1. Introduction
Among the nanostructured materials, the mesoporous silica nanoparticles (MSNs) are a new generation of inorganic platforms for bioapplications. Due their physico-chemical properties such as low toxicity and high drug loading capacity, they are used in controlled and targeted drug delivery, and safe and efficient gene delivery systems [
1,
2].
There are several types of MSNs; one of them is the Mobil Composition of Matter No. 41 (MCM-41) developed in 1996 by researchers at Mobil Oil Corporation [
3]. The MCM-41 nanoparticles possess unique physico-chemical properties, an ordered pore network structure with hundreds of empty channels, tunable pore size (2 to 10 nm diameter of mesopores), high pore volume (~1 cm
3·g
−1), and high surface area (1200 m
2·g
−1) [
2]. Together, these properties allow for the easy functionalization of the silanol containing groups on a surface that is able to absorb or encapsulate relatively large amounts of bioactive molecules [
4].
New approaches show the functionalization of the silanols groups with cross-linked biodegradable polymers has been highlighted as a promising way of obtaining versatile and multifunctional MSNs.
The MSNs can be linked to metals ions to enhance the biological applications and design the copper metal with aminotriazole functionalizing MSNs that inhibits antioxidant defenses and catalyzes the activation of lethal reactive oxygen species (ROS) using the framework of copper ions to kill cancer [
5]. Cu metal was also impregnated in the framework of MSNs for enhancing the doxorubicin (Dox) loading through coordination interactions, which facilitates the release precisely in the tumoral acidic environment [
6].
Furthermore, the biodegradable cationic polymer-modified MSNs nanoparticles have attracted great interest because of the high density of cationic charges on the surface that could be used as nonviral vectors [
7] and drug carriers [
8] and can serve as a biocompatible and effective breast cancer-targeting and theranostic agent [
9,
10,
11,
12].
Hung et al. [
9] show the amino-functionalized MSNs conjugated with glutaraldehyde stabilize enzymes in nanochannels through the formation of covalent imine bonds in prodrug cancer therapy. Another research group reported MSNs functionalized with the monoclonal antibody Herceptin are effective for breast cancer imaging, diagnosis, and treatment [
10].
Liong et al. [
11] reported the preferential uptake of folic acid-conjugated MSNs into pancreatic cancer cell lines was able to release their drug payload into the cytosol, reducing cancer cell survival by 60%. Meng et al. [
12] have demonstrated that MSNs can co-deliver anti-cancer drug and siRNA, thus increasing anti-cancer drug efficacy, mitigating drug resistance via the use of siRNA, and reducing systemic drug release prior to MSNs’ delivery to cancer cells.
The biodegradable cationic polymers are efficient at condensing negatively charged DNA into MSNs nanoparticles via a strong electrostatic interaction and low immunogenicity [
7].
Polycaprolactone (PCL), which is a biodegradable polyester that is easy to synthesize, manipulate, and blend, besides being non-toxic, has been widely used, with amino groups being effective in the drug-controlled and gene-release applications [
13]. PCL easily adheres to surfaces of MSNs by mixing with a wide range of inorganic and organic platforms, enhancing physical–chemical properties and advantageous characteristics such as biodegradability, biocompatibility, and anchoring capacity for bioapplications [
14].
Chitosan (CS) is another biodegradable cationic polymer, which is a structural polysaccharide component naturally occurring in crustacean and insect shells, derived from the partial deacetylation alkaline of chitin, which consists of repeating units of glucosamine and
N-acetylglucosamine [
15].
Several studies have shown that it is one of the most prominent, naturally derived nonviral vectors for gene transfer due to its protonatable amine groups on structure while bearing advantageous characteristics such as biodegradability and biocompatibility [
16].
In order to improve the gene delivery efficiency, different strategies for the structural modification of the biodegradable cationic polymers with MCM-41 nanoparticles have been studied to promote cellular uptake. There are high expectations that gene therapy can cure some diseases caused by genetic disorders, and for this, vehicles for efficient gene delivery are required for release of the payload at target intracellular locations using biodegradable cationic polymers enveloped by MCM-41 nanoparticles. Both the PCL and CS have the ability to form scaffolds, which adhere onto the surface and porous networks of MCM-41.
In this work, we used PCL and CS covalently attached by crosslink molecules to the surface of porous MCM-41 nanoparticles to act as an anchor for siRNA delivery in HeLa cells. The preparation method and main characterizations of the obtained MCM-41 are investigated. The in vitro test of biocompatibility and cytotoxicity suggests the ability of MCM-41 to support passive cellular uptake and consequently indicated its application as potential nanocarrier biomaterials. The results showed that one of the great expectations offered by nanobiotechnology, which through gene therapy can cure some diseases caused by genetic disorders, do not present many treatment alternatives.
3. Results and Discussion
3.1. Thermogravimetric Analysis—TGA
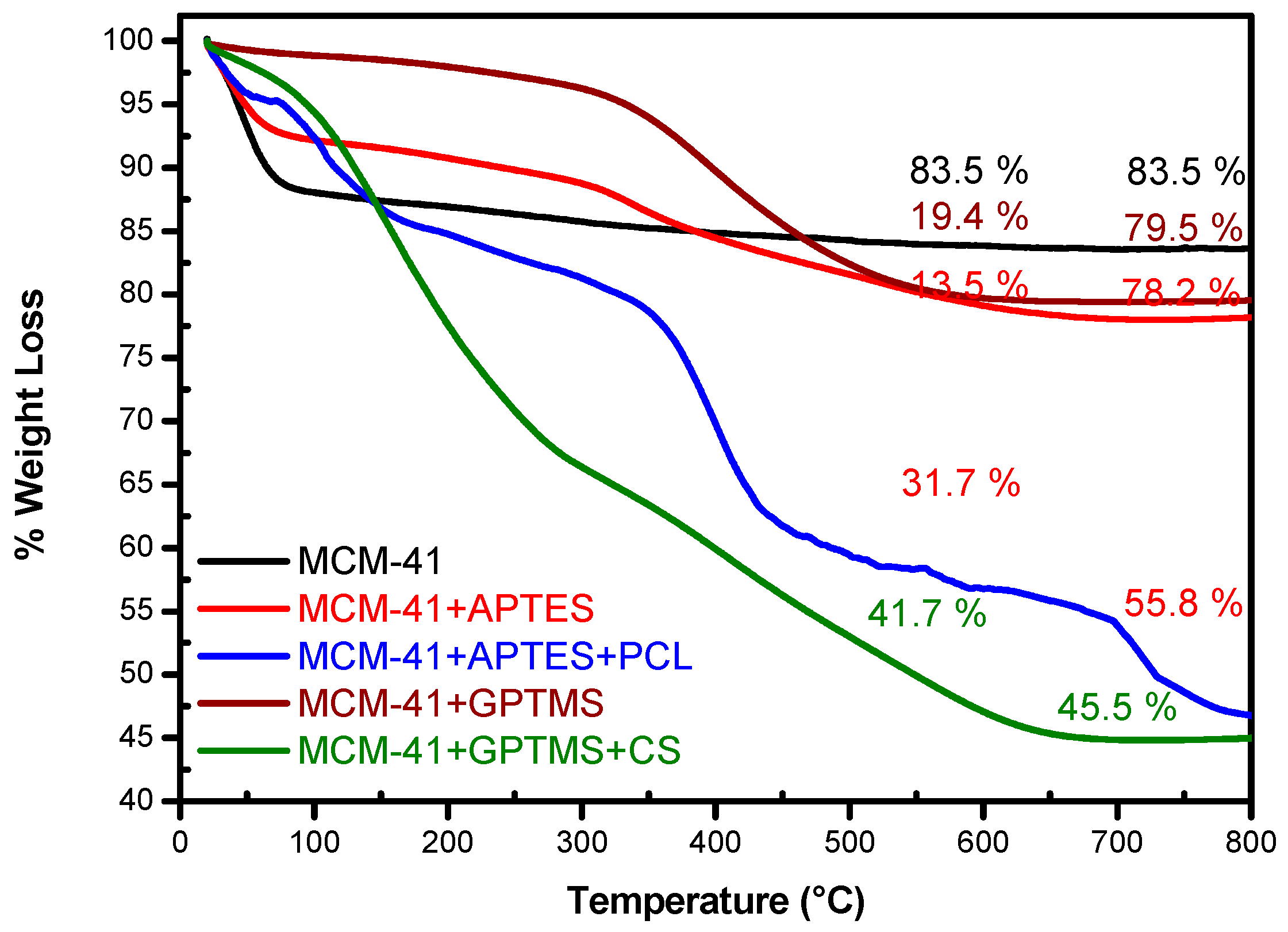
The amount of organic moieties loaded in the MSNs host was determined by thermogravimetry. The TGA curves of all the systems are shown in
Figure 1. The weight losses of MCM-41 and functionalized samples were evaluated in the range of 25–800 °C and are shown in
Table 1. The TGA pattern of MCM-41 demonstrates one weight loss around 12.6% from environment temperature to 150 °C that was attributed to the thermodesorption of physically adsorbed water,
Figure 1. Above this temperature and up to 800 °C, no significant weight loss is verified. In this temperature range, a small weight loss of approximately 3.9% was attributed to the decomposition of residual CTAB surfactant. The total weight loss above 650 °C was 16.5% for pure MCM-41. Studies carried out by Mintzer, and Simanek and Freitas et al., reported similar results to those reported in previous publications [
18,
19,
20].
In case of the functionalized samples, weight loss occurred in distinct regions. The functionalization with APTES that reacted with the silanol groups adding amino groups on the surface of the MCM-41, shows, in TGA analysis, a weight loss of 8.5% in range of 20 °C and 150 °C,
Figure 1. A lower percentage of mass loss is observed in functionalized samples, and a lower water adsorption of these samples can be attributed. This is due to the decrease of free hydroxyl groups present on the surface of the sample when it is chemically modified. Furthermore, MCM-41 was functionalized with groups that promoted a non-polar silica scale. As a consequence, the loss of mass in the first region is much lower for a functionalized than for a pure sample due to surface hydration. In temperature range between 150 °C and 650 °C, this sample loses 13.5% weight, which indicates the total decomposition of the amino groups anchored on the MSNs surface. The residual mass for this sample is 78%; the results indicate that APTES had been successfully grafted onto the surface of MCM-41.
The crosslink APTES serve as bridge between PCL and MCM-41 nanoparticles for the surface functionalization of MCM-41 + APTES with PCL, enhancing the interfacial adhesion between the two phases, adding new functional groups to MCM-41. As shown in TGA analysis, a loss weight of 31.7% could be observed in range between 150 °C and 650 °C, indicating the decomposition of the PCL organic material onto the surface of the MCM-41 + APTES, as can be observed in
Figure 1.
The TGA curves of MCM-41 + GPTMS and MCM-41 + GPTMS + CS samples are shown in
Figure 1, and their percentages of weight loss are shown in
Table 1. The functionalization with GPTMS, which reacted with the silanol groups on the surface of the MCM-41 adding epoxy groups, owing to the characteristic by new functional groups to MCM-41, and samples grafted with CS shows, in TGA analysis, similar behavior to that observed for PCL functionalized samples. In temperature range between 150 °C and 650 °C, this sample loses 19.4% weight, which indicates the total decomposition of the epoxy and glycidoxypropyl groups as related by Hoşgör et al. [
21]. This result indicated that GTPMS had been successfully grafted onto the surface of the MCM-41.
The MCM-41 + GPTMS surface functionalization with CS, shown in TGA analysis, a weight loss of 13.1% in range between 20 °C and 150 °C; between 150 °C and 650 °C, this sample lost 41.7% weight and the total weight loss in temperature above 650 °C was 54.8%, and the residual mass was 45.2%, indicating the decomposition of CS-Suc coating onto the surface of the MCM-41 + GPTMS. TGA results may be associated with the degradation of the polymer chains from MSN surface.
3.2. Fourier Transform Infrared Spectroscopy—FTIR
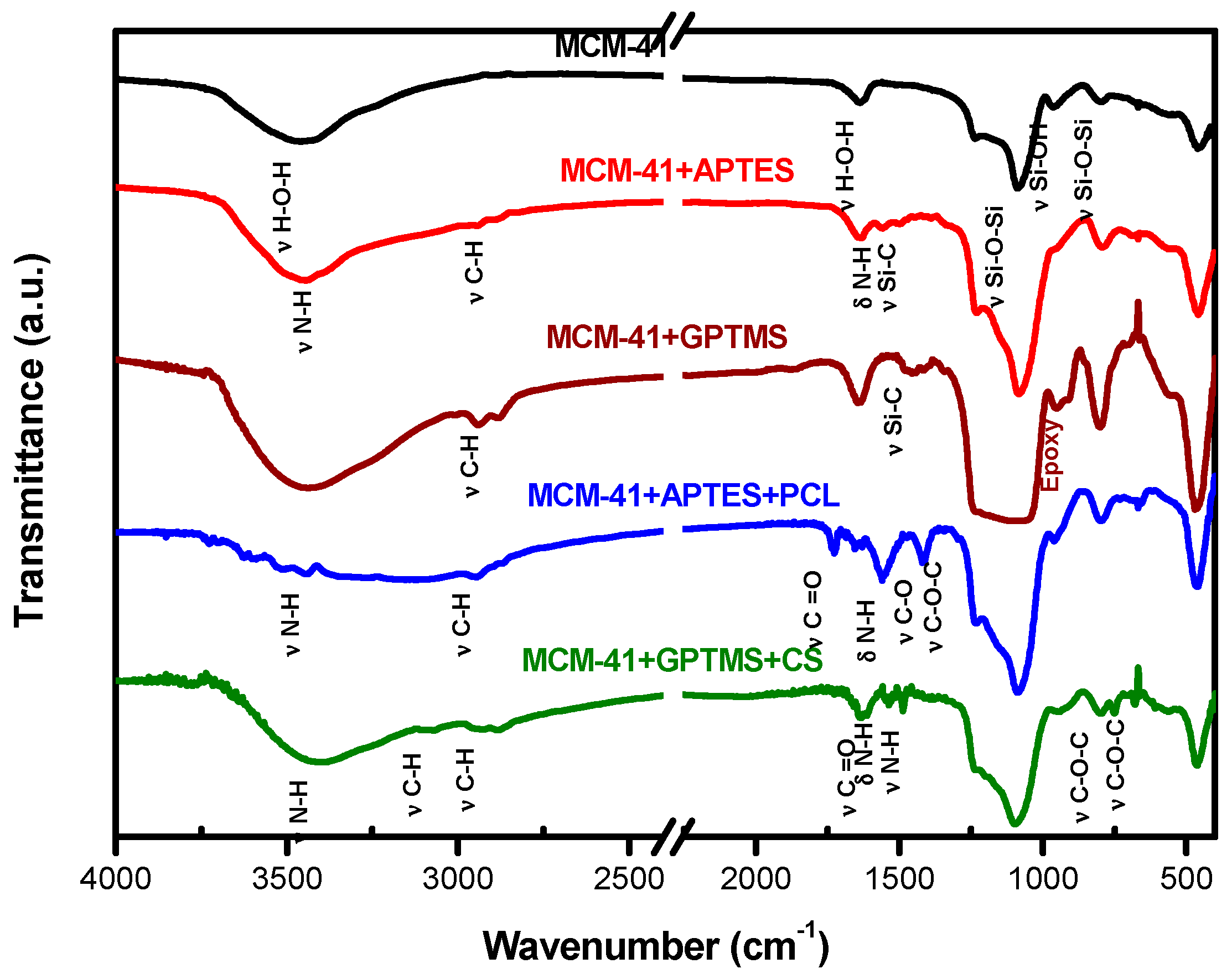
FTIR spectra are a useful tool with which to identify the presence of functional groups in compounds according to the each specific chemical bond.
Figure 2 shows the FTIR spectra of MCM-41, MCM-41 + APTES, and MCM-41 + APTES + PCL samples. Spectrum of MCM-41 shows peak of hydroxyl stretching band for both silanol Si–O–H and the water hydroxyls at 3465 cm
−1, in addition to the peak of H–OH water twisting band at 1640 cm
−1. The Si–O vibration and Si–O bond stretching of surface Si–OH groups are shown at 1090 cm
−1 and 967 cm
−1, respectively, indicating that they are mainly composed of a silica network [
21].
For functionalized samples, additional peaks can be observed; in the first step, in which APTES was grafted into MCM-41 surface, peaks are observed at 2963 cm
−1 and 2886 cm
−1 due to stretching vibration of C–H bond and a peak is observed at 1569 cm
−1 related to the N–H deformation modes of the amine groups, as expected after the amination process [
22]. There is a modification in the 950 cm
−1 peak (Si–OH), showing less definition than in the MCM-41 spectrum. This result indicates that APTES has grafted successfully into the surfaces of the MCM-41.
The spectrum of the second step of functionalization, which refers to surface modification, revealed typical peak for PCL related to 2963 cm
−1 and 2886 cm
−1 due to stretching vibration of C–H bond, peak at 1723 cm
−1 related to stretching vibration of C=O, and asymmetric and symmetric C–O–C stretching vibration around 1240 cm
−1 and 1160 cm
−1, respectively, as related in the literature [
23,
24].
In addition to the bands related to the fundamental functional groups of the silica, the CS functionalized silica samples presented other bands that are assigned to the organic groups bonded to the material surface, as shown in
Figure 2. For MCM-41 + GPTMS and MCM-41 + GPTMS + CS samples, additional peaks can be observed; in the first step, in which GPTMS was anchored into MCM-41 surface, the presence of peaks at 2940 cm
−1 and 2860 cm
−1 due to stretching vibration of C–H bond are observed. Indeed, the peak at 1445 cm
−1 corresponding to Si–C bond stretching present on glycidoxypropyl group, and the peak at 901 cm
−1 due to epoxy group, are identified, indicating that GPTMS has grafted successfully onto the surfaces of the MCM-41 as related in [
25].
The spectrum of the second step of functionalization, which refers to surface modification with chitosan-succinate, revealed typical peaks at 3068 cm
−1, 2940 cm
−1, and 2860 cm
−1 due to stretching vibration of C–H bond. Carboxylic carbonyl stretching in the range of peaks at 1710–1720 cm
−1 is identified, indicating the formation of amide links with succinate moieties. Moreover, the presence of peak around 1660–1670 cm
−1 could be observed, which was attributed to amide carbonyl stretching, the peak of 1536 cm
−1 due to the deformation of N–H (Amine I), the peak of 1467 cm
−1 belonging to the stretching vibration of N–H (amide III), and peaks at 750 cm
−1 and 680 cm
−1 due to stretching vibration of C–O–C bond from the saccharide structure β1-4 as related in [
26].
Therefore, the FTIR results demonstrate that organic moieties have been successfully grafted onto the MCM-41 surface.
3.3. Nitrogen Adsorption/Desorption
The physical properties of these MSNs like pore volume, pore size distribution, and specific surface area were measured by N
2 adsorption–desorption. The surface area of MCM-41, pore volume, and pore size decreased as the extent of modification increased by different steps of functionalization, as shown in
Table 2.
MCM-41 exhibited a sharp step of capillary condensation in primary mesopores at the relative pressure range 0.2 < P/P0 < 0.4, indicating the narrow pore size distribution of the material.
The adsorption and desorption isotherms of N
2 on sample MCM-41 show the typical type IV isotherm according to the IUPAC nomenclature (
Figure 3). At the adsorption branch, the adsorbed amount increased gradually with an increase in relative pressure by adsorption. The desorption branch of the isotherm coincides with the adsorption branch, and the surface area is 987.0 m
2·g
−1 (
Table 2).
The functionalization of the surfaces of MCM-41 with APTES leads to a decrease in the surface area (352.2 m
2·g
−1); the same is observed in the functionalization of the surfaces of MCM-41 + APTES with PCL (45.9 m
2·g
−1). This decrease evidences that the pore wall is indeed covered by organic moieties added in different steps of chemical modifications. Correspondingly, a decrease of pore volume of 0.299 to 0.152 cm
3·g
−1 is observed, which is evidence that organic material is also located within the pores of the matrices (
Table 2). So, after modification, the surface area and pore volume decreased significantly. The extent of such a decrease was dependent on the size of bonded groups, which is expected. These results are in line with reported by Lara et al. [
20].
Similar behavior was observed for samples modified with GPTMS and CS, as shown in
Figure 3 and
Table 2. The functionalization of the surfaces of MCM-41 with GPTMS leads to a decrease in the surface area (411.8 m
2·g
−1); the same is observed in the functionalization of the surfaces of MCM-41 + GPTMS with CS (12.0 m
2·g
−1). This decrease evidences that the pore wall is indeed covered by organic moieties added in different steps of chemical modifications. Correspondingly, a decrease of pore volume of 0.299 to 0.116 cm
3·g
−1 is observed, which is evidence that the organic material is also located within the pores of the matrices (
Table 2).
3.4. Elemental Analysis—CHN
The CHN analysis of bare MSNs matrices and the functionalized ones were used to quantify the organic molecules anchored to its surfaces. The results are shown in
Table 3. The carbon and nitrogen concentrations of functionalized MSNs increased significantly in relation to the bare MSNs, indicating that the bare matrices were successfully functionalized; these results are in line with reports in literature [
17,
19].
MCM-41 + APTES sample presents elemental contents of 8.44% of carbon, 2.99% of hydrogen, and 3.2% of nitrogen. The increasing percentages of carbon, hydrogen, and nitrogen are due to presence of aminopropyl groups functionalized in MCM-41, which is in line with [
19,
20].
The 24.66%, 4.62%, and 2.33% of carbon, hydrogen, and nitrogen, respectively, are the percentages relative to MCM-41 + APTES + PCL sample. They show the increase of carbon and hydrogen, which proves that the PCL was successfully anchored on MCM-41 nanoparticles. The PCL consists of 63.08% of carbon and 9.43% of hydrogen, and these results are consistent with FTIR analyses that shows the peaks bands related to PCL and the BET analyses that show a decrease of surface area, suggesting that the PCL is anchored on MCM-41.
In MCM-41 + GPTMS sample, an increase of 2.0% of carbon and 2.8% of hydrogen is observed. Combining this result with FTIR data, which shows the presence of peak bands of epoxy groups from GPTMS, it is possible to suggest that samples were successfully functionalized with this organic group.
The 18.7%, 2.8%, and 3.2% content of carbon, hydrogen, and nitrogen, respectively, shows the concentration increase of these elements, which demonstrates that the CS was successfully anchored on MCM-41 nanoparticles. The amino groups present in the structure of CS are related by the increase of nitrogen, and are in agreement with the BET analysis that shows the decrease of surface area, indicating the success of functionalizing matrices.
3.5. X-ray Diffraction—XRD
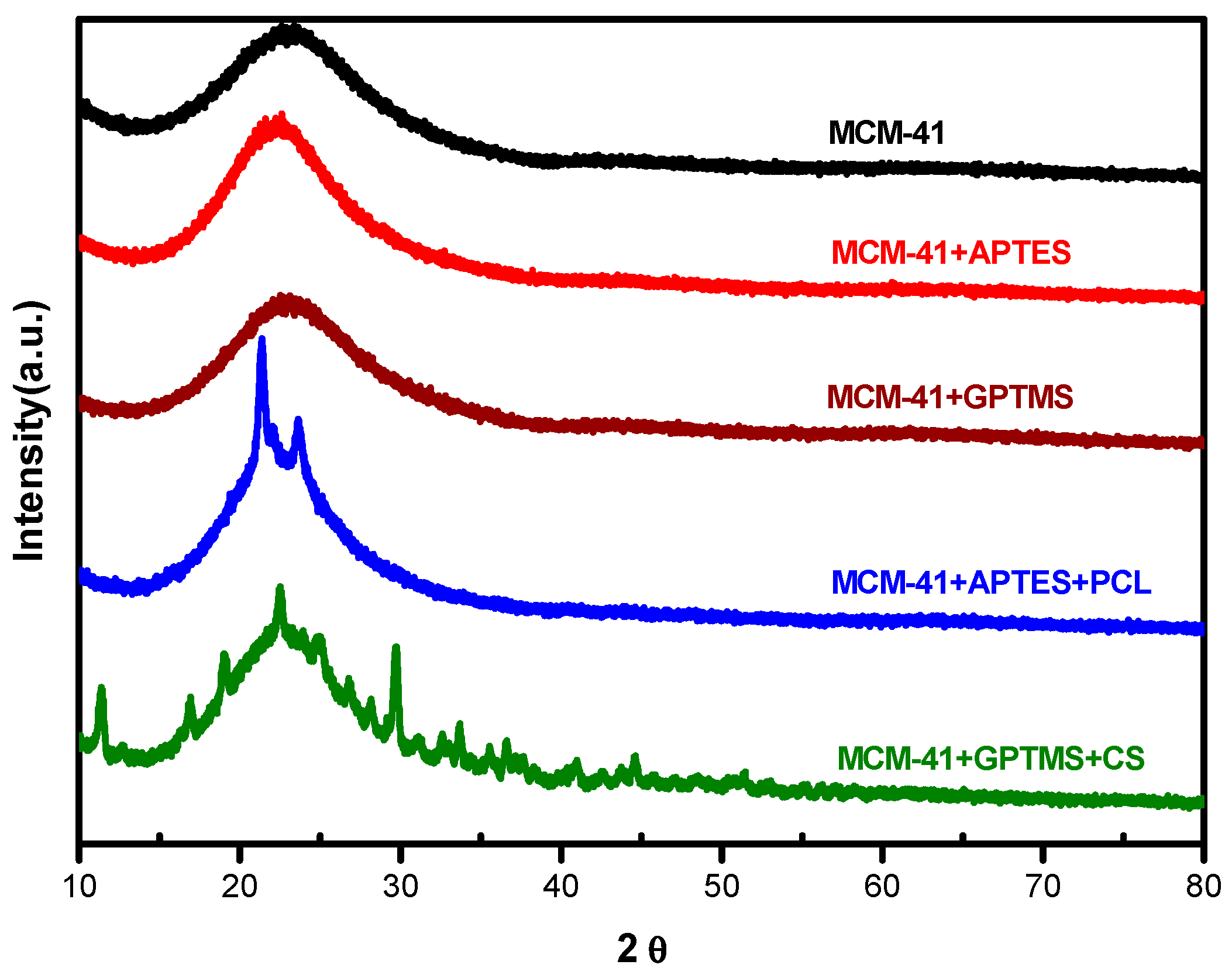
The XRD diffraction patterns (
Figure 4) show two characteristic peaks at angles 2θ = 21.4° and 23.8°, corresponding to the (110) and (200) crystallographic planes of semi-crystalline nature of PCL [
27] anchored in the structure of functionalized MCM-41 nanoparticles. These two peaks characteristic of PCL observed in this nanoparticle indicate miscibility among the nanoparticles and PCL, which means that the functionalization with PCL occurs successfully in the presence of the crystalline structure of PCL. The MCM-41 and MCM-41 + APTES nanoparticles do not present crystallographic planes, indicating the absence of the crystalline structure and, therefore, the presence of an amorphous structure.
As reported by Islam et al. [
28], the XRD pattern of Chitosan shows diffraction peaks at 2θ = 10° and 21° that indicate semi-crystalline structure; similar peaks are observed for MCM-41 + GPTMS + CS sample; probably, the effect of succinic anhydride in nanoparticles appeases the formation of a semi-crystalline structure of MCM-41 + GPTMS + CS sample (
Figure 4).
3.6. Scanning Electron Microscopy—SEM
The morphological characterization of silica nanoparticles was carried out using SEM technique. Images of MCM-41 showed spherical morphology of most nanoparticles and low heterogeneity among them (
Figure 5). As estimated by the Quantikov Image Analyzer data [
29], the particles present an average mean diameter of approximately 110 ± 20 nm (in a total of 161 measurements). As can be seen, the spherical shape was not altered by the presence of organic moieties (
Figure 5b,c), although the particle size distribution was slightly modified.
In the case of MCM-41 + APTES + PCL sample (
Figure 5b), the histogram shows an average diameter of 130 nm (in a total of 124 measurements) and the histogram of MCM-41 + GPTMS + CS sample (
Figure 5c) shows an average diameter of 185 nm (in a total of 75 measurements). The functionalization process with APTES + PCL and GPTMS + CS increased the size of MCM-41 nanoparticles. The MCM-41 nanoparticles have large surface areas and pore volumes that enable the encapsulation and delivery of large quantities of organic molecules through different cell membranes. Furthermore, it has been demonstrated that the mesopores of these nanoparticles can be closed by APTES + PCL, GPTMS + CS, and their surfaces with the same organic matrices capping. As reported by Liberman et al. [
30], the presence of organic moieties in the surface increased the shapes and sizes of nanoparticles. It is worth mentioning that the MCM-41 is within the size expected to be used in biological applications.
3.7. Biological Assays
3.7.1. Cellular Viability
The cytotoxicity is evaluated by MTT assay using fibroblast cell line. As shown in
Figure 6, the fibroblast cells were treated with MCM-41 + APTES + PCL and MCM-41 + GPTMS + CS, and nanoparticles in 100 μg·mL
−1, 75 μg·mL
−1, 50 μg·mL
−1, and 20 μg·mL
−1 concentrations were evaluated. More than 90% of the fibroblast cells are still viable in all concentrations used and showed little statistical difference between MCM-41 + APTES + PCL and MCM-41 + GPTMS + CS nanoparticles, as shown in
Figure 6. The results indicate that nanoparticles have good biocompatibility.
These results indicate that MCM-41 + APTES + PCL and MCM-41 + GPTMS + CS nanoparticles have potential to be used as nonviral vectors. As reported by others authors [
26] using chitosan-silica hollow nanospheres as promising drug nanocarriers consisting of mono-dispersed and pH sensitive suitable for breast cancer therapy, which used concentrations above 100 μg·mL
−1, significant cytotoxicity against the breast cancer cell line (MCF-7 cells) was not shown. As described by Hu et al. [
31], chitosan-capped nanoparticles as pH-Responsive nanocarriers for controlled drug release can exhibit perfect biocompatibility in concentrations above 100 μg·mL
−1.
Thus, the concentrations of nanoparticles that we used in this work are considered ideal for biological applications.
We analyzed the effect of organic molecules on surface of MCM-41. As reported by Kim et al. [
32], the APTES functionalized in nanoparticles were complexed with bone morphogenetic protein-2 (BMP2) plasmid DNA (pDNA) to transfection efficiency in mesenchymal stem cells. The results showed that in concentrations above 100 μg·mL
−1, the presence of APTES is toxic with 50% of viable cells. Other results report [
33] using APTES modified nanoparticles to form dsDNA (double-stranded DNA) complexes, showing that even at a concentration above of 100 μg·mL
−1, the presence of APTES is not toxic with 80% of viable cells. Probably, the toxicity of APTES depends on the cell line. Thus, in this work, the presence of APTES in nanoparticles did not affect cellular viability, and it is considered model for biological applications.
Both PCL and CS are biodegradable cationic polymers, which easily anchor onto surfaces of MCM-41, enhancing the biocompatibility and its presence on the structure of MCM-41, which did not affect the cellular viability as reported in [
31,
33].
3.7.2. In Vitro Cellular Uptake Assays
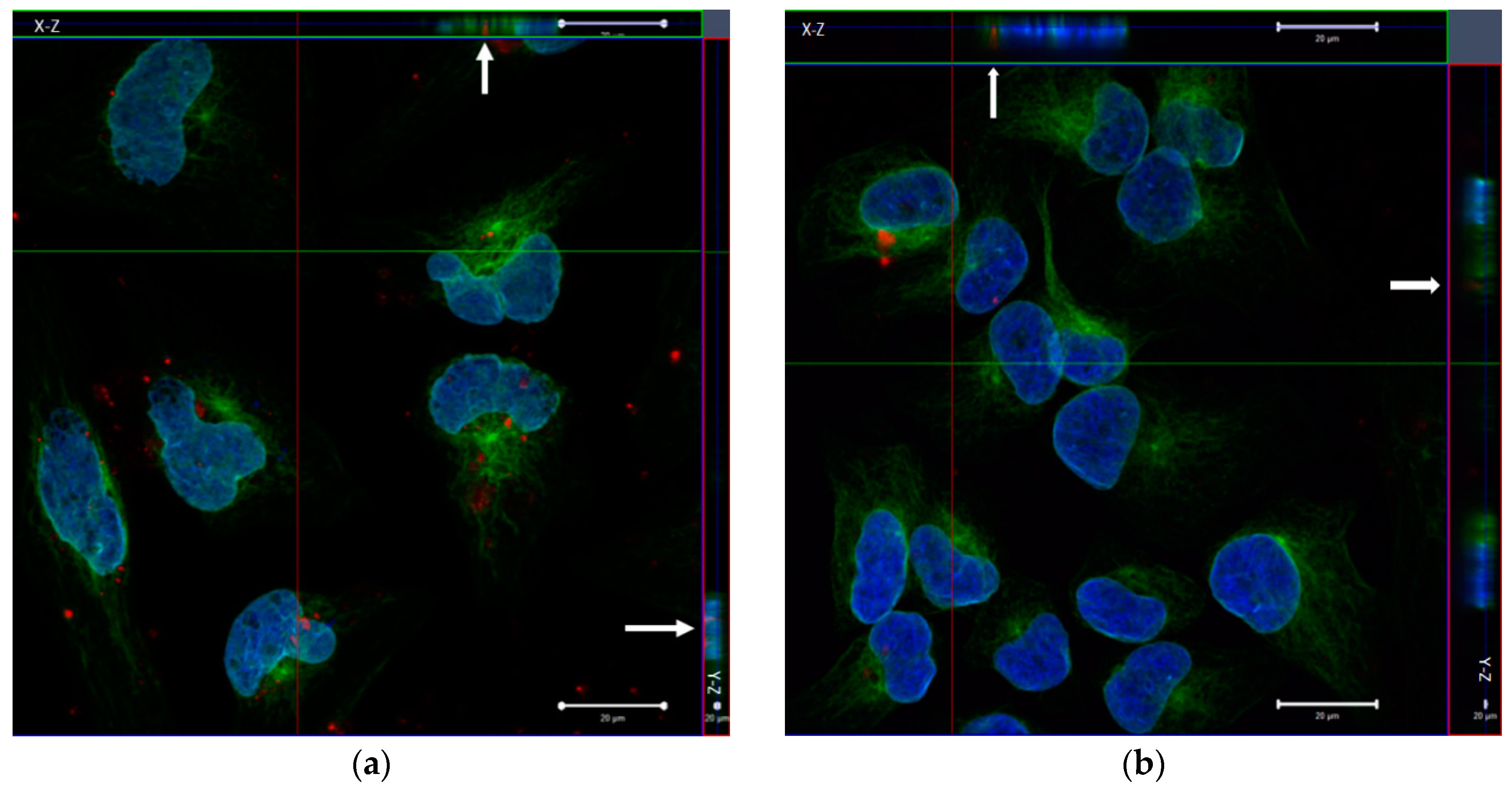
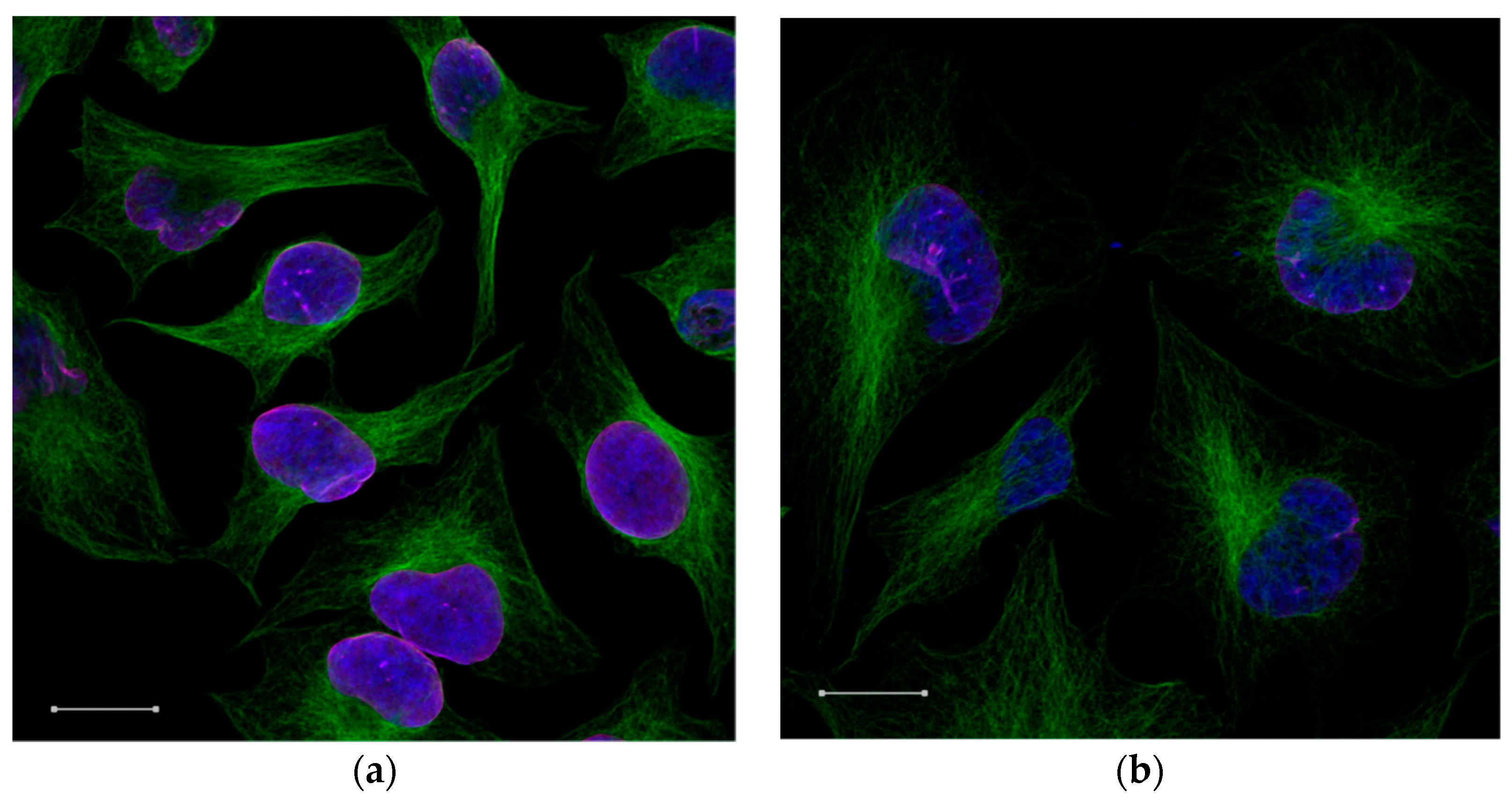
In order to investigate the cellular uptake in HeLa cells, the immunofluorescence assay with confocal microscopy image was used, as shown in
Figure 7. It was observed by the results obtained in the cell viability assay that in concentration of 50 μg·mL
−1 of MCM-41 + APTES + PCL, nanoparticles had high viable cells, which made them favorable for use. We observed the intracellular transport of the nanoparticles by rhodamine-siRNA fluorescent (siRNArod) incorporated in the MCM-41 + APTES + PCL nanoparticles. Cells were labeled with specific antibodies against α-tubulin, a cytoskeletal protein and lamin B1, a nuclear membrane marker to identify the cytosolic and nuclear compartments. The rhodamine-siRNA-MCM-41 + APTES + PCL nanoparticles are bounded and taken up by cells, as shown by fluorescent emission of theses nanoparticles.
The three-dimensional reconstruction of serial confocal immunofluorescence images revealed the intracellular complex localization. Co-localization of rhodamine-siRNA-MCM-41 + APTES + PCL α-tubulin (green) was demonstrated by the appearance of the color yellow (
Figure 7). Together, these findings indicate the internalization and cytosolic localization of MCM-41 + APTES + PCL nanoparticles.
As expected, the surface amino groups allowed for an attractive interaction with the negatively charged siRNArod; in other words, the high positive charge of MCM-41 + APTES + PCL nanoparticles was beneficial for nucleic acid adsorption capacity according Zhang et al. [
34]. According to Xia et al. [
35], PCL showed a lower cytotoxicity and a higher transfection efficiency in HeLa cells, and there was no decrease of the transfection efficiency. These characteristics are favorable for application as gene delivery systems.
The entry of the MCM-41 + APTES + PCL nanoparticles into the cells is evident, since siRNA is adsorbed in these nanoparticles, and it does not cross the cell membrane alone. It was shown that siRNA trapped inside the PCL-coated MCM-41 was efficiently protected against enzymatic degradation. The siRNA could be released into the cytoplasm and escape from the MCM-41 after MCM-41 internalization by A549 cells, as evidenced by fluorescently labeled siRNA and MCM-41 and efficiently knocked-down target genes [
35]. According to Zhang et al. [
36], the siRNA can only be released through systemic delivery using nanoparticles, and it is not readily internalized by cells due to their negative charge (they suffer from poor biological stability and a short half-life).
The same was done for the packaging of siRNA into the 50 μg·mL
−1 of MCM-41 + GPTMS + CS nanoparticles. As shown in
Figure 8, by fluorescent confocal microscopy, fluorescence patterns show the gene transfection in the 50 μg·mL
−1 of MCM-41 + GPTMS + CS nanoparticles. Probably, the siRNA was not being fully protected against degradation by nucleases. As reported in others works [
37,
38], the Chitosan is considered to be a good candidate for a gene delivery carrier with its excellent biocompatibility, biodegradability, low toxicity, and high cationic potential. However, this material shows low transfection efficiency, and it may have been affected by some factors, including chitosan molecular weight, salt form, degree of deacetylation, the pH of the culture medium, and so forth.
4. Conclusions
This study was able to show the surface modification process of MCM-41 with two biodegradable polymers to act as nonviral gene delivery in HeLa cells. The physical–chemical and morphological characterizations, as well as the applicability of functionalized MCM-41 as platforms for gene delivery, were assessed. The results confirmed that MCM-41 was successfully functionalized with PCL, and CS kept its typical morphology and pore arrangement.
MTT analysis in the fibroblast cell line indicated that the presence of materials in concentrations up to 100 µg·mL−1 did not compromise the cell metabolism and, consequently, cell viability. The results confirmed that MCM-41 functionalized with PCL can effectively be used as a nonviral vector in gene delivery systems. The MCM-41 functionalized with CS shows inefficient gene unpacking and therefore low gene transfection efficiency compared with MCM-41 functionalized with PCL. These results are promising for the use of these nanoparticles as siRNA delivery nonviral vectors.