Photodegradation of 1,2,4-Trichlorobenzene on Montmorillonite–TiO2 Nanocomposites
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of the Catalysts
2.2. Characterization Techniques
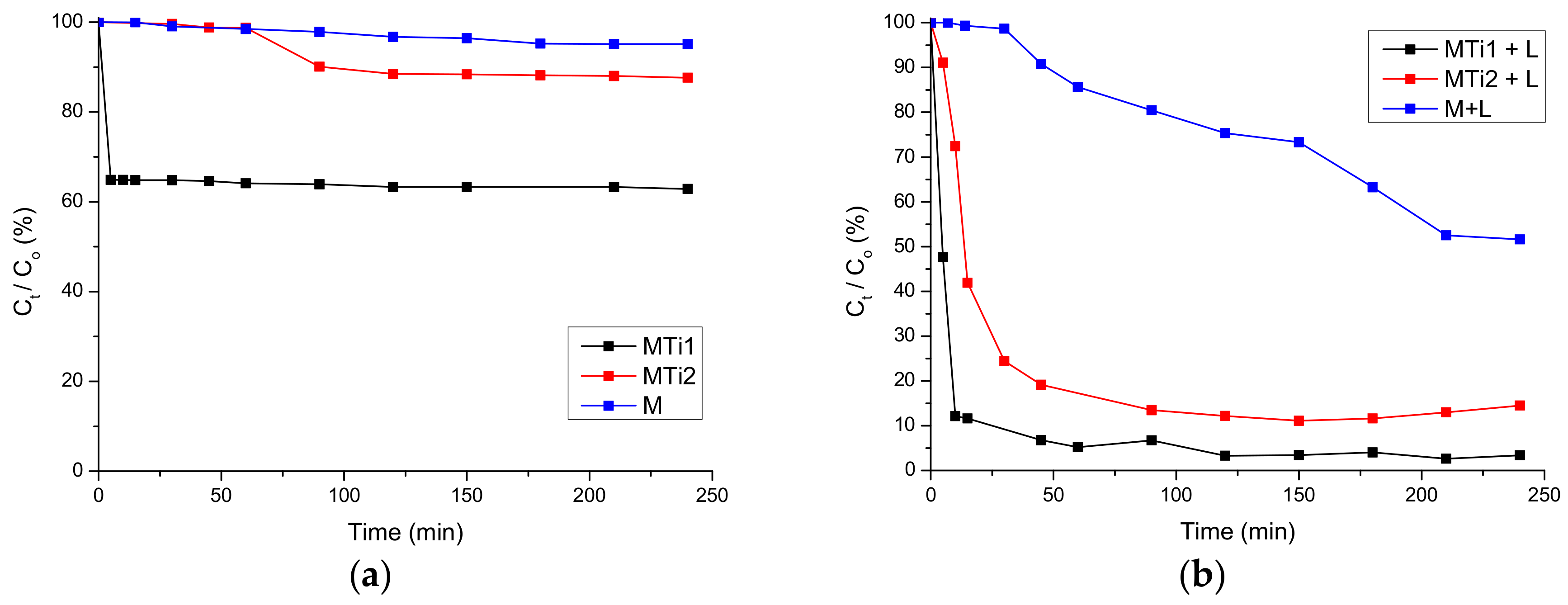
2.3. Reactivity Studies
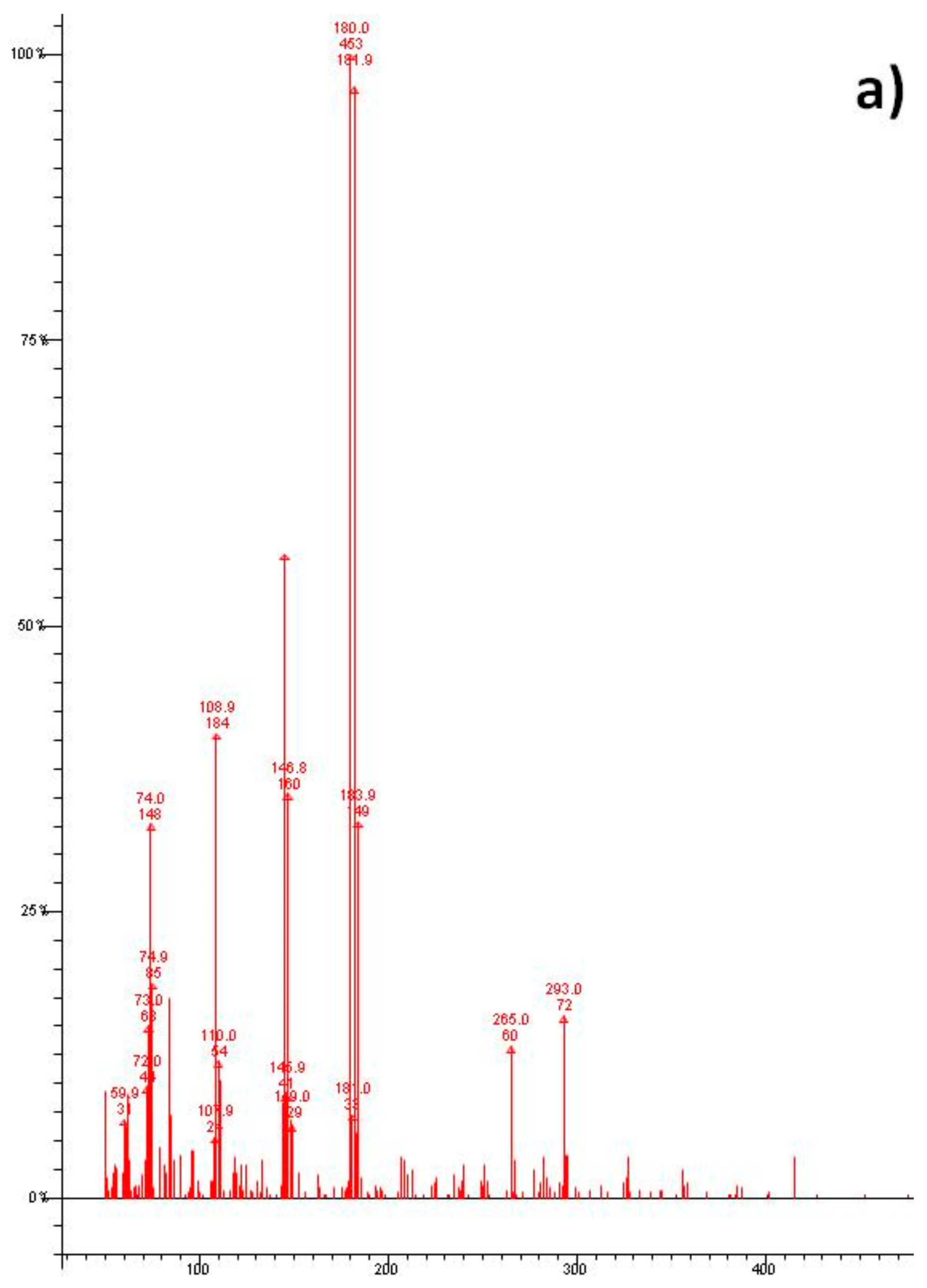
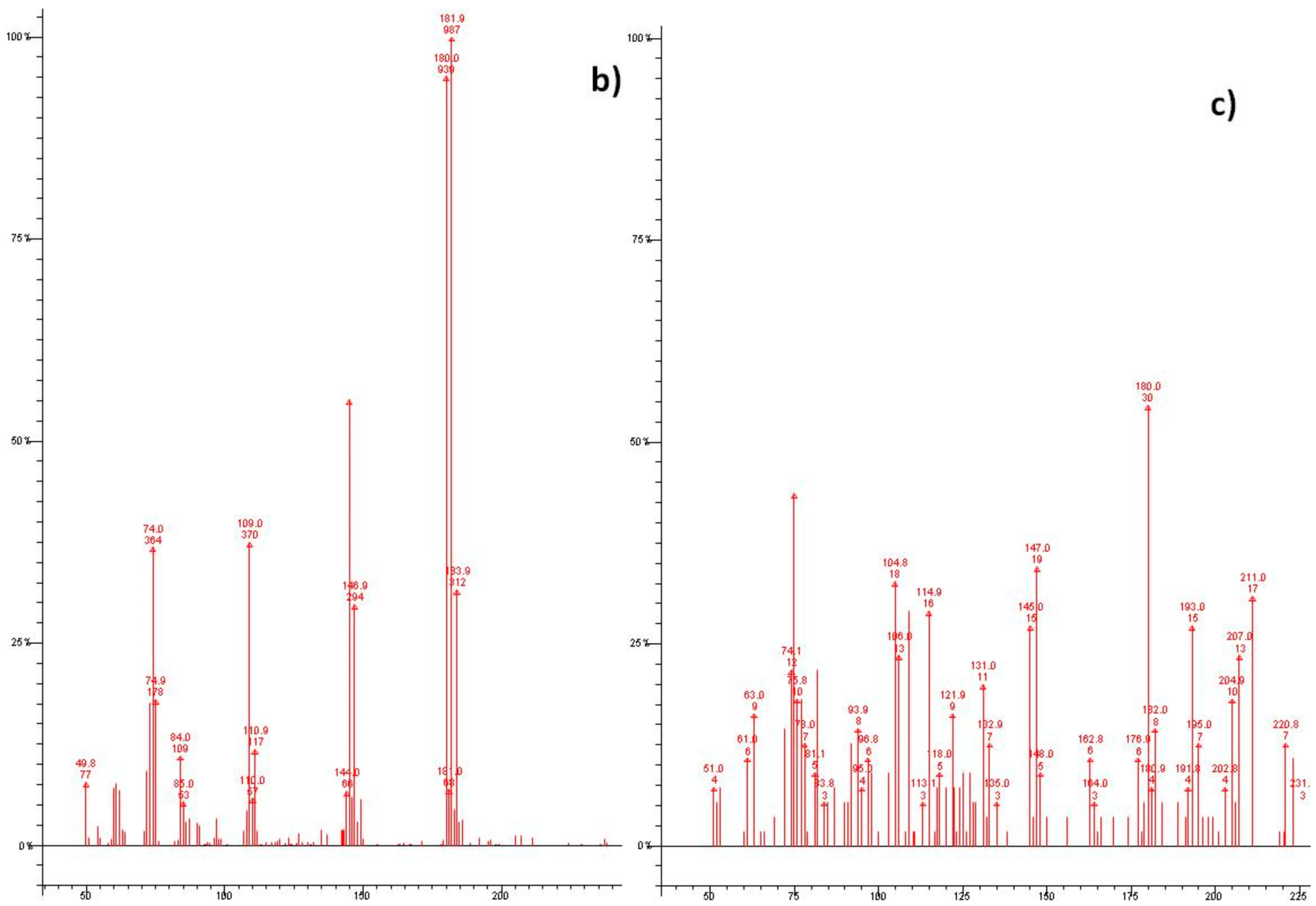
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Adams, J.M.; McCabe, R.W. Clay Minerals as Catalysts. In Handbook of Clay Science; Developments in Clay Science; Bergaya, F., Theng, B.K.G., Lagaly, G., Eds.; Elsevier Ltd.: Amsterdam, The Netherland, 2006; ISBN 9780080457635. [Google Scholar]
- Elbokl, T.A.; Detellier, C. Intercalation of cyclic imides in kaolinite. J. Colloid Interface Sci. 2008, 32, 338–348. [Google Scholar] [CrossRef] [PubMed]
- Gil, A.; Korili, S.A.; Trujillano, R.; Vicente, M.A. (Eds.) Pillared Clays and Related Catalysts; Springer: Berlin, Germany, 2010; ISBN 9781441966698. [Google Scholar]
- Vicente, M.A.; Gil, A.; Bergaya, F. Pillared Clays and Clay Minerals. In Handbook of Clay Science, 2nd ed.; Bergaya, F., Lagaly, G., Eds.; Elsevier: Amsterdam, The Netherlands, 2013; Chapter 10.5; ISBN 9780080993713. [Google Scholar]
- Deng, F.; Zhao, X.; Pei, X.; Luo, X.; Li, W.; Au, C. Sol–Hydrothermal Synthesis of Inorganic Framework Molecularly Imprinted TiO2 Nanoparticle and its Enhanced Photocatalytic Activity for Degradation of Target Pollutant. Sci. Adv. Mater. 2016, 8, 1079–1085. [Google Scholar] [CrossRef]
- Lewis, N.S.; Rosenbluth, M.L. Theory of Semiconductor Materials. In Photocatalysis: Fundamentals and Applications; Serpone, N., Pelizzetti, E., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 1989; pp. 45–99. ISBN 978-0471626039. [Google Scholar]
- Kocí, K.; Matejka, V.; Kovár, P.; Lacny, Z.; Obalová, L. Comparison of the pure TiO2 and kaolinite/TiO2 composite as catalyst for CO2 photocatalytic reduction. Catal. Today 2011, 161, 105–109. [Google Scholar] [CrossRef]
- Mathew, R.; Khan, S.U. Photodegradation of Metolachlor in Water in the Presence of Soil Mineral and Organic Constituents. J. Agric. Food Chem. 1996, 44, 3996–4000. [Google Scholar] [CrossRef]
- Zhang, Y.; Gan, H.; Zhang, G. A novel mixed–phase TiO2/kaolinite composites and their photocatalytic activity for degradation of organic contaminants. Chem. Eng. J. 2011, 172, 936–943. [Google Scholar] [CrossRef]
- Papoulis, D.; Komarneni, S.; Panagiotaras, D.; Stathatos, D.; Christoforidise, K.C.; Fernández–García, M.; Li, H.; Shu, Y.; Sato, T.; Katsuki, H. Three–phase nanocomposites of two nanoclays and TiO2: Synthesis, characterization and photocatalytic activities. Appl. Catal. B 2014, 147, 526–533. [Google Scholar] [CrossRef]
- Mamulová Kutláková, K.; Tokarský, J.; Kovář, P.; Vojtěšková, S.; Kovářová, A.; Smetana, B.; Kukutschová, J.; Čapková, P.; Matějka, V. Preparation and characterization of photoactive composite kaolinite/TiO2. J. Hazard Mater. 2011, 188, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Ray, S.; Sedev, R.; Priest, C.; Ralston, J. Influence of the Work of Adhesion on the Dynamic Wetting of Chemically Heterogeneous Surfaces. Langmuir 2008, 24, 13007–13012. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.C.; Wu, T.X.; Wu, K.Q.; Oikawa, K.; Hidaka, H.; Serpone, N. Photoassisted Degradation of Dye Pollutants. 3. Degradation of the Cationic Dye Rhodamine B in Aqueous Anionic Surfactant/TiO2 Dispersions under Visible Light Irradiation: Evidence for the Need of Substrate Adsorption on TiO2 Particles. Environ. Sci. Technol. 1998, 32, 2394–2400. [Google Scholar] [CrossRef]
- Wang, Q.; Chen, C.C.; Zhao, D.; Ma, W.H.; Zhao, J.C. Change of adsorption modes of dyes on fluorinated TiO2 and its effects on photocatalytic degradation of dyes under visible irradiation. Langmuir 2008, 24, 7338–7345. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.M.; Langford, C.H. Photoactivity of Titanium Dioxide Supported on MCM41, Zeolite X and Zeolite Y. J. Phys. Chem. B 1997, 101, 3115–3121. [Google Scholar] [CrossRef]
- Peng, T.Y.; Zhao, D.; Dai, K.; Shi, W.; Hirao, K. Synthesis of Titanium Dioxide Nanoparticles with Mesoporous Anatase Wall and High Photocatalytic Activity. J. Phys. Chem. B 2005, 109, 4947–4952. [Google Scholar] [CrossRef] [PubMed]
- Miao, S.D.; Liu, Z.M.; Han, B.X.; Zhang, J.L.; Yu, X.; Du, J.M.; Sun, Z.Y. Synthesis and characterization of TiO2–montmorillonite nanocomposites and their application for removal of methylene blue. J. Mater. Chem. 2006, 16, 579–584. [Google Scholar] [CrossRef]
- Hong, H.L.; Jiang, W.T.; Zhang, X.L.; Tie, L.Y.; Li, Z.H. Adsorption of Cr(VI) on STAC–modified rectorite. Appl. Clay Sci. 2008, 42, 292–299. [Google Scholar] [CrossRef]
- Li, Z.H.; Jiang, W.T.; Chen, C.J.; Hong, H.L. Influence of Chain Lengths and Loading Levels on Interlayer Configurations of Intercalated Alkylammonium and Their Transitions in Rectorite. Langmuir 2010, 26, 8289–8294. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.M.; Tang, A.D.; Ouyang, J.; Li, M.; Mann, S. From Natural Attapulgite to Mesoporous Materials: Methodology, Characterization and Structural Evolution. J. Phys. Chem. B 2010, 114, 2390–2398. [Google Scholar] [CrossRef] [PubMed]
- Khataee, A.; Sheydaei, M.; Hassani, A.; Taseidifar, M.; Karaca, S. Sonocatalytic removal of an organic dye using TiO2/Montmorillonite nanocomposite. Ultrason. Sonochem. 2015, 22, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Kun, R.; Mogyorósi, K.; Dékány, I. Synthesis and structural and photocatalytic properties of TiO2/montmorillonite nanocomposites. Appl. Clay Sci. 2006, 32, 99–110. [Google Scholar] [CrossRef]
- Zhang, G.K.; Ding, X.M.; He, F.S.; Yu, X.Y.; Zhou, J.; Hu, Y.J.; Xie, J.W. Low-temperature synthesis and photocatalytic activity of TiO2 pillared montmorillonite. Langmuir 2008, 24, 1026–1030. [Google Scholar] [CrossRef] [PubMed]
- González–Rodríguez, B.; Trujillano, R.; Rives, V.; Vicente, M.A.; Gil, A.; Korili, S.A. Structural, textural and acidic properties of Cu–, Fe– and Cr–doped Ti–pillared montmorillonites. Appl. Clay Sci. 2015, 118, 124–130. [Google Scholar] [CrossRef]
- Rouquerol, F.; Rouquerol, J.; Sing, K. Adsorption by Powders and Porous Solids—Principles, Methodology and Applications; Academic Press: Cambridge, MA, USA, 1998; ISBN 978-0-12-598920-6. [Google Scholar]
- Vicente, M.A.; Belver, C.; Trujillano, R.; Suárez, M.; Bañares, M.A.; Rives, V. Improvement of the properties of natural clays by acid activation. In Applied Study of Cultural Heritage and Clays; Pérez Rodríguez, J.L., Ed.; Biblioteca de Ciencias n° 13; Consejo Superior de Investigaciones Científicas: Madrid, España, 2003; pp. 519–535. ISBN 978-84-00-08197-3. [Google Scholar]
- Hanaor, D.A.H.; Sorrell, C.C. Review of the anatase to rutile phase transformation. J. Mater. Sci. 2011, 46, 855–874. [Google Scholar] [CrossRef]
- Ranade, M.R.; Navrotsky, A.; Zhang, H.Z.; Banfield, J.F.; Elder, S.H.; Zaban, A.; Borse, P.H.; Kulkarni, S.K.; Doran, G.S.; Whitfield, H.J. Energetics of nanocrystalline TiO2. Proc. Natl. Acad. Sci. USA 2002, 99, 6476–6481. [Google Scholar] [CrossRef] [PubMed]
- Porto, S.P.S.; Fleury, P.A.; Damen, T.C. Raman Spectra of TiO2, MgF2, ZnF2, FeF2, and MnF2. Phys. Rev. 1967, 154, 522–526. [Google Scholar] [CrossRef]
- Beattie, I.R.; Gilson, T.R. Single crystal laser Raman spectroscopy. Proc. R. Soc. Lond. A 1968, 307, 407–429. [Google Scholar] [CrossRef]
- Vicente, M.A.; Bañares–Muñoz, M.A.; Toranzo, R.; Gandía, L.M.; Gil, A. Influence of the Ti precursor on the properties of Ti–pillared smectites. Clay Miner. 2001, 36, 125–138. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Everett, D.H.; Haul, R.A.W.; Moscou, L.; Pierotti, R.A.; Rouquerol, J.; Siemieniewska, T. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Barbosa, L.V.; Marçal, L.; Nassar, E.J.; Calefi, P.S.; Vicente, M.A.; Trujillano, R.; Rives, V.; Gil, A.; Korili, S.; Ciuffi, K.J.; et al. Kaolinite–titanium oxide nanocomposites prepared via sol–gel as heterogeneous photocatalysts for dyes degradation. Catal. Today 2015, 246, 133–142. [Google Scholar] [CrossRef]
- Papoulis, D.; Komarneni, S.; Panagiotaras, D.; Stathatos, E.; Toli, D.; Christoforidis, K.C.; Fernandez Garcia, M.; Li, H.; Yin, S.; Sato, T.; et al. Halloysite–TiO2 nanocomposites: Synthesis, characterization and photocatalytic activity. Appl. Catal. B 2013, 132–133, 416–422. [Google Scholar] [CrossRef]
- Papoulis, D.; Komarneni, S.; Nikolopoulou, A.; Tsolis-Katagas, P.; Panagiotaras, D.; Kacandes, H.G.; Zhang, P.; Yin, S.; Satog, T.; Katsuki, H. Palygorskite–and Halloysite–TiO2 nanocomposites: Synthesis and photocatalytic activity. Appl. Clay Sci. 2010, 50, 118–124. [Google Scholar] [CrossRef]
- Lei, S.; Duan, W. Highly active mixed-phase TiO2 photocatalysts fabricated at low temperature and the correlation between phase composition and photocatalytic activity. J. Environ. Sci. 2008, 20, 1263–1267. [Google Scholar] [CrossRef]
- Rives, V. Preparation of titania nanoparticles and relationships between procedures and properties. In New Developments in Metal Oxides Research; Nagy, I., Balogh, A., Eds.; Nova Science Publishers: New York, NY, USA, 2013; Chapter 1; ISBN 9781628081480. [Google Scholar]
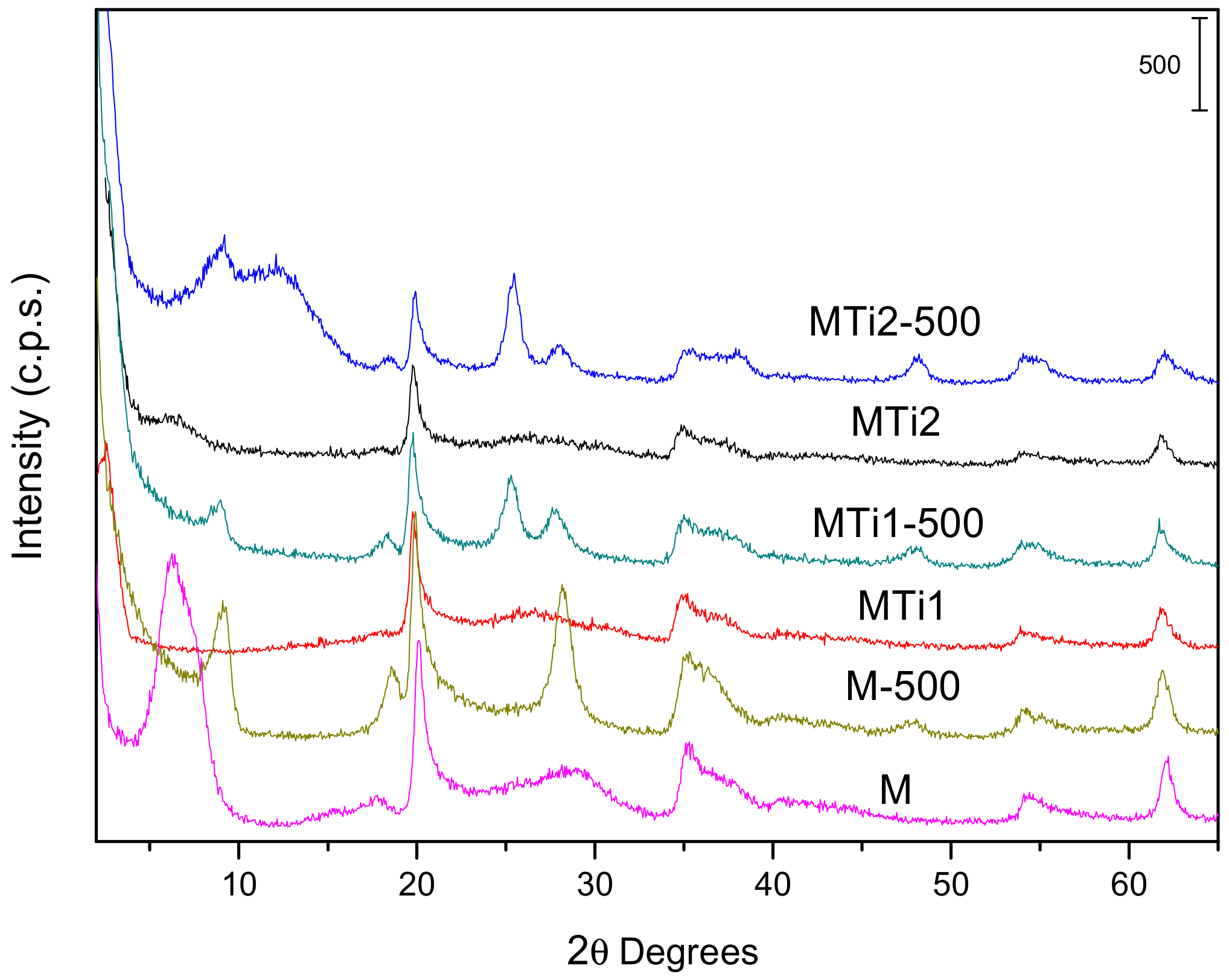
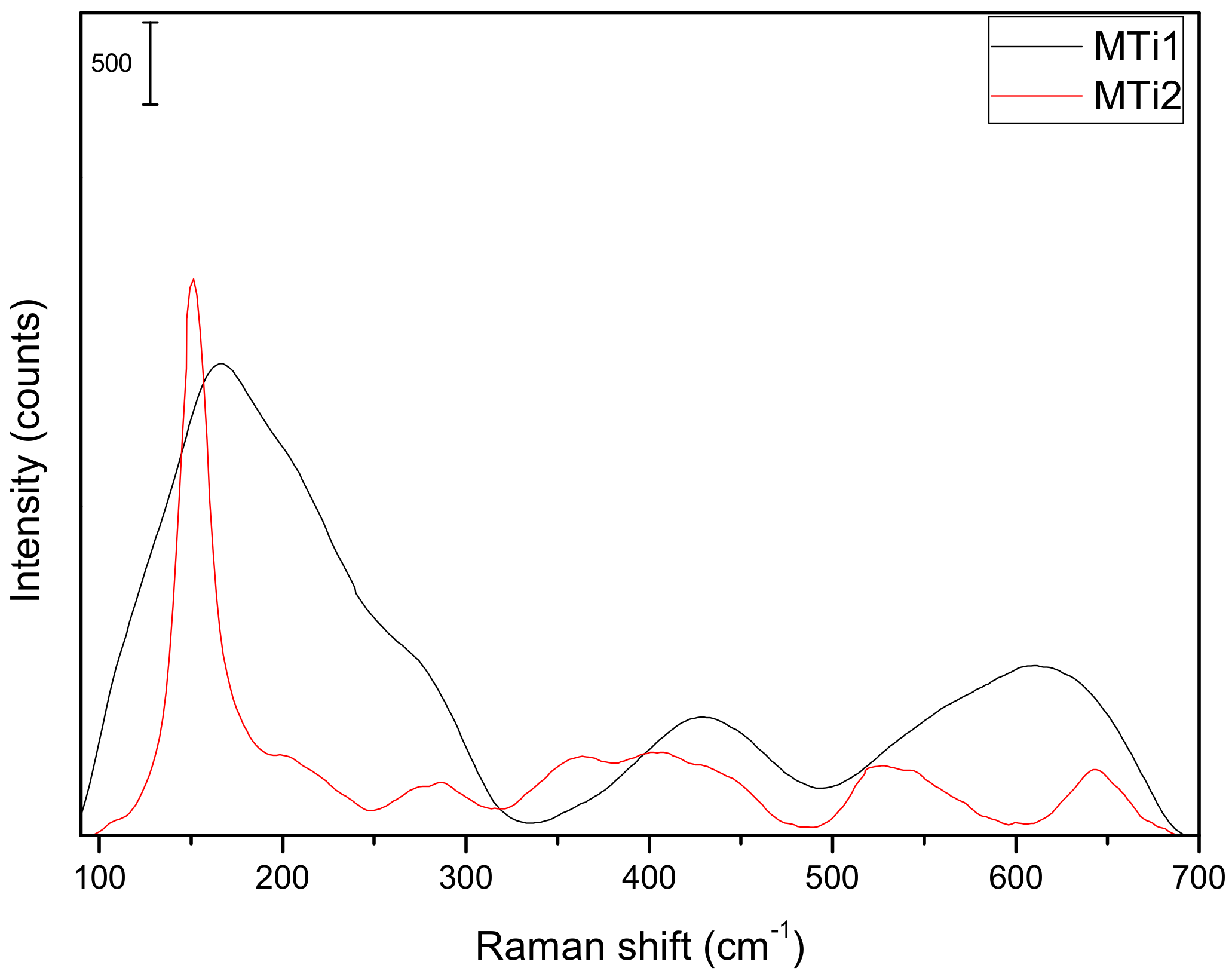
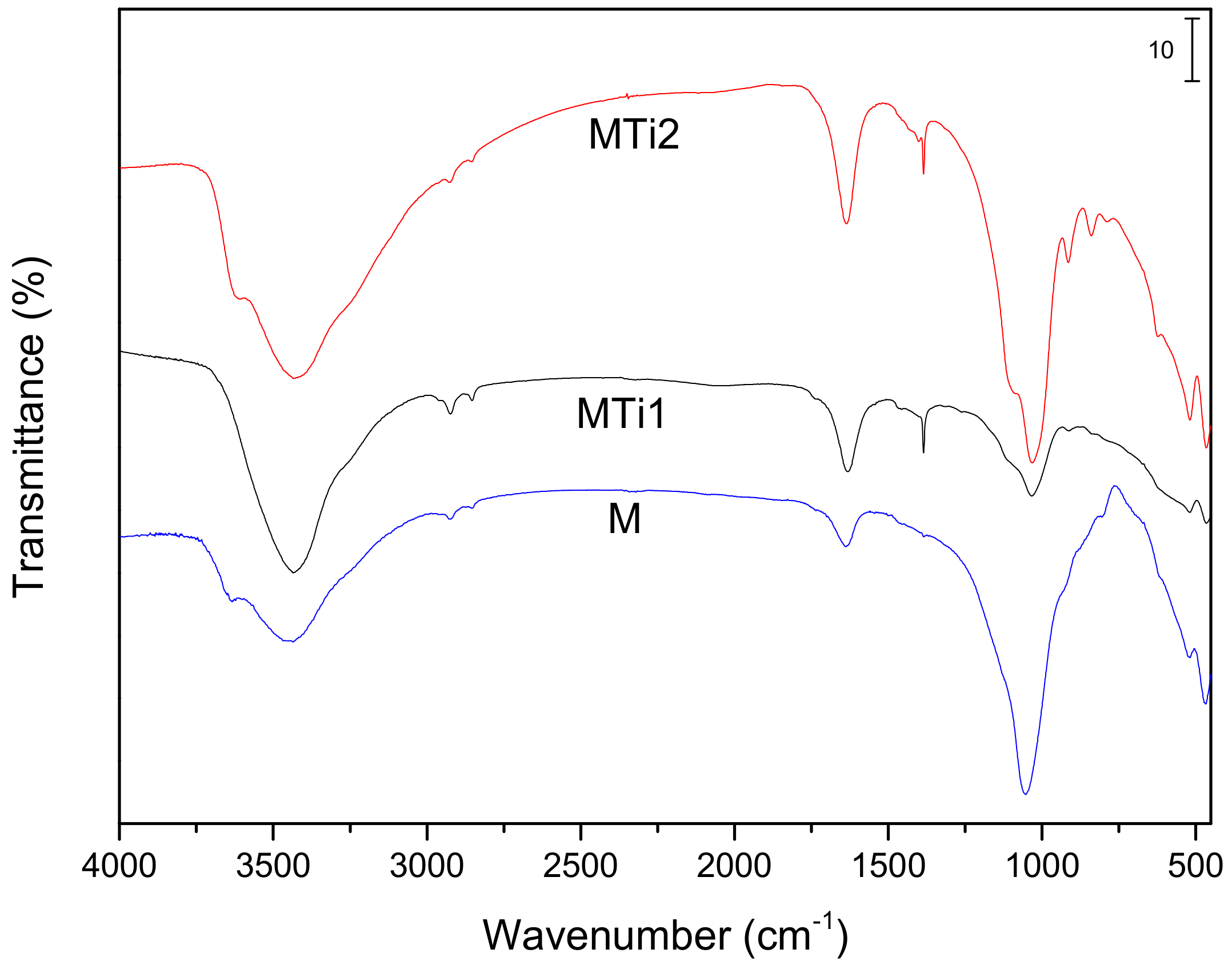
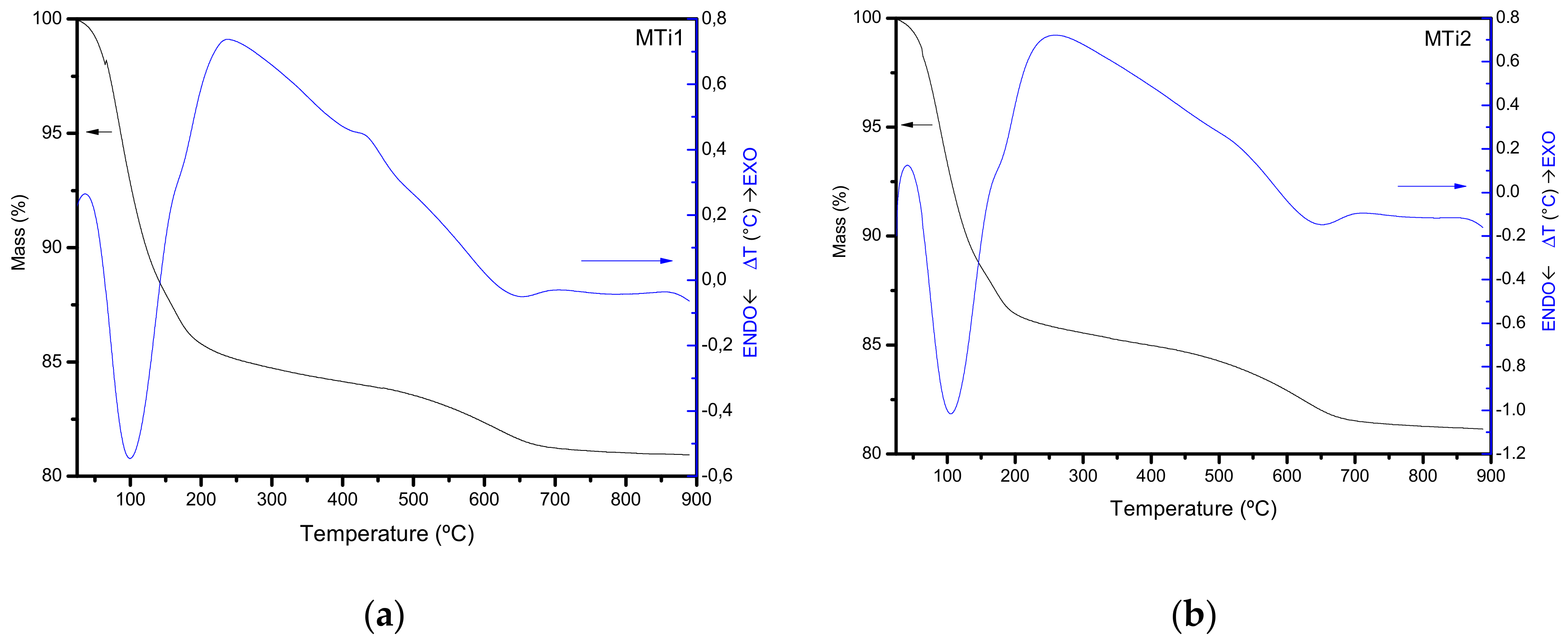
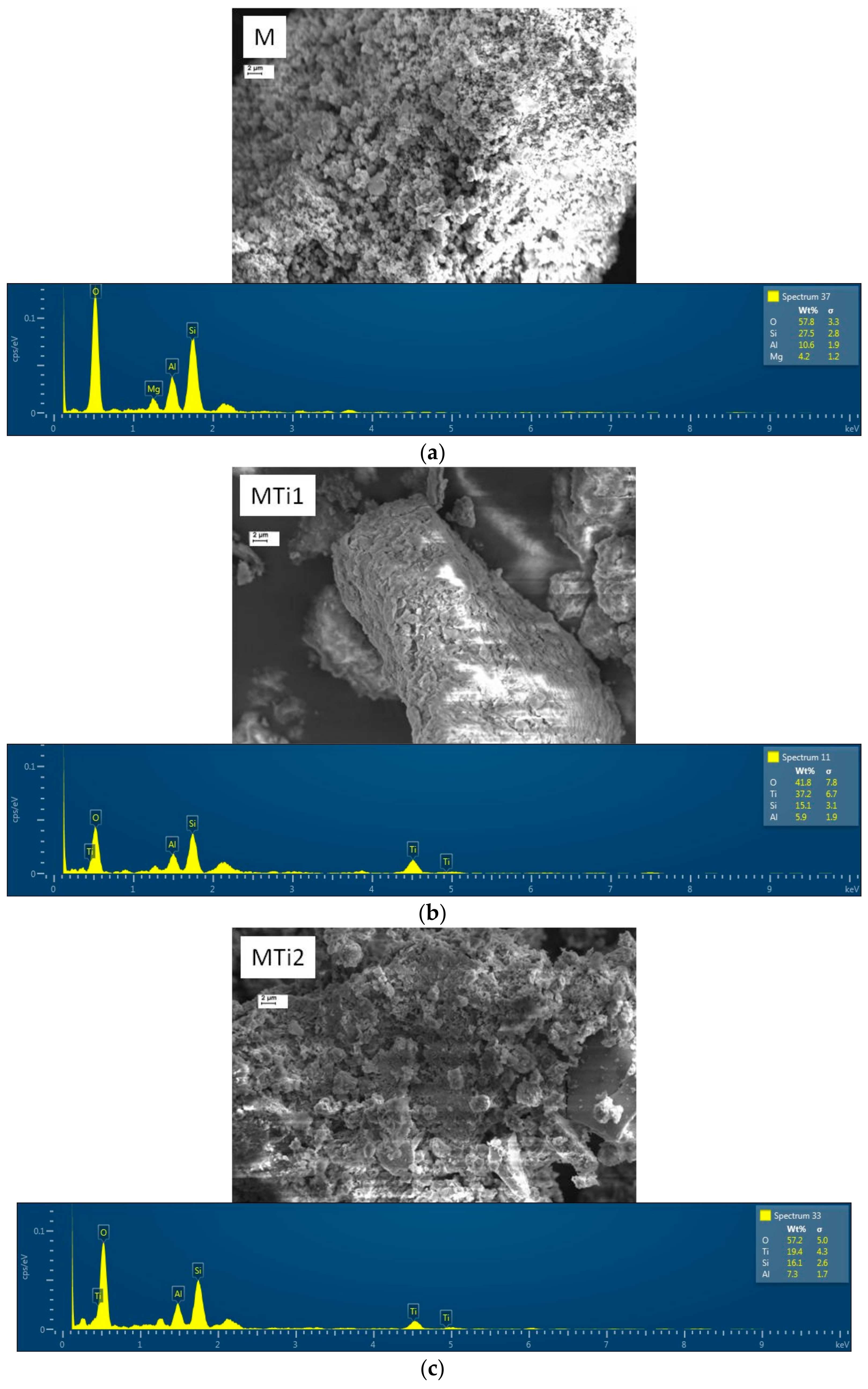
| Sample | SiO2 | Al2O3 | Fe2O3 | MnO | MgO | CaO | Na2O | K2O | TiO2 | Total |
|---|---|---|---|---|---|---|---|---|---|---|
| M | 55.80 | 15.92 | 1.41 | 0.04 | 5.58 | 1.69 | 0.06 | 0.06 | 0.21 | 80.77 |
| MTi1 | 42.65 | 11.48 | 0.98 | 0.04 | 4.08 | 1.18 | 0.17 | 0.07 | 22.71 | 83.36 |
| MTi2 | 49.51 | 13.57 | 1.17 | 0.04 | 4.80 | 1.40 | 0.11 | 0.08 | 14.16 | 84.84 |
| Sample | SiO2 | Al2O3 | Fe2O3 | MnO | MgO | CaO | Na2O | K2O | TiO2 |
|---|---|---|---|---|---|---|---|---|---|
| M | 69.09 | 19.71 | 1.75 | 0.05 | 6.91 | 2.09 | 0.07 | 0.07 | 0.26 |
| MTi1 | 69.09 | 18.60 | 1.59 | 0.06 | 6.61 | 1.91 | 0.28 | 0.11 | 36.79 |
| MTi2 | 69.09 | 18.94 | 1.63 | 0.06 | 6.70 | 1.95 | 0.15 | 0.11 | 19.76 |
| Sample | Dried | Calcined |
|---|---|---|
| M | 13.60 | 9.57 |
| MTi1 | 34.37 | 9.87 |
| MTi2 | 14.01 | 9.66 |
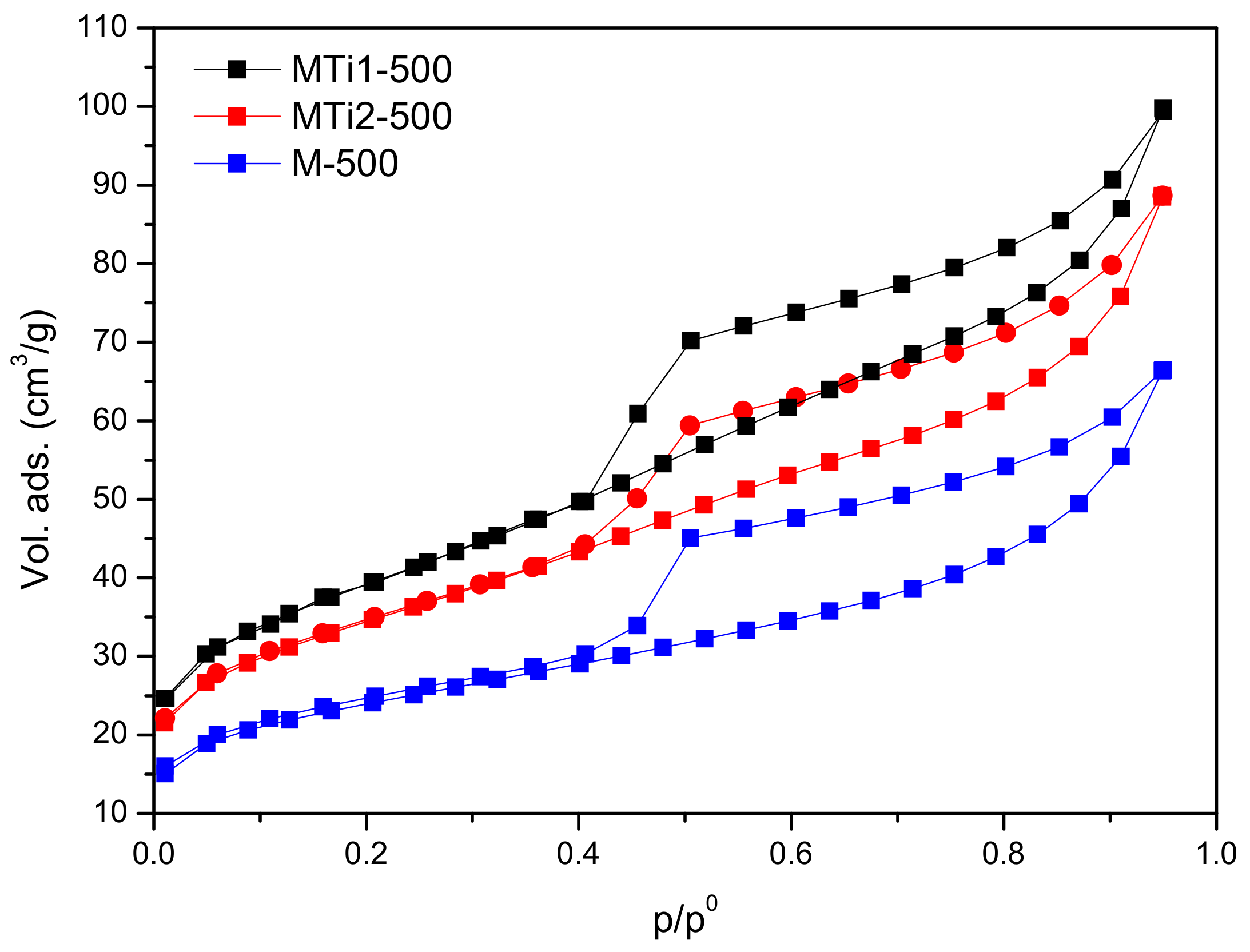
| Sample | Dried | Calcined | ||||
|---|---|---|---|---|---|---|
| SBET (m2/g) | Sext (m2/g) | Vm (cm3/g) | SBET (m2/g) | Sext (m2/g) | Vm (cm3/g) | |
| M | 49 | 49 | 0.001 | 80 | 64 | 0.009 |
| MTi1 | 233 | 226 | 0.007 | 134 | 118 | 0.009 |
| MTi2 | 193 | 173 | 0.012 | 117 | 103 | 0.008 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
González, B.; Muñoz, B.; Vicente, M.A.; Trujillano, R.; Rives, V.; Gil, A.; Korili, S. Photodegradation of 1,2,4-Trichlorobenzene on Montmorillonite–TiO2 Nanocomposites. ChemEngineering 2018, 2, 22. https://doi.org/10.3390/chemengineering2020022
González B, Muñoz B, Vicente MA, Trujillano R, Rives V, Gil A, Korili S. Photodegradation of 1,2,4-Trichlorobenzene on Montmorillonite–TiO2 Nanocomposites. ChemEngineering. 2018; 2(2):22. https://doi.org/10.3390/chemengineering2020022
Chicago/Turabian StyleGonzález, Beatriz, Bárbara Muñoz, Miguel Angel Vicente, Raquel Trujillano, Vicente Rives, Antonio Gil, and Sophia Korili. 2018. "Photodegradation of 1,2,4-Trichlorobenzene on Montmorillonite–TiO2 Nanocomposites" ChemEngineering 2, no. 2: 22. https://doi.org/10.3390/chemengineering2020022









