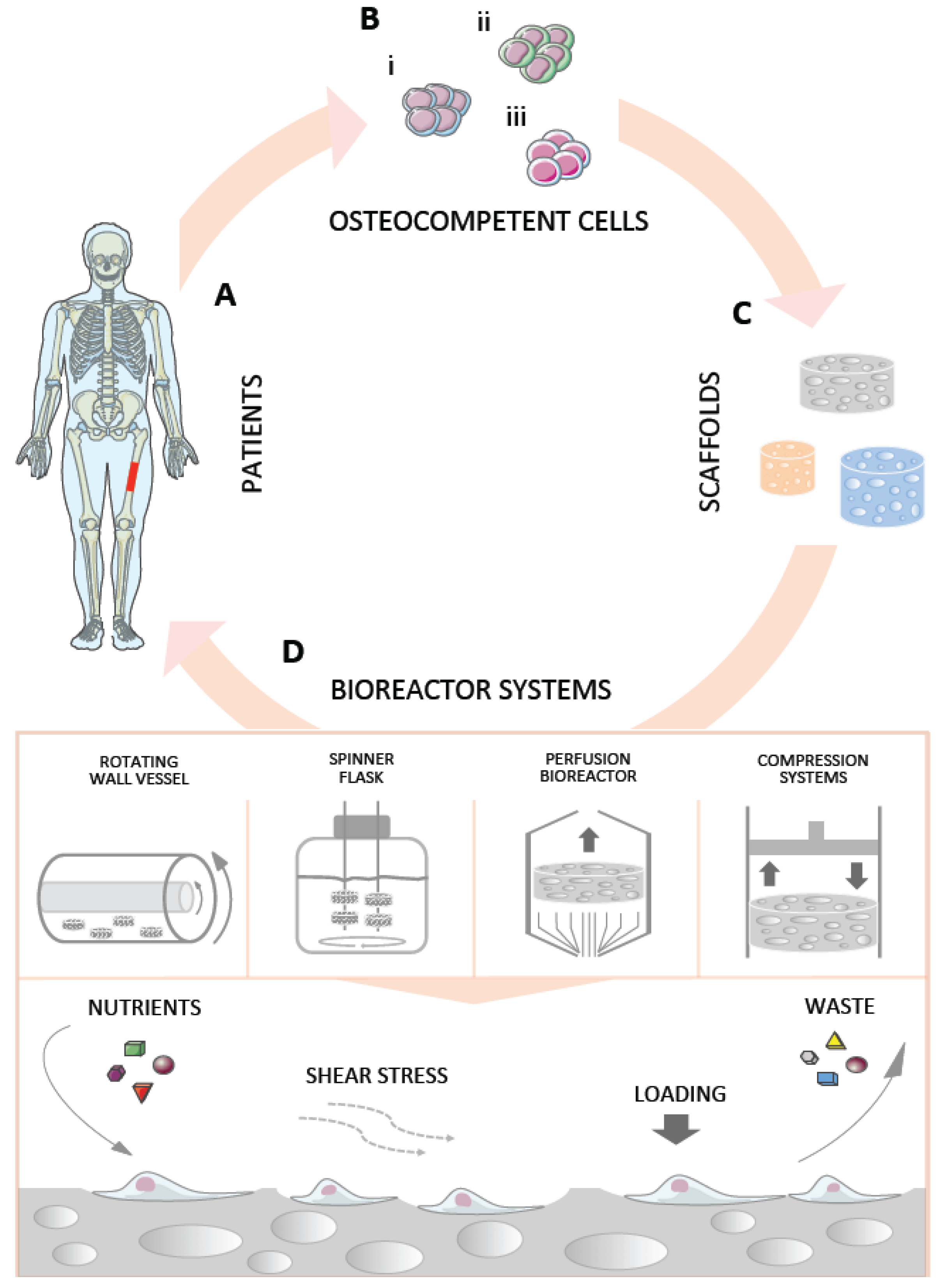
Bioreactor Systems for Human Bone Tissue Engineering
Abstract
:1. Introduction
2. Bioreactors in Bone Tissue Engineering
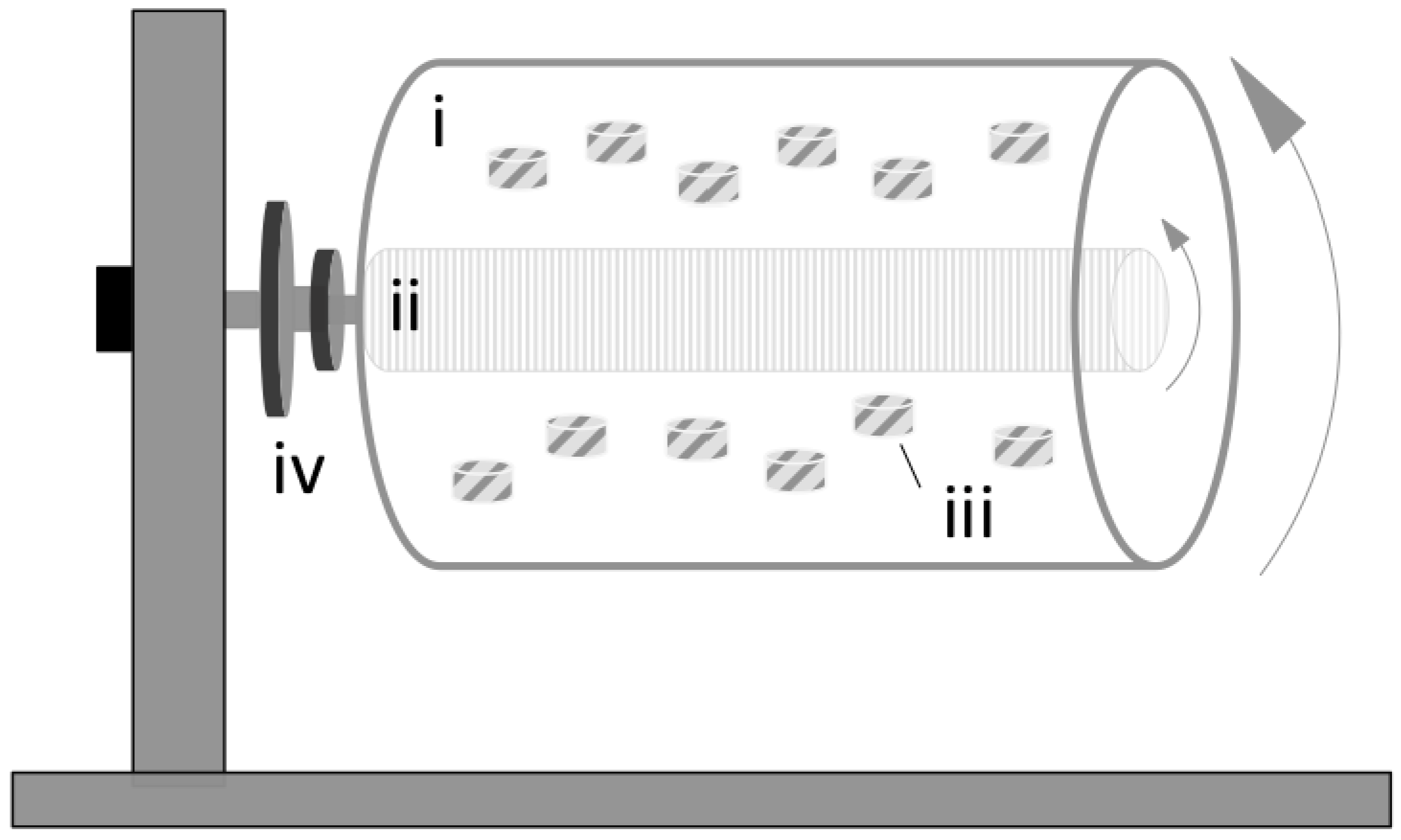
2.1. Rotating Wall Vessel Bioreactors
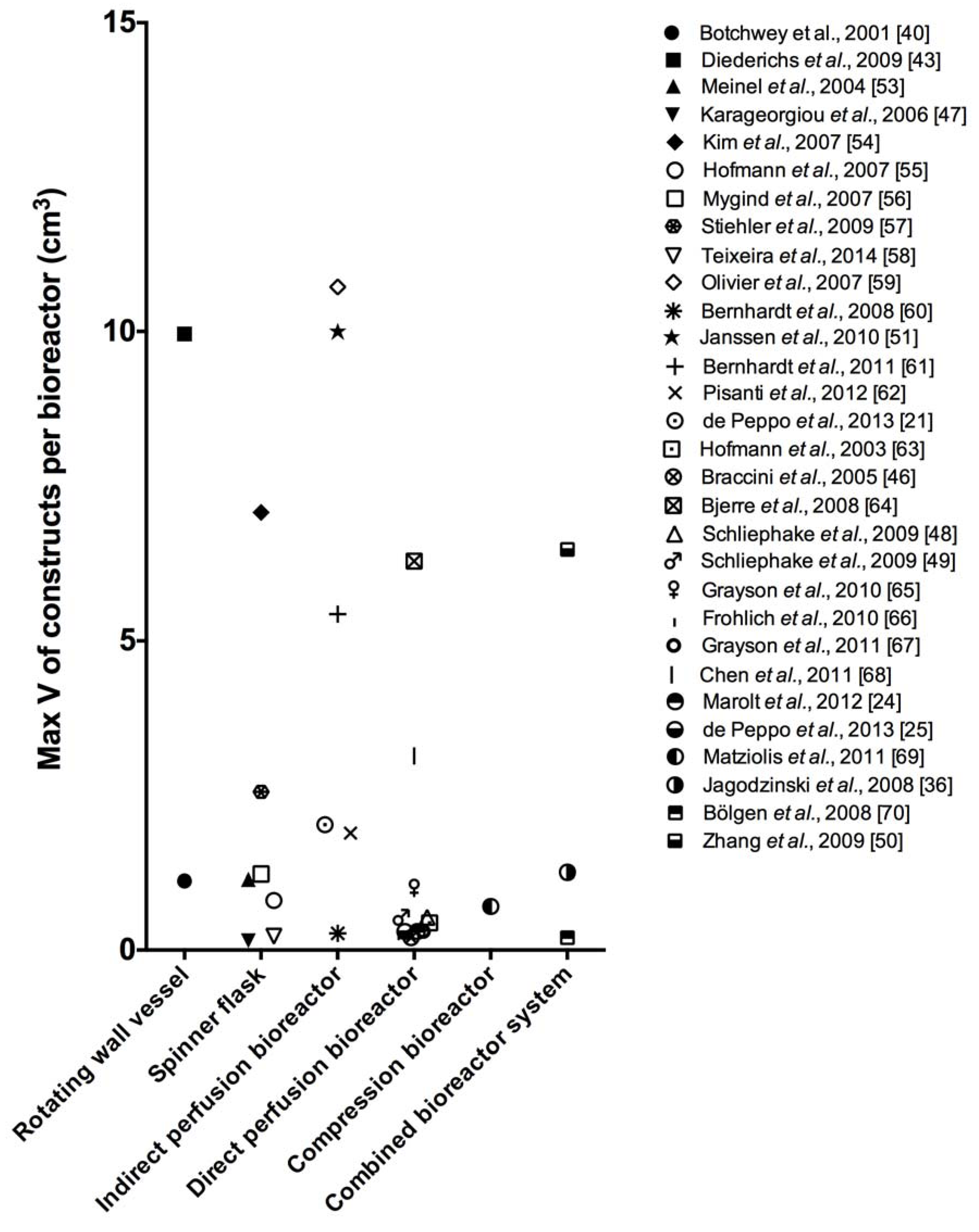
| Reference | Cell type and density | Scaffold type | Max V of constructs/bioreactor | Rotation rate | Shear stress | Culture period and media | Results (vs. static culture) |
|---|---|---|---|---|---|---|---|
| Botchwey et al. 2001 [40] | SaOS-2 (ATCC) ~222 × 103/scaffold | PLGA disc (d 4 × h 2.5 mm) | 1.116 cm3 | 25 rpm | 0.39 Pa | 7 days OS+ medium | Proliferation: ↓ Differentiation: ↑ ALP, Ca |
| Facer et al. 2005 [41] | HEPM (ATCC) 10 × 106/suspension | No scaffold | na | 15 ** rpm | na | 28 days OS− medium OS+ medium | ↑ cell aggregation; ↑ Ca and P deposition |
| Wang et al. 2009 [42] | BM-hMSCs na/scaffold | Gelatin-hyaluronic acid film (1.5 × 1.5 cm) | na | 30 rpm | na | 21 days OS+ medium | Proliferation: ↑ Differentiation: ↓ ALP, OC, COL1 |
| Diederichs et al. 2009 [43] | AT-hMSCs 14 × 106/scaffold | ZrO2 based ceramic (Sponceram®) disc natural HA-coated (d 65 × h 3 mm) | 9.96 cm3 | 1 rpm | na | 47 days OS+ medium | Only in natural scaffolds Proliferation: ↑ Differentiation: ↑ bone specific ECM; ↑ mineralization |
| Araujo et al. 2010 [44] | MG63 (ATCC) 1.5 × 105/cm2 | BCP-PCL nano-mesh PCL nano-mesh (10 × 10 × 0.06 mm) | na | 16 rpm | na | 14 * days OS− medium | Proliferation: ↔ Differentiation: ↑ protein amount |
| Ben-David et al. 2010 [45] | BM-hMSCs 0.5 × 106/scaffold | Gelatin-Coral/HA (ProOsteon™) particles (0.5–1 mm) | na | 7.5 rpm | na | 21 days OS+ medium | na |
| Reference | Cell type | Scaffold type | Bioreactor Pre-culture period | Animal model | Implantation period | Bone formation |
|---|---|---|---|---|---|---|
| Braccini et al. 2005 [46] | BM-hMSCs | HA (d 8 × h 4 mm) | Direct perfusion bioreactor 19 days | Nude mice sc | 8 weeks | Dynamic > Static 52% vs. 9.6% of available pore space |
| Karageorgiou et al. 2006 [47] | BM-hMSCs (Clonetics) | Silk loaded with BMP-2 (d 4 × h 2 mm) | Spinner flask 28 days | Mice Cranial 4 mm defect | 5 weeks | Dynamic ↔ Static 0.28 vs. 0.11 mm2 |
| Schliephake et al. 2009 [48] | hTBCs | CaCO3 (Biocoral®) (d 5 × h 3 mm) | Direct perfusion bioreactor 14 days | Nude rat im | 6 weeks | Dynamic ↔ Static 22% vs. 16% of area |
| Schliephake et al. 2009 [49] | hTBCs | CaCO3 (Biocoral®) Mineralized collagen TCP (Cerasorb®) (d 5 × h 3 mm) | Direct perfusion bioreactor 14 days | Nude rats Mandibular 5 mm defect | 6 weeks | Dynamic ↔ Static CaCO3: 7.2 vs. 8.7% of area Mineralized collagen: 10.3% vs. 12.5% of area TCP: 14.6% vs. 22.8% of area |
| Zhang et al. 2009 [50] | hfMSCs | PCL-TCP (6 × 6 × 4 mm) | Biaxial rotating bioreactor 14 days | NOD/SCID mice sc | 12 weeks | Dynamic > Static Dynamic: 3.2x fold increase |
| Ben-David et al. 2010 [45] | BM-hMSCs | Gelatin-Coral/HA (ProOsteon®) particles (0.5–1 mm) | Rotating wall vessel 21 days | Nude mouse sc | 8 weeks | Bone formation No static group |
| Janssen et al. 2010 [51] | BM-hMSCs | BCP particles (OsSatura™) (2–6 mm) | Indirect perfusion bioreactor 7; 20; 40 days | Nude mice sc | 6 weeks | Dynamic ↔ Static |
| Marolt et al. 2012 [24] | hESC-MPs | Decellularized cow bone (d 4 × h 4 mm) | Direct perfusion bioreactor 35 days | SCID-beige mice sc | 8 weeks | Dynamic > CTL 7% vs. 2% of area |
| de Peppo et al. 2013 [25] | hiPSC-MPs | Decellularized cow bone (d 4 × h 4 mm) | Direct perfusion bioreactor 35 days | SCID-beige mice sc | 12 weeks | Bone-like tissue formation No static group |
| Yeatts et al. 2014 [52] | BM-hMSCs (Lonza) | PLGA/PCL (d 3 × h 3 mm) | Indirect perfusion bioreactor 10 days | Nude rats Femoral condyle 2.5 mm defect | 3 and 6 weeks | Dynamic > Static (6 weeks only) 1.72 vs. 1.26 mm2 |
2.2. Spinner Flasks

| Reference | Cell type and density | Scaffold type | Max V of constructs/bioreactor | Rotation rate | Shear stress | Culture period and media | Results (vs. static culture) |
|---|---|---|---|---|---|---|---|
| Yasuda et al. 2004 [73] | AT-hMSCs 1 × 106/scaffold | PET fiber disc (d 6 mm × h na mm) | na | 50 rpm | na | 21 days OS+ medium | Proliferation: ↑ Differentiation: ↓ ALP |
| Meinel et al. 2004 [53] | BM-hMSCs (Clonetics) 5 × 106/scaffold | Collagen film (Ultrafoam®) (d 11 × h 1.5 mm) | 1.144 cm3 | 50 rpm | na | 35 days OS+ medium | Differentiation: ↑ ALP, Ca |
| Karageorgiou et al. 2006 [47] | BM-hMSCs (Clonetics) 5 × 106/scaffold | Silk loaded with BMP-2 (d 4 × h 2 mm) | 0.151 cm3 | 50 rpm | na | 28 days OS+ medium | na |
| Kim et al. 2007 [54] | BM-hMSCs (Cambrex) 1 × 106/scaffold | Aqueous-derived silk disc (d 15 × h 5 mm) | 7.072 cm3 | 50 rpm | na | 84 days OS+ medium | Proliferation: ↑ Differentiation: ↑ ALP, Ca; ↑ ALP, BSP, COL1, OP; ↑ compressive modulus ↑ compressive strength |
| Hofmann et al. 2007 [55] | BM-hMSCs (Clonetix) 5 × 106/scaffold | Silk fibroin disc (d 8 × h 2 mm) small pores (112–224 µm) large pores (400–500 µm) mixed pores | 0.804 cm3 | 60 rpm | na | 35 days OS− medium; OS+ medium | na |
| Mygind et al. 2007 [56] | BM-hMSCs-TERT 2 × 106/scaffold | Coralline HA disc (ProOsteonTM) (d 10 × h 2 mm) small pores (200 µm) large pores (500 µm) | 1.232 cm3 | 30 rpm | na | 21 days OS− medium | Proliferation: P200 ↔; P500 5 Differentiation: P200 ↑ ALP ↑ ALP, COL1, RUNX2, BMP-2, BSP, Osterix, ON P500 ↔ ALP ↑ ALP, COL1, RUNX2, OP, ON |
| Stiehler et al. 2009 [57] | BM-hMSCs-TERT 2 × 106/scaffold | PLGA block (8 × 8 × 5 mm) | 2.56 cm3 | 30 rpm | na | 21 days OS+ medium | Proliferation: ↔ Differentiation: ↑ ALP, Ca ↑ COL1, BMP-2, RUNX2, ON |
| Wang et al. 2009 [42] | BM-hMSCs
na/scaffold | Gelatin- hyaluronic acid film (1.5 × 1.5 mm) | na | 60 rpm | na | 21 days OS+ medium | Proliferation: ↑ Differentiation: ↑ ALP, OC, COL1 |
| Teixeira et al. 2014 [58] | hMSCs; (Lonza) 0.5 × 106/scaffold | Chitosan disc (d 4 × h 3 mm) | 0.226 cm3 | 50 rpm | na | 14 days; OS− medium; OS+ medium | Proliferation: ↑ Differentiation: ↑ ALP, von Kossa |
2.3. Perfusion Bioreactors
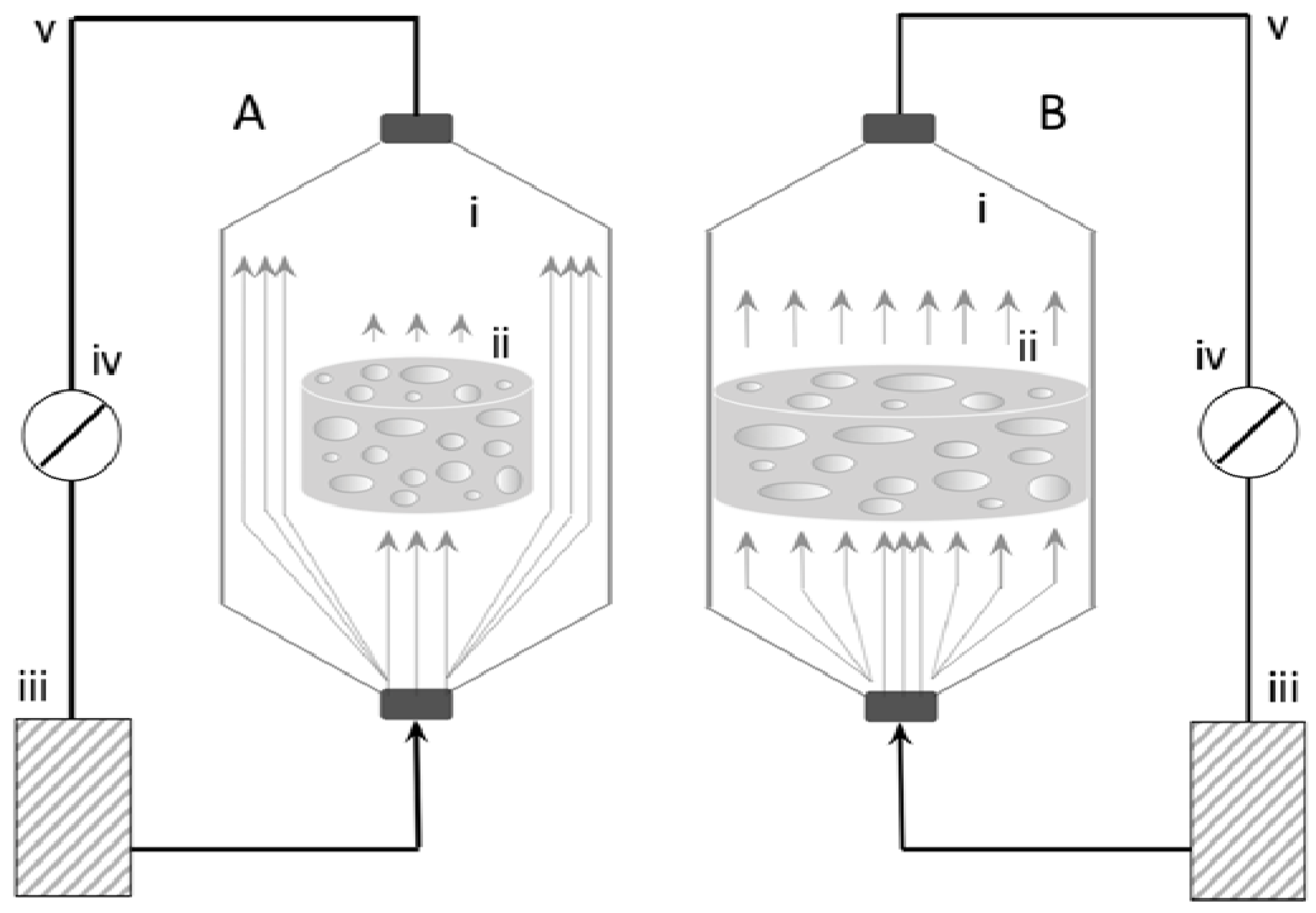
2.3.1. Indirect Perfusion Bioreactors
| Reference | Cell type and density | Scaffold type | Max V of constructs/bioreactor | Perfusion rate | Shear stress | Culture period and media | Results (vs. static culture) |
|---|---|---|---|---|---|---|---|
| Olivier et al. 2007 [59] | MG63 10 × 106/scaffold | ßTCP cylinder (d 14 mm × h 33 mm) | 10.721 cm3 | 3 mL/min
convergent and divergent flow set-up | na | 28 days OS− medium | Proliferation: ↑; Homogeneous cell distribution: Convergent > divergent > static |
| Bernhardt et al. 2008 [60] | BM-hMSCs 0.015 × 106/scaffold | COL1-nanocrystalline HA tape (d 12 × h 0.2 mm) | 0.271 cm3 | 1 mL/min | na | 35 days OS− medium OS+ medium | Proliferation: ↔ OS−; ↔ Os+; Differentiation: ↔ ALP in OS−; ↓ ALP in Os+ |
| Janssen et al. 2010 [51] | BM-hMSCs (1–12) × 106/all scaffolds | BCP particles (OsSatura™) (2–6 mm) | 10 cm3 | 4 mL/min | na | 20 days OS+ medium | Differentiation: ↔ ALP, CBFA1, COL1, OC, ON, S100A4, BMP-2 |
| Bernhardt et al. 2011 [61] | BM-hMSCs 0.2 × 106/scaffold | βTCP (Cerasorb®) (d 12 × h 6 mm)
750 and 1400 µm pore size | 5.43 cm3 | 1.5 mL/min | na | 21 days OS+ medium with 10% serum or 2% serum | Proliferation: ↑ OS+ medium with 10% serum; ↔ OS+ medium with 2% serum; Differentiation: ↔ OS+ medium with 10% serum; ↑ OS+ medium with 2% serum |
| Pisanti et al. 2012 [62] | BM-hMSCs (Lonza) 0.12 × 106/scaffold | PLLA disc (d 4 × h 5 mm)
100, 250, and 500 µm pore size | 1.89 cm3 | 0.3 mL/min | na | 24 days Os+ medium | Differentiation: ↑ ALP, BMP-2 |
| de Peppo et al. 2013 [21] | BM-hMSCs hESC-MPs 0.1 × 106/scaffold | CaCO3 cube (Biocoral®) (3 mm) | 2.025 cm3 | 10 mL/min | 0.001 Pa | 35 days OS+ medium | Proliferation: ↑; hESC-MPs > BM-hMSCs; Differentiation: ↑ RUNX2, COL1, ALP, ON, OP; hESC-MPs > BM-hMSCs |
| Yeatts et al. 2014 [52] | BM-hMSCs (Lonza) 0.25 × 106/scaffold | PLGA-PCL (d 3 × h 3 mm) | na | 1 mL/min | na | 10 days OS+ medium | na |
2.3.2. Direct Perfusion Bioreactors
| Reference | Cell type and density | Scaffold type | Max V of constructs/bioreactor | Perfusion rate | Shear stress | Culture period and media | Results (vs. static culture) |
|---|---|---|---|---|---|---|---|
| Hofmann et al. 2003 [63] | Human osteoblasts 12,500 cells/cm2 | Cancelous human bone HA (Endobon®) (d 7.5 × h 10 mm) | 0.442 cm3 | 1 mL/min | na | 10 days OS− medium | na |
| Braccini et al. 2005 [46] | hBMNCs; ~18 × 106/scaffold | HA (Engipore®) (d 8 × h 4 mm) | 0.201 cm3 | 100 μm/s | na | 19 days; OS+ medium | na |
| Bjerre et al. 2008 [64] | BM-hMSC-TERT 2 × 106/scaffold | 67% Si-TCP/33% HA/ßTCP (SkeliteTM) (d 10 × h 5 mm) | 6.288 cm3 | 0.1 mL/min | na | 21 days OS− medium | Proliferation: ↑; Differentiation: ↑ ALP; ↑ OP, BSP, BMP-2 |
| Schliephake et al. 2009 [48] | hTBCs 5 × 106 cells/cm3 | CaCO3 (Biocoral®) (d 5 × h 3 mm) | 0.530 cm3 | na | na | 14 days OS− medium | na |
| Schliephake et al. 2009 [49] | hTBCs 5 × 106 cells/cm3 | CaCO3 (Biocoral®) Mineralized collagen TCP (Cerasorb®) (d 5 × h 3 mm) | 0.530 cm3 | na | na | 14 days OS− medium | na |
| Grayson et al. 2010 [65] | BM-hMSCs (Cambrex) 3.4 × 106/scaffold | Decellularized cow bone hTMJ-shaped (~15 × 15 × 5 mm) | ~1 cm3 | 1.8 mL/min | na | 35 days OS+ medium | Proliferation: ↑; Differentiation: ↑ bone volume |
| Fröhlich et al. 2010 [66] | AT-hMSCs 1.5 × 106/scaffold | Decellularized cow bone plugs (d 4 × h 4 mm) | 0.302 cm3 | 1.8 mL/min | ~0.01 Pa | 35 days OS− medium; OS+ medium | Proliferation: ↔ OS− ↔ OS+; Differentiation: ↑ bone specific ECM |
| Grayson et al. 2011 [67] | BM-hMSCs (Cambrex) 1.2 × 106/scaffold | Decellularized cow bone plugs (d 4 × h 4 mm) | 0.302 cm3 | 80, 400, 800, 1200 or 1800 µm/s | 0.0006–0.02 Pa | 35 days OS+ medium | na |
| Chen et al. 2011 [68] | AM-hMSCs 5 × 104 cells/mg microcarriers | Porcine gelatin microcarrier (CultiSpher S) (d 20 × h 10 mm) | 3.142 cm3 | 2 mL/min | na | 28 + 7 days; OS− medium; OS+ medium | na |
| Marolt et al. 2012 [24] | hESC-MPs 1.5 × 106/scaffold | Decellularized cow bone plugs (d 4 × h 4 mm) | 0.302 cm3 | 3.6 mL/min | na | 35 days OS+ medium | Proliferation: ↑; Differentiation: ↑ ALP, OP; ↑ bone specific ECM |
| de Peppo et al. 2013 [25] | hiPSC-MPs 1.5 × 106/scaffold | Decellularized cow bone plugs (d 4 × h 4 mm) | 0.302 cm3 | 3.6 mL/min | na | 35 days OS+ medium | Differentiation: ↑ OP; ↑ bone specific ECM |
2.4. Compression Bioreactors and Combined Systems

| Reference | Cell type and density | Scaffold type | Max V of constructs/bioreactor | Compression rate | Culture period and media | Results (vs. static culture) |
|---|---|---|---|---|---|---|
| Matziolis et al. 2011 [69] | BM-hMSCs 1 × 106/scaffold | Human cancellous bone-fibrin composite (d 15 × h 4 mm) | 0.707 cm3 | 4 kPa 25% strain 0.05 Hz | 24 h OS−medium | ↑ OP, integrin-ß-1, TGFßR1, SMAD5, PDGFα, annexin-V |
| Reference | Mechanical stimulation | Cell type and density | Scaffold type | Max V of constructs/bioreactor | Rotation rate | Perfusion rate | Compression rate | Culture period and media | Results (vs. static culture) |
|---|---|---|---|---|---|---|---|---|---|
| Jagodzinski et al. 2008 [36] | Perfusion | BM-hMSCs 1 × 106/scaffold | Decellularized cow bone (Tutobone®) (d 20 × h 4 mm) | 1.257 cm3 | Not done | 10 mL/min | Not done | 21 days OS+ medium | Proliferation: ↑ Differentiation: ↔ OC |
| Compression-perfusion | Not done | 10 mL/min | 10% strain, 0.5 Hz | Proliferation: ↑ Differentiation: ↑ OC; also > to perfusion only | |||||
| Bölgen et al. 2008 [70] | Compression | MG63 (ATTC) 1 × 106/scaffold | HEMA–lactate–dextran (d 8 × h 4 mm) | 0.201 cm3 | Not done | 0.1 mL/min | Not done | 10 days OS− medium | Differentiation: ↔ ALP |
| Compression-perfusion | Not done | 0.1 mL/min | 1.5% strain, 1 Hz, 1 h/day | Differentiation: ↑ ALP | |||||
| Zhang et al. 2009 [50] | Perfusion-rotation | hfMSCs 0.5 × 106/scaffold | PCL-TCP (6 × 6 × 4 mm) | 6.48 cm3 | 5 rpm | 3.8 mL/min | Not done | 28 days OS+ medium | Proliferation: ↑ Differentiation: ↑ ALP, Ca |
| Liu et al. 2012 [76] | Perfusion | BM-hMSCs 6 × 106/scaffold | PU-BDI shape of human meniscus (size na) | na | Not done | 10 mL/min | Not done | 14 days na medium | Proliferation: ↑ Differentiation: ↔ ALP; ↔ mechanical strength |
| Compression-perfusion | Not done | 10 mL/min | 10% strain, 0.5 Hz, 1 time/day, 8 h/time | Proliferation: ↔; Differentiation: ↔ ALP; ↔ mechanical strength | |||||
| Not done | 10 mL/min | 10% strain, 0.5 Hz, 4 times/day, 2 h/time, 4 h of rest | Proliferation: ↑; also > to perfusion only; Differentiation: ↑ ALP; ↑ mechanical strength` |
3. Simulation Techniques for Improved Bioreactor Design
4. Future Directions for Clinical Translation
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- de Peppo, G.M. Human Embryonic Stem Cells for Bone Engineering Applications. Ph.D. Thesis, University of Gothenburg, Gothenburg, Sweden, 7 June 2011. [Google Scholar]
- Braddock, M.; Houston, P.; Campbell, C.; Ashcroft, P. Born again bone: Tissue engineering for bone repair. News Physiol. Sci. 2001, 16, 208–213. [Google Scholar]
- Hollinger, J.O.; Winn, S.; Bonadio, J. Options for tissue engineering to address challenges of the aging skeleton. Tissue Eng. 2000, 6, 341–350. [Google Scholar] [CrossRef]
- U.S. Markets for Orthopedic Biomaterials for Bone Repair and Regeneration; MedTech Insight: Bedminster, NJ, USA, 2013.
- Albert, A.; Leemrijse, T.; Druez, V.; Delloye, C.; Cornu, O. Are bone autografts still necessary in 2006? A three-year retrospective study of bone grafting. Acta Orthop. Belg. 2006, 72, 734–740. [Google Scholar]
- de Peppo, G.M.; Marolt, D. Modulating the biochemical and biophysical culture environment to enhance osteogenic differentiation and maturation of human pluripotent stem cell-derived mesenchymal progenitors. Stem Cell Res. Ther. 2013, 4, 106. [Google Scholar]
- Frohlich, M.; Grayson, W.L.; Wan, L.Q.; Marolt, D.; Drobnic, M.; Vunjak-Novakovic, G. Tissue engineered bone grafts: Biological requirements, tissue culture and clinical relevance. Curr. Stem Cell Res. Ther. 2008, 3, 254–264. [Google Scholar] [CrossRef]
- Marolt, D.; Knezevic, M.; Vunjak-Novakovic, G. Bone tissue engineering with human stem cells. Stem Cell Res. Ther. 2010, 1, 10. [Google Scholar] [CrossRef]
- Murphy, M.B.; Moncivais, K.; Caplan, A.I. Mesenchymal stem cells: Environmentally responsive therapeutics for regenerative medicine. Exp. Mol. Med. 2013, 45, e54. [Google Scholar]
- Duffy, G.P.; Ahsan, T.; O’Brien, T.; Barry, F.; Nerem, R.M. Bone marrow-derived mesenchymal stem cells promote angiogenic processes in a time- and dose-dependent manner in vitro. Tissue Eng. Part A 2009, 15, 2459–2470. [Google Scholar] [CrossRef]
- de Peppo, G.M.; Svensson, S.; Lennerås, M.; Synnergren, J.; Stenberg, J.; Strehl, R.; Hyllner, J.; Thomsen, P.; Karlsson, C. Human embryonic mesodermal progenitors highly resemble human mesenchymal stem cells and display high potential for tissue engineering applications. Tissue Eng. Part A 2010, 16, 2161–2182. [Google Scholar] [CrossRef]
- Wagner, W.; Horn, P.; Castoldi, M.; Diehlmann, A.; Bork, S.; Saffrich, R.; Benes, V.; Blake, J.; Pfister, S.; Eckstein, V.; et al. Replicative senescence of mesenchymal stem cells: A continuous and organized process. PLoS One 2008, 3, e2213. [Google Scholar] [CrossRef]
- Zhou, S.; Greenberger, J.S.; Epperly, M.W.; Goff, J.P.; Adler, C.; Leboff, M.S.; Glowacki, J. Age-related intrinsic changes in human bone-marrow-derived mesenchymal stem cells and their differentiation to osteoblasts. Aging Cell 2008, 7, 335–343. [Google Scholar] [CrossRef]
- Bertram, H.; Mayer, H.; Schliephake, H. Effect of donor characteristics, technique of harvesting and in vitro processing on culturing of human marrow stroma cells for tissue engineered growth of bone. Clin. Oral Implants Res. 2005, 16, 524–531. [Google Scholar] [CrossRef]
- Thomson, J.A.; Itskovitz-Eldor, J.; Shapiro, S.S.; Waknitz, M.A.; Swiergiel, J.J.; Marshall, V.S.; Jones, J.M. Embryonic stem cell lines derived from human blastocysts. Science 1998, 282, 1145–1147. [Google Scholar] [CrossRef]
- Tachibana, M.; Amato, P.; Sparman, M.; Gutierrez, N.M.; Tippner-Hedges, R.; Ma, H.; Kang, E.; Fulati, A.; Lee, H.S.; Sritanaudomchai, H.; et al. Human Embryonic Stem Cells Derived by Somatic Cell Nuclear Transfer. Cell 2013, 153, 1228–1238. [Google Scholar] [CrossRef]
- Takahashi, K.; Tanabe, K.; Ohnuki, M.; Narita, M.; Ichisaka, T.; Tomoda, K.; Yamanaka, S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 2007, 131, 861–872. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.; Kim, S.S.; Lee, S.H.; Eun Ahn, S.; Gwak, S.J.; Song, J.H.; Kim, B.S.; Chung, H.M. In vivo bone formation from human embryonic stem cell-derived osteogenic cells in poly(d,l-lactic-co-glycolic acid)/hydroxyapatite composite scaffolds. Biomaterials 2008, 29, 1043–1053. [Google Scholar] [CrossRef]
- Bigdeli, N.; de Peppo, G.M.; Lennerås, M.; Sjövall, P.; Lindahl, A.; Hyllner, J.; Karlsson, C. Superior osteogenic capacity of human embryonic stem cells adapted to matrix-free growth compared to human mesenchymal stem cells. Tissue Eng. Part A 2010, 16, 3427–3440. [Google Scholar] [CrossRef]
- de Peppo, G.M.; Sjovall, P.; Lennerås, M.; Strehl, R.; Hyllner, J.; Thomsen, P.; Karlsson, C. Osteogenic potential of human mesenchymal stem cells and human embryonic stem cell-derived mesodermal progenitors: A tissue engineering perspective. Tissue Eng. Part A 2010, 16, 3413–3426. [Google Scholar] [CrossRef]
- de Peppo, G.M.; Sladkova, M.; Sjövall, P.; Palmquist, A.; Oudina, K.; Hyllner, J.; Thomsen, P.; Petite, H.; Karlsson, C. Human embryonic stem cell-derived mesodermal progenitors display substantially increased tissue formation compared to human mesenchymal stem cells under dynamic culture conditions in a packed bed/column bioreactor. Tissue Eng. Part A 2013, 19, 175–187. [Google Scholar] [CrossRef]
- Kuznetsov, S.A.; Cherman, N.; Robey, P.G. In vivo bone formation by progeny of human embryonic stem cells. Stem Cells Dev. 2011, 20, 269–287. [Google Scholar] [CrossRef]
- Levi, B.; Hyun, J.S.; Montoro, D.T.; Lo, D.D.; Chan, C.K.; Hu, S.; Sun, N.; Lee, M.; Grova, M.; Connolly, A.J.; et al. In vivo directed differentiation of pluripotent stem cells for skeletal regeneration. Proc. Natl. Acad. Sci. USA 2012, 109, 20379–20384. [Google Scholar] [CrossRef]
- Marolt, D.; Campos, I.M.; Bhumiratana, S.; Koren, A.; Petridis, P.; Zhang, G.; Spitalnik, P.F.; Grayson, W.L.; Vunjak-Novakovic, G. Engineering bone tissue from human embryonic stem cells. Proc. Natl. Acad. Sci. USA 2012, 109, 8705–8709. [Google Scholar] [CrossRef]
- de Peppo, G.M.; Marcos-Campos, I.; Kahler, D.J.; Alsalman, D.; Shang, L.; Vunjak-Novakovic, G.; Marolt, D. Engineering bone tissue substitutes from human induced pluripotent stem cells. Proc. Natl. Acad. Sci. USA 2013, 110, 8680–8685. [Google Scholar] [CrossRef]
- Goldstein, A.S.; Juarez, T.M.; Helmke, C.D.; Gustin, M.C.; Mikos, A.G. Effect of convection on osteoblastic cell growth and function in biodegradable polymer foam scaffolds. Biomaterials 2001, 22, 1279–1288. [Google Scholar] [CrossRef]
- Yeatts, A.B.; Fisher, J.P. Bone tissue engineering bioreactors: Dynamic culture and the influence of shear stress. Bone 2011, 48, 171–181. [Google Scholar] [CrossRef]
- Klein-Nulend, J.; Bakker, A.D.; Bacabac, R.G.; Vatsa, A.; Weinbaum, S. Mechanosensation and transduction in osteocytes. Bone 2013, 54, 182–190. [Google Scholar] [CrossRef]
- Klein-Nulend, J.; Bacabac, R.G.; Mullender, M.G. Mechanobiology of bone tissue. Pathol. Biol. 2005, 53, 576–580. [Google Scholar] [CrossRef]
- Huang, C.; Ogawa, R. Mechanotransduction in bone repair and regeneration. FASEB J. 2010, 24, 3625–3632. [Google Scholar] [CrossRef]
- McCoy, R.J.; O’Brien, F.J. Influence of shear stress in perfusion bioreactor cultures for the development of three-dimensional bone tissue constructs: A review. Tissue Eng. Part B Rev. 2010, 16, 587–601. [Google Scholar] [CrossRef]
- Hoffman, B.D.; Crocker, J.C. Cell mechanics: Dissecting the physical responses of cells to force. Annu. Rev. Biomed. Eng. 2009, 11, 259–288. [Google Scholar] [CrossRef]
- Salter, E.; Goh, B.; Hung, B.; Hutton, D.; Ghone, N.; Grayson, W.L. Bone tissue engineering bioreactors: A role in the clinic? Tissue Eng. Part B Rev. 2012, 18, 62–75. [Google Scholar]
- de Peppo, G.M.; Thomsen, P.; Karlsson, C.; Strehl, R.; Lindahl, A.; Hyllner, J. Human progenitor cells for bone engineering applications. Curr. Mol. Med. 2013, 13, 723–734. [Google Scholar] [CrossRef]
- Martin, I.; Wendt, D.; Heberer, M. The role of bioreactors in tissue engineering. Trends Biotechnol. 2004, 22, 80–86. [Google Scholar] [CrossRef]
- Jagodzinski, M.; Breitbart, A.; Wehmeier, M.; Hesse, E.; Haasper, C.; Krettek, C.; Zeichen, J.; Hankemeier, S. Influence of perfusion and cyclic compression on proliferation and differentiation of bone marrow stromal cells in 3-dimensional culture. J. Biomech. 2008, 41, 1885–1891. [Google Scholar] [CrossRef]
- Schwarz, R.P.; Thomas, J.; Wolf, D.A. Cell culture for three-dimensional modeling in rotating-wall vessels: An application of simulated microgravity. J. Tissue Cult. Methods 1992, 14, 51–55. [Google Scholar] [CrossRef]
- Vunjak-Novakovic, G.; Martin, I.; Obradovic, B.; Treppo, S.; Grodzinsky, A.J.; Langer, R.; Freed, L.E. Bioreactor cultivation conditions modulate the composition and mechanical properties of tissue-engineered cartilage. J. Orthop. Res. 1999, 17, 130–138. [Google Scholar] [CrossRef]
- Freed, L.E.; Vunjak-Novakovic, G. Spaceflight bioreactor studies of cells and tissues. Adv. Space Biol. Med. 2002, 8, 177–195. [Google Scholar] [CrossRef]
- Botchwey, E.A.; Pollack, S.R.; Levine, E.M.; Laurencin, C.T. Bone tissue engineering in a rotating bioreactor using a microcarrier matrix system. J. Biomed. Mater. Res. 2001, 55, 242–253. [Google Scholar] [CrossRef]
- Facer, S.R.; Zaharias, R.S.; Andracki, M.E.; Lafoon, J.; Hunter, S.K.; Schneider, G.B. Rotary culture enhances pre-osteoblast aggregation and mineralization. J. Dent. Res. 2005, 84, 542–547. [Google Scholar] [CrossRef]
- Wang, T.W.; Wu, H.C.; Wang, H.Y.; Lin, F.H.; Sun, J.S. Regulation of adult human mesenchymal stem cells into osteogenic and chondrogenic lineages by different bioreactor systems. J. Biomed. Mater. Res. A 2009, 88, 935–946. [Google Scholar]
- Diederichs, S.; Röker, S.; Marten, D.; Peterbauer, A.; Scheper, T.; van Griensven, M.; Kasper, C. Dynamic cultivation of human mesenchymal stem cells in a rotating bed bioreactor system based on the Z RP platform. Biotechnol. Prog. 2009, 25, 1762–1771. [Google Scholar]
- Araujo, J.V.; Cunha-Reis, C.; Rada, T.; da Silva, M.A.; Gomes, M.E.; Yang, Y.; Ashammakhi, N.; Reis, R.L.; El-Haj, A.J.; Neves, N.M. Dynamic culture of osteogenic cells in biomimetically coated poly(caprolactone) nanofibre mesh constructs. Tissue Eng. Part A 2010, 16, 557–563. [Google Scholar] [CrossRef] [Green Version]
- Ben-David, D.; Kizhner, T.; Livne, E.; Srouji, S. A tissue-like construct of human bone marrow MSCs composite scaffold support in vivo ectopic bone formation. J. Tissue Eng. Regen. Med. 2010, 4, 30–37. [Google Scholar]
- Braccini, A.; Wendt, D.; Jaquiery, C.; Jakob, M.; Heberer, M.; Kenins, L.; Wodnar-Filipowicz, A.; Quarto, R.; Martin, I. Three-dimensional perfusion culture of human bone marrow cells and generation of osteoinductive grafts. Stem Cells 2005, 23, 1066–1072. [Google Scholar] [CrossRef]
- Karageorgiou, V.; Tomkins, M.; Fajardo, R.; Meinel, L.; Snyder, B.; Wade, K.; Chen, J.; Vunjak-Novakovic, G.; Kaplan, D.L. Porous silk fibroin 3-D scaffolds for delivery of bone morphogenetic protein-2 in vitro and in vivo. J. Biomed. Mater. Res. A 2006, 78, 324–334. [Google Scholar]
- Schliephake, H.; Zghoul, N.; Jäger, V.; van Griensven, M.; Zeichen, J.; Gelinsky, M.; Wülfing, T. Effect of seeding technique and scaffold material on bone formation in tissue-engineered constructs. J. Biomed. Mater. Res. A 2009, 90, 429–437. [Google Scholar]
- Schliephake, H.; Zghoul, N.; Jäger, V.; van Griensven, M.; Zeichen, J.; Gelinsky, M.; Szubtarsky, N. Bone formation in trabecular bone cell seeded scaffolds used for reconstruction of the rat mandible. Int. J. Oral Maxillofac. Surg. 2009, 38, 166–172. [Google Scholar] [CrossRef]
- Zhang, Z.; Teoh, S.H.; Chong, W.S.; Foo, T.T.; Chng, Y.C.; Choolani, M.; Chan, J. A biaxial rotating bioreactor for the culture of fetal mesenchymal stem cells for bone tissue engineering. Biomaterials 2009, 30, 2694–2704. [Google Scholar] [CrossRef]
- Janssen, F.W.; van Dijkhuizen-Radersma, R.; van Oorschot, A.; Oostra, J.; de Bruijn, J.D.; van Blitterswijk, C.A. Human tissue-engineered bone produced in clinically relevant amounts using a semi-automated perfusion bioreactor system: A preliminary study. J. Tissue Eng. Regen. Med. 2010, 4, 12–24. [Google Scholar]
- Yeatts, A.B.; Both, S.K.; Yang, W.; Alghamdi, H.S.; Yang, F.; Fisher, J.P.; Jansen, J.A. In vivo bone regeneration using tubular perfusion system bioreactor cultured nanofibrous scaffolds. Tissue Eng. Part A 2014, 20, 139–146. [Google Scholar] [CrossRef]
- Meinel, L.; Karageorgiou, V.; Fajardo, R.; Snyder, B.; Shinde-Patil, V.; Zichner, L.; Kaplan, D.; Langer, R.; Vunjak-Novakovic, G. Bone tissue engineering using human mesenchymal stem cells: Effects of scaffold material and medium flow. Ann. Biomed. Eng. 2004, 32, 112–122. [Google Scholar] [CrossRef]
- Kim, H.J.; Kim, U.J.; Leisk, G.G.; Bayan, C.; Georgakoudi, I.; Kaplan, D.L. Bone regeneration on macroporous aqueous-derived silk 3-D scaffolds. Macromol. Biosci. 2007, 7, 643–655. [Google Scholar] [CrossRef]
- Hofmann, S.; Hagenmüller, H.; Koch, A.M.; Müller, R.; Vunjak-Novakovic, G.; Kaplan, D.L.; Merkle, H.P.; Meinel, L. Control of in vitro tissue-engineered bone-like structures using human mesenchymal stem cells and porous silk scaffolds. Biomaterials 2007, 28, 1152–1162. [Google Scholar] [CrossRef]
- Mygind, T.; Stiehler, M.; Baatrup, A.; Li, H.; Zou, X.; Flyvbjerg, A.; Kassem, M.; Bünger, C. Mesenchymal stem cell ingrowth and differentiation on coralline hydroxyapatite scaffolds. Biomaterials 2007, 28, 1036–1047. [Google Scholar] [CrossRef]
- Stiehler, M.; Bünger, C.; Baatrup, A.; Lind, M.; Kassem, M.; Mygind, T. Effect of dynamic 3-D culture on proliferation, distribution, and osteogenic differentiation of human mesenchymal stem cells. J. Biomed. Mater. Res. A 2009, 89, 96–107. [Google Scholar]
- Teixeira, G.Q.; Barrias, C.C.; Lourenço, A.H.; Gonçalves, R.M. A multi-compartment holder for spinner flasks improves expansion and osteogenic differentiation of mesenchymal stem cells in 3D scaffolds. Tissue Eng. Part C Methods 2014. [Google Scholar] [CrossRef]
- Olivier, V.; Hivart, P.; Descamps, M.; Hardouin, P. In vitro culture of large bone substitutes in a new bioreactor: Importance of the flow direction. Biomed. Mater. 2007, 2, 174–180. [Google Scholar] [CrossRef]
- Bernhardt, A.; Lode, A.; Boxberger, S.; Pompe, W.; Gelinsky, M. Mineralised collagen—An artificial, extracellular bone matrix—Improves osteogenic differentiation of bone marrow stromal cells. J. Mater. Sci. Mater. Med. 2008, 19, 269–275. [Google Scholar]
- Bernhardt, A.; Lode, A.; Peters, F.; Gelinsky, M. Optimization of culture conditions for osteogenically-induced mesenchymal stem cells in β-tricalcium phosphate ceramics with large interconnected channels. J. Tissue Eng. Regen. Med. 2011, 5, 444–453. [Google Scholar] [CrossRef]
- Pisanti, P.; Yeatts, A.B.; Cardea, S.; Fisher, J.P.; Reverchon, E. Tubular perfusion system culture of human mesenchymal stem cells on poly-L-lactic acid scaffolds produced using a supercritical carbon dioxide-assisted process. J. Biomed. Mater. Res. A 2012, 100, 2563–2572. [Google Scholar]
- Hofmann, A.; Konrad, L.; Gotzen, L.; Printz, H.; Ramaswamy, A.; Hofmann, C. Bioengineered human bone tissue using autogenous osteoblasts cultured on different biomatrices. J. Biomed. Mater. Res. A 2003, 67, 191–199. [Google Scholar]
- Bjerre, L.; Bünger, C.E.; Kassem, M.; Mygind, T. Flow perfusion culture of human mesenchymal stem cells on silicate-substituted tricalcium phosphate scaffolds. Biomaterials 2008, 29, 2616–2627. [Google Scholar] [CrossRef]
- Grayson, W.L.; Fröhlich, M.; Yeager, K.; Bhumiratana, S.; Chan, M.E.; Cannizzaro, C.; Wan, L.Q.; Liu, X.S.; Guo, X.E.; Vunjak-Novakovic, G. Engineering anatomically shaped human bone grafts. Proc. Natl. Acad. Sci. USA 2010, 107, 3299–3304. [Google Scholar] [CrossRef]
- Fröhlich, M.; Grayson, W.L.; Marolt, D.; Gimble, J.M.; Kregar-Velikonja, N.; Vunjak-Novakovic, G. Bone grafts engineered from human adipose-derived stem cells in perfusion bioreactor culture. Tissue Eng. Part A 2010, 16, 179–189. [Google Scholar] [CrossRef]
- Grayson, W.L.; Marolt, D.; Bhumiratana, S.; Fröhlich, M.; Guo, X.E.; Vunjak-Novakovic, G. Optimizing the medium perfusion rate in bone tissue engineering bioreactors. Biotechnol. Bioeng. 2011, 108, 1159–1170. [Google Scholar] [CrossRef]
- Chen, M.; Wang, X.; Ye, Z.; Zhang, Y.; Zhou, Y.; Tan, W.S. A modular approach to the engineering of a centimeter-sized bone tissue construct with human amniotic mesenchymal stem cells-laden microcarriers. Biomaterials 2011, 32, 7532–7542. [Google Scholar] [CrossRef]
- Matziolis, D.; Tuischer, J.; Matziolis, G.; Kasper, G.; Duda, G.; Perka, C. Osteogenic Predifferentiation of Human Bone Marrow-Derived Stem Cells by Short-Term Mechanical Stimulation. Open Orthop. J. 2011, 5, 1–6. [Google Scholar] [CrossRef]
- Bölgen, N.; Yang, Y.; Korkusuz, P.; Güzel, E.; El Haj, A.J.; Pişkin, E. Three-Dimensional Ingrowth of Bone Cells within Biodegradable Cryogel Scaffolds in Bioreactors at Different Regimes. Tissue Eng. Part A 2008, 14, 1743–1750. [Google Scholar] [CrossRef]
- Sucosky, P.; Osorio, D.F.; Brown, J.B.; Neitzel, G.P. Fluid mechanics of a spinner-flask bioreactor. Biotechnol. Bioeng. 2004, 85, 34–46. [Google Scholar] [CrossRef]
- Martin, Y.; Vermette, P. Bioreactors for tissue mass culture: Design, characterization, and recent advances. Biomaterials 2005, 26, 7481–7503. [Google Scholar] [CrossRef]
- Yasuda, K.; Inoue, S.; Tabata, Y. Influence of culture method on the proliferation and osteogenic differentiation of human adipo-stromal cells in nonwoven fabrics. Tissue Eng. 2004, 10, 1587–1596. [Google Scholar] [CrossRef]
- Boschetti, F.; Raimondi, M.T.; Migliavacca, F.; Dubini, G. Prediction of the micro-fluid dynamic environment imposed to three-dimensional engineered cell systems in bioreactors. J. Biomech. 2006, 39, 418–425. [Google Scholar]
- David, B.; Bonnefont-Rousselot, D.; Oudina, K.; Degat, M.C.; Deschepper, M.; Viateau, V.; Bensidhoum, M.; Oddou, C.; Petite, H. A Perfusion Bioreactor for Engineering Bone Constructs: An in vitro and in vivo Study. Tissue Eng. Part C Methods 2011, 17, 505–516. [Google Scholar] [CrossRef]
- Liu, C.; Abedian, R.; Meister, R.; Haasper, C.; Hurschler, C.; Krettek, C.; von Lewinski, G.; Jagodzinski, M. Influence of perfusion and compression on the proliferation and differentiation of bone mesenchymal stromal cells seeded on polyurethane scaffolds. Biomaterials 2012, 33, 1052–1064. [Google Scholar] [CrossRef]
- Hutmacher, D.W.; Singh, H. Computational fluid dynamics for improved bioreactor design and 3D culture. Trends Biotechnol. 2008, 26, 166–172. [Google Scholar] [CrossRef]
- Vetsch, J.R.; Müller, R.; Hofmann, S. The evolution of simulation techniques for dynamic bone tissue engineering in bioreactors. J. Tissue Eng. Regen. Med. 2013. [Google Scholar] [CrossRef]
- Milan, J.L.; Planell, J.A.; Lacroix, D. Simulation of bone tissue formation within a porous scaffold under dynamic compression. Biomech. Model. Mech. 2010, 9, 583–596. [Google Scholar] [CrossRef]
- Galbusera, F.; Cioffi, M.; Raimondi, M.T.; Pietrabissa, R. Computational modeling of combined cell population dynamics and oxygen transport in engineered tissue subject to interstitial perfusion. Comput. Methods Biomech. Biomed. Eng. 2007, 10, 279–287. [Google Scholar] [CrossRef]
- Lappa, M. Organic tissues in rotating bioreactors: Fluid-mechanical aspects, dynamic growth models, and morphological evolution. Biotechnol. Bioeng. 2003, 84, 518–532. [Google Scholar] [CrossRef]
- Verbruggen, S.W.; Vaughan, T.J.; McNamara, L.M. Fluid flow in the osteocyte mechanical environment: A fluid-structure interaction approach. Biomech. Model. Mechanobiol. 2014, 13, 85–97. [Google Scholar] [CrossRef]
- Brennan, M.A.; Jean-Michel Davaine, J.M.; Layrolle, P. Pre-vascularization of bone tissue-engineered constructs. Stem Cell Res. Ther. 2013, 4, 96. [Google Scholar]
- Rivron, N.C.; Liu, J.J.; Rouwkema, J.; de Boer, J.; van Blitterswijk, C.A. Engineering vascularised tissues in vitro. Eur. Cells Mater. 2008, 21, 27–40. [Google Scholar]
- Laurent, C.P.; Vaquette, C.; Martin, C.; Guedon, E.; Wu, X.; Delconte, A.; Dumas, D.; Hupont, S.; Isla, N.D.; Rahouadj, R.; Wang, X. Towards a tissue-engineered ligament: Design and preliminary evaluation of a dedicated multi-chamber tension-torsion bioreactor. Processes 2014, 2, 167–179. [Google Scholar] [CrossRef]
- Figallo, E.; Cannizzaro, C.; Gerecht, S.; Burdick, J.A.; Langer, R.; Elvassore, N.; Vunjak-Novakovic, G. Micro-bioreactor array for controlling cellular microenvironments. Lab Chip 2007, 7, 710–719. [Google Scholar] [CrossRef]
- Rauh, J.; Milan, F.; Günther, K.P.; Stiehler, M. Bioreactor systems for bone tissue engineering. Tissue Eng. Part B Rev. 2011, 17, 263–280. [Google Scholar] [CrossRef]
- de Peppo, G.M.; Marolt, D. Make no bones about it: Cells could soon be reprogrammed to grow replacement bones? Expert Opin. Biol. Ther. 2014, 14, 1–5. [Google Scholar] [CrossRef]
- Archer, R.; Williams, D.J. Why tissue engineering needs process engineering. Nat. Biotechnol. 2005, 23, 1353–1355. [Google Scholar] [CrossRef]
- Martin, I.; Smith, T.; Wendt, D. Bioreactor-based roadmap for the translation of tissue engineering strategies into clinical products. Trends Biotechnol. 2009, 27, 495–502. [Google Scholar] [CrossRef] [Green Version]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sladkova, M.; De Peppo, G.M. Bioreactor Systems for Human Bone Tissue Engineering. Processes 2014, 2, 494-525. https://doi.org/10.3390/pr2020494
Sladkova M, De Peppo GM. Bioreactor Systems for Human Bone Tissue Engineering. Processes. 2014; 2(2):494-525. https://doi.org/10.3390/pr2020494
Chicago/Turabian StyleSladkova, Martina, and Giuseppe Maria De Peppo. 2014. "Bioreactor Systems for Human Bone Tissue Engineering" Processes 2, no. 2: 494-525. https://doi.org/10.3390/pr2020494




