Gluten-Free Bread: Influence of Sourdough and Compressed Yeast on Proofing and Baking Properties
Abstract
:1. Introduction
2. Materials and Methods
2.1. Gluten-Free Bread Recipe
2.2. Gluten-Free Dough Characterization
- -
- SD230 (SD, having a farinographic consistency of 230 ± 10 BU);
- -
- SD180 (SD, having a farinographic consistency of 180 ± 10 BU);
- -
- CY180 (CY, having a farinographic consistency of 180 ± 10 BU);
- -
- SDCY180 (SDCY, having a farinographic consistency of 180 ± 10 BU).
2.3. Breadmaking Process
2.4. Gluten-Free Bread Characterization
2.5. Statistical Analysis
3. Results and Discussion
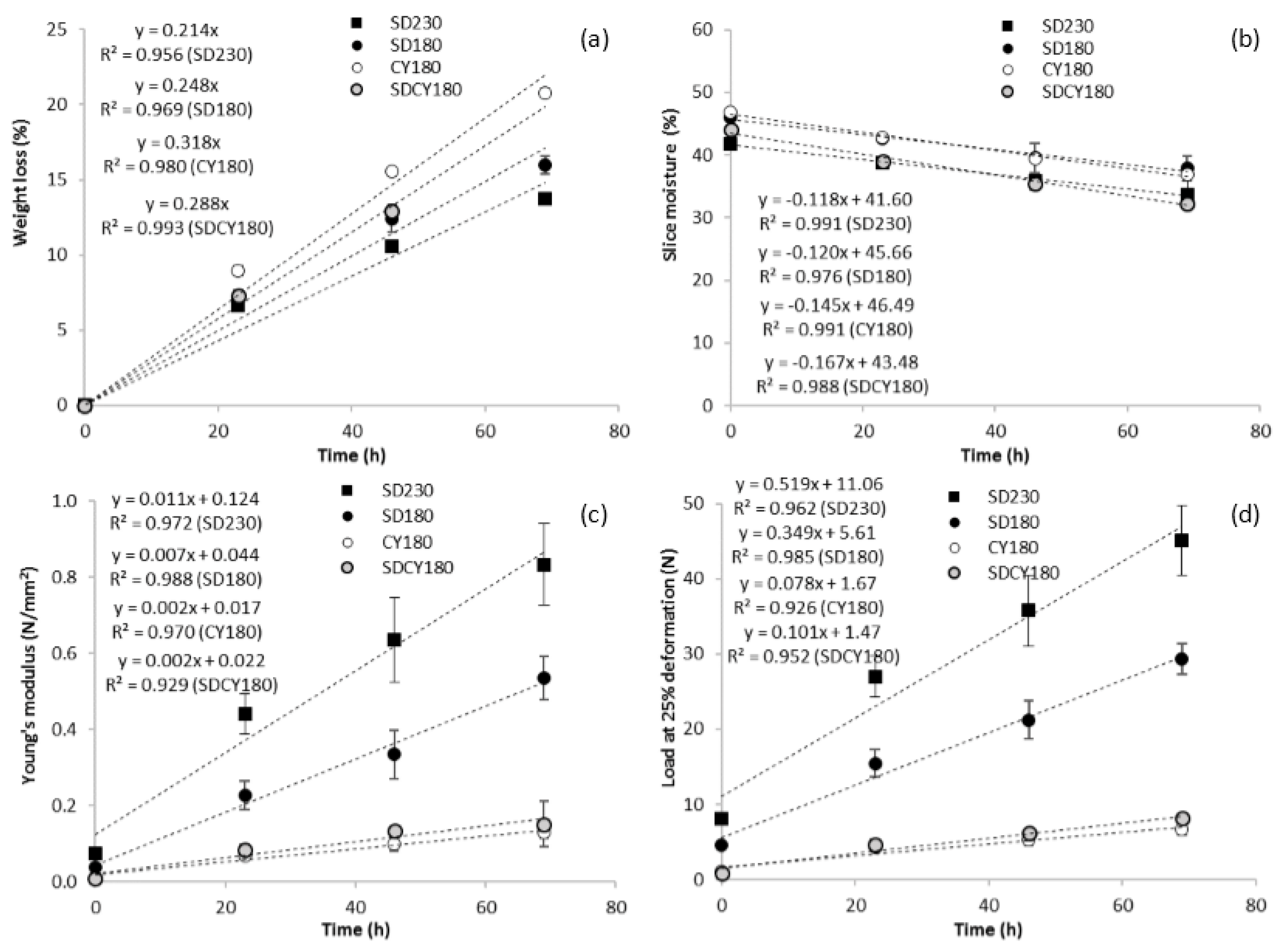
3.1. Gluten-Free Dough Properties
3.2. Gluten-Free Bread Properties
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mariotti, M. Valorizzazione tecnologica di avena e grano saraceno. In Tesi di Dottorato di Ricerca in Biotecnologia degli Alimenti; Facoltà di Agraria, Università degli Studi di Milano: Milan, Italy, 2004. [Google Scholar]
- Masure, H.G.; Fierens, E.; Delcour, J.A. Current and forward looking experimental approaches in gluten-free bread making research. J. Cereal Sci. 2016, 67, 92–111. [Google Scholar] [CrossRef]
- Cappa, C. Gluten-free bread: Optimization of formulation and process conditions. In Tesi di Dottorato di Ricerca in Innovazione Tecnologica per le Scienze Agro-Alimentari e Ambientali; Facoltà di Agraria, Università degli Studi di Milano: Milan, Italy, 2011. [Google Scholar]
- Gallagher, E.; Gormley, T.R.; Arendt, E.K. Recent advances in the formulation of gluten-free cereal-based products. Trends Food Sci. Technol. 2004, 15, 143–152. [Google Scholar] [CrossRef]
- Kobylañski, J.R.; Pérez, O.E.; Pilosof, A.M. Thermal transitions of gluten-free doughs as affected by water, egg white and hydroxypropylmethylcellulose. Thermochim. Acta 2004, 411, 81–89. [Google Scholar] [CrossRef]
- Lazaridou, A.; Duta, D.; Papageorgiou, M.; Belc, N.; Biliaderis, C.G. Effects of hydrocolloids on dough rheology and bread quality parameters in gluten-free formulations. J. Food Eng. 2007, 79, 1033–1047. [Google Scholar] [CrossRef]
- Moroni, A.; Dal Bello, F.; Arendt, E.K. Sourdough in gluten-free bread-making: An ancient technology to solve a novel issue? Food Microbiol. 2009, 26, 676–684. [Google Scholar] [CrossRef] [PubMed]
- Bárcenas, M.E.; Altamirano-Fortoul, R.; Rosell, C.M. Effect of high pressure processing on wheat dough and bread characteristics. Food Sci. Technol. LEB 2010, 43, 12–19. [Google Scholar] [CrossRef]
- Vallons, K.J.; Ryan, L.A.; Koehler, P.; Arendt, E.K. High pressure—treated sorghum flour as a functional ingredient in the production of sorghum bread. Eur. Food Res. Technol. 2010, 231, 711–717. [Google Scholar] [CrossRef]
- Cappa, C.; Lucisano, M.; Barbosa-Cánovas, G.V.; Mariotti, M. Physical and structural changes induced by high pressure on corn starch, rice flour and waxy rice flour. Food Res. Int. 2016, 85, 95–103. [Google Scholar] [CrossRef]
- Moore, M.M.; Dal Bello, F.; Arendt, E.K. Sourdough fermented by Lactobacillus plantarum FST 1.7 improves the quality and shelf life of gluten-free bread. Eur. Food Res. Technol. 2008, 226, 1309–1316. [Google Scholar] [CrossRef]
- Gobbetti, M.; Rizzello, C.G.; Di Cagno, R.; De Angelis, M. How the sourdough may affect the functional features of leavened baked goods. Food Microbiol. 2014, 37, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Picozzi, C.; Mariotti, M.; Cappa, C.; Tedesco, B.; Vigentini, I.; Foschino, R.; Lucisano, M. Development of a Type I gluten-free sourdough. Lett. Appl. Microbiol. 2015, 62, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Picozzi, C.; Bonacina, G.; Vigentini, I.; Foschino, R. Genetic diversity in Italian Lactobacillus sanfranciscensis strains assessed by multilocus sequence typing and pulsed-field gel electrophoresis analyses. Microbiology 2010, 156, 2035–2045. [Google Scholar] [CrossRef] [PubMed]
- Vigentini, I.; Antoniani, D.; Roscini, L.; Comasio, A.; Galafassi, S.; Picozzi, C.; Corte, L.; Compagno, C.; Dal Bello, F.; Cardinali, G.; et al. Candida milleri species reveals intraspecific genetic and metabolic polymorphisms. Food Microbiol. 2014, 42, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Gobbetti, M.; De Angelis, M.; Di Cagno, R.; Rizzello, C.G. Sourdough lactic/acid bacteria. In Gluten Free Cereals Products and Beverages; Arendt, E.K., Dal Bello, F., Eds.; Academic Press: New York, NY, USA, 2008; pp. 267–288. [Google Scholar]
- Barber, B.; Ortolá, C.; Barber, S.; Fernandez, F. Storage of packaged white bread. Z. Lebensm. Unters Forsch. 1992, 194, 442–449. [Google Scholar] [CrossRef]
- De Vuyst, L.; Vancanneyt, M. Biodiversity and identification of sourdough lactic acid bacteria. Food Microbiol. 2007, 24, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Lattanzi, A.; Minervini, F.; Di Cagno, R.; Diviccaro, A.; Antonielli, L.; Cardinali, G.; Cappelle, S.; De Angelis, M.; Gobbetti, M. The lactic acid bacteria and yeast microbiota of eighteen sourdoughs used for the manufacture of traditional Italian sweet leavened baked goods. Int. J. Food Microbiol. 2013, 163, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Huttner, E.K.; Dal Bello, F.; Arendt, E.K. Identification of lactic acid bacteria isolated from oat sourdoughs and investigation into their potential for the improvement of oat bread quality. Eur. Food Res. Technol. 2010, 230, 849–857. [Google Scholar] [CrossRef]
- Wolter, A.; Hager, A.S.; Zannini, E.; Czerny, M.; Arendt, E.K. Impact of sourdough fermented with Lactobacillus plantarum FST 1.7 on baking and sensory properties of gluten-free breads. Eur. Food Res. Technol. 2014, 239, 1–12. [Google Scholar] [CrossRef]
- Novotni, D.; Cukelj, N.; Smerdel, B.; Bituh, M.; Dujmic, F.; Curic, D. Glycemic index and firming kinetics of partially baked frozen gluten-free bread with sourdough. J. Cereal Sci. 2012, 55, 120–125. [Google Scholar] [CrossRef]
- Rozylo, R.; Rudy, S.; Krzykowski, A.; Dziki, D.; Grawlik-Dziki, U.; Rozylo, K.; Stonecki, S. Effect of adding fresh and freeze-dried buckwheat sourdough on gluten-free bread quality. Int. J. Food Sci. Technol. 2015, 50, 313–322. [Google Scholar] [CrossRef]
- Falade, A.; Naushad Emmambux, M.; Buys, E.M.; Taylor, J.R.N. Improvement of maize bread quality through modification of dough rheological properties by lactic acid bacteria fermentation. J. Cereal Sci. 2014, 60, 471–476. [Google Scholar] [CrossRef]
- Galle, S.; Schwab, C.; Dal Bello, F.; Coffey, A.; Ganzle, M.; Arendt, E.K. Influence of in-situ synthesized exopolysaccharides on the quality of gluten-free sorghum sourdough bread. Int. J. Food Microbiol. 2012, 155, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Edema, M.O.; Naushad Emmambux, M.; Taylor, J.R.N. Improvement of fonio dough properties through starch modification by sourdough fermentation. Starch/Starke 2013, 65, 730–737. [Google Scholar] [CrossRef]
- Mariotti, M.; Cappa, C.; Picozzi, C.; Tedesco, B.; Fongaro, L.; Lucisano, M. Compressed yeast and Type I gluten-free sourdough in gluten-free breadmaking. J. Food Bioprocess Technol. 2016, submitted. [Google Scholar]
- Corsetti, A. Technology of sourdough fermentation and sourdough applications. In Handbook on Sourdough Biotechnology; Gobbetti, M., Ganzle, M., Eds.; Springer: New York, NY, USA, 2013; pp. 85–103. [Google Scholar]
- Hager, A.S.; Wolter, A.; Czerny, M.; Bez, J.; Zannini, E.; Arendt, E.K.; Czerny, M. Investigation of product quality, sensory profile and ultrastructure of breads made from a range of commercial gluten-free flours compared to their wheat counterparts. Eur. Food Res. Technol. 2012, 235, 333–344. [Google Scholar] [CrossRef]
- Matos, M.E.; Rosell, C.M. Understanding gluten-free dough for reaching breads with physical quality and nutritional balance. J. Sci. Food Agric. 2015, 95, 653–661. [Google Scholar] [CrossRef] [PubMed]
- Raineri, A. Messa a Punto di Una Formulazione per Pane Gluten-Free a Lievitazione Naturale. Bachelor’s Thesis, Facoltà di Scienze Agrarie e Alimentari, Università degli Studi di Milano, Milan, Italy, 2011. [Google Scholar]
- Cappa, C.; Barbosa-Cánovas, G.V.; Lucisano, M.; Mariotti, M. Effect of high pressure processing on the baking aptitude of corn starch and rice flour. LWT Food Sci. Technol. 2016, 73, 20–27. [Google Scholar] [CrossRef]
- Cappa, C.; Lucisano, M.; Mariotti, M. Influence of Psyllium, sugar beet fibre and water on gluten-free dough properties and bread quality. Carbohydr. Polym. 2013, 98, 1657–1666. [Google Scholar] [CrossRef] [PubMed]
- Wolter, A.; Hager, A.S.; Zannini, E.; Czerny, M.; Arendt, E.K. Influence of dextran-producing Weissella cibaria on baking properties and sensory profile of gluten-free and wheat breads. Int. J. Food Microbiol. 2014, 172, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Axel, C.; Rocker, B.; Brosnan, B.; Zannini, E.; Furey, A.; Coffey, A.; Arendt, E.K. Application of Lactobacillus amylovorus DSM19280 in gluten-free sourdough bread to improve the microbial shelf life. Food Microbiol. 2015, 47, 36–44. [Google Scholar] [CrossRef] [PubMed]


| Ingredient (%) | SD230 | SD180 | CY180 | SDCY180 |
|---|---|---|---|---|
| Corn starch | 31 | 31 | 38 | 30 |
| Rice flour | 31 | 31 | 38 | 30 |
| Isolated pea protein | 5 | 5 | 6 | 5 |
| Psyllium fiber | 1.2 | 1.2 | 1.5 | 1.2 |
| Hydroxyl propyl methylcellulose | 1.2 | 1.2 | 1.5 | 1.2 |
| Emulsifiers | 0.4 | 0.4 | 0.5 | 0.4 |
| Oil | 5.0 | 5.0 | 6.1 | 4.9 |
| Maltose | 3.3 | 3.3 | 4.1 | 3.2 |
| NaCl | 1.7 | 1.7 | 2.0 | 1.6 |
| SD | 20 | 20 | 0 | 20 |
| CY | 0 | 0 | 2 | 2 |
| SD230 | SD180 | CY180 | SDCY180 | |
|---|---|---|---|---|
| Bread weight (g) | 130.4 ± 1.4 b | 135.0 ± 1.03 c | 134.9 ± 1.2 c | 123.1 ± 1.3 a |
| Height max (cm) | 3.9 ± 0.1 a | 4.7 ± 0.2 b | 5.6 ± 0.1 c | 6.0 ± 0.2 d |
| Height min (cm) | 2.3 ± 0.10 a | 2.7 ± 0.1 b | 4.0 ± 0.2 c | 4.3 ± 0.3 d |
| Volume (mL) | 231.9 ± 9.5 a | 255.4 ± 6.2 b | 347.0 ± 8.4 c | 376.0 ± 9.1 d |
| Specific volume (mL/g) | 1.78 ± 0.07 a | 1.89 ± 0.05 b | 2.57 ± 0.08 c | 3.07 ± 0.09 d |
| L*-crust | 79.2 ± 3.2 b | 82.2 ± 2.3 c | 81.3 ± 1.5 c | 77.3 ± 2.4 a |
| a*-crust | 0.8 ± 0.9 c | −1.5 ± 0.2 b | −1.9 ± 0.2 a | 1.1 ± 0.7 c |
| b*-crust | 20.0 ± 2.9 b | 16.9 ± 2.8 a | 17.0 ± 2.4 a | 20.4 ± 1.4 b |
| L*-crumb | 74.3 ± 2.0 b | 75.1 ± 1.1 c | 72.6 ± 1.1 a | 74.3 ± 0.9 b |
| a*-crumb | −2.06 ± 0.3 bc | −1.96 ± 0.2 c | −2.16 ± 0.2 ab | −2.29 ± 0.2 a |
| b*-crumb | 16.4 ± 0.4 c | 14.9 ± 0.3 b | 12.6 ± 0.9 a | 12.8 ± 0.6 a |
| Water activity | 0.980 ± 0.001 a | 0.992 ± 0.001 c | 0.997 ± 0.001 d | 0.989 ± 0.001 b |
| Slice moisture (%) | 41.86 ± 0.14 a | 46.06 ± 0.02 c | 46.85 ± 0.14 d | 43.99 ± 0.25 b |
| Crumb moisture (%) | 47.98 ± 0.55 a | 50.40 ± 0.04 b | 51.05 ± 0.42 b | 53.01 ± 0.36 c |
| Young’s modulus (N/mm2) | 0.073 ± 0.002 c | 0.038 ± 0.003 b | 0.009 ± 0.001 a | 0.007 ± 0.002 a |
| Load 25% deformation (N) | 8.16 ± 0.073 c | 4.52 ± 0.28 b | 1.06 ± 0.05 a | 0.84 ± 0.12 a |
| Sample | Holes Features | |||
|---|---|---|---|---|
| Size (mm2) | Distribution (%) | Area (%) | Diameter (mm) | |
| SD230-bread | 0.1 < x ≤ 1 | 90.2 ± 1.6 b | 62.50 ± 4.5 b | 0.6 ± 0.1 b |
| 1 < x ≤ 3 | 8.8 ± 1.8 b | 27.63 ± 5.2 b | 1.4 ± 0.1 ab | |
| x > 3 | 1.1 ± 0.6 a | 9.86 ± 5.5 a | 2.2 ± 0.6 a | |
| SD180-bread | 0.1 < x ≤ 1 | 94.0 ± 1.5 c | 67.9 ± 8.6 c | 0.6 ± 0.1 a |
| 1 < x ≤ 3 | 5.3 ± 1.5 a | 20.2 ± 5.1 a | 1.4 ± 0.1 ab | |
| x > 3 | 0.7 ± 0.3 a | 13.0 ± 10.4 ab | 2.7 ± 0.7 b | |
| CY180-bread | 0.1 < x ≤ 1 | 80.7 ± 3.6 a | 44.5 ± 6.6 a | 0.6 ± 0.1 c |
| 1 < x ≤ 3 | 16.8 ± 3.0 c | 38.0 ± 5.9 c | 1.4 ± 0.1 b | |
| x > 3 | 2.5 ± 1.1 b | 17.5 ± 8.6 b | 2.3 ± 0.2 a | |
| SDCY180-bread | 0.1 < x ≤ 1 | 80.8 ± 3.5 a | 47.0 ± 6.4 a | 0.7 ± 0.1 d |
| 1 < x ≤ 3 | 17.1 ± 3.3 c | 37.8 ± 6.1 c | 1.4 ± 0.1 a | |
| x > 3 | 2.1 ± 0.8 b | 15.2 ± 7.7 ab | 2.3 ± 0.2 ab | |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cappa, C.; Lucisano, M.; Raineri, A.; Fongaro, L.; Foschino, R.; Mariotti, M. Gluten-Free Bread: Influence of Sourdough and Compressed Yeast on Proofing and Baking Properties. Foods 2016, 5, 69. https://doi.org/10.3390/foods5040069
Cappa C, Lucisano M, Raineri A, Fongaro L, Foschino R, Mariotti M. Gluten-Free Bread: Influence of Sourdough and Compressed Yeast on Proofing and Baking Properties. Foods. 2016; 5(4):69. https://doi.org/10.3390/foods5040069
Chicago/Turabian StyleCappa, Carola, Mara Lucisano, Andrea Raineri, Lorenzo Fongaro, Roberto Foschino, and Manuela Mariotti. 2016. "Gluten-Free Bread: Influence of Sourdough and Compressed Yeast on Proofing and Baking Properties" Foods 5, no. 4: 69. https://doi.org/10.3390/foods5040069
APA StyleCappa, C., Lucisano, M., Raineri, A., Fongaro, L., Foschino, R., & Mariotti, M. (2016). Gluten-Free Bread: Influence of Sourdough and Compressed Yeast on Proofing and Baking Properties. Foods, 5(4), 69. https://doi.org/10.3390/foods5040069








