Effects of Rosemary Oil (Rosmarinus officinalis) on the Shelf-Life of Minced Rainbow Trout (Oncorhynchus mykiss) during Refrigerated Storage
Abstract
:1. Introduction
2. Experimental Section
2.1. Preparation of Minced Fish Muscle Samples
2.2. pH Measurement
2.3. TBARS Measurement
2.4. Color Measurement
2.5. Gas-Chromatographic Analysis of the Fatty Acids
2.6. Sample Preparation for HPLC-MS/MS and GC/MS Analyses
2.7. Determination of Biogenic Amines
2.8. Gas-Chromatographic Analysis of Terpenes and Sesquiterpenes
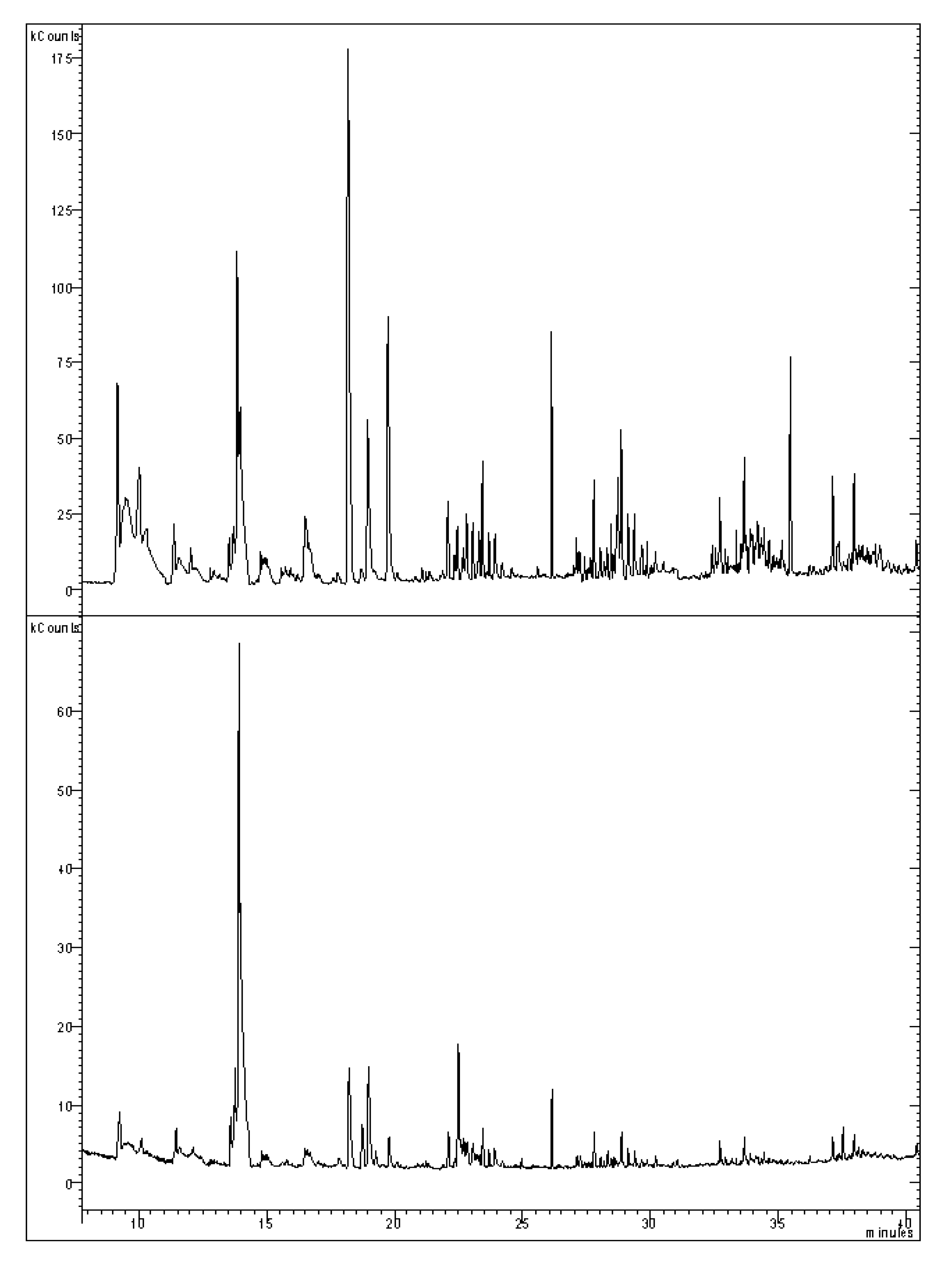
2.9. Statistical Analysis
3. Results and Discussion
3.1. Freshness Indicators
| C | RO 0.2 | RO 1.0 | RO 3.0 | SEM | Significance | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Days | 3 | 9 | 3 | 9 | 3 | 9 | 3 | 9 | Treatment | Time | Interaction | |
| pH | 6.75 | 7.04 | 6.70 | 7.01 | 6.73 | 6.84 | 6.65 | 6.89 | 0.003 | 0.007 | 0.000 | 0.044 |
| TBARS | 0.41 | 0.23 | 0.27 | 0.13 | 0.19 | 0.18 | 0.90 | 0.26 | 0.017 | 0.000 | 0.000 | 0.004 |
| L * | 70.7 | 71.2 | 70.6 | 75.8 | 75.2 | 76.1 | 80.2 | 79.9 | 0.328 | 0.000 | 0.000 | 0.000 |
| a * | 2.66 | 2.72 | 3.01 | 2.79 | 1.31 | 2.51 | 0.79 | 0.29 | 0.020 | 0.000 | 0.030 | 0.000 |
| b * | 12.1 | 11.1 | 11.6 | 13.4 | 12.6 | 11.9 | 12.2 | 13.2 | 0.120 | 0.000 | 0.075 | 0.000 |
| Chroma | 12.4 | 11.4 | 12.0 | 13.7 | 12.7 | 12.2 | 12.2 | 13.2 | 0.123 | 0.001 | 0.053 | 0.000 |
| Hue | 77.6 | 76.2 | 75.5 | 78.3 | 84.1 | 78.1 | 86.3 | 88.7 | 0.345 | 0.000 | 0.035 | 0.000 |
3.2. Fatty Acid Profile
| C | RO 0.2 | RO 1.0 | RO 3.0 | SEM | Significance | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Days | 3 | 9 | 3 | 9 | 3 | 9 | 3 | 9 | Treatment | Time | Interaction | |
| C14:0 | 3.76 | 3.12 | 3.63 | 2.41 | 3.02 | 2.81 | 2.50 | 3.05 | 0.067 | 0.003 | 0.002 | 0.000 |
| C16:0 | 20.0 | 15.6 | 20.0 | 11.9 | 15.2 | 12.8 | 14.7 | 14.7 | 1.319 | 0.000 | 0.000 | 0.000 |
| C16:1 | 4.33 | 3.77 | 4.23 | 3.20 | 4.13 | 3.37 | 3.33 | 4.17 | 0.127 | 0.356 | 0.018 | 0.002 |
| C18:0 | 6.17 | 5.30 | 6.20 | 3.87 | 4.37 | 3.80 | 4.40 | 4.03 | 0.077 | 0.000 | 0.000 | 0.000 |
| C18:1n9 | 16.9 | 14.1 | 17.0 | 12.3 | 15.3 | 11.8 | 13.2 | 15.9 | 1.739 | 0.179 | 0.001 | 0.001 |
| C18:1n7 | 2.90 | 2.40 | 2.83 | 1.93 | 2.37 | 2.03 | 2.07 | 2.40 | 0.038 | 0.004 | 0.000 | 0.001 |
| C18:2n6 | 14.0 | 13.4 | 15.4 | 15.3 | 19.4 | 9.50 | 17.1 | 20.9 | 2.424 | 0.000 | 0.015 | 0.000 |
| C18:3n3 | 1.27 | 1.40 | 2.77 | 1.83 | 2.23 | 0.70 | 2.07 | 2.40 | 0.618 | 0.100 | 0.139 | 0.170 |
| C18:4n3 | 0.00 | 0.27 | 0.00 | 0.12 | 0.00 | 0.00 | 0.32 | 0.06 | 0.022 | 0.001 | 0.018 | 0.000 |
| C20:1 | 0.77 | 0.87 | 0.73 | 0.60 | 0.33 | 0.00 | 0.60 | 0.63 | 0.011 | 0.000 | 0.068 | 0.011 |
| C20:2n6 | 0.63 | 0.60 | 0.00 | 0.67 | 0.73 | 0.00 | 0.73 | 0.80 | 0.003 | 0.000 | 0.710 | 0.000 |
| C20:3n3 | 1.30 | 0.50 | 0.77 | 1.23 | 0.37 | 2.53 | 0.30 | 0.40 | 0.292 | 0.023 | 0.043 | 0.002 |
| C20:4n6 | 0.00 | 0.00 | 0.00 | 0.33 | 0.70 | 0.00 | 0.70 | 0.70 | 0.012 | 0.000 | 0.054 | 0.000 |
| C20:4n3 | 0.00 | 0.00 | 0.00 | 0.17 | 0.73 | 0.00 | 0.63 | 0.70 | 0.011 | 0.000 | 0.011 | 0.000 |
| C20:5n3 | 2.03 | 2.57 | 2.13 | 3.50 | 6.33 | 1.10 | 6.33 | 5.90 | 0.132 | 0.000 | 0.000 | 0.000 |
| C22:5n3 | 0.00 | 0.00 | 0.00 | 1.40 | 2.23 | 0.00 | 2.17 | 2.30 | 0.010 | 0.000 | 0.000 | 0.000 |
| C22:6n3 | 5.83 | 5.63 | 5.60 | 8.57 | 14.93 | 12.47 | 17.83 | 13.97 | 0.835 | 0.000 | 0.000 | 0.000 |
3.3. Biogenic Amines Content
| C | RO 0.2 | RO 1.0 | RO 3.0 | SEM | Significance | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Days | 3 | 9 | 3 | 9 | 3 | 9 | 3 | 9 | Treatment | Time | Interaction | |
| Putrescine | 27.2 | 46.5 | 24.6 | 37.0 | 10.7 | 24.9 | 4.7 | 6.5 | 18.8 | 0.000 | 0.000 | 0.052 |
| Cadaverine | 16.9 | 29.6 | 15.6 | 22.2 | 6.1 | 18.0 | 2.2 | 3.8 | 35.5 | 0.007 | 0.025 | 0.559 |
| Spermidine | 2.1 | 1.8 | 1.7 | 2.4 | 1.6 | 1.4 | 1.1 | 1.2 | 0.57 | 0.293 | 0.930 | 0.759 |
| Tyramine | 13.4 | 16.0 | 17.0 | 13.5 | 2.8 | 14.9 | 2.8 | 4.6 | 6.4 | 0.000 | 0.015 | 0.004 |
| Histamine | 17.3 | 320.1 | 103.4 | 271.2 | 5.0 | 50.9 | 8.0 | 16.5 | 2808 | 0.000 | 0.000 | 0.001 |
| Spermine | ND | ND | ND | ND | ND | ND | ND | ND | - | - | - | - |
| Dopamine | ND | ND | ND | ND | ND | ND | ND | ND | - | - | - | - |
3.4. Characterization and Content of Terpenoid Component
| RO 0.2 | RO 1.0 | RO 3.0 | SEM | Significance | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Days | 3 | 9 | 3 | 9 | 3 | 9 | Treatment | Time | Interaction | |
| α-Pinene | 10,609 | 14,498 | 16,228 | 12,228 | 334,740 | 555,058 | 2.1 × 109 | 0.000 | 0.005 | 0.002 |
| Eucalyptol | 4366 | 30,740 | 133,773 | 224,546 | 927,153 | 1,293,565 | 3.5 × 109 | 0.000 | 0.000 | 0.001 |
| Camphor | 18,831 | 71,698 | 158,171 | 243,992 | 1,125,807 | 146,497 | 3.8 × 109 | 0.000 | 0.000 | 0.000 |
| Borneol | 547 | 3106 | 13,458 | 26,446 | 43,900 | 38,061 | 8.4 × 106 | 0.000 | 0.036 | 0.000 |
| Isoborneol | 4525 | 36,386 | 49,713 | 66,694 | 398,769 | 409,514 | 1.7 × 108 | 0.000 | 0.007 | 0.382 |
| α-Terpineol | ND | ND | 36,132 | 40,596 | 391,096 | 495,470 | 2.7 × 108 | 0.000 | 0.001 | 0.000 |
| Bornylacetate | ND | 5493 | 2278 | 1598 | 69,125 | 50,527 | 5.0 × 106 | 0.000 | 0.001 | 0.000 |
| β-Caryophyllene | ND | ND | 5332 | 3084 | 250,712 | 176,756 | 2.2 × 108 | 0.000 | 0.004 | 0.001 |
4. Conclusions
Acknowledgment
Conflicts of Interest
References
- Lindberg Madsen, H.; Bertelsen, G. Spices as antioxidants. Trends Food Sci. Technol. 1995, 6, 271–278. [Google Scholar] [CrossRef]
- Liu, H.F.; Booren, A.M.; Gray, J.I.; Crackel, R.L. Antioxidant efficacy of oleoresin rosemary and sodium tripolyphosphate in restructured pork steaks. J. Food Sci. 1992, 57, 803–806. [Google Scholar] [CrossRef]
- Bousbia, N.; Abert Vian, M.; Ferhat, M.A.; Petitcolas, E.; Meklati, B.Y.; Chemat, F. Comparison of two isolation methods for essential oil from rosemary leaves: Hydrodistillation and microwave hydrodiffusion and gravity. Food Chem. 2009, 114, 355–362. [Google Scholar] [CrossRef]
- Erkan, N.; Ayranci, G.; Ayranci, E. Antioxidant activities of rosemary (Rosmarinus officinalis L.) extract, blackseed (Nigella sativa L.) essential oil, carnosic acid, rosmarinic acid and sesamol. Food Chem. 2008, 110, 76–82. [Google Scholar] [CrossRef]
- Akhtar, P.; Gray, J.I.; Booren, A.M.; Garling, D.L. Effect of dietary components and surface application of oleoresin rosemary on lipid stability of rainbow trout (Oncorhynchus mykiss) muscle during refrigerated and frozen storage. J. Food Lipids 1998, 5, 43–58. [Google Scholar] [CrossRef]
- Djenane, D.; Sánchez-Escalante, A.; Beltrán, J.A.; Roncalés, P. Extension of the shelf-life of beef steaks packaged in a modified atmosphere by treatment with rosemary and displayed under UV-free lighting. Meat Sci. 2003, 64, 417–426. [Google Scholar] [CrossRef]
- Serdaroğlu, M.; Felekoğlu, E. Effects of using rosemary extract and onion juice on oxidative stability of sardine (Sardina pilchardus) mince. J. Food Qual. 2005, 28, 109–120. [Google Scholar] [CrossRef]
- Cuvelier, M.E.; Richard, H.; Berset, C. Antioxidative activity of phenolic composition of pilot-plant and commercial extracts of sage and rosemary. J. Am. Oil Chem. Soc. 1996, 73, 645–652. [Google Scholar] [CrossRef]
- Frankel, E.N.; Huang, S.W.; Aeschbach, R.; Prior, E. Antioxidant activity of a rosemary extract and its constituents, carnosic acid, carnosol, and rosmarinic acid, in bulk oil and oil-in-water emulsion. J. Agric. Food Chem. 1996, 44, 131–135. [Google Scholar] [CrossRef]
- Fu, Y.; Zu, Y.; Chen, L.; Shi, X.; Wang, Z.; Sun, S.; Efferth, T. Antimicrobial activity of clove and rosemary essential oils alone and in combination. Phytother. Res. 2007, 21, 989–994. [Google Scholar] [CrossRef]
- Murphy, A.; Kerry, J.P.; Buckley, J.; Gray, I. The antioxidative properties of rosemary oleoresin and inhibition of off-flavours in precooked roast beef slices. J. Sci. Food Agric. 1998, 77, 235–243. [Google Scholar] [CrossRef]
- Corbo, M.R.; Speranza, B.; Filippone, A.; Granatiero, S.; Conte, A.; Sinigaglia, M.; Del Nobile, M.A. Study on the synergic effect of natural compounds on the microbial quality decay of packed fish hamburger. Int. J. Food Microbiol. 2008, 127, 261–267. [Google Scholar] [CrossRef]
- Gram, L.; Dalgaard, P. Fish spoilage bacteria-problems and solutions. Curr. Opin. Biotechnol. 2002, 13, 262–266. [Google Scholar] [CrossRef]
- zogul, F.; Kuley, E.; Kenar, M. Effects of rosemary and sage tea extract on biogenic amines formation of sardine (Sardina pilchardus) fillets. Int. J. Food Sci. Technol. 2011, 46, 761–766. [Google Scholar] [CrossRef]
- Emir Çoban, Ö.; Özpolat, E. The effects of different concentrations of rosemary (Rosmarinus officinalis) extract on the shelf-life of hot-smoked and vacuum-packed Luciobarbus esocinus fillets. J. Food Process. Preserv. 2012. [Google Scholar] [CrossRef]
- Özyurt, G.; Kuley, E.; Balikçi, E.; Kaçar, Ç.; Gökdogan, S.; Etyemez, M.; Özogul, F. Effect of the icing with rosemary extract on the oxidative stability and biogenic amine formation in sardine (Sardinella aurita) during chilled storage. Food Bioprocess Technol. 2012, 5, 2777–2786. [Google Scholar] [CrossRef]
- Bao, H.N.; Ushio, H.; Ohshima, T. Antioxidative activities of mushroom (Flammulina velutipes) extract added to big eye tuna meat: Dose-dependent efficacy and comparison with other biological antioxidants. J. Food Sci. 2009, 74, 162–169. [Google Scholar]
- Sárraga, C.; Carreras, I.; Garcia Regueiro, J.A.; Guardia, M.D.; Guerriero, L. Effects of alpha-tocopheryl acetate and beta-carotene dietary supplementation on the antioxidant enzymes, TBARS and sensory attributes of turkey meat. Br. Poult. Sci. 2006, 47, 700–707. [Google Scholar]
- Commission Internationale de L’Eclairage (CIE), Colorimetry; CIE Central Bureau: Vienna, Austria, 1976.
- Boccard, R.; Butcher, L.; Casleels, E.; Cosentino, E.; Dransfìeld, E.; Hood, D.E.; Joseph, R.L.; Macdougall, D.B.; Rhodes, D.N.; Schön, I.; et al. Procedures for measuring meat quality characteristics in beef production experiments. Report of a working group in the commission of the European communities (CEC), beef production research programme. Livest. Prod. Sci. 1981, 8, 385–397. [Google Scholar] [CrossRef]
- Peiretti, P.G.; Meineri, G. Effects on growth performance, carcass characteristics, and the fat and meat fatty acid profile of rabbits fed diets with chia (Salvia hispanica L.) seed supplements. Meat Sci. 2008, 80, 1116–1121. [Google Scholar] [CrossRef]
- Calbiani, F.; Careri, M.; Elviri, L.; Mangia, A.; Pistara, L.; Zagnoni, I. Rapid assay for analyzing biogenic amines in cheese: Matrix solid-phase dispersion followed by liquid chromatography-electrospray-tandem mass spectrometry. J. Agric. Food Chem. 2005, 53, 3779–3783. [Google Scholar]
- Beretta, G.; Artali, R.; Facino, R.M.; Gelmini, F. An analytical and theoretical approach for the profiling of the antioxidant activity of essential oils: The case of Rosmarinus officinalis L. J. Pharm. Biomed. Anal. 2011, 55, 1255–1264. [Google Scholar] [CrossRef]
- Formanek, Z.; Kerry, J.P.; Higgins, F.M.; Buckley, D.J.; Morrissey, P.A.; Farkas, J. Addition of synthetic and natural antioxidants to α-tocopheryl acetate supplemented beef patties: Effects of antioxidants and packaging on lipid oxidation. Meat Sci. 2001, 58, 337–341. [Google Scholar] [CrossRef]
- Tironi, V.A.; Tomás, M.C.; Añón, M.C. Quality loss during the frozen storage of sea salmon (Pseudopercis semifasciata). Effect of rosemary (Rosmarinus officinalis L.) extract. Food Sci. Technol. 2010, 43, 263–272. [Google Scholar] [CrossRef]
- Tironi, V.; Tomás, M.; Añón, M. Lipid and protein changes in chilled sea salmon (Pseudopercis semifasciata): Effect of previous rosemary extract (Rosmarinus officinalis L.) application. Int. J. Food Sci. Technol. 2009, 44, 1254–1262. [Google Scholar] [CrossRef]
- Food and Drug Administration (FDA), Scombrotoxin (Histamine) Formation. In Fish and Fishery Products Hazards and Control Guide; FDA: Washington, DC, USA, 1998; pp. 73–90.
- Council of the European Communities. Council Directive of 22 July 1991 laying down the health conditions for the productions and the placing on the market of fishery products (91/493/EEC). Off. J. Eur. Communities 1991, 34, 15–34.
- Özogul, F.; Özogul, Y. Biogenic amine content and biogenic amine quality indices of sardines (Sardina pilchardus) stored in modified atmosphere packaging and vacuum packaging. Food Chem. 2006, 99, 574–578. [Google Scholar] [CrossRef]
- Ababouch, L.; Afilal, M.E.; Benabdeljelil, H.; Busta, F.F. Quantitative changes in bacteria, amino acids and biogenic amines in sardine (Sardina pilchardus) stored at ambient temperature (25–28 °C) and in ice. Int. J. Food Sci. Technol. 1991, 26, 297–306. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Peiretti, P.G.; Gai, F.; Ortoffi, M.; Aigotti, R.; Medana, C. Effects of Rosemary Oil (Rosmarinus officinalis) on the Shelf-Life of Minced Rainbow Trout (Oncorhynchus mykiss) during Refrigerated Storage. Foods 2012, 1, 28-39. https://doi.org/10.3390/foods1010028
Peiretti PG, Gai F, Ortoffi M, Aigotti R, Medana C. Effects of Rosemary Oil (Rosmarinus officinalis) on the Shelf-Life of Minced Rainbow Trout (Oncorhynchus mykiss) during Refrigerated Storage. Foods. 2012; 1(1):28-39. https://doi.org/10.3390/foods1010028
Chicago/Turabian StylePeiretti, Pier Giorgio, Francesco Gai, Marco Ortoffi, Riccardo Aigotti, and Claudio Medana. 2012. "Effects of Rosemary Oil (Rosmarinus officinalis) on the Shelf-Life of Minced Rainbow Trout (Oncorhynchus mykiss) during Refrigerated Storage" Foods 1, no. 1: 28-39. https://doi.org/10.3390/foods1010028
APA StylePeiretti, P. G., Gai, F., Ortoffi, M., Aigotti, R., & Medana, C. (2012). Effects of Rosemary Oil (Rosmarinus officinalis) on the Shelf-Life of Minced Rainbow Trout (Oncorhynchus mykiss) during Refrigerated Storage. Foods, 1(1), 28-39. https://doi.org/10.3390/foods1010028








