Qualifying the Role of Indium in the Multiple-Filled Ce0.1InxYb0.2Co4Sb12 Skutterudite
Abstract
:1. Introduction
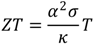
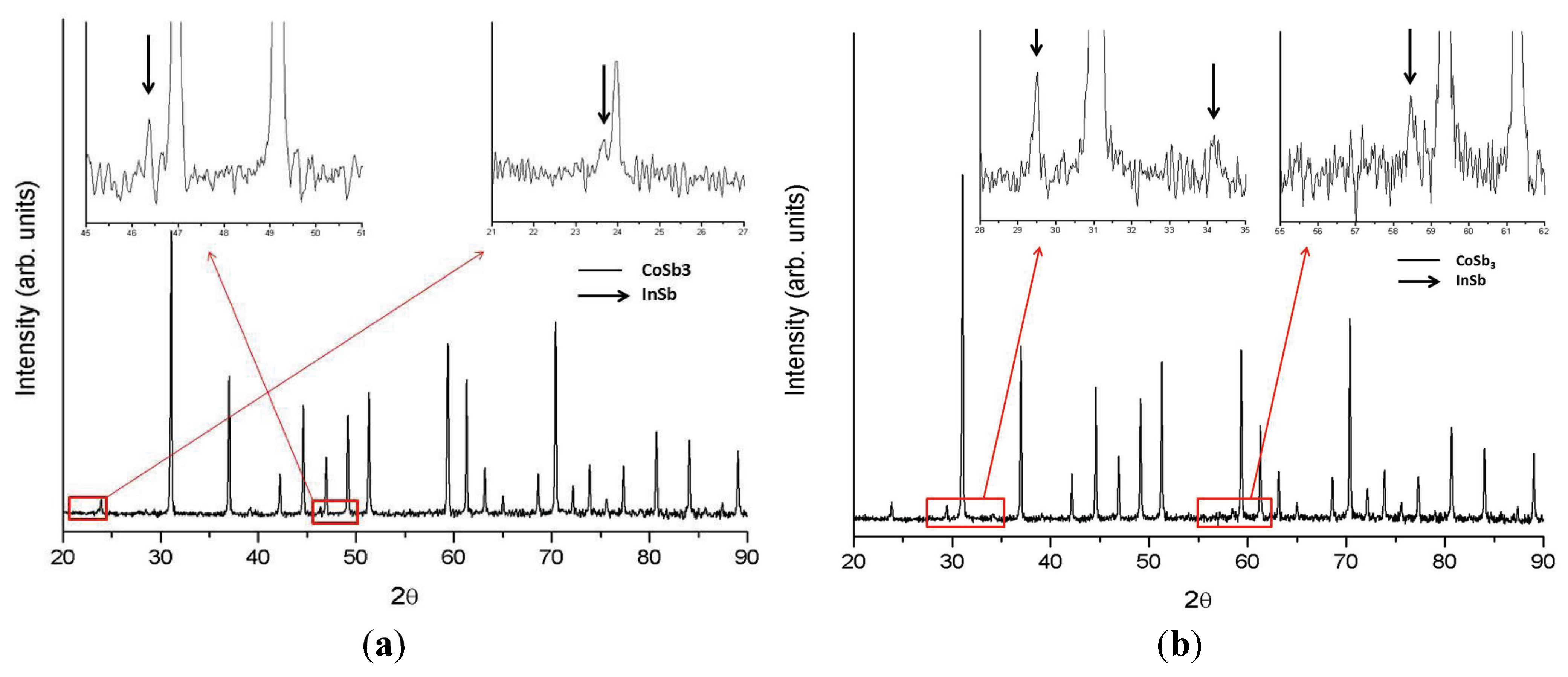
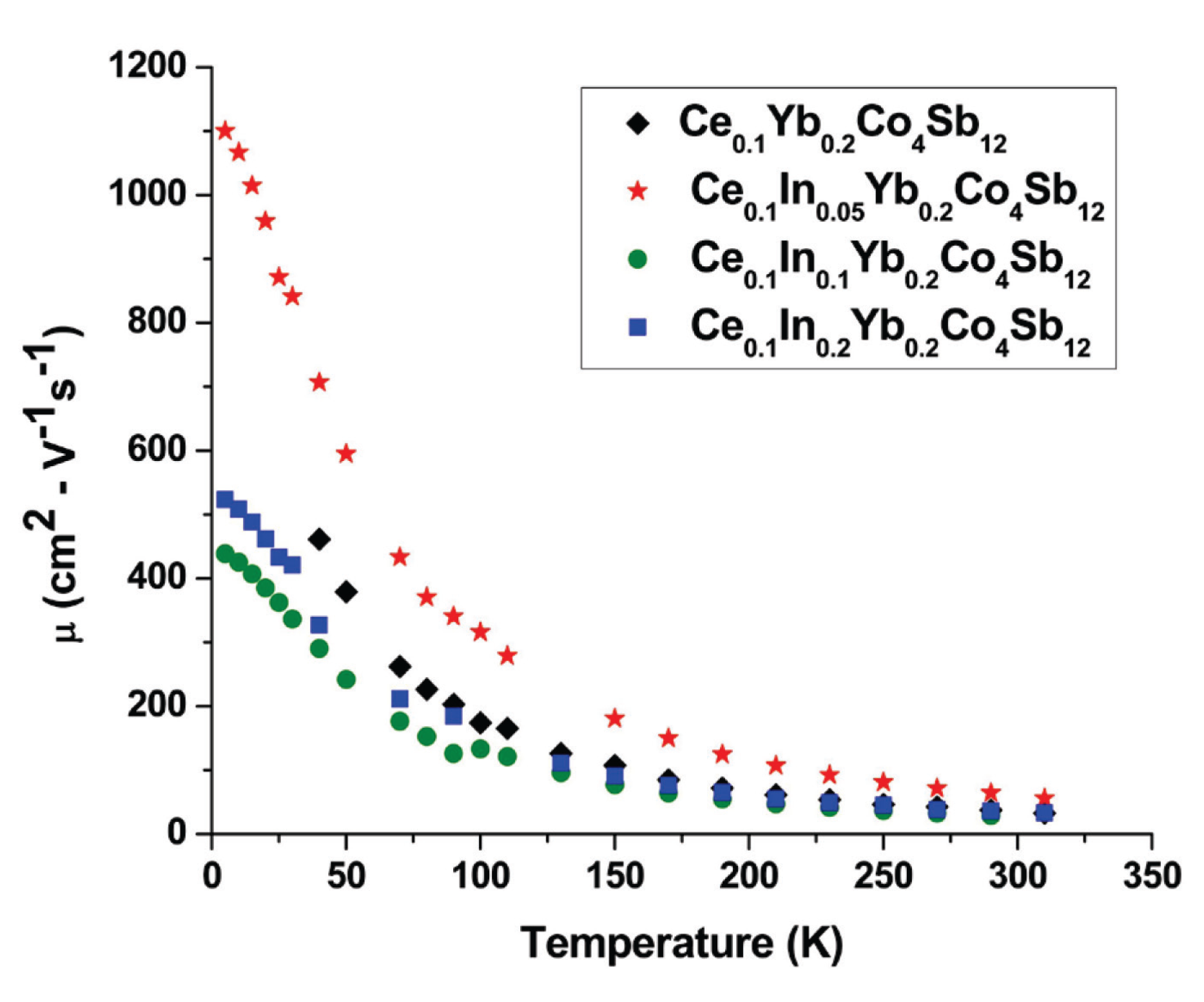
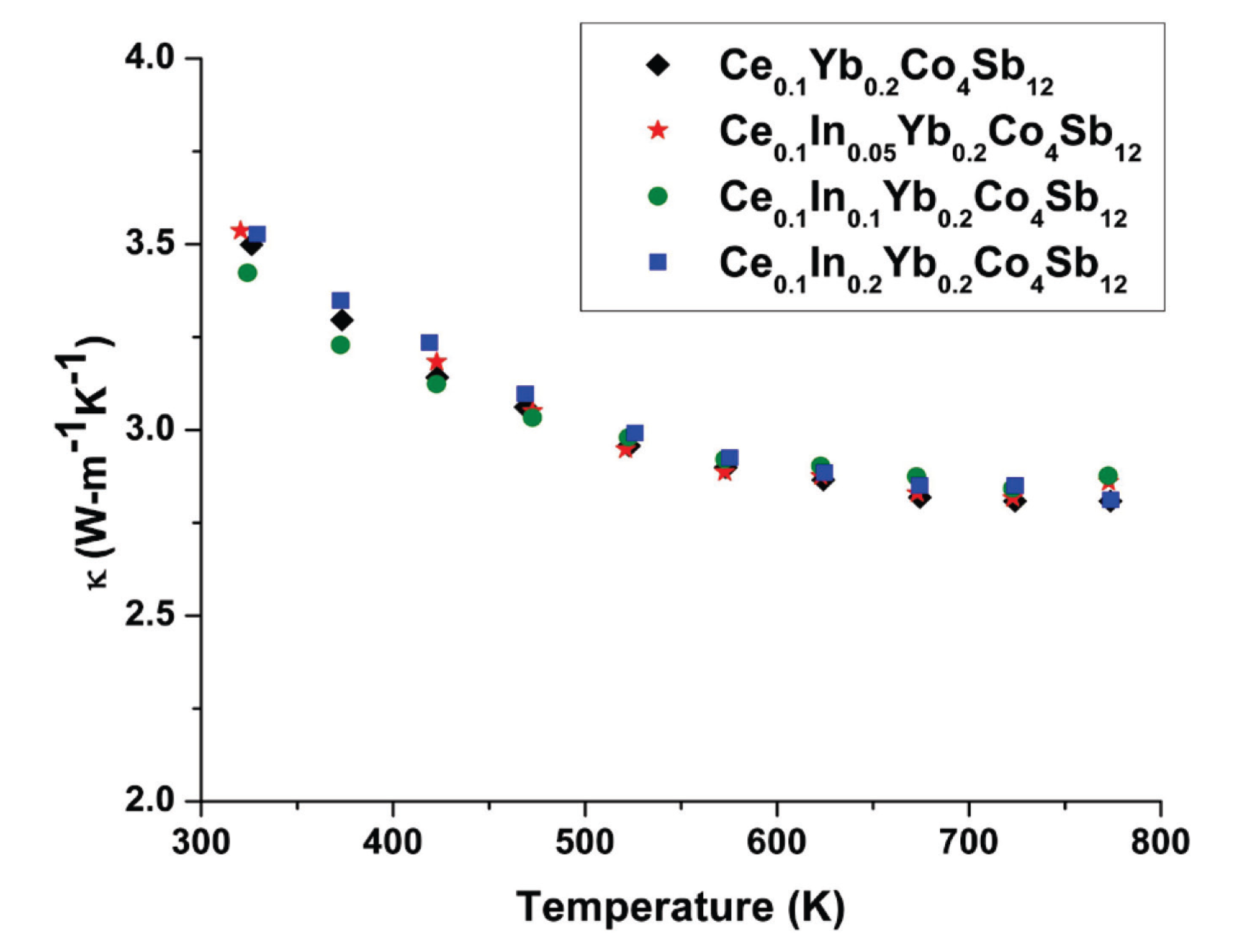
2. Results and Discussion






3. Experimental Section
4. Conclusions
Acknowledgments
Conflicts of Interest
References and Notes
- Slack, G.A. CRC Handbook of Thermoelectrics; Rowe, D.M., Ed.; CRC Press: Boca Raton, FL, USA, 1995. [Google Scholar]
- Klein, C.; Hurlbut, C.S., Jr. Manual of Mineralogy, 20th ed.; John Wiley: New York, NY, USA, 1985. [Google Scholar]
- Nolas, G.S.; Slack, G.S.; Morelli, D.T.; Tritt, T.M.; Ehrhich, A.C. The effect of rare-earth filling on the lattice thermal conductivity of skutterudites. J. Appl. Phys. 1996, 79, 4002. [Google Scholar]
- Zhao, X.Y.; Shi, X.; Chen, L.D.; Zhang, W.Q.; Zhang, W.B.; Pei, Y.Z. Synthesis and thermoelectric properties of Sr-filled skutterudite SryCo4Sb12. J. Appl. Phys. 2006, 99, 053711. [Google Scholar] [CrossRef]
- Nolas, G.S.; Takizawa, H.; Endo, T.; Sellinschegg, H.; Johnson, D.C. Thermoelectric properties of Sn-filled skutterudite. Appl. Phys. Lett. 2000, 77, 52. [Google Scholar] [CrossRef]
- Kuznetsov, V.L.; Kuznetsova, L.A.; Rowe, D.M. Effect of partial void filling on the transport properties of NdxCo4Sb12 skutterudites. J. Phys. 2003, 15, 5035–5048. [Google Scholar]
- Zhang, J.; Xu, B.; Wang, L.M.; Yu, D.; Yang, J.; Yu, F.; Liu, Z.; He, J.; Wen, B.; Tian, Y. High-pressure synthesis of phonon-glass electron-crystal featured thermoelectric LixCo4Sb12. Acta Mater. 2012, 60, 1246–1251. [Google Scholar]
- Nolas, G.S.; Cohn, J.L.; Slack, G.S. Effect of partial void filling on the lattice thermal conductivity of skutterudites. Phys. Rev. B 1998, 58, 164. [Google Scholar] [CrossRef]
- Lamberton, G.A., Jr.; Bhattacharya, S.; Littleton IV, R.T.; Kaeser, M.A.; Tedstrom, R.H.; Tritt, T.M. High figure of merit in Eu-filled CoSb3-based skutterudites. Appl. Phys. Lett. 2002, 80, 598. [Google Scholar] [CrossRef]
- Tritt, T.M. Thermal Conductivity: Theory, Properties, and Applications; Springer: New York, NY, USA, 2004. [Google Scholar]
- Bai, S.Q.; Pei, Y.Z.; Chen, L.D.; Zhang, W.Q.; Zhao, X.Y.; Yang, J. Enhanced thermoelectric performance of dual-element-filled skutterudites BaxCeyCo4Sb12. Acta Mater. 2009, 57, 3135–3139. [Google Scholar] [CrossRef]
- Ballikaya, S.; Wang, G.; Sun, K.; Uher, C. Thermoelectric Properties of Triple-Filled BaxYbyInzCo4Sb12. J. Electron. Mater. 2010, 40, 570–576. [Google Scholar] [CrossRef]
- Graff, J.W.; Zhu, S.; Holgate, T.; Peng, J.; He, J.; Tritt, T.M. High-Temperature Thermoelectric Properties of Co4Sb12-Based Skutterdites with Multiple Filler Atoms: Ce0.1InxYbyCo4Sb12. J. Electron. Mater. 2011, 40, 696–701. [Google Scholar] [CrossRef]
- Morelli, D.T.; Meisner, G.P.; Chen, B.; Hu, S.; Uher, C. Cerium filling and doping of coblat triantimonide. Phys. Rev. B 1997, 56, 7376. [Google Scholar] [CrossRef]
- Peng, J.; He, J.; Alboni, P.N.; Tritt, T.M. Synthesis and Thermoelectric Properties of the Double-Filled Skutterudite Yb0.2InyCo4Sb12. J. Electron. Mater. 2009, 38, 981–984. [Google Scholar] [CrossRef]
- Salvador, J.R.; Yang, J.; Wang, H.; Shi, X. Double-filled skutterudites of the type YbxCayCo4Sb12: Synthesis and properties. J. Appl. Phys. 2010, 107, 043705. [Google Scholar] [CrossRef]
- Shi, X.; Kong, H.; Li, C.P.; Uher, C.; Yang, J.; Salvador, J.R.; Wang, H.; Chen, L.; Zhang, W. Low thermal conductivity and high thermoelectric figure of merit in n-type BaxYbyCo4Sb12. Appl. Phys. Lett. 2008, 92, 182101. [Google Scholar]
- Shi, X.; Yang, J.; Salvador, J.R.; Chi, M.; Cho, J.Y.; Wang, H.; Bai, S.; Yang, J.; Zhang, W.; Chen, L. Multiple-Filled Skutterudites: High Thermoelectric Figure of Merit through Separately Optimizing Electrical and Thermal Transports. J. Am. Chem. Soc. 2011, 133, 7837–7846. [Google Scholar] [CrossRef]
- Zhang, L.; Grytsiv, A.; Rogl, P.; Bauer, E.; Zehetbauer, M. High thermoelectric performance of triple-filled n-type skutterudites (Sr,Ba,Yb)yCo4Sb12. J. Phys. D 2009, 42, 225405–225413. [Google Scholar] [CrossRef]
- He, T.; Chen, J.; Rosenfeld, D.; Subramanian, M.A. Thermoelectric Properties of Indium-Filled Skutterudites. Chem. Mater. 2006, 18, 759–762. [Google Scholar] [CrossRef]
- Peng, J.Y.; Alboni, P.N.; He, J.; Zhang, B.; Su, Z.; Holgate, T.; Gothard, N.; Tritt, T.M. Thermoelectric properties of (In,Yb) double-filled CoSb3 skutterudite. J. Appl. Phys. 2008, 104, 053710. [Google Scholar] [CrossRef]
- Li, H.; Tang, X.F.; Zhang, Q.; Uher, C. High performance InxCeyCo4Sb12 thermoelectric materials with in situ forming nanostructured InSb phase. Appl. Phys. Lett. 2009, 94, 102114. [Google Scholar] [CrossRef]
- Qi, W.H.; Wang, M.P. Size effect on the cohesive energy of nanoparticle. J. Mat. Sci. Lett. 2002, 21, 1743. [Google Scholar] [CrossRef]
- Nanda, K.K.; Sahu, S.N.; Behera, S.N. Liquid-drop model for the size-dependent melting of low-dimensional systems. Phys. Rev. A 2002, 66, 013208. [Google Scholar] [CrossRef]
- Frenken, J.W.M.; van der Veen, J.F. Observation of Surface Melting. Phys. Rev. Lett. 1985, 54, 134. [Google Scholar] [CrossRef]
- Pope, A.L.; Littleton, R.T., IV; Tritt, T.M. Apparatus for the rapid measurement of electrical transport properties for both “needle-like” and bulk materials. Rev. Sci. Instru. 2001, 72, 3129. [Google Scholar] [CrossRef]
- Pope, A.L.; Zawilski, B.; Tritt, T.M. Description of removable sample mount apparatus for rapid thermal conductivity measurement. Cryogenics 2001, 41, 725–731. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Graff, J.; He, J.; Tritt, T.M. Qualifying the Role of Indium in the Multiple-Filled Ce0.1InxYb0.2Co4Sb12 Skutterudite. Inorganics 2014, 2, 168-176. https://doi.org/10.3390/inorganics2020168
Graff J, He J, Tritt TM. Qualifying the Role of Indium in the Multiple-Filled Ce0.1InxYb0.2Co4Sb12 Skutterudite. Inorganics. 2014; 2(2):168-176. https://doi.org/10.3390/inorganics2020168
Chicago/Turabian StyleGraff, Jennifer, Jian He, and Terry M. Tritt. 2014. "Qualifying the Role of Indium in the Multiple-Filled Ce0.1InxYb0.2Co4Sb12 Skutterudite" Inorganics 2, no. 2: 168-176. https://doi.org/10.3390/inorganics2020168



