scholar] [CrossRef]
- Sosa, V.; Barceló, C.; Serrano, N.; Ariño, C.; Díaz-Cruz, J.M.; Esteban, M. Antimony film screen-printed carbon electrode for stripping analysis of Cd(II), Pb(II), and Cu(II) in natural samples. Anal. Chim. Acta 2015, 855, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Bailey, T.S.; Pluth, M.D. Chemiluminiscent detection of enzimatically produced hydrogen sulfide: substrate hydrogen bonding influences selectivity for H2S over biological thiols. J. Am. Chem. Soc. 2013, 135, 16697–16704. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, N.S.; Davis, J.; Compton, R.G. Analytical strategies for the detection of sulfide: a review. Talanta 2000, 52, 771–784. [Google Scholar] [CrossRef]
- Pandey, S.K.; Kim, K.-H.; Tang, K.-T. A review of sensor-based methods for monitoring hydrogen sulfide. TrAC Trends Anal. Chem. 2012, 32, 87–99. [Google Scholar] [CrossRef]
- Figueiredo-Filho, L.C.S.; Brownson, D.A.C.; Fatibello-Filho, O.; Banks, C.E. Exploring the origins of the apparent "electrocatalytic" oxidation of kojic acid at graphene modified electrodes. Analyst 2013, 138, 4436–4442. [Google Scholar] [CrossRef] [PubMed]
- Brownson, D.A.C.; Foster, C.W.; Banks, C.E. The electrochemical performance of graphene modified electrodes: An analytical perspective. Analyst 2012, 137, 1815–1823. [Google Scholar] [CrossRef] [PubMed]
- Ramdani, O.; Metters, J.P.; Figueiredo-Filho, L.C.S.; Fatibello-Filho, O.; Banks, C.E. Forensic electrochemistry: Sensing the molecule of murder atropine. Analyst 2013, 138, 1053–1059. [Google Scholar] [CrossRef] [PubMed]
- Brownson, D.A.C.; Banks, C.E. Graphene electrochemistry: Fabricating amperometric biosensors. Analyst 2011, 136, 2084–2089. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Ibáñez, N.; García-Cruz, L.; Montiel, V.; Foster, C.W.; Banks, C.E.; Iniesta, J. Electrochemical lactate biosensor based upon chitosan/carbon nanotubes modified screen-printed graohite electrodes for the determination of lactate in embryonic cell cultures. Biosens. Bioelectron. 2016, 77, 1168–1174. [Google Scholar] [CrossRef] [PubMed]
- Brownson, D.A.C.; Gorbachev, R.V.; Haigh, S.J.; Banks, C.E. CVD graphene vs. highly ordered pyrolitic graphite for use in electroanalytical sensing. Analyst 2012, 137, 833–839. [Google Scholar] [CrossRef] [PubMed]
- Thakur, B.; Bernalte, E.; Smith, J.P.; Foster, C.W.; Linton, P.E.; Sawant, S.N.; Banks, C.E. Utilising copper screen-printed electrodes (CuSPE) for the electroanalytical sensing of sulfide. Analyst 2016, 141, 1233–1238. [Google Scholar] [CrossRef] [PubMed]
- Hart, J.P.; Wring, S.A. Screen-printed voltammetric and amperometric electrochemical sensors for decentralized testing. Electroanalysis 1994, 6, 617. [Google Scholar] [CrossRef]
- Li, M.; Li, Y.; Li, D.; Long, Y. Recent developments and applications of screen-printed electrodes in environmental assays-A review. Anal. Chim. Acta 2011, 734, 31. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.S.; Zen, J. Electrochemical investigation of glucose sensor fabricated at copper-plated screen-printed carbon electrodes. Electroanalysis 2002, 14, 671. [Google Scholar] [CrossRef]
- Honeychurch, K.C.; Hart, J.P. Screen-printed electrochemical sensors for monitoring metal pollutants. TrAC Trends Anal. Chem. 2003, 22, 456. [Google Scholar] [CrossRef]
- De Souza, A.P.R.; Foster, C.W.; Kolliopoulos, A.V.; Bertotti, M.; Banks, C.E. Screen-printed back-to-back electroanalytical sensors: Heavy metal ion sensing. Analyst 2015, 140, 4130–4136. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.P.; Metters, J.P.; Kampouris, D.K.; Lledo-Fernandez, C.; Sutcliffe, O.B.; Banks, C.E. Forensic electrochemistry: The electroanalytical sensing of Rohypnol® (flunitrazepam) using screen-printed graphite electrodes without recourse for electrode or sample pre-treatment. Analyst 2013, 138, 6185–6191. [Google Scholar] [CrossRef] [PubMed]
- Ruas de Souza, A.P.; Bertotti, M.; Foster, C.W.; Banks, C.E. Back-to-back screen-printed electroanalytical sensors: Extending the pottential applications of the simplistic design. Electroanalysis 2015, 27, 2295–2301. [Google Scholar] [CrossRef]
- Robinson, K.L.; Lawrence, N.S. A vinylanthracene and vinylferrocene-containing copolymer: A new dual pH/sulfide sensor. Electroanalysis 2006, 18, 677–683. [Google Scholar] [CrossRef]
- Lawrence, N.S. Amperometric detection of sulfide: An electrocatalytic reactiuon with ferrocene carboxylate. Electroanalysis 2006, 18, 1658–1663. [Google Scholar] [CrossRef]
- Dilgin, Y.; Kizilkaya, B.; Ertek, B.; Eren, N.; Dilgin, D.G. Amperometric detection of sulfide based on its elecctrocatalytic oxidation at a pencil graphite electrode modified with quercetin. Talanta 2012, 89, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Prodromidis, M.I.; Veltsistas, P.G.; Karayannis, M.I. Electrochemical study of chemically modified and screen-printed graphite electrodes with [SbVO(CHL)2]Hex. Application for the selective determination of sulfide. Anal. Chem. 2000, 72, 3995–4002. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, N.S.; Tustin, G.J.; Faulkner, M.; Jones, T.G.J. Ferrocene sulfonates as electrocatalysts for sulfide detection. Electrochim. Acta 2006, 52, 499–503. [Google Scholar] [CrossRef]
- Chang, J.-L.; Wei, G.-T.; Chen, T.-Y.; Zen, J.-M. Highly stable polymeric ionic liquid modified electrode to immobilize ferricyanide for electroanalysis of sulfide. Electroanalysis 2013, 25, 845–849. [Google Scholar] [CrossRef]
- Lawrence, N.S.; Deo, R.P.; Wang, J. Electrochemical determination of hydrogen sulfide at carbon nanotube modified electrodes. Anal. Chim. Acta 2004, 517, 131–137. [Google Scholar] [CrossRef]
- Banks, C.E.; Yashina, A.S.; Tustin, G.J.; Lafitte, V.G.H.; Jones, T.G.J.; Lawrence, N.S. Exploring alkylated ferrocene sulfonates as electrocatalysts for sulfide detection. Electroanalysis 2007, 19, 2518–2522. [Google Scholar] [CrossRef]
- Lawrence, N.S.; Davis, J.; Jiang, L.; Jones, T.G.J.; Davies, S.N.; Compton, R.G. Electrochemically initiated reactions of dyphenilamines with sulfide: Application to the voltammetric detection of hydrogen sulfide. Electroanalysis 2001, 13, 143–148. [Google Scholar] [CrossRef]
- Lawrence, N.S.; Davis, J.; Jiang, L.; Jones, T.G.J.; Davies, S.N.; Compton, R.G. Electrochemically initiated 1,4-nucleophilic substitutions: A general strategy for the analytical detection of hydrgen sulfide. Electroanalysis 2001, 13, 432–436. [Google Scholar] [CrossRef]
- Long Vu, D.; Červenka, L. Determination of sulfide by hematoxylin multiwalled carbon nanotubes modified carbon paste electrodes. Electroanalysis 2013, 25, 1967–1973. [Google Scholar] [CrossRef]
- Giovanelli, D.; Lawrence, N.S.; Wilkins, S.J.; Jiang, L.; Jones, T.G.J.; Compton, R.G. Anodic stripping voltammetry of sulphide at a nickel film: Towards the development of a reagentless sensor. Talanta 2003, 61, 211–220. [Google Scholar] [CrossRef]
- Lawrence, N.S.; Davis, J.; Jiang, L.; Jones, T.G.J.; Davies, S.N.; Compton, R.G. The electrochemical analog of the methylene blue reaction: A novel amperometric approach to the detection of hydrogen sulfide. Electroanalysis 2000, 12, 1453–1460. [Google Scholar] [CrossRef]
- Bitziou, E.; Joseph, M.B.; Read, T.L.; Palmer, N.; Mollart, T.; Newton, M.E.; Macpherson, J.V. In situ optimization of pH for parts-per-billion electrochemical detection of dissolved hydrogen sulfide using boron doped diamond flow electrodes. Anal. Chem. 2014, 86, 10834–10840. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, J.; Robinson, K.L.; Lawrence, N.S. Electrochemical determination of sulfide at various carbon substrates: A comparative study. Anal. Sci. 2007, 23, 673–676. [Google Scholar] [CrossRef] [PubMed]
- Brownson, D.A.C.; Kelly, P.J.; Banks, C.E. In situ electrochemical characterisation of graphene and various carbon-based electrode materials: An internal standard approach. RSC Adv. 2015, 5, 37281–37286. [Google Scholar] [CrossRef]
- Brownson, D.A.C.; Varey, S.A.; Hussain, F.; Haigh, S.J.; Banks, C.E. Electrochemical properties of CVD grown pristine graphene: Monolayer- vs. quasi-graphene. Nanoscale 2014, 6, 1607–1621. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, R.S. Theory and application of cyclic voltammetry for measurement of electron reaction kinetic. Anal. Chem. 1965, 37, 1351. [Google Scholar] [CrossRef]
- Tan, F.; Metters, J.P.; Banks, C.E. Electroanalytical applications of screen printed microelectrode arrays. Sens. Actuators B Chem. 2013, 181, 454. [Google Scholar] [CrossRef]
- Lavagnini, I.; Antiochia, R.; Magno, F. An extended method for the practical evaluation of the standard rate constant from cyclic voltammetric data. Electroanalysis 2004, 16, 505. [Google Scholar] [CrossRef]
- Banks, C.E.; Compton, R.G.; Fisher, A.C.; Henley, I.E. The transport limited currents at insonated electrodes. Phys. Chem. Chem. Phys. 2004, 6, 3147–3152. [Google Scholar] [CrossRef]
- Cumba, L.R.; Foster, C.W.; Brownson, D.A.C.; Smith, J.P.; Iniesta, J.; Thakur, B.; Carmo, D.R.D.; Banks, C.E. Can the mechanical activation (polishing) of screen-printed electrodes enhance their electroanalytical response? Analyst 2016. [Google Scholar] [CrossRef] [PubMed]
- Foster, C.W.; Metters, J.P.; Kampouris, D.K.; Banks, C.E. Ultraflexible screen-printed graphitic electroanalytical sensing platforms. Electroanalysis 2014, 26, 262–274. [Google Scholar] [CrossRef]
- Rowley-Neale, S.J.; Brownson, D.A.C.; Smith, G.C.; Sawtell, D.A.G.; Kelly, P.J.; Banks, C.E. 2D nanosheet molybdenum disulphide (MoS2) modified electrodes explored towards the hydrogen evolution reaction. Nanoscale 2015, 7, 18152–18168. [Google Scholar] [CrossRef] [PubMed]
- Randviir, E.P.; Brownson, D.A.C.; Metters, J.P.; Kadara, R.O.; Banks, C.E. The fabrication, characterisation and electrochemical investigation of screen-printed graphene electrodes. Phys. Chem. Chem. Phys. 2014, 16, 4598–4611. [Google Scholar] [CrossRef] [PubMed]
- Giovanelli, D.; Lawrence, N.S.; Jiang, L.; Jones, T.G.J.; Compton, R.G. Amperometric determination of sulfide at pre-oxidized nickel electrode in acidic media. Analyst 2003, 128, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Foster, C.W.; Metters, J.P.; Banks, C.E. Ultra flexible paper based electrochemical sensors: effect of mechanical contortion upon electrochemical performance. Electroanalysis 2013, 25, 2275–2282. [Google Scholar] [CrossRef]
- Galdino, F.E.; Foster, C.W.; Bonacin, J.A.; Banks, C.E. Exploring the electrical wiring of screen-printed configurations utilised in electroanalysis. Anal. Methods 2015, 7, 1208–1214. [Google Scholar] [CrossRef]
- Gomis-Berenguer, A.; Gomez-Mingot, M.; Montiel, V.; Canals, A.; Thiemann, T.; Kadara, R.O.; Banks, C.E.; Iniesta, J. Exploring the electrochemical behavior of screen printed graphite electrodes in a room temperature ionic liquid. RSC Adv. 2012, 2, 7735–7742. [Google Scholar] [CrossRef]

| Electrode Modification | Underlying Electrode | Technique | Sensitivity/µA·mM−1·cm2 | LOD (3σ)/µM | Linear Range/µM | Comments | Reference |
|---|---|---|---|---|---|---|---|
| Copolymer of anthracene and ferrocene | BPPG | CV | γ | γ | 200–2000 | Dual sensor for pH and sulfide. | [22] |
| Ferrocene carboxylate | GCE and BDDE | CV | 81.4 × 103 | 2.0 | 200–1000 | Ferrocene carboxylate used as electrocatalyst. Detection of sulfide within river water samples reported. | [23] |
| Quercetin | Pencil graphite electrode | CV | 41.0 × 103 | 0.3 | 1–20 | Quercetin used as electrocatalyst for sulfide oxidation. Sulfide detected in waste waters and validated with an independent spectrophotometric method. | [24] |
| Hexadecylpyridiniumbis (chloranilato)-antimonyl(V) | SPE | CV | γ | γ | 0.01–0.70 | Hexadecylpyridinium-bis(chloranilato)-antimonyl(V) acted as an electrocatalyst for the sensing of sulfide. | [25] |
| Ferrocene sulfonate | BDDE and GCE | CV | 70.7 | 14.0 | 20–1000 | Ferrocene sulfonate acted as used as an electrocatalyst for the sensing of sulfide. | [26] |
| Ionic liquid | SPE | Amperometry | 41.0 | 12.9 × 10−3 | 1–3000 | Ionic liquid used to immobilize ferricyanide as an electrocatalyst. Sulfide detected in hot spring water and ground water. | [27] |
| CNT | GCE | Amperometry | 16.2 × 102 | 0.3 | 1–112 | Sulfide detected in river water samples | [28] |
| Alkylated ferrocene | BDDE | CV | γ | γ | γ | Alkylated ferrocene sulfonates used as electrocatalyst. | [29] |
| DMPD | GCE | CV | 54 × 10−5 | 3.0 | 3–150 | γ | [30] |
| DPSA | 11.3 × 10−5 | 5.3 | 5–150 | ||||
| DDPS | 22.85 × 10−5 | 12.3 | 12–163 | ||||
| Hydroquinone | GCE | CV | 7.2 × 10−5 | 3.0 | 3–150 | Detection of sulfide performed at pH 4. | [31] |
| Catechol | 41.4 × 10−5 | 10.0 | 10–192 | ||||
| DPPD | 54 × 10−5 | 14.0 | 14–91 | ||||
| Dopamine | 17.4 × 10−5 | 22.0 | 22–262 | ||||
| Aminophenol | 51.6 × 10−5 | 50.0 | 50–181 | ||||
| Hematoxylin MWCNT | CPE | Amperometry | 14.6 × 102 | 0.2 | 0.5–150 | Detection of sulfide performed in river water. | [32] |
| Prussian blue | GCE | Amperometry | 1.35 × 102 | 0.3 | 0.5–100 | Detection of sulfide performed in waste water and validated with independent spectrophotometric method. | [1] |
| Nickel oxide | GCE | LSV | 5.5 × 103 | 5.0 | 20–90 | Ni modified SPEs demonstrate the possible development of inexpensive and disposable sensors for sulfide. | [33] |
| SPE | 3.1 × 103 | 40–100 | |||||
| DMPD | GCE, Pt, CPE and ITO | CV | γ | γ | 10–300 | DMPD used as electrocatalyst. Various electrode substrates were used (Pt, ITO, carbon paste), which gave similar response like GCE. | [34] |
| Unmodified | BDDE | Amperometry | 13.03 × 102 | 0.4 | γ | Detection performed at in-situ generated pH 10. | [35] |
| Unmodified | BDDE | CV and Square Wave | 1.12 × 102 | 4.9 | 5–60 | EPPGE detection compared to a CNT modified GCE, performed at pH 7. | [36] |
| BPPGE | |||||||
| EPPGE | |||||||
| GCE | |||||||
| Unmodified | GCE | CV and Amperometry | 1.69 × 102 | 37.5 | 25–400 | Mediatorless sulfide sensing with unmodified electrodes. | This work |
| SPE | 1.52 × 102 | 51.8 | 25–700 |
| Electrode | Analyte | |
|---|---|---|
| Hexammineruthenium(III) Chloride | Ammonium Iron(II) Sulfate | |
| ΔEp/mV | ΔEp/mV | |
| EPPGE | 76 | 117 |
| BPPGE | 150 | 964 |
| GCE | 86 | 537 |
| SPE | 115 | 605 |
| BDDE | 120 | 1162 |
| Electrode | Slope/µA·mM−1 | Sensitivity/µA·mM−1·cm−2 | Linear Range/μM | LOD */μM |
|---|---|---|---|---|
| SPE | 10.755 | 152.23 | 25–700 | 32.5 |
| GCE—Unpolished | 6.0880 | 86.170 | 25–400 | 51.8 |
| GCE—Polished | 11.989 | 169.69 | 25–1000 | 37.5 |
| Experiment Number | Spiked/µM | Detected/µM | % RSD (n = 3) | % Apparent Recovery |
|---|---|---|---|---|
| 1 | 50 | 51 (±0.005) | 0.52 | 101.6 |
| 2 | 150 | 148 (±0.007) | 0.79 | 98.8 |
| 3 | 650 | 630 (±0.018) | 1.81 | 96.9 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thakur, B.; Bernalte, E.; Smith, J.P.; Linton, P.E.; Sawant, S.N.; Banks, C.E.; Foster, C.W. The Mediatorless Electroanalytical Sensing of Sulfide Utilizing Unmodified Graphitic Electrode Materials. C 2016, 2, 14. https://doi.org/10.3390/c2020014
Thakur B, Bernalte E, Smith JP, Linton PE, Sawant SN, Banks CE, Foster CW. The Mediatorless Electroanalytical Sensing of Sulfide Utilizing Unmodified Graphitic Electrode Materials. C. 2016; 2(2):14. https://doi.org/10.3390/c2020014
Chicago/Turabian StyleThakur, Bhawana, Elena Bernalte, Jamie P. Smith, Patricia E. Linton, Shilpa N. Sawant, Craig. E. Banks, and Christopher W. Foster. 2016. "The Mediatorless Electroanalytical Sensing of Sulfide Utilizing Unmodified Graphitic Electrode Materials" C 2, no. 2: 14. https://doi.org/10.3390/c2020014







