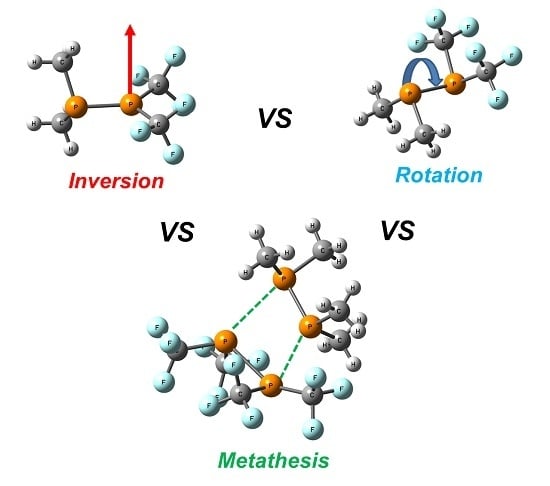
PP-Rotation, P-Inversion and Metathesis in Diphosphines Studied by DFT Calculations: Comments on Some Literature Conflicts
Abstract
:1. Introduction
2. Results and Discussion
2.1. Rotational Isomerization
2.2. Stereomutation by Pyramidal Inversion
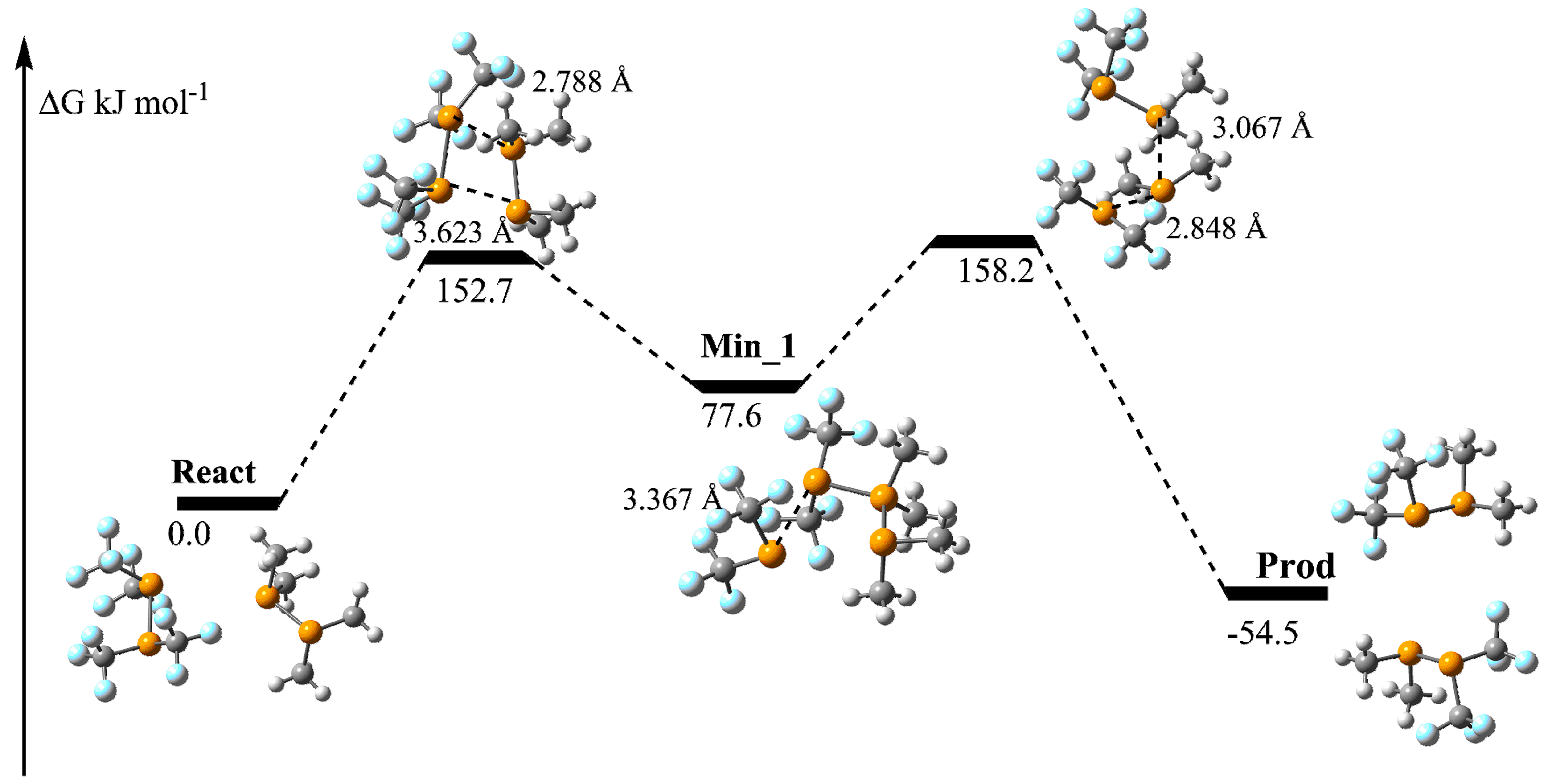
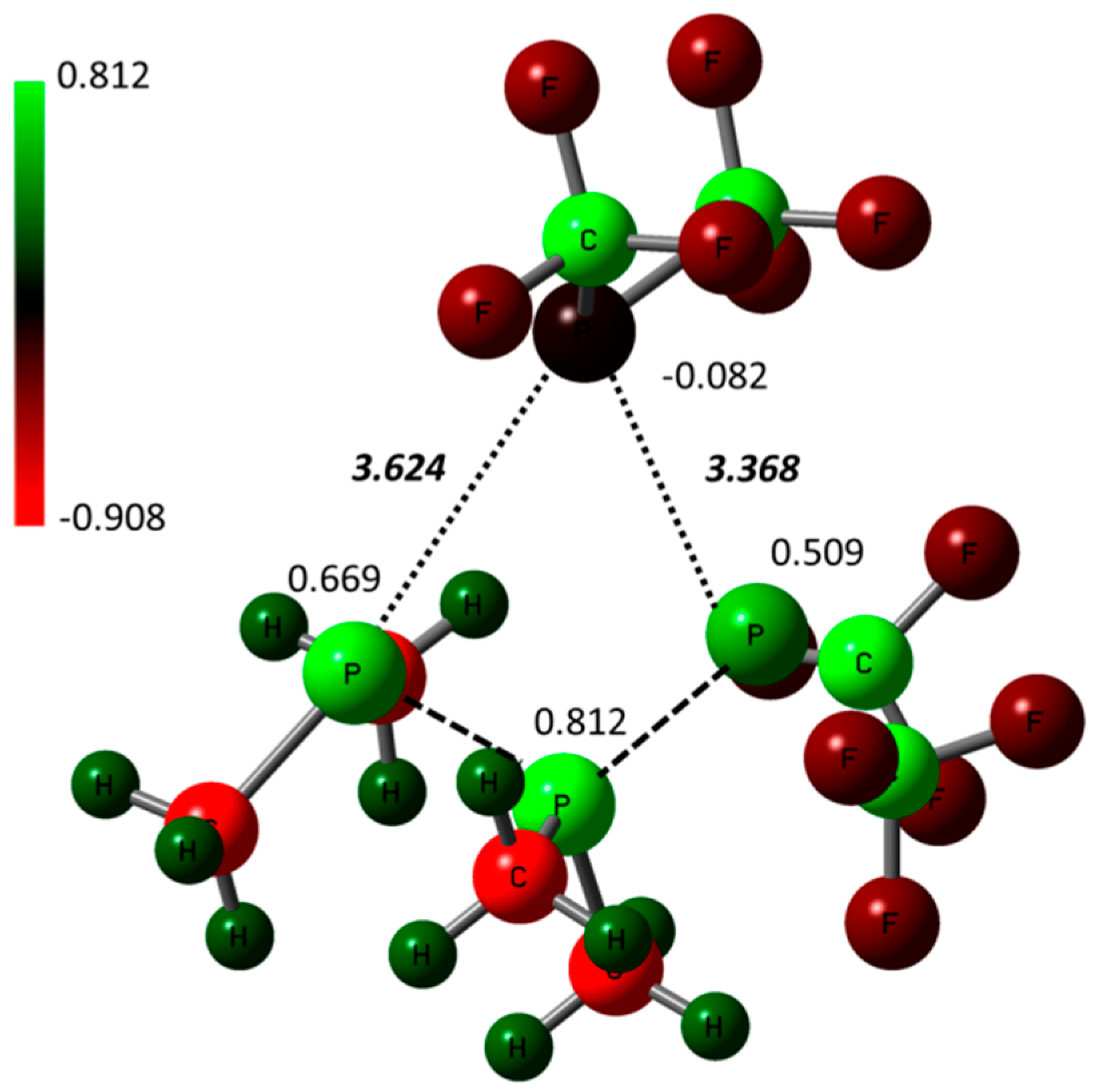
2.3. Stereomutation by Metathesis Self-Reaction
3. Methods
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Maier, L. Direkt synthese von Alkyl-halogen-phosphinen. Angew. Chem. 1959, 71, 574–575. [Google Scholar] [CrossRef]
- Mayer, L. Organische Phosphorverbindungen, I. Darstellung asymmetrischer Tetraalkyl- und Dialkyl-diaryl-diphosphindisulfide. Chem. Ber. 1961, 94, 3043–3050. [Google Scholar] [CrossRef]
- Fluck, E.; Issleib, K. Untersuchungen der kernmagnetischen Resonanzspektren von Phosphorverbindungen, VIII: 31P-Resonanzspektren von Phosphinen, Biphosphinen und Biphosphindisulfiden. Chem. Ber. 1965, 98, 2674–2680. [Google Scholar] [CrossRef]
- Lambert, J.B.; Mueller, D.C. The Inversion of Diphosphines. J. Am. Chem. Soc. 1966, 88, 3669–3670. [Google Scholar] [CrossRef]
- Lambert, J.B.; Jackson, G.F.; Mueller, D.C. The stereochemical lability of diphosphines and diarsines. J. Am. Chem. Soc. 1968, 90, 6401–6405. [Google Scholar] [CrossRef]
- Rauk, A.; Allen, L.C.; Mislow, K. Pyramidal Inversion. Angew. Chem. Int. Ed. Engl. 1970, 9, 400–414. [Google Scholar] [CrossRef]
- McFarlane, H.C.E.; McFarlane, W. Intermolecular association of biphosphines. J. Chem. Soc. Chem. Commun. 1972, 1189–1190. [Google Scholar] [CrossRef]
- Harris, R.K.; Norval, E.M.; Fild, M. Nuclear magnetic resonance studies of scrambling reactions in solutions of tetra-alkyl- and tetra-aryl-diphosphanes. J. Chem. Soc. Dalton Trans. 1979, 826–831. [Google Scholar] [CrossRef]
- McFarlane, H.C.E.; McFarlane, W. Sign and magnitude of 1J(PP) in some biphosphines with bulky substituents. J. Chem. Soc. Chem. Commun. 1975, 582–583. [Google Scholar] [CrossRef]
- Issleib, K.; Krech, K. Alkali-Phosphorverbindungen und ihr reaktives Verhalten, XXXI: Unsymmetrische Biphosphine des Typs R2PPR′2. Chem. Ber. 1965, 98, 1093–1096. [Google Scholar] [CrossRef]
- Cavell, R.G.; Dobbie, R.C. Preparation and properties of some bis(trifluoromethyl)-phosphorus and -arsenic derivatives containing P–P, P–As, and As–As bonds. J. Chem. Soc. A Inorg. Phys. Theor. 1968, 1406–1410. [Google Scholar] [CrossRef]
- Avens, L.R.; Wolcott, R.A.; Cribbs, L.V.; Mills, J.L. Some perfluoroalkyl-substituted tripnicogens and their hydrolysis to yield chiral dipnicogens. Inorg. Chem. 1989, 28, 200–205. [Google Scholar] [CrossRef]
- Avens, L.R.; Cribbs, L.V.; Mills, J.L. Reactions of secondary phosphines with a phosphorus–phosphorus bond and related reactions. Inorg. Chem. 1989, 28, 205–211. [Google Scholar] [CrossRef]
- Avens, L.R.; Cribbs, L.V.; Mills, J.L. Exchange reactions of tetrakis(trifluoromethyl)diphosphine with pnicogen–pnicogen, phosphorus–hydrogen, and phosphorus–chlorine bonds. Inorg. Chem. 1989, 28, 211–214. [Google Scholar] [CrossRef]
- Cowley, A.H. The chemistry of the phosphorus–phosphorus bond. Chem. Rev. 1965, 65, 617–634. [Google Scholar] [CrossRef]
- Borisenko, K.B.; Rankin, D.W.H. P–P bond cleavage; energetics and structural changes in tetramethyldiphosphine and tetrasilyldiphosphine from ab initio MO calculations. J. Chem. Soc. Dalton Trans. 2002, 3135–3141. [Google Scholar] [CrossRef]
- Balabin, R.M. Enthalpy difference between conformations of normal alkanes: Raman spectroscopy study of n-Pentane and n-Butane. J. Phys. Chem. A 2009, 113, 1012–1019. [Google Scholar] [CrossRef] [PubMed]
- Durig, J.R.; DiYorio, J.S. Vibrational spectra and structure of organophosphorus compounds. VIII. Tetramethylbiphosphine. Inorg. Chem. 1969, 8, 2796–2802. [Google Scholar] [CrossRef]
- Durig, J.R.; MacNamee, R.W. Raman spectra of gases. J. Mol. Struct. 1973, 17, 426–428. [Google Scholar] [CrossRef]
- Azofra, L.M.; Alkorta, I.; Elguero, J. Chiral discrimination in dimers of diphosphines PH2PH2 and PH2PHF. ChemPhysChem 2014, 15, 3663–3670. [Google Scholar] [CrossRef] [PubMed]
- Durig, J.R.; Shen, Z.; Zhao, W. Conformational stability, structural parameters, and vibrational frequencies from ab initio calculations for biphosphine. J. Mol. Struct. THEOCHEM 1996, 375, 95–104. [Google Scholar]
- Becker, G.; Golla, W.; Grobe, J.; Klinkhammer, K.W.; Le Van, D.; Maulitz, A.H.; Mundt, O.; Oberhammer, H.; Sachs, M. Element−element bonds. IX.1 Structures of tetrakis(trifluoromethyl)diphosphane and -diarsane: Experimental and theoretical investigations. Inorg. Chem. 1999, 38, 1099–1107. [Google Scholar] [CrossRef] [PubMed]
- Katsyuba, S.; Schmutzler, R. Comparative studies of geometric and quasielastic characteristics of PP and SS bonds. Dalton Trans. 2008, 1465–1470. [Google Scholar] [CrossRef] [PubMed]
- Albrand, J.P.; Cogne, A.; Taieb, C. 13C, 1H and 31P dynamic NMR studies of tetraalkyldiphosphanes: Barrier to internal rotation about the P–P Bond in tetraisopropyldiphosphane. Org. Magn. Reson. 1983, 21, 246–251. [Google Scholar] [CrossRef]
- Cowley, A.H.; Dewar, M.J.S.; Goodman, D.W.; Padolina, M.C. Detection of rotational isomerism in diphosphines and diarsines by photoelectron spectroscopy. J. Am. Chem. Soc. 1974, 96, 2648–2650. [Google Scholar] [CrossRef]
- Becke, A.D. Density-Functional Thermochemistry 3. The Role of Exact Exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Lee, C.T.; Yang, W.T.; Parr, R.G. Development of the Colle-Salvetti Correlation-Energy Formula into a Functional of the Electron-Density. Phys. Rev. B 1998, 37, 785–789. [Google Scholar] [CrossRef]
- Møller, C.; Plesset, M.S. Note on an Approximation Treatment for Many-Electron Systems. Phys Rev. 1934, 46, 618–622. [Google Scholar] [CrossRef]
- Frisch, M.J.; Pople, J.A.; Binkley, J.S. Self-Consistent Molecular-Orbital Methods 25. Supplementary Functions for Gaussian-Basis Sets. J. Chem. Phys. 1984, 80, 3265–3269. [Google Scholar] [CrossRef]
- Reed, A.E.; Curtiss, L.A.; Weinhold, F. Intermolecular interactions from a natural bond orbital, donor–acceptor viewpoint. Chem. Rev. 1988, 88, 899–926. [Google Scholar] [CrossRef]
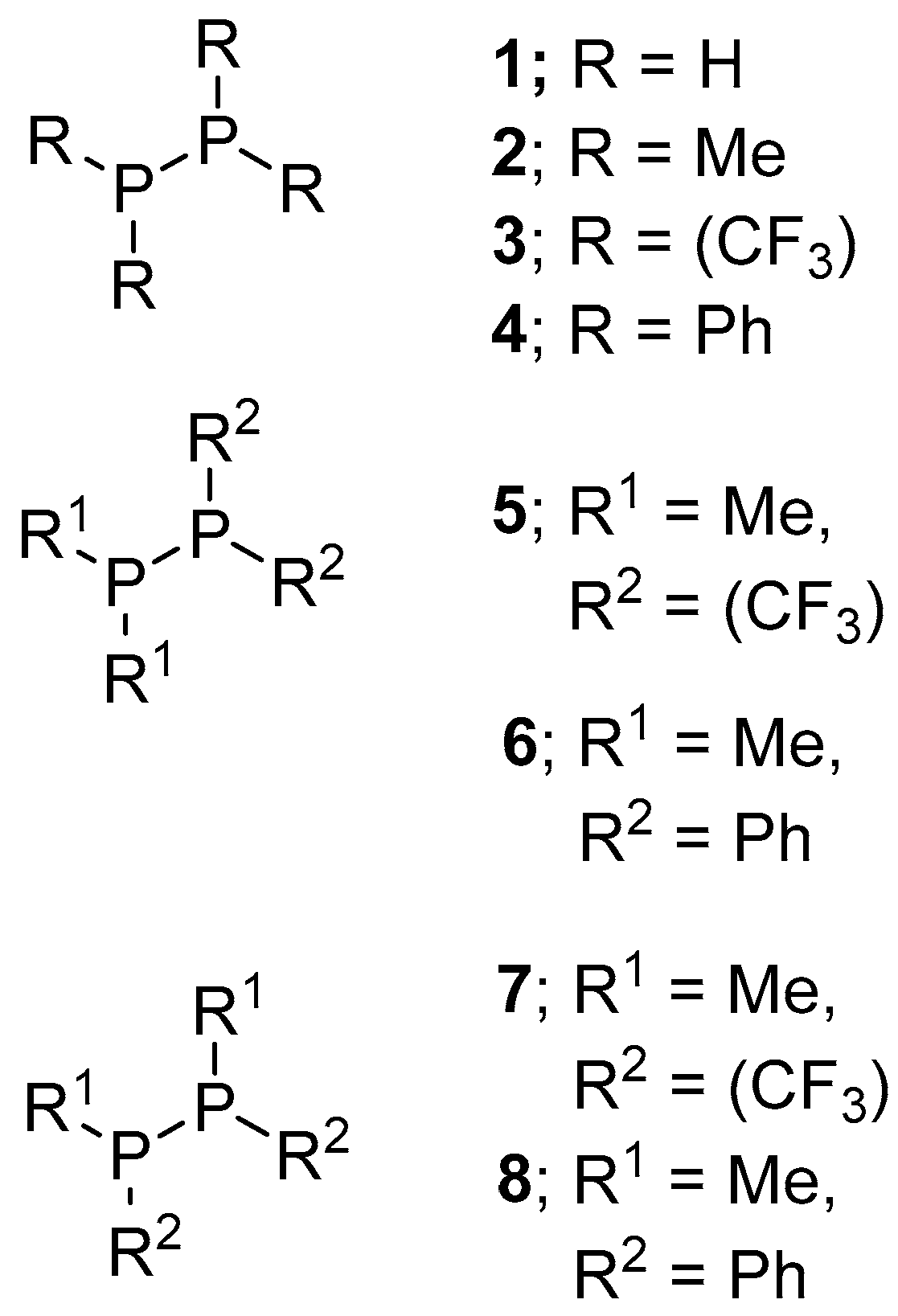

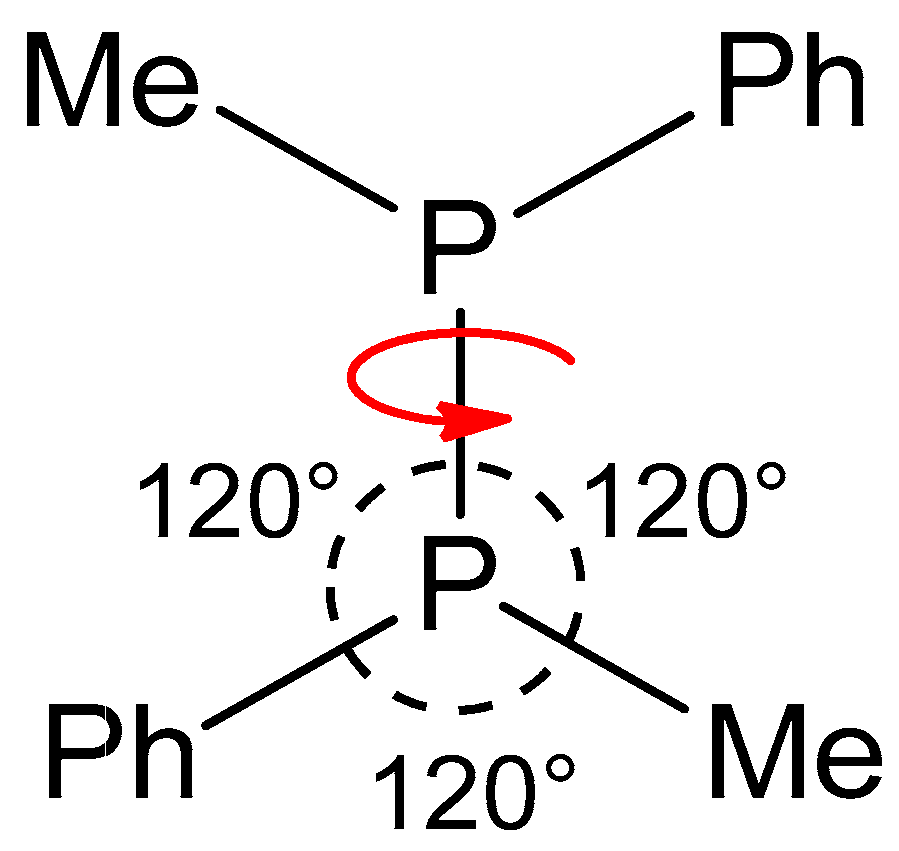
| Technique | Conclusion |
|---|---|
| Raman spectroscopy [18] | 60:40 gauche:anti (liquid) |
| IR spectroscopy [19] | 60:40 gauche:trans |
| Electron diffraction | exclusively distorted trans |
| Photoelectron spectroscopy | 16:84 gauche:trans |
| VT PE spectroscopy | trans more stable than gauche 2.1 ± 0.4 kJ/mol |
| MS fragmentation analysis | exclusively trans |
| Computational | trans more stable than gauche 6.1 vs. 5.4 kJ/mol: MP2 6-311+G* vs. B3LYP 6-311+G* |
| Compound | Compound | anti b | eclipsed (TS) c | gauche b | syn (TS) c | ||
| H4P2 | 1 | 0.0 | 3.9 | 0.9 | 15.0 | ||
| Me4P2 | 2 | 0.0 | 13.0 | 4.4 | 29.8 | ||
| (CF3)4P2 | 3 | 0.0 | 7.7 | 1.5 | 45.9 | ||
| Ph4P2 | 4 | 0.0 | 22.8 | 11.1 | 52.6 | ||
| Me2P–(CF3)2P | 5 | 1.0 | 11.0 | 0.0 | 31.2 | ||
| Me2P–Ph2P | 6 | 0.0 | 13.6 | 5.9 | 42.9 | ||
| Me(CF3)P–P(CF3)Me | 7-meso | 0.0 | 9.1 | 2.8 | 41.4 | ||
| MePhP–PPhMe | 8-meso | 0.0 | 15.2 | 4.5 | 37.4 | ||
| Compound | Compound | anti | eclipsed (R) | gauche (R) | syn | gauche (Me) | eclipsed (Me) |
| Me(CF3)P–P(CF3)Me | 7-dl | 1.1 | 23.7 | 8.6 | 35.7 | 0.0 | 5.1 |
| MePhP–PPhMe | 8-dl | 0.0 | 16.2 | 4.1 | 29.5 | 5.3 | 17.6 |
| Compound | This Work: anti:gauche Boltzmann Distribution at B3LYP/6-31G* | Literature Values anti:gauche |
|---|---|---|
| H4P2 (1) | 59:41 | - |
| Me4P2 (2) | 86:14 | 84:16 [25] |
| (CF3)4P2 (3) | 64:36 | 85:15 ±10 [22] 90:10 [25] |
| Ph4P2 (4) | 99:1 | - |
| Compound | Compound | Inversion (R) | ||
| H4P2 | 1 | 110.7 | ||
| Me4P2 | 2 | 114.0 | ||
| (CF3)4P2 | 3 | 100.9 | ||
| Ph4P2 | 4 | 84.6 | ||
| Compound | Compound | Inversion (R) | Inversion (Me) | |
| Me2P–(CF3)2P | 5 | 106.4 | 100.8 | |
| Me2P–Ph2P | 6 | 86.5 | 106.9 | |
| Compound | Compound | (R)-gauche-meso a | (R)-gauche (Me)-dl b | (R)-gauche (X)-dl c |
| Me(CF3)P–P(CF3)Me | 7 | 102.8 | 107.4 | 99.1 |
| MePhP–PPhMe | 8 | 103.1 | 100.8 | 101.8 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molloy, A.D.; Sánchez-Sanz, G.; Gilheany, D.G. PP-Rotation, P-Inversion and Metathesis in Diphosphines Studied by DFT Calculations: Comments on Some Literature Conflicts. Inorganics 2016, 4, 36. https://doi.org/10.3390/inorganics4040036
Molloy AD, Sánchez-Sanz G, Gilheany DG. PP-Rotation, P-Inversion and Metathesis in Diphosphines Studied by DFT Calculations: Comments on Some Literature Conflicts. Inorganics. 2016; 4(4):36. https://doi.org/10.3390/inorganics4040036
Chicago/Turabian StyleMolloy, Adam D., Goar Sánchez-Sanz, and Declan G. Gilheany. 2016. "PP-Rotation, P-Inversion and Metathesis in Diphosphines Studied by DFT Calculations: Comments on Some Literature Conflicts" Inorganics 4, no. 4: 36. https://doi.org/10.3390/inorganics4040036