Enrichment and Purification of Casein Glycomacropeptide from Whey Protein Isolate Using Supercritical Carbon Dioxide Processing and Membrane Ultrafiltration
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Enrichment of β-LG and GMP Using Supercritical CO2
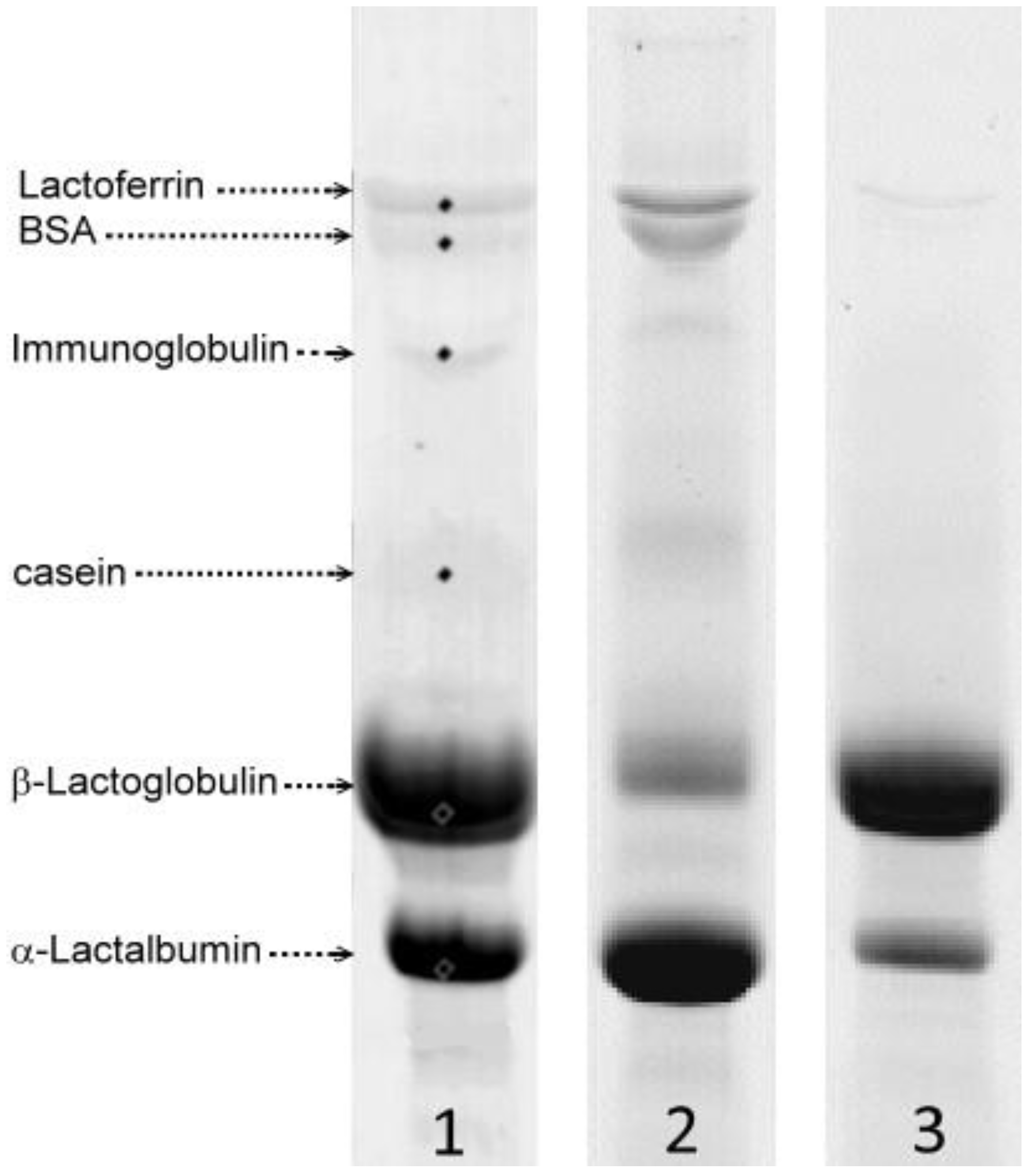
2.3. Separation and Quantification of the Protein Fractions
2.4. Ultrafiltration of Soluble Fractions
2.5. Compositional Analysis of the Enriched Protein Fraction
3. Results and Discussion
3.1. Fractionation of WPI with SCO2
| No. | T (°C) | CWPI (g·L−1) | pH | t (min) | Csolids (g·L−1) | Xprotein (wt%) | CA (g·L−1) | CB (g·L−1) | Cother (g·L−1) | CGMP (g·L−1) | Recovery | HPLC Composition | Ratio β-LG/α-LA | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| recA (%) | recB (%) | α-LA (wt%) | β-LG (wt%) | GMP (wt%) | HPLC | SDS-PAGE | |||||||||||
| 0. | WPI Feed | - | - | - | 18.3 | 50.1 | - | 16.1 | - | - | 20.3 | 55.7 | 17.9 | 2.74 | 2.71 | ||
| 1. | 60 | 100 | 5.0 | 215 | 85.3 | 80 | 6.6 | 45.1 | 16.6 | 14.9 | 36.1 | 90.0 | 9.9 | 67.8 | 22.3 | 6.8 | 7.1 |
| 2. | 100 | 4.9 | 185 | 85.7 | 80.9 | 6.6 | 45.7 | 17.0 | 14.6 | 36.1 | 91.2 | 9.9 | 68.3 | 21.8 | 6.9 | 7.8 | |
| 3. | 70 | 4.6 | 155 | 51.6 | 80 | 4.8 | 26.9 | 9.5 | 8.7 | 37.4 | 76.7 | 11.8 | 66.7 | 21.5 | 5.7 | 5.7 | |
| 4. | 20 | 4.2 | 120 | 18.9 | 83.6 | 3.0 | 9.2 | 3.6 | 3.5 | 82.0 | 91.8 | 19.1 | 58.8 | 22.1 | 3.1 | 2.6 | |
| 5. | 62 | 100 | 5.0 | 180 | 81.6 | 81.1 | 6 | 49.2 | 11.0 | 14.4 | 32.8 | 98.2 | 8.6 | 70.7 | 20.7 | 8.2 | 8.5 |
| 6. | 100 | 4.7 | 310 | 81.5 | 80 | 3.6 | 43.2 | 18.4 | 13.5 | 19.7 | 86.2 | 6.0 | 71.6 | 22.4 | 12.0 | 10.2 | |
| 7. | 100 | 4.6 | 120 | 77.0 | 80 | 5.9 | 42.8 | 12.9 | 13.8 | 32.2 | 85.4 | 9.4 | 68.5 | 22.1 | 7.3 | 7.0 | |
| 8. | 70 | 4.6 | 180 | 48.8 | 80 | 3.7 | 25.1 | 10.2 | 9.2 | 28.9 | 71.6 | 9.7 | 66.1 | 24.2 | 6.8 | 5.7 | |
| 9. | 65 | 100 | 5.0 | 160 | 75.1 | 78.3 | 2.4 | 36.9 | 19.5 | 13.0 | 13.1 | 73.6 | 4.6 | 70.6 | 24.8 | 15.4 | 16.3 |
| 10. | 100 | 4.6 | 140 | 58.7 | 79 | 1.8 | 24.6 | 20.0 | 12.2 | 9.8 | 49.1 | 4.7 | 63.6 | 31.7 | 13.6 | 9.5 | |
| 11. | 70 | 100 | 4.9 | 305 | 43.6 | 80 | 3.0 | 6.6 | - | 13.6 | 16.4 | 13.2 | 13.0 | 28.3 | 58.7 | 2.2 | 2.0 |
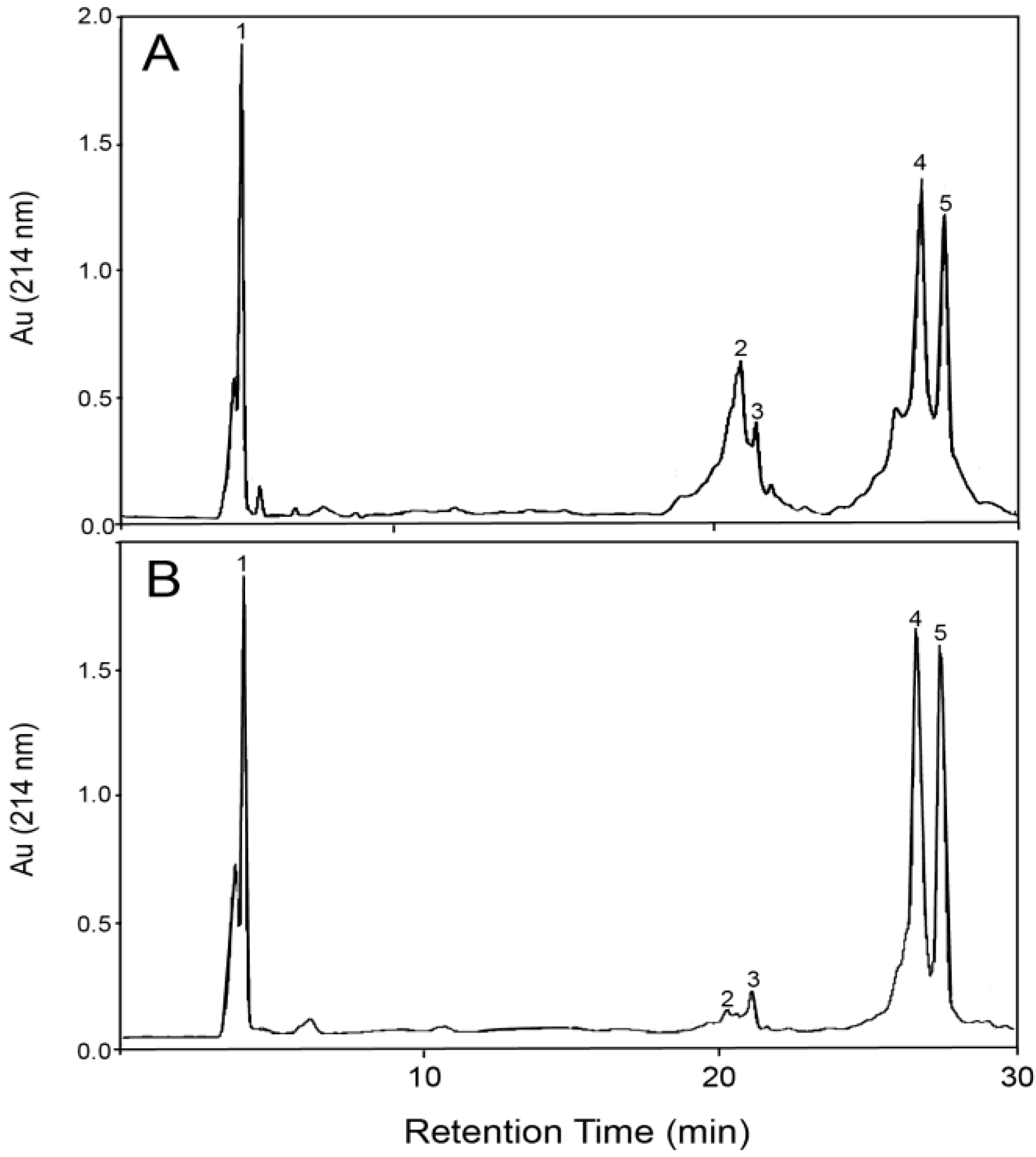
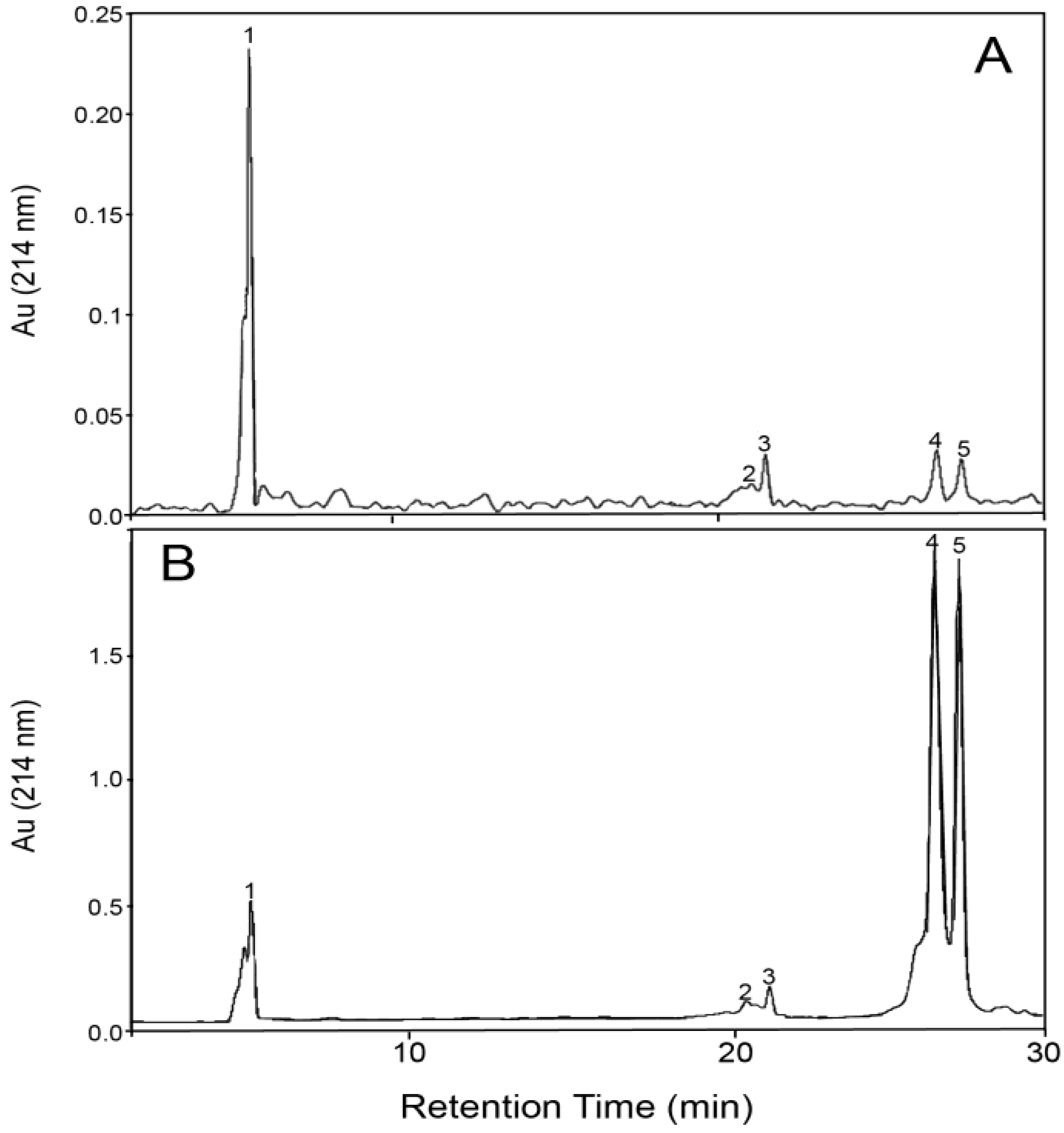
3.2. Fractionation of β-LG and GMP with Membrane Filtration
| Starting Composition | Membrane Pore Size (kDa) | Ultrafiltration | RP-HPLC Analysis | GMP Recovery (%) | GMP Purity (%) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Filtration Stage | Volume Solution (mL) | Volume Filtrate (mL) | Volume Retentate (mL) | Dilution Factor | GMP Peak Area (×10−6) | β-LG Peak Area (×10−6) | Ratio GMP:β-LG | ||||
| 31.7% GMP 63.6% β-LG 4.7% α-LA (65 °C, pH 4.6) | - | Initial | - | - | - | 80 | 39.2 | 78.6 | 0.50 | - | 31.7 |
| 10 | Filtration | 50 | 30 | 20 | 80 | 16.5 | ~0 | - | 25 | - | |
| 30 | Filtration | 50 | 34 | 16 | 80 | 25.0 | 6.0 | 4.19 | 43 | 59.7 | |
| 1st Wash | 50 | 85 | 15 | 40 | 20.3 | 3.7 | 5.42 | 44 | 57.8 | ||
| 2nd Wash | 50 | 138 | 12 | 20 | 33.7 | 9.5 | 3.53 | 59 | 58.1 | ||
| Retentate | 12 | 40 | 117.3 | 880.6 | 0.13 | 36 | 11.2 | ||||
| 58.7% GMP 28.3% β-LG 13.0% α-LA (70 °C, pH 4.9) | - | Initial | - | - | - | 80 | 40.7 | 19.6 | 2.07 | - | 58.7 |
| 30 | Filtration | 100 | 85 | 15 | 40 | 43.8 | ~0 | - | 46 | 94.0 | |
| 1st Wash | 100 | 189 | 11 | 20 | 64.2 | 1.2 | 53.8 | 75 | 87.0 | ||
| 2nd Wash | 100 | 294 | 6 | 10 | 90.9 | 1.5 | 59.4 | 82 | 80.4 | ||
| Retentate | 6 | 100 | 73.9 | 163.3 | 0.45 | 14 | 27.1 | ||||
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Bonnaillie, L.M.; Tomasula, P.M. Whey Protein Fractionation. In Whey Processing, Functionality and Health Benefits; Onwulata, C.I., Huth, P.J., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2008; pp. 15–38. [Google Scholar]
- Onwulata, C.I.; Konstance, R.P.; Tomasula, P.M. Minimizing variations in functionality of whey protein concentrates from different sources. J. Dairy Sci. 2004, 87, 749–756. [Google Scholar] [CrossRef]
- Yver, A.L.; Bonnaillie, L.M.; Yee, W.; McAloon, A.; Tomasula, P.M. Fractionation of whey protein isolate with supercritical carbon dioxide—Process modeling and cost estimation. Int. J. Mol. Sci. 2011, 13, 240–259. [Google Scholar] [CrossRef]
- Bonnaillie, L.M.; Tomasula, P.M. Kinetics, aggregation behavior and optimization of the fractionation of whey protein isolate with hydrochloric acid. Food Bioprod. Process. 2012, 90, 737–747. [Google Scholar] [CrossRef]
- Bonnaillie, L.M.; Tomasula, P.M. Fractionation of whey protein isolate with supercritical carbon dioxide to produce enriched α-lactalbumin and β-lactoglobulin food ingredients. J. Agric. Food Chem. 2012, 60, 5257–5266. [Google Scholar] [CrossRef]
- Bruck, W.M.; Redgrave, M.; Tuohy, K.M.; Lonnerdal, B.; Graverholt, G.; Hernell, O.; Gibson, G.R. Effects of bovine alpha-lactalbumin and casein glycomacropeptide-enriched infant formulae on faecal microbiota in healthy term infants. J. Pediatr. Gastroenterol. Nutr. 2006, 43, 673–679. [Google Scholar] [CrossRef]
- Sandstrom, O.; Lonnerdal, B.; Graverholt, G.; Hernell, O. Effects of alpha-lactalbumin-enriched formula containing different concentrations of glycomacropeptide on infant nutrition. Am. J. Clin. Nutr. 2008, 87, 921–928. [Google Scholar]
- Heine, W.; Radke, M.; Wietzke, K.D.; Polars, E.; Kundt, G. A-lactalbumin-enriched low-protein infant formulas: A comparison to breast milk feeding. Acta Pediatr. 1996, 85, 1024–1028. [Google Scholar] [CrossRef]
- Heine, W.E.; Klein, P.D.; Miyashita, C. Method for Isolating Alpha-Lactalbumin from Whey. Patent #WO 92/03468, 5 March 1992. [Google Scholar]
- Etzel, M.R. Manufacture and use of dairy protein fractions. J. Nutr. 2004, 134, 996S–1002S. [Google Scholar]
- Farrell, H.M., Jr.; Jimenez-Flores, R.; Bleck, G.T.; Brown, E.M.; Butler, J.E.; Creamer, L.K.; Hicks, C.L.; Hollar, C.M.; Ng-Kwai-Hang, K.F.; Swaisgood, H.E. Nomenclature of the proteins of cows’ milk—Sixth revision. J. Dairy Sci. 2004, 87, 1641–1674. [Google Scholar] [CrossRef]
- Tomasula, P.M.; Craig, J.C., Jr.; Boswell, R.T.; Cook, R.D.; Kurantz, M.J.; Maxwell, M. Preparation of casein using carbon dioxide. J. Dairy Sci. 1995, 78, 506–514. [Google Scholar] [CrossRef]
- Tomasula, P.M.; Parris, N.; Boswell, R.T.; Moten, R.O. Preparation of enriched fractions of α-lactalbumin and β-lactoglobulin from cheese whey using carbon dioxide. J. Food Process. Preserv. 1998, 22, 463–476. [Google Scholar] [CrossRef]
- Green, D.; Perry, R. Perry’s Chemical Engineers’ Handbook, 8th ed.; McGraw-Hill Professional: New York, NY, USA, 2007. [Google Scholar]
- Fox, P.F.; McSweeney, P. Advanced Dairy Chemistry: Volume 1: Proteins, Parts A & B, 3rd ed.; Springer: Berlin, Germany, 2003. [Google Scholar]
- Lopez-Fandino, R.; Ramos, M. Bovine caseinomacropeptide. 1. Physicochemical characteristics and biological-activity—Review. Rev. Esp. Cienc. Tecnol. Aliment. 1992, 32, 575–588. [Google Scholar]
- ElSalam, M.H.A.; ElShibiny, S.; Buchheim, W. Characteristics and potential uses of the casein macropeptide. Int. Dairy J. 1996, 6, 327–341. [Google Scholar] [CrossRef]
- LaClair, C.E.; Ney, D.M.; MacLeod, E.L.; Etzel, M.R. Purification and use of glycomacropeptide for nutritional management of phenylketonuria. J. Food Sci. 2009, 74, E199–E206. [Google Scholar] [CrossRef]
- Thoma-Worringer, C.; Sorensen, J.; Lopez-Fandino, R. Health effects and technological features of caseinomacropeptide. Int. Dairy J. 2006, 16, 1324–1333. [Google Scholar] [CrossRef]
- Nejad, A.S.; Kanekanian, A.; Tatham, A. The inhibitory effect of glycomacropeptide on dental erosion. Dairy Sci. Technol. 2009, 89, 233–239. [Google Scholar] [CrossRef]
- Setarehnejad, A.; Kanekanian, A.; Tatham, A.; Abedi, A.H. The protective effect of caseinomacropeptide against dental erosion using hydroxyapatite as a model system. Int. Dairy J. 2010, 20, 652–656. [Google Scholar] [CrossRef]
- Requena, P.; Gonzalez, R.; Lopez-Posadas, R.; Abadia-Molina, A.; Suarez, M.D.; Zarzuelo, A.; de Medina, F.S.; Martinez-Augustin, O. The intestinal antiinflammatory agent glycomacropeptide has immunomodulatory actions on rat splenocytes. Biochem. Pharm. 2010, 79, 1797–1804. [Google Scholar] [CrossRef]
- Hursel, R.; van der Zee, L.; Westerterp-Plantenga, M.S. Effects of a breakfast yoghurt, with additional total whey protein or caseinomacropeptide-depleted alpha-lactalbumin-enriched whey protein, on diet-induced thermogenesis and appetite suppression. Br. J. Nutr. 2010, 103, 775–780. [Google Scholar] [CrossRef]
- Li, E.W.Y.; Mine, Y. Technical note: Comparison of chromatographic profile of glycomacropeptide from cheese whey isolated using different methods. J. Dairy Sci. 2004, 87, 174–177. [Google Scholar] [CrossRef]
- Da Silva, C.A.S.; Coimbra, J.S.R.; Rojas, E.E.G.; Teixeira, J.A.C. Partitioning of glycomacropeptide in aqueous two-phase systems. Process Biochem. 2009, 44, 1213–1216. [Google Scholar] [CrossRef]
- Casal, E.; Corzo, N.; Moreno, F.J.; Olano, A. Selective recovery of glycosylated caseinmacropeptide with chitosan. J. Agric. Food Chem. 2005, 53, 1201–1204. [Google Scholar] [CrossRef]
- Tolkach, A.; Kulozik, U. Fractionation of whey proteins and caseinomacropeptide by means of enzymatic crosslinking and membrane separation techniques. J. Food Eng. 2005, 67, 13–20. [Google Scholar] [CrossRef]
- Nakano, T.; Ikawa, N.; Ozimek, L. Separation of glycomacropeptide from sweet whey by using chitosan and a centrifugal filter. Milchwissenschaft 2006, 61, 191–193. [Google Scholar]
- Chu, L.; Macleod, A.; Ozimek, L. Isolation of glycomacropeptide from sodium caseinate hydrolysate solution by ultrafiltration. Milchwissenschaft 1996, 51, 303–306. [Google Scholar]
- Tanimoto, M.; Kawasaki, Y.; Shinmoto, H.; Dosako, S.; Tomizawa, A. Process for Producing κ-Casein Glycomacropeptides. US Patent No. 5,075,424, 24 December 1991. [Google Scholar]
- Kawasaki, Y.; Kawakami, M.; Tanimoto, M.; Dosako, S.; Tomizawa, A.; Kotake, M.; Naka-jima, I. pH-dependent molecular weight changes of κ-casein glyco-macropeptide and its preparation by ultrafiltration. Milchwissenschaft 1993, 48, 191–196. [Google Scholar]
- Martin-Diana, A.B.; Fraga, M.J.; Fontecha, J. Isolation and characterisation of caseinmacropeptide from bovine, ovine, and caprine cheese whey. Eur. Food Res. Technol. 2002, 214, 282–286. [Google Scholar] [CrossRef]
- Kreuss, M.; Kulozik, U. Separation of glycosylated caseinomacropeptide at pilot scale using membrane adsorption in direct-capture mode. J. Chromatogr. A 2009, 1216, 8771–8777. [Google Scholar]
- Lieske, B.; Konrad, G.; Kleinschmidt, T. Isolation of caseinomacropeptide from rennet whey by a multistage ultrafiltration process. III. Influence of pH in the first ultrafiltration-step on the chemical properties of isolated caseinomacropeptide. Milchwissenschaft 2004, 59, 408–410. [Google Scholar]
- Ayers, J.S.; Elgar, D.F.; Palmano, K.P.; Pritchard, M.; Bhaskar, G.B. Process for Separation of Whey Proteins Using a Novel Anion Exchanger. Patent CA2393270 A1, 28 March 2006. [Google Scholar]
- Outinen, M.; Tossavainen, O.; Syvaoja, E.L.; Korhonen, H. Chromatographic isolation of kappa-casein macropeptide from cheese whey with a strong basic anion-exchange resin. Milchwissenschaft 1995, 50, 570–574. [Google Scholar]
- Doultani, S.; Turhan, K.N.; Etzel, M.R. Whey protein isolate and glycomacropeptide recovery from whey using ion exchange chromatography. J. Food Sci. 2003, 68, 1389–1395. [Google Scholar] [CrossRef]
- Tek, H.N.; Turhan, K.N.; Etzel, M.R. Effect of conductivity, pH, and elution buffer salinity on glycomacropeptide recovery from whey using anion exchange chromatography. J. Food Sci. 2005, 70, E295–E300. [Google Scholar]
- Kreuss, M.; Krause, I.; Kulozik, U. Separation of a glycosylated and non-glycosylated fraction of caseinomacropeptide using different anion-exchange stationary phases. J. Chromatogr. A 2008, 1208, 126–132. [Google Scholar] [CrossRef]
- Holland, B.; Yazdi, S.R.; Titapiccolo, G.I.; Corredig, M. Short communication: Separation and quantification of caseins and casein macropeptide using ion-exchange chromatography. J. Dairy Sci. 2010, 93, 893–900. [Google Scholar] [CrossRef]
- Kawakami, H.; Kawasaki, Y.; Dosako, S.; Tanimoto, M.; Nakajima, I. Determination of κ-casein glycomacropeptide by high-performance liquid-chromatography without trichloroacetic-acid pretreatment. Milchwissenschaft 1992, 47, 688–693. [Google Scholar]
- Shammet, K.M.; Mcmahon, D.J.; Brown, R.J. Characteristics of macropeptide fraction isolated from whole casein and purified κ-casein. Milchwissenschaft 1992, 47, 615–619. [Google Scholar]
- Minkiewicz, P.; Slangen, C.J.; Lagerwerf, F.M.; Haverkamp, J.; Rollema, H.S.; Visser, S. Reversed-phase high-performance liquid chromatographic separation of bovine κ-casein macropeptide and characterization of isolated fractions. J. Chromatogr. A 1996, 743, 123–135. [Google Scholar] [CrossRef]
- Elgar, D.F.; Norris, C.S.; Ayers, J.S.; Pritchard, M.; Otter, D.E.; Palmano, K.P. Simultaneous separation and quantitation of the major bovine whey proteins including proteose peptone and caseinomacropeptide by reversed-phase high-performance liquid chromatography on polystyrene-divinylbenzene. J. Chromatogr. A 2000, 878, 183–196. [Google Scholar] [CrossRef]
- Ferreira, I.M.P.L.V.O.; Oliveira, M.B.P.P. Determination of caseinomacropeptide by an RP-HPLC method and monitoring of the addition of rennet whey to powdered milk. J. Liq. Chromatogr. Relat. Technol. 2003, 26, 99–107. [Google Scholar] [CrossRef]
- Thoma, C.; Krause, I.; Kulozik, U. Precipitation behaviour of caseinomacropeptides and their simultaneous determination with whey proteins by RP-HPLC. Int. Dairy J. 2006, 16, 285–293. [Google Scholar] [CrossRef]
- Vanriel, J.; Olieman, C. Determination of caseinomacropeptide with capillary zone electrophoresis and its application to the detection and estimation of rennet whey solids in milk and buttermilk powder. Electrophoresis 1995, 16, 529–533. [Google Scholar] [CrossRef]
- Molle, D.; Leonil, J. Quantitative determination of bovine κ-casein macropeptide in dairy products by liquid chromatography/electrospray coupled to mass spectrometry (LC-ESI/MS) and liquid chromatography/electrospray coupled to tamdem mass spectrometry (LC-ESI/MS/MS). Int. Dairy J. 2005, 15, 419–428. [Google Scholar] [CrossRef]
- Galindo-Amaya, L.; Valbuena-Colmenares, E.; Rojas-Villarroel, E. Standardization of glycomacropeptide detection with SDS-PAGE as a milk adulteration index. Rev. Cient. 2006, 16, 308–314. [Google Scholar]
- Grindrod, J.; Nickerso, T.A. Glycomacropeptide identification by disc gel electrophoresis. Abstracts of papers presented at the Sixtieth Annual Meeting of the American Dairy Science Association: University of Kentucky, Lexington, June 20–23, 1965. J. Dairy Sci. 1965, 48, 762–830. [Google Scholar] [CrossRef]
- Parris, N.; White, A.E.; Farrell, H.M. Identification of altered proteins in nonfat dry milk powder prepared from heat-treated skim milk. J. Agric. Food Chem. 1990, 38, 824–829. [Google Scholar] [CrossRef]
- Barbano, D.M.; Lynch, J.M.; Fleming, J.R. Direct and indirect determination of true protein content of milk by kjeldahl analysis: Collaborative study. J. Assoc. Off. Anal. Chem. 1991, 74, 281–288. [Google Scholar]
- McGuffey, M.K.; Epting, K.L.; Kelly, R.M.; Foegeding, E.A. Denaturation and aggregation of three alpha-lactalbumin preparations at neutral pH. J. Agric. Food Chem. 2005, 53, 3182–3190. [Google Scholar]
- De Wit, J.N. Functional Properties of Whey Proteins. In Developments in Dairy Chemistry; Fox, P.F., Ed.; Elsevier Applied Science: New York, NY, USA, 1989; Volume 4. [Google Scholar]
- Verheul, M.; Pedersen, J.S.; Roefs, S.P.F.M.; de Kruif, K.G. Association behavior of native β-lactoglobulin. Biopolymers 1999, 49, 11–20. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bonnaillie, L.M.; Qi, P.; Wickham, E.; Tomasula, P.M. Enrichment and Purification of Casein Glycomacropeptide from Whey Protein Isolate Using Supercritical Carbon Dioxide Processing and Membrane Ultrafiltration. Foods 2014, 3, 94-109. https://doi.org/10.3390/foods3010094
Bonnaillie LM, Qi P, Wickham E, Tomasula PM. Enrichment and Purification of Casein Glycomacropeptide from Whey Protein Isolate Using Supercritical Carbon Dioxide Processing and Membrane Ultrafiltration. Foods. 2014; 3(1):94-109. https://doi.org/10.3390/foods3010094
Chicago/Turabian StyleBonnaillie, Laetitia M., Phoebe Qi, Edward Wickham, and Peggy M. Tomasula. 2014. "Enrichment and Purification of Casein Glycomacropeptide from Whey Protein Isolate Using Supercritical Carbon Dioxide Processing and Membrane Ultrafiltration" Foods 3, no. 1: 94-109. https://doi.org/10.3390/foods3010094
APA StyleBonnaillie, L. M., Qi, P., Wickham, E., & Tomasula, P. M. (2014). Enrichment and Purification of Casein Glycomacropeptide from Whey Protein Isolate Using Supercritical Carbon Dioxide Processing and Membrane Ultrafiltration. Foods, 3(1), 94-109. https://doi.org/10.3390/foods3010094





