Biodiversity of Pigmented Fungi Isolated from Marine Environment in La Réunion Island, Indian Ocean: New Resources for Colored Metabolites
Abstract
:1. Introduction
2. Materials and Methods
2.1. Samples Collection
2.2. Culture and Purification of Fungi
2.3. Fungal Identification
2.3.1. Fungal DNA Extraction
2.3.2. Primers, PCR Amplification and Sequencing
2.3.3. Sequence Analysis
2.4. Culture Conditions for Pigment Production and Separation of Biomass from Liquid Medium
2.4.1. Culture Conditions
2.4.2. Separation of Biomass from Culture Liquid
2.5. Production of Pigments
2.5.1. Determination of Pigments Production in Liquid Cultures
2.5.2. Extraction of Pigments
3. Results
3.1. Diversity of Isolated Fungi
3.2. Pigment Production
3.2.1. In Culture Broth
3.2.2. Pigmented Contents from Mycelium
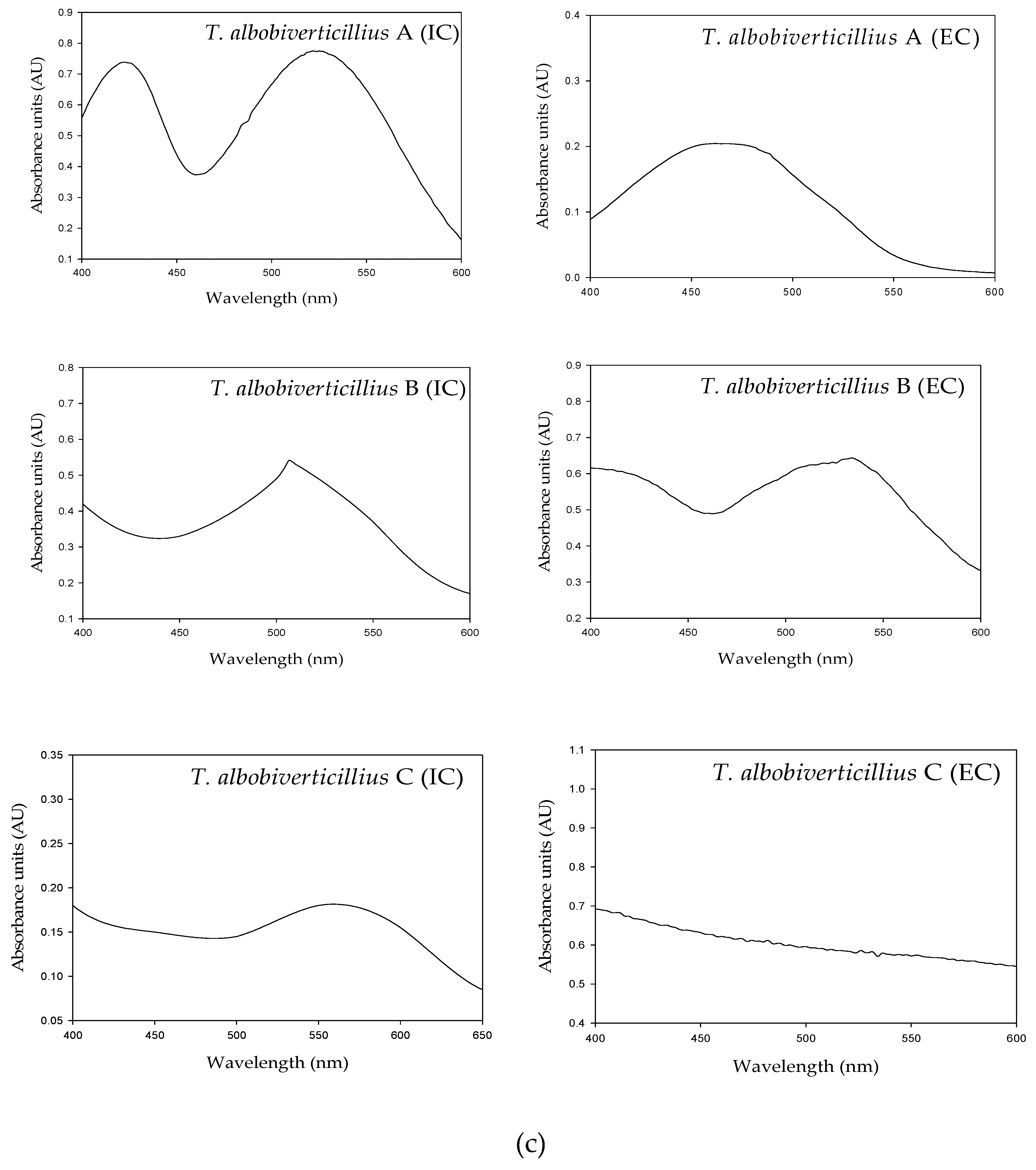
3.3. Spectrophotometric Characterization of Pigments
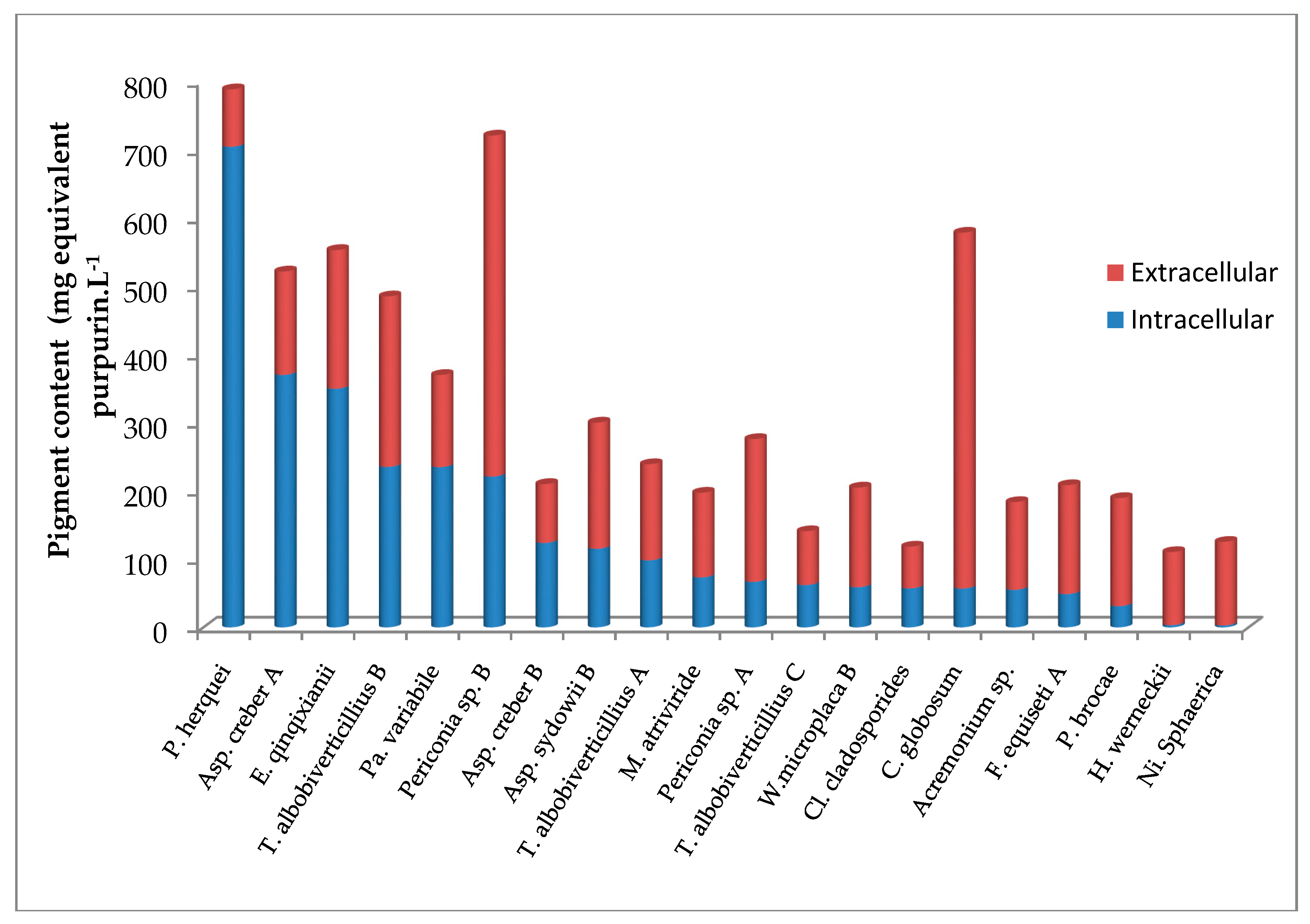
3.4. Evaluation of Intracellular and Extracellular Contents in Pigments
4. Discussion
4.1. Biodiversity of Marine-Derived Fungi around La Réunion Island
4.2. Qualitative Aspect of the Pigment Production
4.3. Quantitative Aspect of Pigment Production
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dufossé, L.; Galaup, P.; Yaron, A.; Arad, S.M.; Blanc, P.; Chidambara Murthy, K.N.; Ravishankar, G.A. Microorganisms and microalgae as sources of pigments for food use: A scientific oddity or an industrial reality? Trends Food Sci. Technol. 2005, 16, 389–406. [Google Scholar] [CrossRef]
- Mayer, A.M.; Rodriguez, A.D.; Taglialatela-Scafati, O.; Fusetani, N. Marine pharmacology in 2009–2011: Marine compounds with antibacterial, antidiabetic, antifungal, anti-inflammatory, antiprotozoal, antituberculosis, and antiviral activities; affecting the immune and nervous systems, and other miscellaneous mechanisms of action. Mar. Drugs 2013, 11, 2510–2573. [Google Scholar] [PubMed]
- Mapari, S.A.S.; Nielsen, K.F.; Larsen, T.O.; Frisvad, J.C.; Meyer, A.S.; Thrane, U. Exploring fungal biodiversity for the production of water-soluble pigments as potential natural food colorants. Curr. Opin. Biotechnol. 2005, 16, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Hohmann, C.; Schneider, K.; Bruntner, C.; Irran, E.; Nicholson, G.; Bull, A.T.; Jones, A.L.; Brown, R.; Stach, J.E.; Goodfellow, M.; et al. Caboxamycin, a new antibiotic of the benzoxazole family produced by the deep-sea strain Streptomyces sp. Ntk 937. J. Antibiot. 2009, 62, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Costantino, V.; Fattorusso, E.; Mangoni, A.; Perinu, C.; Cirino, G.; De Gruttola, L.; Roviezzo, F. Tedanol: A potent anti-inflammatory ent-pimarane diterpene from the caribbean sponge Tedania ignis. Bioorg. Med. Chem. 2009, 17, 7542–7547. [Google Scholar] [CrossRef] [PubMed]
- Bonugli-Santos, R.C.; dos Santos Vasconcelos, M.R.; Passarini, M.R.Z.; Vieira, G.A.L.; Lopes, V.C.P.; Mainardi, P.H.; dos Santos, J.A.; de Azevedo Duarte, L.; Otero, I.V.R.; da Silva Yoshida, A.M.; et al. Marine-derived fungi: Diversity of enzymes and biotechnological applications. Front. Microbiol. 2015, 6, 269. [Google Scholar] [CrossRef] [PubMed]
- Panno, L.; Bruno, M.; Voyron, S.; Anastasi, A.; Gnavi, G.; Miserere, L.; Varese, G.C. Diversity, ecological role and potential biotechnological applications of marine fungi associated to the seagrass Posidonia oceanica. New Biotechnol. 2013, 30, 685–694. [Google Scholar] [CrossRef] [PubMed]
- Cathrine, S.J.; Raghukumar, C. Anaerobic denitrification in fungi from the coastal marine sediments off Goa, India. Mycol. Res. 2009, 113, 100–109. [Google Scholar] [CrossRef] [PubMed]
- Holguin, G.; Vazquez, P.; Bashan, Y. The role of sediment microorganisms in the productivity, conservation, and rehabilitation of mangrove ecosystems: An overview. Biol. Fertil. Soils 2001, 33, 265–278. [Google Scholar] [CrossRef]
- Bugni, T.S.; Ireland, C.M. Marine-derived fungi: A chemically and biologically diverse group of microorganisms. Nat. Prod. Rep. 2004, 21, 143–163. [Google Scholar] [CrossRef] [PubMed]
- Jones, E.B.G.; Pang, K.L. Marine Fungi: And Fungal-Like Organisms; De Gruyter: Berlin, Germany, 2012. [Google Scholar]
- Kathiresan, K.; Bingham, B.L. Biology of mangroves and mangrove ecosystems. In Advances in Marine Biology; Southward, A., Young, C., Fuiman, L., Tyler, P., Eds.; Academic Press: San Diego, CA, USA, 2001; Volume 40, pp. 81–251. [Google Scholar]
- Kohlmeyer, J.; Kohlmeyer, E. Marine Mycology; Elsvier Inc.: London, UK, 1979; p. 704. [Google Scholar]
- Jones, E.B.G. Marine fungi: Some factors affecting biodiversity. Fungal Divers. 2000, 4, 53–73. [Google Scholar] [CrossRef]
- Saleem, M.; Nazir, M. Bioactive natural products from marine-derived fungi: An update. In Studies in Natural Products Chemistry; Atta-ur-Rahman, Ed.; Elsevier: Amsterdam, The Netherlands, 2015; Volume 45, pp. 297–361. [Google Scholar]
- Imhoff, J.F. Natural products from marine fungi—Still an underrepresented resource. Mar. Drugs 2016, 14, 19. [Google Scholar] [CrossRef] [PubMed]
- Debashish, G.; Malay, S.; Barindra, S.; Joydeep, M. Marine enzymes. Adv. Biochem. Eng./Biotechnol. 2005, 96, 189–218. [Google Scholar]
- Zhang, C.; Kim, S.K. Application of marine microbial enzymes in the food and pharmaceutical industries. Adv. Food Nutr. Res. 2012, 65, 423–435. [Google Scholar] [PubMed]
- Fouillaud, M.; Venkatachalam, M.; Girard-Valenciennes, E.; Caro, Y.; Dufossé, L. Anthraquinones and derivatives from marine-derived fungi: Structural diversity and selected biological activities. Mar. Drugs 2016, 14, 64. [Google Scholar] [CrossRef] [PubMed]
- Fouillaud, M.; Venkatachalam, M.; Girard-Valenciennes, E.; Caro, Y.; Dufossé, L. Marine-derived fungi producing red anthraquinones: New resources for natural colors? In Proceedings of the 8th International Conference of Pigments in Food, “Coloured Foods for Health Benefits”, Cluj-Napoca, Romania, 28 June—1 July 2016. [Google Scholar]
- Caro, Y.; Venkatachalam, M.; Lebeau, J.; Fouillaud, M.; Dufossé, L. Pigments and colorants from filamentous fungi. In Fungal Metabolites; Merillon, J.-M., Ramawat, G.K., Eds.; Springer: Cham, Switzerland, 2016; pp. 1–70. [Google Scholar]
- Ebel, R. Natural product diversity from marine fungi. In Comprehensive Natural Products II: Chemistry and Biology; Mander, L., Liu, H.-W., Eds.; Elsevier: Oxford, UK, 2010; Volume 2, pp. 223–262. [Google Scholar]
- Calvo, A.M.; Wilson, R.A.; Bok, J.W.; Keller, N.P. Relationship between secondary metabolism and fungal development. Microbiol. Mol. Biol. Rev. 2002, 66, 447–459. [Google Scholar] [CrossRef] [PubMed]
- Margalith, P. Pigment Microbiology; Springer: London, UK; New York, NY, USA, 1992; p. 156. [Google Scholar]
- Demain, A.L.; Fang, A. The natural functions of secondary metabolites. In History of Modern Biotechnology I; Fiechter, A., Ed.; Springer: Berlin/Heidelberg, Germany, 2000; pp. 1–39. [Google Scholar]
- Réunion’s Coral Reef. Available online: https://en.wikipedia.org/wiki/R%C3%A9union%27s_coral_reef (accessed on 1 May 2017).
- Peyrot-Clausade, M.; Chazottes, V.; Pari, N.; Peyrot-Clausade, M.; Chazottes, V.; Pari, N. Bioerosion in the carbonate budget of two indo-pacific reefs: La Réunion (Indian Ocean) and moorea (Pacific Ocean). Bull. Geol. Soc. Denmark 1999, 1999, 1–30. [Google Scholar]
- Conand, C.; Chabanet, P.; Cuet, P.; Letourneur, Y. The carbonate budget of a fringing reef in La Reunion Island (Indian Ocean): Sea urchin and fish bioerosion and net calcification. In Proceedings of the 8th International Coral Reef Symposium, Panama City, Panama, 24–29 June 1997; Lessios, H.A., Macintyre, I.G., Eds.; pp. 953–958. [Google Scholar]
- Naim, O.; Cuet, P.; Mangar, V. Coral reefs of the mascarene archipelago. In Coral Reefs of the Indian Ocean: Their Ecology and Conservation; McClanahan, T.R., Sheppard, C., Obura, D.O., Eds.; Oxford University Press: New York, NY, USA, 2000; pp. 353–381. [Google Scholar]
- Turner, J.; Klaus, R. Coral reefs of the mascarenes, western indian ocean. Philos. Trans. R. Soc. Lond. A Math. Phys. Eng. Sci. 2005, 363, 229–250. [Google Scholar] [CrossRef] [PubMed]
- Montaggioni, L.; Faure, G. Les Récifs Coralliens des Mascareignes (Océan Indien); Université Francaise de l’Océan Indien, Centre Universitaire de La Réunion: Réunion, France, 1980; p. 151. [Google Scholar]
- Sanders, E.R. Aseptic laboratory techniques: Plating methods. J. Vis. Exp. JoVE 2011, 63, e3064. [Google Scholar] [CrossRef] [PubMed]
- Jong, S.; Dugan, F.; Edwards, M. ATCC Filamentous Fungi, 19th ed.; Rockville, MD American Type Culture Collection: Manassas, VA, USA, 1996. [Google Scholar]
- Dahmen, H.; Staub, T.; Schwinn, F. Technique for long-term preservation of phytopathogenic fungi in liquid nitrogen. Phytopathology 1983, 73, 241–246. [Google Scholar] [CrossRef]
- Knebelsberger, T.; Stoger, I. DNA extraction, preservation, and amplification. Methods Mol. Biol. 2012, 858, 311–338. [Google Scholar] [PubMed]
- Toju, H.; Tanabe, A.S.; Yamamoto, S.; Sato, H. High-coverage its primers for the DNA-based identification of ascomycetes and basidiomycetes in environmental samples. PLoS ONE 2012, 7, e40863. [Google Scholar] [CrossRef] [PubMed]
- Samson, R.A.; Visagie, C.M.; Houbraken, J.; Hong, S.B.; Hubka, V.; Klaassen, C.H.W.; Perrone, G.; Seifert, K.A.; Susca, A.; Tanney, J.B.; et al. Phylogeny, identification and nomenclature of the genus aspergillus. Stud. Mycol. 2014, 78, 141–173. [Google Scholar] [CrossRef] [PubMed]
- Samson, R.A.; Yilmaz, N.; Houbraken, J.; Spierenburg, H.; Seifert, K.A.; Peterson, S.W.; Varga, J.; Frisvad, J.C. Phylogeny and nomenclature of the genus talaromyces and taxa accommodated in penicillium subgenus biverticillium. Stud. Mycol. 2011, 70, 159–183. [Google Scholar] [CrossRef] [PubMed]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR Protoc. Guide Methods Appl. 1990, 18, 315–322. [Google Scholar]
- Romanelli, A.M.; Sutton, D.A.; Thompson, E.H.; Rinaldi, M.G.; Wickes, B.L. Sequence-based identification of filamentous basidiomycetous fungi from clinical specimens: A cautionary note. J. Clin. Microbiol. 2010, 48, 741–752. [Google Scholar] [CrossRef] [PubMed]
- Sutton, D.A.; Marín, Y.; Thompson, E.H.; Wickes, B.L.; Fu, J.; García, D.; Swinford, A.; de Maar, T.; Guarro, J. Isolation and characterization of a new fungal genus and species, aphanoascella galapagosensis, from carapace keratitis of a galapagos tortoise (chelonoidis nigra microphyes). Med. Mycol. 2013, 51, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Li, Y.; Tang, Y. Cyclization of aromatic polyketides from bacteria and fungi. Nat. Prod. Rep. 2010, 27, 839–868. [Google Scholar] [CrossRef] [PubMed]
- Caro, Y.; Anamale, L.; Fouillaud, M.; Laurent, P.; Petit, T.; Dufosse, L. Natural hydroxyanthraquinoid pigments as potent food grade colorants: An overview. Nat. Prod. Bioprospect. 2012, 2, 174–193. [Google Scholar] [CrossRef]
- Machatová, Z.; Barbieriková, Z.; Poliak, P.; Jančovičová, V.; Lukeš, V.; Brezová, V. Study of natural anthraquinone colorants by epr and uv/vis spectroscopy. Dyes Pigments 2016, 132, 79–93. [Google Scholar] [CrossRef]
- Geyer, C.J. Fuzzy p-Values and Ties in Nonparametric Tests. Avaliable online: http://www.stat.umn.edu/geyer/fuzz (accessed on 30 June 2017).
- Golubic, S.; Radtke, G.; Le Campion-Alsumard, T. Endolithic fungi in marine ecosystems. Trends Microbiol. 2005, 13, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Priess, K.; Le Campion-Alsumard, T.; Golubic, S.; Gadel, F.; Thomassin, B. Fungi in corals: Black bands and density-banding of Porites lutea and P. lobata skeleton. Mar. Biol. 2000, 136, 19–27. [Google Scholar] [CrossRef]
- Nicoletti, R.; Trincone, A. Bioactive compounds produced by strains of penicillium and Talaromyces of marine origin. Mar. Drugs 2016, 14, 37. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, M.; Kasai, Y.; Komatsu, K.; Sone, T.; Tanaka, M.; Mikami, Y.; Kobayashi, J.i. Citrinadin a, a novel pentacyclic alkaloid from marine-derived fungus Penicillium citrinum. Org. Lett. 2004, 6, 3087–3089. [Google Scholar] [CrossRef] [PubMed]
- Malmstrøm, J.; Christophersen, C.; Frisvad, J.C. Secondary metabolites characteristic of Penicillium citrinum, Penicillium steckii and related species. Phytochemistry 2000, 54, 301–309. [Google Scholar] [CrossRef]
- Endo, A.; Kuroda, M.; Tsujita, Y. Ml-236a, ml-236b, and ml-236c, new inhibitors of cholesterogensis produced by Penicillium citrinum. J. Antibiot. 1976, 29, 1346–1348. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Raghukumar, C.; Verma, P.; Shouche, Y. Assessment of fungal diversity in deep-sea sediments by multiple primer approach. World J. Microbiol. Biotechnol. 2012, 28, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Brunati, M.; Rojas, J.L.; Sponga, F.; Ciciliato, I.; Losi, D.; Gottlich, E.; de Hoog, S.; Genilloud, O.; Marinelli, F. Diversity and pharmaceutical screening of fungi from benthic mats of antarctic lakes. Mar. Genom. 2009, 2, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Yahr, R.; Schoch, C.L.; Dentinger, B.T. Scaling up discovery of hidden diversity in fungi: Impacts of barcoding approaches. Philos. Trans. R. Soc. B 2016, 371, 20150336. [Google Scholar] [CrossRef] [PubMed]
- An, X.; Feng, B.-M.; Chen, G.; Chen, S.-F.; Wang, H.-F.; Pei, Y.-H. Isolation and identification of two new compounds from marine-derived fungus Acremonium fusidioides rz01. Chin. J. Nat. Med. 2016, 14, 934–938. [Google Scholar] [CrossRef]
- Afiyatullov, S.S.; Kalinovsky, A.I.; Antonov, A.S.; Zhuravleva, O.I.; Khudyakova, Y.V.; Aminin, D.L.; Yurchenko, A.N.; Pivkin, M.V. Isolation and structures of virescenosides from the marine-derived fungus Acremonium striatisporum. Phytochem. Lett. 2016, 15, 66–71. [Google Scholar] [CrossRef]
- Gallardo, G.L.; Butler, M.; Gallo, M.L.; Rodríguez, M.A.; Eberlin, M.N.; Cabrera, G.M. Antimicrobial metabolites produced by an intertidal acremonium furcatum. Phytochemistry 2006, 67, 2403–2410. [Google Scholar] [CrossRef] [PubMed]
- Hiort, J.; Maksimenka, K.; Reichert, M.; Perovic-Ottstadt, S.; Lin, W.H.; Wray, V.; Steube, K.; Schaumann, K.; Weber, H.; Proksch, P.; et al. New natural products from the sponge-derived fungus Aspergillus niger. J. Nat. Prod. 2004, 67, 1532–1543. [Google Scholar] [CrossRef] [PubMed]
- Pagano, M.C.; Rosa, L.H. Fungal molecular taxonomy. In Fungal Biomolecules; John Wiley & Sons, Ltd.: Chichester, UK, 2015; pp. 311–321. [Google Scholar]
- Slater, G.; Haskins, R.; Hogge, L.; Nesbitt, L. Metabolic products from a Trichoderma viride pers. Ex fries. Can. J. Chem. 1967, 45, 92–96. [Google Scholar] [CrossRef]
- Duran, N.; Teixeira, M.F.; De Conti, R.; Esposito, E. Ecological-friendly pigments from fungi. Crit. Rev. Food Sci. Nutr. 2002, 42, 53–66. [Google Scholar] [CrossRef] [PubMed]
- Butinar, L.; Frisvad, J.C.; Gunde-Cimerman, N. Hypersaline waters–a potential source of foodborne toxigenic Aspergilli and Penicillia. FEMS Microbiol. Ecol. 2011, 77, 186–199. [Google Scholar] [CrossRef] [PubMed]
- Woo, P.C.; Lam, C.W.; Tam, E.W.; Lee, K.C.; Yung, K.K.; Leung, C.K.; Sze, K.H.; Lau, S.K.; Yuen, K.Y. The biosynthetic pathway for a thousand-year-old natural food colorant and citrinin in Penicillium marneffei. Sci. Rep. 2014, 4, 6728. [Google Scholar] [CrossRef] [PubMed]
- Arai, T.; Koganei, K.; Umemura, S.; Kojima, R.; Kato, J.; Kasumi, T.; Ogihara, J. Importance of the ammonia assimilation by Penicillium purpurogenum in amino derivative monascus pigment, PP-V, production. AMB Express 2013, 3, 19. [Google Scholar] [CrossRef] [PubMed]
- Ogihara, J.; Kato, J.; Oishi, K.; Fujimoto, Y. Pp-r, 7-(2-hydroxyethyl)-monascorubramine, a red pigment produced in the mycelia of Penicillium sp. AZ. J. Biosci. Bioeng. 2001, 91, 44–47. [Google Scholar] [CrossRef]
- Frisvad, J.C.; Yilmaz, N.; Thrane, U.; Rasmussen, K.B.; Houbraken, J.; Samson, R.A. Talaromyces atroroseus, a new species efficiently producing industrially relevant red pigments. PLoS ONE 2013, 8, e84102. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, N.; Houbraken, J.; Hoekstra, E.S.; Frisvad, J.C.; Visagie, C.M.; Samson, R.A. Delimitation and characterisation of Talaromyces purpurogenus and related species. Persoonia 2012, 29, 39–54. [Google Scholar] [CrossRef] [PubMed]
- Thomson, R.H. Naturally Occurring Quinones IV: Recent Advances; Blackie Academic & Professional: London, UK; New York, NY, USA, 1997. [Google Scholar]
- Takahashi, M.; Koyama, K.; Natori, S. Four new azaphilones from Chaetomium globosum var. Flavo-viridae. Chem. Pharm. Bull. 1990, 38, 625–628. [Google Scholar] [CrossRef]
- McMullin, D.R. Structural Characterization of Secondary Metabolites Produced by Fungi Obtained from Damp Canadian Building. Ph.D. Thesis, Ottawa-Carleton University, Ottawa, ON, Canada, 2008. [Google Scholar]
- McMullin, D.R.; Sumarah, M.W.; Miller, J.D. Chaetoglobosins and azaphilones produced by Canadian strains of Chaetomium globosum isolated from the indoor environment. Mycotoxin Res. 2013, 29, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Ming Ge, H.; Yun Zhang, W.; Ding, G.; Saparpakorn, P.; Chun Song, Y.; Hannongbua, S.; Xiang Tan, R. Chaetoglobins A and B, two unusual alkaloids from endophytic Chaetomium globosum culture. Chem. Commun. 2008, 5978–5980. [Google Scholar] [CrossRef] [PubMed]
- Brewer, D.; Jerram, W.A.; Taylor, A. The production of cochliodinol and a related metabolite by chaetomium species. Can. J. Microbiol. 1968, 14, 861–866. [Google Scholar] [CrossRef] [PubMed]
- Prado-Cabrero, A.; Schaub, P.; Diaz-Sanchez, V.; Estrada, A.F.; Al-Babili, S.; Avalos, J. Deviation of the neurosporaxanthin pathway towards beta-carotene biosynthesis in Fusarium fujikuroi by a point mutation in the phytoene desaturase gene. FEBS J. 2009, 276, 4582–4597. [Google Scholar] [CrossRef] [PubMed]
- Kohlmeyer, J. New genera and species of higher fungi from the deep sea (1615–5315 m). Revue de Mycologie 1977, 41, 189–206. [Google Scholar]
- Usami, Y.; Ichikawa, H.; Arimoto, M. Synthetic efforts for stereo structure determination of cytotoxic marine natural product pericosines as metabolites of Periconia sp. From sea hare. Int. J. Mol. Sci. 2008, 9, 401–421. [Google Scholar] [CrossRef] [PubMed]
- Dighton, J.; White, J.F. The Fungal Community: Its Organization and Role in the Ecosystem, 3rd ed.; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Ogbonna, C.N. Production of food colourants by filamentous fungi. Afr. J. Microbiol. Res. 2016, 10, 960–971. [Google Scholar]
- Kaufmann, B.; Christen, P. Recent extraction techniques for natural products: Microwave-assisted extraction and pressurised solvent extraction. Phytochem. Anal. PCA 2002, 13, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Debbab, A.; Aly, A.H.; Proksch, P. Bioactive secondary metabolites from endophytes and associated marine derived fungi. Fungal Divers. 2011, 49, 1–12. [Google Scholar] [CrossRef]
- Gunatilaka, A.L.; Wijeratne, E.K. Natural Products from Bacteria and Fungi; Elsevier: Amsterdam, The Netherlands, 2000. [Google Scholar]
- Kim, S.-K. Marine Microbiology: Bioactive Compounds and Biotechnological Applications; Wiley & Sons: Weinheim, Germany, 2013; p. 550. [Google Scholar]
- Kjer, J.; Debbab, A.; Aly, A.H.; Proksch, P. Methods for isolation of marine-derived endophytic fungi and their bioactive secondary products. Nat. Protoc. 2010, 5, 479–490. [Google Scholar] [CrossRef] [PubMed]
- Rateb, M.E.; Ebel, R. Secondary metabolites of fungi from marine habitats. Nat. Prod. Rep. 2011, 28, 290–344. [Google Scholar] [CrossRef] [PubMed]





| Primers | Direction | Sequences (5’→ 3’) | Note | Hybrid. T °C | Refs. |
|---|---|---|---|---|---|
| ITS1-F_KYO2 | Forward | TAGAGGAAGTAAAAGTCGTAA | Small sub-unit, ITS 1, 5.8S, ITS 2, Largest sub unit rDNA | 56 | [36] |
| ITS2_KYO2 | Reverse | TTYRCTRCGTTCTTCATC | 47 | ||
| ITS3_KYO2 | Forward | GATGAAGAACGYAGYRAA | 47 | ||
| ITS 1 | Forward | TCCGTAGGTGAACCTGCGG | 55 | [39] | |
| ITS 2 | Reverse | GCTGCGTTCTTCATCGATGC | 55 | ||
| ITS 3 | Forward | GCATCGATGAAGAACGCAGC | 55 | ||
| ITS 4 | Reverse | TCCTCCGCTTATTGATATGC | 55 | ||
| V9G | Forward | TTACGTCCCTGCCCTTTGTA | Large sub unit D1/D2 for basidiomycetous yeast | 55 | [38] |
| LR3 | Reverse | TGACCATTACGCCAGCATCC | 57 | ||
| Cmd 5 | Forward | CCGAGTACAAGGARGCCTTC | Calmodulin, specific for Aspergillus | 52 | |
| Cmd 6 | Reverse | CCGATRGAGGTCATRACGTGG | 52 | ||
| T 10 | Forward | ACGATAGGTTCACCTCCAGAC | β- tubulin, specific for Penicillium | 55 | [38] |
| Bt2b | Reverse | ACCCTCAGTGTAGTGACCCTTGGC | 55 | ||
| EF1-728F | Forward | CATCGAGAAGTTCGAGAAGG | Elongation factor 1 for Trichoderma | 55 | [38] |
| TEF1-LLErev | Reverse | AACTTGCAGGCAATGTGG | 55 |
| Family | Fungal Species | Sampling Site | Gene Accession Number |
|---|---|---|---|
| Water Bodies | |||
| Davidiellaceae | Cladosporium Cladosporioides | Lava flow (−70 m) | JF949719.1 |
| Didymellaceae | Peyronellaea glomerata (syn: Phoma glomerata) | Lava flow (−70 m) | JQ936163.1 |
| Nectriaceae | Nectria haematococca A | Lava flow (−70 m) | XM_003053163.1 |
| Pleosporales Incertae Sedis | Periconia sp. A | Lava flow (−70 m) | HQ608027.1 |
| Periconia sp. B | Lava flow (−70 m) | HQ608027.1 | |
| Sporidiobolaceae | Rhodosporidium paludigenum | Lava flow (−70 m) | AF444493.1 |
| Stachybotryaceae | Myrothecium atroviride | Lava flow (−70 m) | AJ302002.1 |
| Teratospheriaceae | Hortaea werneckii (syn: Cladosporium werneckii) | Lava flow (−70 m) | JN997372.1 |
| Trichocomaceae | Aspergillus sydowii B | Lava flow (−70 m) | KC253961.1 |
| Emericella qinqixianii | TDE outer slope | AB249008.1 | |
| Penicillium brocae NRRL 32599 | TDE outer slope | DQ123642.1 | |
| Penicillium viticola B | TDE inner reef | AB606414.1 | |
| Talaromyces rotundus | TDE inner reef | EU497950.1 | |
| Talaromyces verruculosus | PA inner reef | KC416631.1 | |
| Wallemiaceae | Wallemia sebi | Lava flow (−70 m) | Morphological Identification |
| Living Coral Pocillopora sp. | |||
| Hypocreaceae | Acremonium sp. | PA inner reef | FJ770373.1 |
| Hypocrea koningii | TDE inner reef | JX174420.1 | |
| Trichocomaceae | Aspergillus creber A | TDE inner reef | JN854049.1 |
| Aspergillus creber B | TDE inner reef | JN854049.1 | |
| Aspergillus sydowii A | PA inner reef | JN854052.1 | |
| Eurotium amstelodami | TDE outer slope | FR727111.1 | |
| Penicillium viticola C | PA inner reef | AB606414.1 | |
| Coral Rubbles | |||
| Chaetomiaceae | Chaetomium globosum or Chaetomium murorum | TDE outer slope | JN209898.1 |
| Trichocomaceae | Penicillium herquei | TDE outer slope | JN246042.1 |
| Talaromyces albobiverticillius B | TDE outer slope | JN899313.1 | |
| Talaromyces albobiverticillius C | TDE outer slope | JN899313.1 | |
| Hard Substrate/Rock Substrate | |||
| Nectriaceae | Fusarium equiseti A | Lava flow (−25 m) | JQ936153.1 |
| Fusarium equiseti B | Lava flow (−25 m) | JF311925.1 | |
| Fusarium equiseti C | Lava flow (−25 m) | JQ936153.1 | |
| Nectria haematococca B | Lava flow (−25 m) | XM_003053163.1 | |
| Pleosporaceae | Epicoccum sorghi (syn: Phoma sorghina; Peyronellaea stemphylioides) | Lava flow (−25 m) | KC106717.1 |
| Sordariomycetes | Nigrospora sphaerica (or Env. sample from marine air) | TDE outer slope | KC505176.1 |
| Sporidiobolaceae | Rhodotorula mucilaginosa | TDE outer slope | KC515367.1 |
| Trichocomaceae | Penicillium citrinum | Lava flow (−25 m) | EU030332.1 |
| Penicillium viticola A | Lava flow (−25 m) | AB606414.1 | |
| Ustilaginaceae | Sporisorium exsertum | TDE outer slope | JN367293.1 |
| Xylariaceae | Biscogniauxia sp. | PA inner reef | FJ884075.1 |
| Whalleya microplaca A | TDE outer slope | JQ760548.1 | |
| Sordariomycete (or Whalleya microplaca B) | TDE outer slope | FJ416301.1 | |
| Sediments | |||
| Didymosphaeriaceae | Paraconiothyrium variabile | TDE outer slope | JQ936271.1 |
| Hypocreaceae | Trichoderma atroviride | TDE outer slope | KC008065.1 |
| Trichocomaceae | Talaromyces albobiverticillius A | TDE outer slope | JN899313.1 |
| Fungal Isolates | Approximate Hues | Fungal Isolates | Approximate Hues | |||||
|---|---|---|---|---|---|---|---|---|
| Isolates with Intense Hues (Purple/Red/Maroon) | Isolates with Orange Hues | |||||||
| Broth | EC | IC | Broth | EC | IC | |||
| Acremonium sp. | Penicillium viticola A | |||||||
| Talaromyces albobiverticillius A | Penicillium viticola B | |||||||
| Talaromyces albobiverticillius B | Epicoccum sorghi | |||||||
| Talaromyces albobiverticillius C | Penicillium brocae NRRL 32599 | |||||||
| Aspergillus sydowii A | Penicillium herquei | |||||||
| Aspergillus creber A | Aspergillus sydowii B | |||||||
| Aspergillus creber B | Chaetomium globosum or C. murorum | |||||||
| Emericella qinqixianii | Penicillium viticola C | |||||||
| Trichoderma atroviride | Penicillium citrinum | |||||||
| Biscogniauxia sp. | Hypocrea koningii | |||||||
| Paraconiothyrium variabile | ||||||||
| Myrothecium atroviride | ||||||||
| Isolates with Yellow Hues | Isolates with Green/Brown Hues | |||||||
| Broth | EC | IC | Broth | EC | IC | |||
| Peyronellaea glomerata | Talaromyces verruculosus | |||||||
| Eurotium amstelodami | Talaromyces rotundus | |||||||
| Rhodosporidium paludigenum | Wallemia sebi | |||||||
| Periconia sp. A | Sporisorium exertum | |||||||
| Periconia sp. B | Hortea werneckii | |||||||
| Rhodotorula mucilaginosa | Whalleya microplaca A | |||||||
| Fusarium equiseti A | Whalleya microplaca B | |||||||
| Fusarium equiseti B | Nigrospora sphaerica or Env. sample | |||||||
| Fusarium equiseti C | from marine air | |||||||
| Nectria haematococca A | Cladosporium cladosporioïdes | |||||||
| Nectria haematococca B | ||||||||
| T. albobiverticillius | Sample | Peaks in the UV Region (nm) | Peaks in the Visible Region (nm) | ||
|---|---|---|---|---|---|
| 200–250 | 250–300 | 300–400 | >400 | ||
| A | IC | 235 | 286 | 362 | 422, 425, 511, 525 |
| EC | 265 | 365 | 458, 469.8, 480 | ||
| B | IC | 232 | 268, 292 | 410, 440, 460, 500 | |
| EC | 288 | 412, 524, 532 | |||
| C | IC | 222 | 283 | 385 | 520–580 |
| EC | 283 | 370, 385 | 436 | ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fouillaud, M.; Venkatachalam, M.; Llorente, M.; Magalon, H.; Cuet, P.; Dufossé, L. Biodiversity of Pigmented Fungi Isolated from Marine Environment in La Réunion Island, Indian Ocean: New Resources for Colored Metabolites. J. Fungi 2017, 3, 36. https://doi.org/10.3390/jof3030036
Fouillaud M, Venkatachalam M, Llorente M, Magalon H, Cuet P, Dufossé L. Biodiversity of Pigmented Fungi Isolated from Marine Environment in La Réunion Island, Indian Ocean: New Resources for Colored Metabolites. Journal of Fungi. 2017; 3(3):36. https://doi.org/10.3390/jof3030036
Chicago/Turabian StyleFouillaud, Mireille, Mekala Venkatachalam, Melissa Llorente, Helene Magalon, Pascale Cuet, and Laurent Dufossé. 2017. "Biodiversity of Pigmented Fungi Isolated from Marine Environment in La Réunion Island, Indian Ocean: New Resources for Colored Metabolites" Journal of Fungi 3, no. 3: 36. https://doi.org/10.3390/jof3030036









