Improving Bread Quality with the Application of a Newly Purified Thermostable α-Amylase from Rhizopus oryzae FSIS4
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microorganism and Inoculum Preparation
2.2. Medium Composition and Fermentation
2.3. Partial Purification and Enzyme Formulation
2.4. Amylase Activity and Protein Content
2.5. Determination of Carbohydrate Content and Analysis of Hydrolysis Products
2.6. Bread-Making
2.7. Bread Quality Evaluation
2.8. Statistical Analysis
3. Results and Discussion
3.1. Purification of α-Amylase
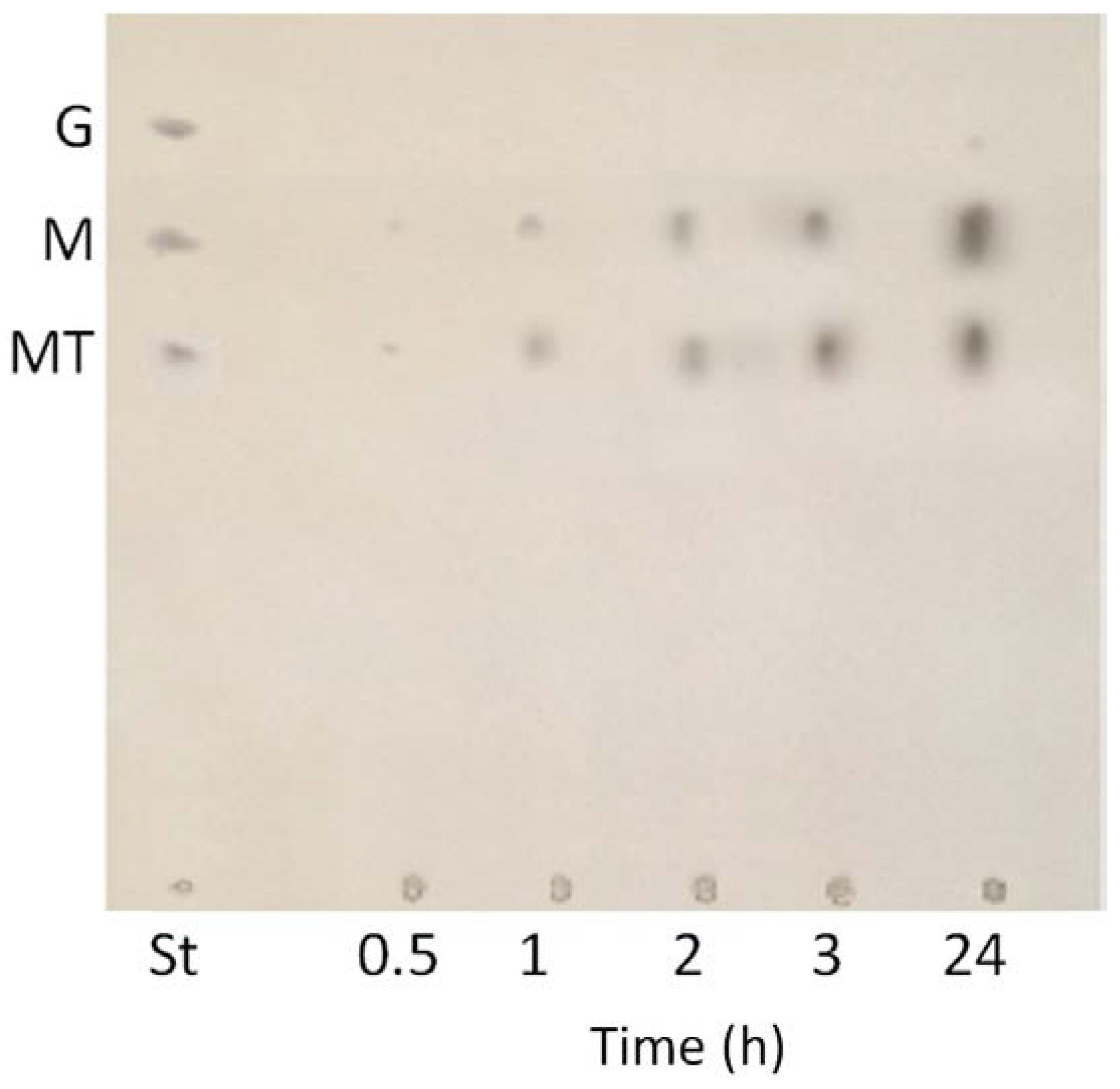
3.2. TLC of Hydrolysis Products
3.3. Bread-Making Performances
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gupta, R.; Gigras, P.; Mohapatra, H.; Goswami, V.K.; Chauhan, B. Microbial α-amylases: A biotechnological perspective. Process Biochem. 2003, 38, 1599–1616. [Google Scholar] [CrossRef]
- Goesaert, H.; Gebruers, K.; Courtin, C.M.; Brijs, K.; Delcour, J.A. Enzymes in Breadmaking. In Bakery Products; Blackwell Publishing: Oxford, UK, 2007; pp. 337–364. [Google Scholar]
- Goesaert, H.; Brijs, K.; Veraverbeke, W.S.; Courtin, C.M.; Gebruers, K.; Delcour, J.A. Wheat flour constituents: How they impact bread quality, and how to impact their functionality. Trends Food Sci. Technol. 2005, 16, 12–30. [Google Scholar] [CrossRef]
- Gagaoua, M.; Hafid, K. Three phase partitioning system, an emerging non-chromatographic tool for proteolytic enzymes recovery and purification. Biosens. J. 2016, 5. [Google Scholar] [CrossRef]
- Gagaoua, M.; Hafid, K.; Hoggas, N. Data in support of three phase partitioning of zingibain, a milk-clotting enzyme from Zingiber officinale Roscoe rhizomes. Data Brief 2016, 6, 634–639. [Google Scholar] [CrossRef] [PubMed]
- Gagaoua, M.; Hoggas, N.; Hafid, K. Three phase partitioning of zingibain, a milk-clotting enzyme from Zingiber officinale Roscoe rhizomes. Int. J. Biol. Macromol. 2015, 73, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Djekrif-Dakhmouche, S.; Gheribi-Aoulmi, Z.; Meraihi, Z.; Bennamoun, L. Application of a statistical design to the optimization of culture medium for α-amylase production by Aspergillus niger ATCC 16404 grown on orange waste powder. J. Food Eng. 2006, 73, 190–197. [Google Scholar] [CrossRef]
- Ray, R.C.; Kar, S. Statistical optimization of α-amylase production by Bacillus brevis MTCC 7521 in solid-state fermentation using cassava bagasse. Biologia 2009, 64, 864–870. [Google Scholar] [CrossRef]
- Ait Kaki El-Hadef El-Okki, A.; Gagaoua, M.; Bennamoun, L.; Djekrif, S.; Hafid, K.; El-Hadef El-Okki, M.; Meraihi, Z. Statistical optimization of thermostable α-amylase production by a newly isolated Rhizopus oryzae strain FSIS4 using decommissioned dates. Waste Biomass Valor. 2016, in press. [Google Scholar] [CrossRef]
- Dennison, C.; Lovrien, R. Three phase partitioning: Concentration and purification of proteins. Protein Expr. Purif. 1997, 11, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Gagaoua, M.; Boucherba, N.; Bouanane-Darenfed, A.; Ziane, F.; Nait-Rabah, S.; Hafid, K.; Boudechicha, H.-R. Three-phase partitioning as an efficient method for the purification and recovery of ficin from Mediterranean fig (Ficus carica L.) latex. Sep. Purif. Technol. 2014, 132, 461–467. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef] [PubMed]
- Sahnoun, M.; Naili, B.; Elgharbi, F.; Kammoun, R.; Gabsi, K.; Bejar, S. Effect of Aspergillus oryzae CBS 819.72 α-amylase on rheological dough properties and bread quality. Biologia 2013, 68, 808–815. [Google Scholar] [CrossRef]
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Fontana, J.D.; Gebara, M.; Blumel, M.; Schneider, H.; MacKenzie, C.R.; Johnson, K.G. α-4-O-methyl-d-glucuronidase Component of Xylanolytic Complexes. In Methods in Enzymology; Academic Press: New York, NY, USA, 1988; Volume 160, pp. 560–571. [Google Scholar]
- Ndangui, C.B.; Petit, J.; Gaiani, C.; Nzikou, J.-M.; Scher, J. Impact of thermal and chemical pretreatments on physicochemical, rheological, and functional properties of sweet potato (Ipomea batatas Lam) flour. Food Bioprocess Technol. 2014, 7, 3618–3628. [Google Scholar] [CrossRef]
- American Association of Cereal Chemists. AACC International. Approved Methods of Analysis, 11th Ed. Method 10–05.01. Guidelines for Measurement of Volume by Rapeseed Displacement, 11th ed.; AACC International: St. Paul, MN, USA, 2009. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; Version 3.2; R Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Mondal, K.; Sharma, A.; Lata; Gupta, M.N. Macroaffinity ligand-facilitated three-phase partitioning (MLFTPP) of α-amylases using a modified alginate. Biotechnol. Prog. 2003, 19, 493–494. [Google Scholar] [CrossRef] [PubMed]
- Sagu, S.T.; Nso, E.J.; Homann, T.; Kapseu, C.; Rawel, H.M. Extraction and purification of beta-amylase from stems of Abrus precatorius by three phase partitioning. Food Chem. 2015, 183, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Veluppillai, S.; Nithyanantharajah, K.; Vasantharuba, S.; Balakumar, S.; Arasaratnam, V. Optimization of bread preparation from wheat flour and malted rice flour. Rice Sci. 2010, 17, 51–59. [Google Scholar] [CrossRef]
- Goesaert, H.; Leman, P.; Bijttebier, A.; Delcour, J.A. Antifirming effects of starch degrading enzymes in bread crumb. J. Agric. Food Chem. 2009, 57, 2346–2355. [Google Scholar] [CrossRef] [PubMed]
- Pongjaruvat, W.; Methacanon, P.; Seetapan, N.; Fuongfuchat, A.; Gamonpilas, C. Influence of pregelatinised tapioca starch and transglutaminase on dough rheology and quality of gluten-free jasmine rice breads. Food Hydrocoll. 2014, 36, 143–150. [Google Scholar] [CrossRef]


| Specific Volume (cm3/g) | Height/Width (Ratio) | |
|---|---|---|
| Without α-amylase | 1.99 ± 0.21 a | 0.79 ± 0.01 a |
| With commercial α-amylase | 2.48 ± 0.08 b | 0.89 ± 0.02 b |
| With α-amylase of R. oryzae FSIS4 | 2.71 ± 0.14 c | 0.98 ± 0.02 c |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ait Kaki El-Hadef El-Okki, A.; Gagaoua, M.; Bourekoua, H.; Hafid, K.; Bennamoun, L.; Djekrif-Dakhmouche, S.; El-Hadef El-Okki, M.; Meraihi, Z. Improving Bread Quality with the Application of a Newly Purified Thermostable α-Amylase from Rhizopus oryzae FSIS4. Foods 2017, 6, 1. https://doi.org/10.3390/foods6010001
Ait Kaki El-Hadef El-Okki A, Gagaoua M, Bourekoua H, Hafid K, Bennamoun L, Djekrif-Dakhmouche S, El-Hadef El-Okki M, Meraihi Z. Improving Bread Quality with the Application of a Newly Purified Thermostable α-Amylase from Rhizopus oryzae FSIS4. Foods. 2017; 6(1):1. https://doi.org/10.3390/foods6010001
Chicago/Turabian StyleAit Kaki El-Hadef El-Okki, Amel, Mohammed Gagaoua, Hayat Bourekoua, Kahina Hafid, Leila Bennamoun, Shahrazed Djekrif-Dakhmouche, Mohamed El-Hadef El-Okki, and Zahia Meraihi. 2017. "Improving Bread Quality with the Application of a Newly Purified Thermostable α-Amylase from Rhizopus oryzae FSIS4" Foods 6, no. 1: 1. https://doi.org/10.3390/foods6010001








