Molecular Features of Wheat Endosperm Arabinoxylan Inclusion in Functional Bread
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Chemicals
2.3. Methods
2.3.1. Bread Making
| White bread (g) | Bread + AEM (g) | Bread + AEM + 360 ppm endoxylanase (g) | Bread + AEM + 600 ppm endoxylanase (g) | |
|---|---|---|---|---|
| Strong flour | 600 | 408 | 408 | 408 |
| Salt | 8.6 | 8.6 | 8.6 | 8.6 |
| Sugar | 6 | 6 | 6 | 6 |
| Fat | 6.6 | 6.6 | 6.6 | 6.6 |
| Compact yeast | 37.3 | 37.3 | 37.3 | 37.3 |
| Bread improver | 6 | 6 | 6 | 6 |
| Arabinoxylan enriched material | 0 | 192 | 192 | 192 |
| Water | 336 | 390 | 390 | 390 |
| Endoxylanase | 0 | 0 | 0.036 | 0.06 |
2.3.2. Bread Firmness Determination
2.3.3. Water-Extractable AX (WE-AX) Preparation
2.3.4. Determination of AX Content
2.3.5. Characterization of Molecule Weights of AXs
2.3.5.1. Construction of Standard Curve
| Samples | Molecular weight (Da) |
|---|---|
| P-800 | 708,000 |
| P-400 | 375,000 |
| P-200 | 200,000 |
| P-100 | 107,000 |
| P-50 | 47,100 |
| P-20 | 21,100 |
| P-10 | 11,000 |
| P-5 | 5900 |
2.3.5.2. Determination of AXs Samples
3. Results and Discussion
3.1. Chemical Composition of AEM

3.2. Incorporation of AEM into Bread
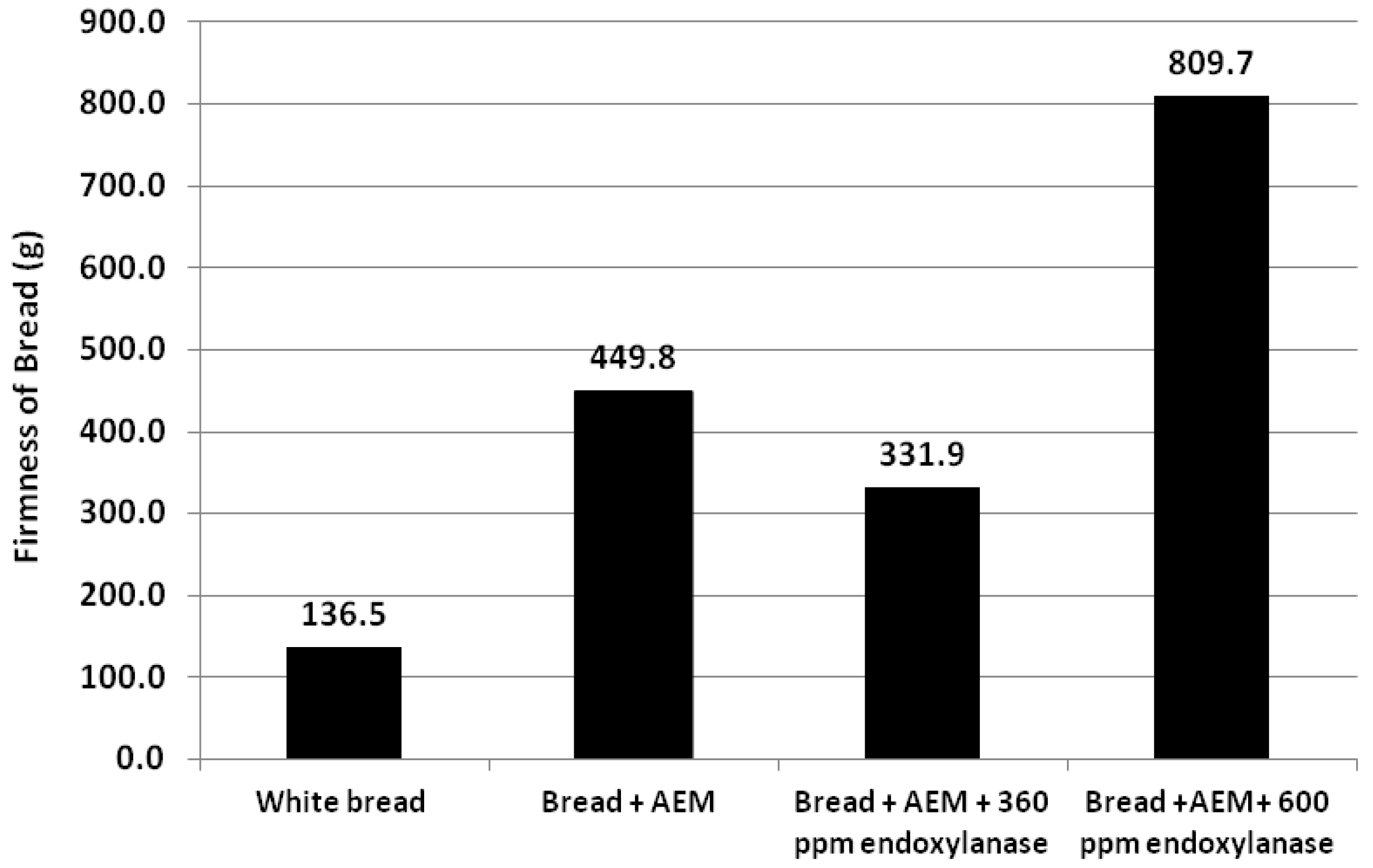

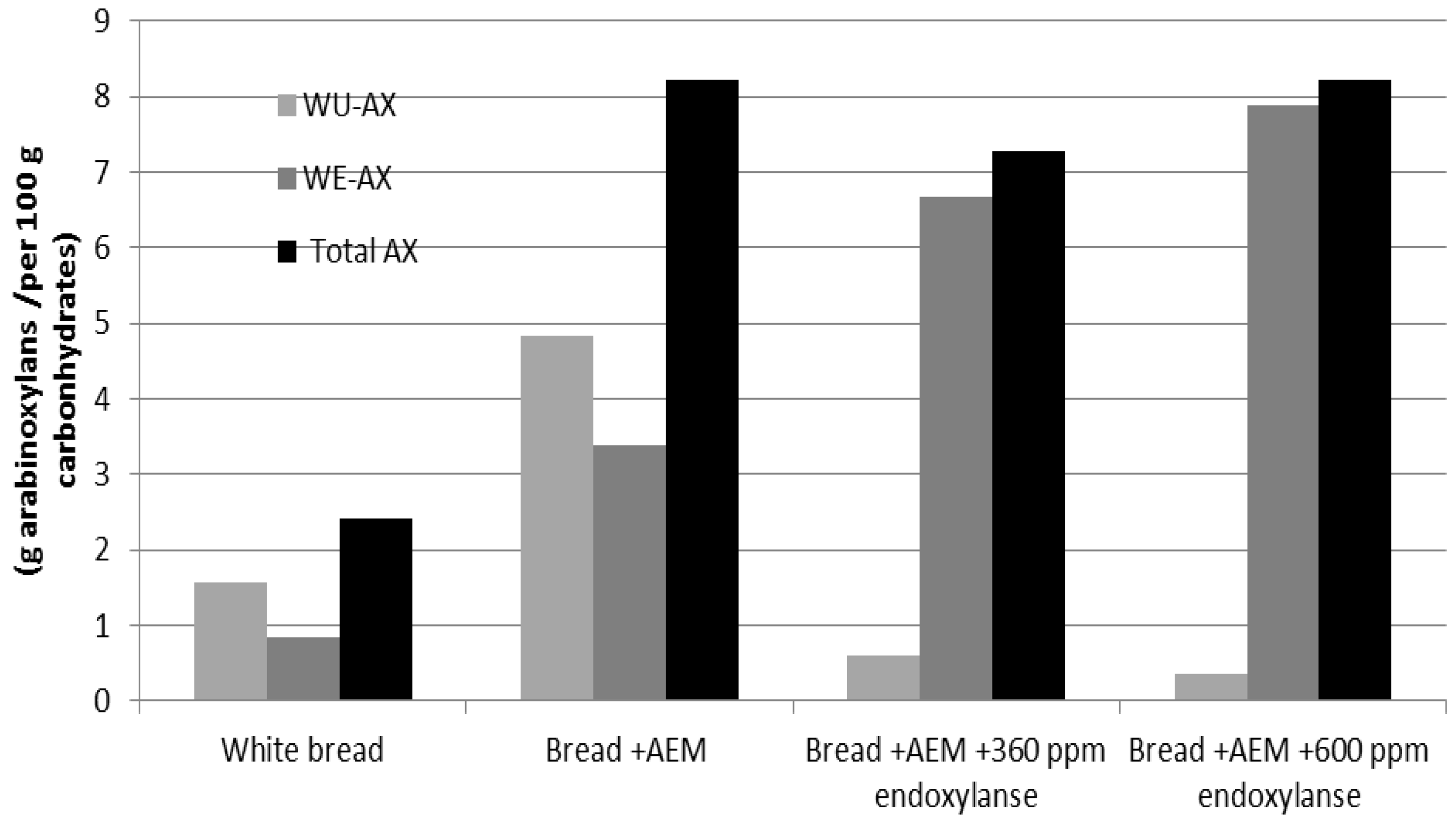
3.3. Extractability of AXs in Bread
3.4. Characterization of Mw of the WE-AX of the AEM and Tested Breads

4. Conclusions
Conflicts of Interest
References
- Anderson, W.J.; Baird, P.; Davis, H.R., Jr.; Ferreri, S.; Knudtson, M.; Koraym, A.; Waters, V.; Williams, L.C. Health benefits of dietary fiber. Nutr. Rev. 2009, 67, 188–205. [Google Scholar] [CrossRef]
- Yadav, P.M.; Johnston, B.D.; Hotchkiss, T.A., Jr.; Hicks, B.K. Corn fiber gum: A potential gum arabic replacer for beverage flavor emulsification. Food Hydrocol. 2007, 21, 1022–1030. [Google Scholar] [CrossRef]
- Broekaert, W.F.; Courtin, C.M.; Verbeke, K.; van de Wiele, T.; Verstraete, W.; Delcour, J.A. Prebiotic and other health-related effects of cereal-derived arabinoxylans, arabinoxylan-oligosaccharides, and xylooligosaccharides. Crit. Rev. Food Sci. Nutr. 2011, 51, 178–194. [Google Scholar] [CrossRef]
- Saeed, F.; Pasha, I.; Anjuma, M.F.; Sultan, T.M. Arabinoxylans and arabinogalactans: A comprehensive treatise. Crit. Rev. Food Sci. Nutr. 2011, 51, 467–476. [Google Scholar] [CrossRef]
- François, I.E; Lescroart, O.; Veraverbeke, W.S.; Marzorati, M.; Possemiers, S.; Evenepoel, P.; Hamer, H.; Houben, E.; Windey, K.; Welling, G.W.; et al. Effects of a wheat bran extract containing arabinoxylan oligosaccharides on gastrointestinal health parameters in healthy adult human volunteers: A double-blind, randomised, placebo-controlled, cross-over trial. Br. J. Nutr. 2012, 108, 2229–2242. [Google Scholar]
- Toole, G.A.; le Gall, G.; Colquhoun, I.J.; Johnson, P.; Bedo, Z.; Saulnier, L.; Shewry, P.R.; Mills, E.N.C. Spectroscopic analysis of diversity of Arabinoxylan structures in endosperm cell walls of wheat cultivars (Triticum aestivum) in the HEALTHGRAIN diversity collection. J. Agric. Food Chem. 2011, 59, 7075–7082. [Google Scholar] [CrossRef]
- Damen, B.; Pollet, A.; Dornez, E.; Broekaert, W.F.; Haesendonck, I.V.; Trogh, I.; Arnaut, F.; Delcour, J.A.; Courtin, C.M. Xylanase-mediated in situ production of arabinoxylan oligosaccharides with prebiotic potential in whole meal breads and breads enriched with arabinoxylan rich material. Food Chem. 2012, 131, 111–118. [Google Scholar] [CrossRef]
- Lu, Z.X.; Walker, K.Z.; Muir, J.G.; Mascara, T.; O’Dea, K. Arabinoxylan fiber, a byproduct of wheat flour processing, reduces the postprandial glucose response in normoglycemic subjects. Am. J. Clin. Nutr. 2000, 71, 1123–1128. [Google Scholar]
- Lu, Z.X.; Walker, K.Z.; Muir, J.G.; O’Dea, K. Arabinoxylan fibre improves metabolic control in people with Type II diabetes. Eur. J. Clin. Nutr. 2004, 58, 621–628. [Google Scholar] [CrossRef]
- Zhou, S.; Liu, X.; Guo, Y.; Wang, Q.; Peng, D.; Cao, L. Comparison of the immunological activities of arabinoxylans from wheat bran with alkali and xylanase-aided extraction. Carbohydr. Polym. 2010, 81, 784–789. [Google Scholar] [CrossRef]
- Cao, L.; Liu, X.Z.; Qian, T.X.; Sun, G.B.; Guo, Y.; Chang, F.J.; Zhou, S.M.; Sun, X.B. Antitumor and immunomodulatory activity of arabinoxylans: A major constituent of wheat bran. Int. J. Biol. Macromol. 2011, 48, 160–164. [Google Scholar] [CrossRef]
- Cloetens, L.; Swennen, K.; de Preter, V.; Broekaert, W.F.; Courtin, C.M.; Delcour, J.A.; Rutgeerts, P.; Verbeke, K. Effect of arabinoxylo-oligosaccharides on proximal gastrointestinal motility and digestion in healthy volunteers. e-SPEN 2008, 35, e220–e225. [Google Scholar]
- Grootaert, C.; Delcour, J.A.; Courtin, C.M.; Broekaert, W.F.; Verstraete, W.; Wiele van de, T. Microbial metabolism and prebiotic potency of arabinoxylan oligosaccharides in the human intestine. Trends Food Sci. Technol. 2007, 18, 64–71. [Google Scholar] [CrossRef]
- Grootaert, C.; van den Abbeele1, P.; Marzorati1, M.; Broekaert, W.F.; Courtin, M.C.; Delcour, J.A.; Verstraete1, W.; Wiele van de, T. Comparison of prebiotic effects of arabinoxylan oligosaccharides and inulin in a simulator of the human intestinal microbial ecosystem. FEMS Microbiol. Ecol. 2009, 69, 231–242. [Google Scholar] [CrossRef]
- Maki, K.C.; Gibson, G.R.; Dickmann, R.S.; Kendall, C.W.C.; Chen, C.Y.O.; Costabile, A.; Comelli, E.M.; McKay, D.L.; Almeida, N.G.; Jenkins, D.; et al. Digestive and physiologic effects of a wheat bran extract, arabino-xylan-oligosaccharide, in breakfast cereal. Nutrition 2012, 28, 1115–1121. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. Agency Response Letter GRAS Notice No. GRN 000343. CFSAN/Office of Food Additive Safety. Available online: http://www.fda.gov/Food/ IngredientsPackagingLabeling/GRAS/NoticeInventory/ucm235936.htm (access on 15 February 2013).
- AACC Internationl, Approved Methods of Analysis, 11th Ed. Method 74-09.01. Measurement of Bread Firmness by Universal Testing Machine; AACC International: St. Paul, MN, USA, 1999.
- Douglas, S.G. A rapid method for the determination of pentosans in wheat flour. Food Chem. 1980, 7, 139–145. [Google Scholar] [CrossRef]
- Hollmann, J.; Lindhauer, M.G. Pilot-scale isolation of glucuronoarabinoxylans from wheat bran. Carbohydr. Polym. 2005, 59, 225–230. [Google Scholar] [CrossRef]
- Bataillon, M.; Mathaly, P.; Crdinali, A.P.N.; Duchiron, F. Extraction and purification ofarabinoxylan from destarched wheat bran in a pilot scale. Ind. Crop. Prod. 1998, 8, 37–43. [Google Scholar] [CrossRef]
- Courtin, C.M.; Delcour, J.A. Arabinoxylans and endoxylanases in wheat flour bread-making. J. Cereal Sci. 2002, 35, 225–243. [Google Scholar] [CrossRef]
- Redgwell, R.J.; de Michieli, J.-H.; Fischer, M.; Reymond, S.; Nicolas, P.; Sievert, D. Xylanase induced changes to water- and alkaliextractable arabinoxylans in wheat flour: Their role in lowering batter viscosity. J. Cereal Sci. 2001, 33, 83–96. [Google Scholar] [CrossRef]
- Skendi, A.; Biliaderis, C.G; Izydorczyk, M.S.; Zervou, M.; Zoumpoulakis, P. Structural variation and rheological properties of water-extractable arabinoxylans from six Greek wheat cultivars. Food Chem. 2011, 126, 526–536. [Google Scholar]
- Courtin, C.M.; Delcour, J.A. Physicochemical and bread-making properties of low molecular weight wheat derived arabinoxylans. J. Agri. Food Chem. 1998, 46, 4066–4073. [Google Scholar] [CrossRef]
- Cleemput, G.; Booij, C.; Hessing, M.; Gruppen, H.; Delcour, J.A. Solubilisation and changes in molecular weight distribution of arabinoxylan and protein in wheat flours during bread-making, and the effects of endogenous arabinoxylan hydrolysing enzymes. J. Cereal Sci. 2000, 26, 55–66. [Google Scholar]
- Goesaert, H.; Brijs, K.; Veraverbeke, W.S.; Courtin, C.M.; Gebruers, K.J.; Delcour, J.A. Wheat flour constituents: How they impact bread quality, and how to impact their functionality. Trends Food Sci. Technol. 2005, 16, 12–30. [Google Scholar]
- Courtin, C.M.; Gelders, G.G.; Delcour, J.A. The use of two endoxylanases with different substrate selectivity provides insight into the functionality of arabinoxylans in wheat flour breadmaking. Cereal Chem. 2001, 78, 564–571. [Google Scholar] [CrossRef]
- Henrissat, B.; Bairoch, A. New families in the classification of glycosyl hydrolases based on amino acid sequence similarities. Biochem. J. 1993, 293, 781–788. [Google Scholar]
- Reilly, P.J. Xylanases, Structure and Function. In Trends in the Biology of Fermentation for Fuels and Chemicals; Hollaender, A., Ed.; Plenum Press: New York, NY, USA, 1981; pp. 111–129. [Google Scholar]
- Li, K.; Azadi, P.; Collins, R.; Tolan, J.; Kim, J.S.; Eriksson, K.L. Relationships between activities of xylanases and xylan structures. Enzyme Microb. Technol. 2000, 27, 89–94. [Google Scholar]
- Vardakou, M.; Katapodis, P.; Samiotaki, M.; Kekos, D.; Panayotou, G.; Christakopoulos, P. Mode of action of family 10 and 11 endoxylanases on water-unextractable arabinoxylan. Int. J. Biol. Macromol. 2003, 33, 129–134. [Google Scholar] [CrossRef]
- Pitkanen, L.; Tuomainen, P.; Virkki, L.; Tenkanen, M. Molecular characterization and solution properties of enzymatically tailored arabinoxylans. Int. J. Biol. Macromol. 2011, 49, 963–969. [Google Scholar] [CrossRef]
- Courtin, C.M.; Roelants, A.; Delcour, J.A. Fractionation-reconstitution experiments provide insight into the role of endoxylanases in bread-making. J. Agric. Food Chem. 1999, 47, 1870–1877. [Google Scholar] [CrossRef]
- Arnaut, F.; Willem, F.; Broekaert, W.F.; Courtin, C.; Delcour, J.A.; Georis, J.; van Haesendonck, I.P.H. Bread with Increased Arabinoxylo-Oligosaccharide Content. U.S. Patent WO2008087167 A, 24 July 2008. [Google Scholar]
- Makaravicius, T.; Basinskiene, L.; Juodeikiene, G.; van Gool, M.P.; Schols, H.A. Production of oligosaccharides from extruded wheat and rye biomass using enzymatic treatment. Catal. Today 2012, 196, 16–25. [Google Scholar]
- Möhlig, M.; Koebnick, C.; Weickert, M.O; Lueder, W.; Otto, B.; Steiniger, J.; Twilfert, M.; Meuser, F.; Pfeiffer, A.F.; Zunft, H.J. Arabinoxylan-enriched meal increases serum ghrelin levels in healthy humans. Horm. Metab. Res. 2005, 37, 303–308. [Google Scholar]
- European Food Safety Authority. Scientific Opinion on the substantiation of health claims related to arabinoxylan produced from wheat endosperm and reduction of post-prandial glycaemic responses (ID 830) pursuant to Article 13(1) of Regulation (EC) No 1924/2006. EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). EFSA J. 2011, 9, 2205.
- Hughes, S.A.; Shewry, P.R.; Li, L.; Gibson, G.R.; Sanz, M.L.; Rastall, R.A. In vitro fermentation by human fecal microflora of wheat arabinoxylans. J. Agric. Food Chem. 2007, 55, 4589–4595. [Google Scholar]
- Vardakou, M.; Palop, C.N.; Christakopoulos, P.; Faulds, C.B.; Gasson, M.A.; Narbad, A. Evaluation of the prebiotic properties of wheat arabinoxylan fractions and induction of hydrolase activity in gut microflora. Int. J. Food Microbiol. 2008, 123, 166–170. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, W.; Hu, H.; Wang, Q.; Brennan, C.S. Molecular Features of Wheat Endosperm Arabinoxylan Inclusion in Functional Bread. Foods 2013, 2, 225-237. https://doi.org/10.3390/foods2020225
Li W, Hu H, Wang Q, Brennan CS. Molecular Features of Wheat Endosperm Arabinoxylan Inclusion in Functional Bread. Foods. 2013; 2(2):225-237. https://doi.org/10.3390/foods2020225
Chicago/Turabian StyleLi, Weili, Hui Hu, Qi Wang, and Charles S. Brennan. 2013. "Molecular Features of Wheat Endosperm Arabinoxylan Inclusion in Functional Bread" Foods 2, no. 2: 225-237. https://doi.org/10.3390/foods2020225






