Molecular Pathophysiology of Epithelial Barrier Dysfunction in Inflammatory Bowel Diseases
Abstract
:1. Introduction
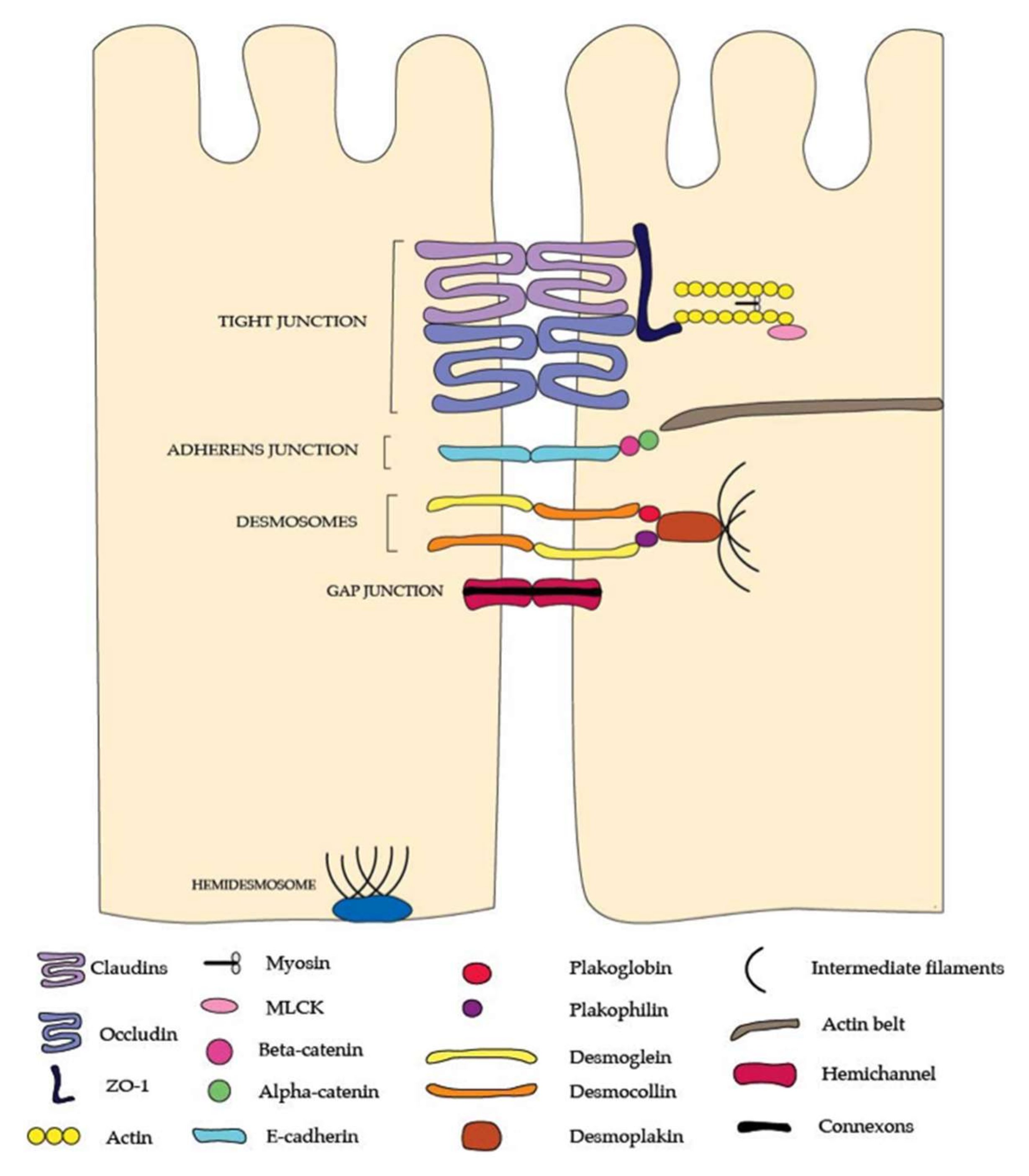
2. The Intestinal Epithelial Barrier
3. Intracellular Regulators of Paracellular Permeability
4. Epithelial Restitution and Healing
5. Clinical Implications of Impaired Intestinal Permeability in IBD
6. Assessing Barrier Function in Clinical Practice Today
7. Biomarkers of Intestinal Barrier Function
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yau, Y.; Leong, R.W.; Zeng, M.; Wasinger, V.C. Proteomics and metabolomics in inflammatory bowel disease. J. Gastroenterol. Hepatol. 2013, 28, 1076–1086. [Google Scholar] [CrossRef] [PubMed]
- Antoni, L.; Nuding, S.; Wehkamp, J.; Stange, E.F. Intestinal barrier in inflammatory bowel disease. World J. Gastroenterol. WJG 2014, 20, 1165–1179. [Google Scholar] [CrossRef] [PubMed]
- Yau, Y.Y.; Leong, R.W.L.; Pudipeddi, A.; Redmond, D.; Wasinger, V.C. Serological Epithelial Component Proteins Identify Intestinal Complications in Crohn’s Disease. Mol. Cell. Proteom. MCP 2017, 16, 1244–1257. [Google Scholar] [CrossRef] [PubMed]
- Wasinger, V.C.; Yau, Y.; Duo, X.; Zeng, M.; Campbell, B.; Shin, S.; Luber, R.; Redmond, D.; Leong, R.W. Low Mass Blood Peptides Discriminative of Inflammatory Bowel Disease (IBD) Severity: A Quantitative Proteomic Perspective. Mol. Cell. Proteom. 2016, 15, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Leong, R.W.; Wasinger, V.C.; Ip, M.; Yang, M.; Phan, T.G. Impaired Intestinal Permeability Contributes to Ongoing Bowel Symptoms in Patients With Inflammatory Bowel Disease and Mucosal Healing. Gastroenterology. 2017, 153, 723–731. [Google Scholar] [CrossRef] [PubMed]
- Arnott, I.D.R.; Kingstone, K.; Ghosh, S. Abnormal intestinal permeability predicts relapse in inactive Crohn disease. Scand. J. Gastroenterol. 2000, 35, 1163–1169. [Google Scholar] [PubMed]
- De Arcangelis, A.; Hamade, H.; Alpy, F.; Normand, S.; Bruyere, E.; Lefebvre, O.; Mechine-Neuville, A.; Siebert, S.; Pfister, V.; Lepage, P.; et al. Hemidesmosome integrity protects the colon against colitis and colorectal cancer. Gut 2017, 66, 1748–1760. [Google Scholar] [CrossRef] [PubMed]
- D’Inca, R.; Di Leo, V.; Corrao, G.; Martines, D.; D’Odorico, A.; Mestriner, C.; Venturi, C.; Longo, G.; Sturniolo, G.C. Intestinal permeability test as a predictor of clinical course in Crohn’s disease. Am. J. Gastroenterol. 1999, 94, 2956–2960. [Google Scholar] [CrossRef] [PubMed]
- Gassler, N.; Rohr, C.; Schneider, A.; Kartenbeck, J.; Bach, A.; Obermuller, N.; Otto, H.F.; Autschbach, F. Inflammatory bowel disease is associated with changes of enterocytic junctions. Am. J. Physiol. Gastrointest. Liver Physiol. 2001, 281, G216–G228. [Google Scholar] [CrossRef] [PubMed]
- Goswami, P.; Das, P.; Verma, A.K.; Prakash, S.; Das, T.K.; Nag, T.C.; Ahuja, V.; Gupta, S.D.; Makharia, G.K. Are alterations of tight junctions at molecular and ultrastructural level different in duodenal biopsies of patients with celiac disease and crohn's disease? Virchows Arch. 2014, 465, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Gu, P.; Theiss, A.; Han, J.; Feagins, L.A. Increased pecam-1 predicts increased risk for flare in patients with IBD. Am. J. Gastroenterol. 2015, 110, S790–S791. [Google Scholar]
- Hollander, D.; Vadheim, C.M.; Brettholz, E.; Petersen, G.M.; Delahunty, T.; Rotter, J.I. Increased Intestinal Permeability in Patients with Crohn’s Disease and Their RelativesA Possible Etiologic Factor. Ann. Intern. Med. 1986, 105, 883–885. [Google Scholar] [CrossRef] [PubMed]
- Kiesslich, R.; Duckworth, C.A.; Moussata, D.; Gloeckner, A.; Lim, L.G.; Goetz, M.; Pritchard, D.M.; Galle, P.R.; Neurath, M.F.; Watson, A.J.M. Local barrier dysfunction identified by confocal laser endomicroscopy predicts relapse in inflammatory bowel disease. Gut 2012, 61, 1146–1153. [Google Scholar] [CrossRef] [PubMed]
- Ni, J.; Wu, G.D.; Albenberg, L.; Tomov, V.T. Gut microbiota and IBD: Causation or correlation? Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 573–584. [Google Scholar] [CrossRef] [PubMed]
- Shawki, A.; McCole, D.F. Mechanisms of Intestinal Epithelial Barrier Dysfunction by Adherent-Invasive Escherichia coli. Cell. Mol. Gastroenterol. Hepatol. 2017, 3, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Hager, C.L.; Ghannoum, M.A. The mycobiome: Role in health and disease, and as a potential probiotic target in gastrointestinal disease. Dig. Liver Dis. 2017, 49, 1171–1176. [Google Scholar] [CrossRef] [PubMed]
- Bischoff, S.C.; Barbara, G.; Buurman, W.; Ockhuizen, T.; Schulzke, J.-D.; Serino, M.; Tilg, H.; Watson, A.; Wells, J.M. Intestinal permeability - a new target for disease prevention and therapy. BMC Gastroenterol. 2014, 14, 189. [Google Scholar] [CrossRef] [PubMed]
- Turner, J.R. Intestinal mucosal barrier function in health and disease. Nat. Rev. Immunol. 2009, 9, 799–809. [Google Scholar] [CrossRef] [PubMed]
- Krug, S.M.; Schulzke, J.D.; Fromm, M. Tight junction, selective permeability, and related diseases. Semin. Cell Dev. Biol. 2014, 36, 166–176. [Google Scholar] [CrossRef] [PubMed]
- Raleigh, D.R.; Marchiando, A.M.; Zhang, Y.; Shen, L.; Sasaki, H.; Wang, Y.; Long, M.; Turner, J.R. Tight Junction–associated MARVEL Proteins MarvelD3, Tricellulin, and Occludin Have Distinct but Overlapping Functions. Mol. Biol. Cell 2010, 21, 1200–1213. [Google Scholar] [CrossRef] [PubMed]
- Edelblum, K.L.; Turner, J.R. The Tight Junction in Inflammatory Disease: Communication Breakdown. Curr. Opin. Pharmacol. 2009, 9, 715–720. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Turner, J.R. Role of Epithelial Cells in Initiation and Propagation of Intestinal Inflammation. Eliminating the static: Tight junction dynamics exposed. Am. J. Physiol. 2006, 290, G577–G582. [Google Scholar] [CrossRef] [PubMed]
- Petit, C.S.V.; Barreau, F.; Besnier, L.; Gandille, P.; Riveau, B.; Chateau, D.; Roy, M.; Berrebi, D.; Svrcek, M.; Cardot, P.; et al. Requirement of Cellular Prion Protein for Intestinal Barrier Function and Mislocalization in Patients With Inflammatory Bowel Disease. Gastroenterology 2012, 143, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Zeissig, S.; Burgel, N.; Gunzel, D.; Richter, J.; Mankertz, J.; Wahnschaffe, U.; Kroesen, A.J.; Zeitz, M.; Fromm, M.; Schulzke, J.D. Changes in expression and distribution of claudin 2, 5 and 8 lead to discontinuous tight junctions and barrier dysfunction in active Crohn’s disease. Gut 2007, 56, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Oshima, T.; Miwa, H.; Joh, T. Changes in the expression of claudins in active ulcerative colitis. J. Gastroenterol. Hepatol. 2008, 23, S146–S150. [Google Scholar] [CrossRef] [PubMed]
- Heller, F.; Fromm, A.; Gitter, A.H.; Mankertz, J.; Schulzke, J.D. Epithelial apoptosis is a prominent feature of the epithelial barrier disturbance in intestinal inflammation: Effect of pro-inflammatory interleukin-13 on epithelial cell function. Mucosal Immunol. 2008, 1, S58–S61. [Google Scholar] [CrossRef] [PubMed]
- Thuijls, G.; Derikx, J.P.M.; de Haan, J.-J.; Grootjans, J.; de Bruine, A.; Masclee, A.A.M.; Heineman, E.; Buurman, W.A. Urine-based detection of intestinal tight junction loss. J. Clin. Gastroenterol. 2010, 44, e14–e19. [Google Scholar] [CrossRef] [PubMed]
- Das, P.; Goswami, P.; Das, T.K.; Nag, T.; Sreenivas, V.; Ahuja, V.; Panda, S.K.; Gupta, S.D.; Makharia, G.K. Comparative tight junction protein expressions in colonic Crohn’s disease, ulcerative colitis, and tuberculosis: A new perspective. Virchows Archiv 2012, 460, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Poritz, L.S.; Harris, I.L.; Kelly, A.A.; Koltun, W.A. Increase in the tight junction protein claudin-1 in intestinal inflammation. Dig. Dis. Sci. 2011, 56, 2802–2809. [Google Scholar] [CrossRef] [PubMed]
- Kucharzik, T.; Walsh, S.V.; Chen, J.; Parkos, C.A.; Nusrat, A. Neutrophil transmigration in inflammatory bowel disease is associated with differential expression of epithelial intercellular junction proteins. Am. J. Pathol. 2001, 159, 2001–2009. [Google Scholar] [CrossRef]
- Heller, F.; Florian, P.; Bojarski, C.; Richter, J.; Christ, M.; Hillenbrand, B.; Mankertz, J.; Gitter, A.H.; Bürgel, N.; Fromm, M.; et al. Interleukin-13 Is the Key Effector Th2 Cytokine in Ulcerative Colitis That Affects Epithelial Tight Junctions, Apoptosis, and Cell Restitution. Gastroenterology 2005, 129, 550–564. [Google Scholar] [CrossRef] [PubMed]
- Muise, A.M.; Walters, T.D.; Glowacka, W.K.; Griffiths, A.M.; Ngan, B.Y.; Lan, H.; Xu, W.; Silverberg, M.S.; Rotin, D. Polymorphisms in E-cadherin (CDH1) result in a mislocalised cytoplasmic protein that is associated with Crohn’s disease. Gut 2009, 58, 1121–1127. [Google Scholar] [CrossRef] [PubMed]
- Karstensen, J. Evaluation of confocal laser endomicroscopy for assessment and monitoring of therapeutic response in patients with inflammatory bowel disease. Dan. Med. J. 2016, 63, 1–23. [Google Scholar]
- Kiela, P.R.; Ghishan, F.K. Physiology of Intestinal Absorption and Secretion. Best Pract. Res. Clin. Gastroenterol. 2016, 30, 145–159. [Google Scholar] [CrossRef] [PubMed]
- Michielan, A.; D’Incà, R. Intestinal Permeability in Inflammatory Bowel Disease: Pathogenesis, Clinical Evaluation, and Therapy of Leaky Gut. Mediat. Inflamm. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Shen, L. Myosin light chain phosphorylation regulates barrier function by remodeling tight junction structure. J. Cell Sci. 2006, 119, 2095–2106. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Nalle, S.C.; Shen, L.; Turner, E.S.; Singh, G.; Breskin, L.A.; Khramtsova, E.A.; Khramtsova, G.; Tsai, P.-Y.; Fu, Y.-X.; et al. TNFR2 Activates MLCK-Dependent Tight Junction Dysregulation to Cause Apoptosis-Mediated Barrier Loss and Experimental Colitis. Gastroenterology 2013, 145, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, K.E.; Turner, J.R. Myosin light chain kinase: Pulling the strings of epithelial tight junction function. Ann. N. Y. Acad. Sci. 2012, 1258, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Blair, S.A.; Kane, S.V.; Clayburgh, D.R.; Turner, J.R. Epithelial myosin light chain kinase expression and activity are upregulated in inflammatory bowel disease. Lab. Investig. 2006, 86, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Weber, C.R.; Raleigh, D.R.; Su, L.; Shen, L.; Sullivan, E.A.; Wang, Y.; Turner, J.R. Epithelial myosin light chain kinase activation induces mucosal interleukin-13 expression to alter tight junction ion selectivity. J. Biol. Chem. 2010, 285, 12037–12046. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Graham, W.V.; Wang, Y.; Witkowski, E.D.; Schwarz, B.T.; Turner, J.R. Interferon-γ and Tumor Necrosis Factor-α Synergize to Induce Intestinal Epithelial Barrier Dysfunction by Up-Regulating Myosin Light Chain Kinase Expression. Am. J. Pathol. 2005, 166, 409–419. [Google Scholar] [CrossRef]
- Zolotarevsky, Y.; Hecht, G.; Koutsouris, A.; Gonzalez, D.E.; Quan, C.; Tom, J.; Mrsny, R.J.; Turner, J.R. A membrane-permeant peptide that inhibits MLC kinase restores barrier function in in vitro models of intestinal disease. Gastroenterology 2002, 123, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Clayburgh, D.R.; Barrett, T.A.; Tang, Y.; Meddings, J.B.; Van Eldik, L.J.; Watterson, D.M.; Clarke, L.L.; Mrsny, R.J.; Turner, J.R. Epithelial myosin light chain kinase–dependent barrier dysfunction mediates T cell activation–induced diarrhea in vivo. J. Clin. Investig. 2005, 115, 2702–2715. [Google Scholar] [CrossRef] [PubMed]
- Walsh, S.V.; Hopkins, A.M.; Chen, J.; Narumiya, S.; Parkos, C.A.; Nusrat, A. Rho kinase regulates tight junction function and is necessary for tight junction assembly in polarized intestinal epithelia. Gastroenterology 2001, 121, 566–579. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Shen, L.; Clayburgh, D.R.; Nalle, S.C.; Sullivan, E.A.; Meddings, J.B.; Abraham, C.; Turner, J.R. Targeted epithelial tight junction dysfunction causes immune activation and contributes to development of experimental colitis. Gastroenterology 2009, 136, 551–563. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Turner, J.R. Actin Depolymerization Disrupts Tight Junctions via Caveolae-mediated Endocytosis. Mol. Biol. Cell 2005, 16, 3919–3936. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Kim, J.J.; Shen, J.; Dai, N. Crosstalk between Inflammation and ROCK/MLCK Signaling Pathways in Gastrointestinal Disorders with Intestinal Hyperpermeability. Gastroenterol. Res. Pract. 2016, 2016, 9. [Google Scholar] [CrossRef] [PubMed]
- Hanna, S.; El-Sibai, M. Signaling networks of Rho GTPases in cell motility. Cell. Signal. 2013, 25, 1955–1961. [Google Scholar] [CrossRef] [PubMed]
- Sturm, A.; Dignass, A.U. Epithelial restitution and wound healing in inflammatory bowel disease. World J. Gastroenterol. 2008, 14, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Eisenhoffer, G.T.; Loftus, P.D.; Yoshigi, M.; Otsuna, H.; Chien, C.-B.; Morcos, P.A.; Rosenblatt, J. Crowding induces live cell extrusion to maintain homeostatic cell numbers in epithelia. Nature 2012, 484, 546–549. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.J.; Kay, T.M.; Davis, E.M.; Lou, Y.; Kao, D.; Claggett, B.; Fedorak, R.N.; Irvin, R.T. Epithelial Cell Extrusion Zones Observed on Confocal Laser Endomicroscopy Correlates with Immunohistochemical Staining of Mucosal Biopsy Samples. Dig. Dis. Sci. 2016, 61, 1895–1902. [Google Scholar] [CrossRef] [PubMed]
- Neurath, M.F. New targets for mucosal healing and therapy in inflammatory bowel diseases. Mucosal Immunol. 2014, 7, 6–19. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.J.; Wong, K.; Thiesen, A.L.; Mah, S.J.; Dieleman, L.A.; Claggett, B.; Saltzman, J.R.; Fedorak, R.N. Increased epithelial gaps in the small intestines of patients with inflammatory bowel disease: Density matters. Gastrointest. Endosc. 2011, 73, 1174–1180. [Google Scholar] [CrossRef] [PubMed]
- Quigley, E.M.M. Leaky gut- concept or clinical entity? Curr. Opin. Gastroenterol. 2016, 32, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Wyatt, J.; Vogelsang, H.; Hubl, W.; Waldhoer, T.; Lochs, H. Intestinal permeability and the prediction of relapse in Crohn’s disease. Lancet 1993, 341, 1437–1439. [Google Scholar] [CrossRef]
- Vivinus-Nebot, M.; Frin-Mathy, G.; Bzioueche, H.; Dainese, R.; Bernard, G.; Anty, R.; Filippi, J.; Saint-Paul, M.C.; Tulic, M.K.; Verhasselt, V.; et al. Functional bowel symptoms in quiescent inflammatory bowel diseases: Role of epithelial barrier disruption and low-grade inflammation. Gut 2014, 63, 744–752. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, D.N.; Karstensen, J.G.; Riis, L.B.; Brynskov, J.; Vilmann, P. Confocal Laser Endomicroscopy in Inflammatory Bowel Disease—A Systematic Review. J. Crohns Colitis 2015, 9, 1152–1159. [Google Scholar] [CrossRef] [PubMed]
- Lim, L.G.; Neumann, J.; Hansen, T.; Goetz, M.; Hoffman, A.; Neurath, M.F.; Galle, P.R.; Chan, Y.H.; Kiesslich, R.; Watson, A.J. Confocal endomicroscopy identifies loss of local barrier function in the duodenum of patients with Crohn’s disease and ulcerative colitis. Inflamm. Bowel Dis. 2014, 20, 892–900. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, D.; Bording-Jorgensen, M.; Huynh, H.Q.; Carroll, M.W.; Turcotte, J.-F.; Sergi, C.; Liu, J.; Wine, E. Increased Epithelial Gap Density in the Noninflamed Duodenum of Children With Inflammatory Bowel Diseases. J. Pediatric Gastroenterol. Nutr. 2016, 63, 644–650. [Google Scholar] [CrossRef] [PubMed]
- Karstensen, J.G.; Săftoiu, A.; Brynskov, J.; Hendel, J.; Klausen, P.; Cartan, A.T.; Klausen, T.W.; Riis, L.B.; Vilmann, P. Confocal laser endomicroscopy: A novel method for prediction of relapse in Crohn’s disease. Endoscopy 2016, 48, 364–372. [Google Scholar] [PubMed]
- Paramsothy, S.; Leong, R. Fluorescein contrast in confocal laser endomicroscopy. Nat. Rev. Gastroenterol. Hepatol. 2010, 7, 366–368. [Google Scholar] [CrossRef] [PubMed]
- Turcotte, J.F.; Wong, K.; Mah, S.J.; Dieleman, L.A.; Fedorak, R.N.; Wine, E.; Kroeker, K.I.; Liu, J.J. Increased epithelial gap density as measured by probe-based confocal laser endomicroscopy predicts the need for future hospitalization in patients with inflammatory bowel disease (IBD). Gastroenterology 2012, 142, S6–S7. [Google Scholar] [CrossRef]
- Li, C.-Q.; Liu, J.; Ji, R.; Li, Z.; Xie, X.J.; Li, Y.Q. Use of confocal laser endomicroscopy to predict relapse of ulcerative colitis. BMC Gastroenterol. 2014, 14, 45. [Google Scholar] [CrossRef] [PubMed]
- Buda, A.; Hatem, G.; Neumann, H.; Inca, R.; Mescoli, C.; Piselli, P.; Jackson, J.; Bruno, M.; Sturniolo, G. Confocal laser endomicroscopy for prediction of disease relapse in ulcerative colitis: A pilot study. J. Crohns Colitis 2014, 8, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Shavrov, A.; Kharitonova, A.Y.; Davis, E.M.; Claggett, B.; Morozov, D.A.; Brown, D.K.; Shavrov, A.A.; Liu, J.J. A pilot study of confocal laser endomicroscopy to predict barrier dysfunction and relapse in pediatric inflammatory bowel disease. J. Pediatr. Gastroenterol. Nutr. 2016, 62, 873–878. [Google Scholar] [CrossRef] [PubMed]
- Soderholm, J.D.; Olaison, G.; Lindberg, E.; Hannestad, U.; Vindels, A.; Tysk, C.; Jarnerot, G.; Sjodahl, R. Different intestinal permeability patterns in relatives and spouses of patients with Crohn’s disease: An inherited defect in mucosal defence? Gut 1999, 44, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Breslin, N.P.; Nash, C.; Hilsden, R.J.; Hershfield, N.B.; Price, L.M.; Meddings, J.B.; Sutherland, L.R. Intestinal permeability is increased in a proportion of spouses of patients with Crohn’s disease. Am. J. Gastroenterol. 2001, 96, 2934–2938. [Google Scholar] [CrossRef] [PubMed]
- Mace, V.; Ahluwalia, A.; Coron, E.; Le Rhun, M.; Boureille, A.; Bossard, C.; Mosnier, J.-F.; Matysiak-Budnik, T.; Tarnawski, A.S. Confocal laser endomicroscopy: A new gold standard for the assessment of mucosal healing in ulcerative colitis. J. Gastroenterol. Hepatol. 2015, 30, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Ip, M.; Yang, M.; Wong, B.; Power, T.; Lin, L.; Xuan, W.; Phan, T.G.; Leong, R.W. The learning curve, interobserver, and intraobserver agreement of endoscopic confocal laser endomicroscopy in the assessment of mucosal barrier defects. Gastrointest. Endosc. 2016, 83, 785–791. [Google Scholar] [CrossRef] [PubMed]
- Anderson, A.D.G.; Jain, P.K.; Fleming, S.; Poon, P.; Mitchell, C.J.; MacFie, J. Evaluation of a triple sugar test of colonic permeability in humans. Acta Physiol. Scand. 2004, 182, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Gianolio, E.; Boffa, C.; Orecchia, V.; Bardini, P.; Catanzaro, V.; Poli, V. A relaxometric method for the assessment of intestinal permeability based on the oral administration of gadolinium-based MRI contrast agents. NMR Biomed. 2016, 29, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Grootjans, J.; Thuijls, G.; Verdam, F.; Derikx, J.P.M.; Lenaerts, K.; Buurman, W.A. Non-invasive assessment of barrier integrity and function of the human gut. World J. Gastrointest. Surg. 2010, 2, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Vetrano, S.; Ploplis, V.A.; Sala, E.; Sandoval-Cooper, M.; Donahue, D.L.; Correale, C.; Arena, V.; Spinelli, A.; Repici, A.; Malesci, A.; et al. Unexpected role of anticoagulant protein C in controlling epithelial barrier integrity and intestinal inflammation. Proc. Natl. Acad. Sci. USA 2011, 108, 19830–19835. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Feo, J.A.; Puerto, M.; Fernandez-Mena, C.; Verdejo, C.; Lara, J.M.; Diaz-Sanchez, M.; Alvarez, E.; Vaquero, J.; Marin-Jimenez, I.; Banares, R.; et al. A new role for reticulon-4B/NOGO-B in the intestinal epithelial barrier function and inflammatory bowel disease. Am. J. Physiol. 2015, 308, 981–993. [Google Scholar] [CrossRef] [PubMed]
- Vetrano, S.; Rescigno, M.; Rosaria Cera, M.; Correale, C.; Rumio, C.; Doni, A.; Fantini, M.; Sturm, A.; Borroni, E.; Repici, A.; et al. Unique Role of Junctional Adhesion Molecule-A in Maintaining Mucosal Homeostasis in Inflammatory Bowel Disease. Gastroenterology 2008, 135, 173–184. [Google Scholar] [CrossRef] [PubMed]
- Gu, A.P.; Theiss, A.A.; Han, A.J.; Feagins, A.L. Increased Cell Adhesion Molecules, PECAM-1, ICAM-3, or VCAM-1, Predict Increased Risk for Flare in Patients With Quiescent Inflammatory Bowel Disease. J. Clin. Gastroenterol. 2017, 51, 522–527. [Google Scholar] [CrossRef] [PubMed]
| Action | Regulatory Factors |
|---|---|
| Inhibit cell proliferation | TGF-β |
| Activin A | |
| Promote epithelial restitution via TGF-β dependent pathway | Epidermal growth factor (EGF) |
| Glucagon-like-peptide-2 (GLP-2) | |
| IL-1 | |
| IFN-ϒ | |
| IL-2 | |
| HGF | |
| VEGF | |
| FGF | |
| Promote epithelial restitution via TGF-β independent pathway | Trefoil peptides |
| Galectin-2 | |
| Galectin-4 | |
| Keratinocyte growth factor (KGF) | |
| Decrease epithelial restitution velocity | IL-13 [32] |
| Promote epithelial proliferation | Epidermal growth factor (EGF) |
| TGF-α | |
| IL-6IL-22 | |
| Induce cell apoptosis | TNF-α |
| Prevent cell apoptosis | Prostaglandin E2 |
| Technique | General Principle | Test Site | Test Method | Limitations |
|---|---|---|---|---|
| Molecular Probes | ||||
| Lactulose/mannitol | Oligosaccharides of different sizes | Small intestine | Urine | Time-consuming. Metabolised in the colon so limited application in assessing the large intestine (e.g., ulcerative colitis (UC)). Does not show permeation of bacterial components. Mannitol is contraindicated with blood transfusions. |
| Sucralose | Sucralose | Colon | Urine | Time-consuming. Does not show permeation of bacterial components. |
| Multi-sugar test | Sucrose, lactulose, sucralose, erythritol, rhamnase | Whole intestine | Urine | Time-consuming. Does not show permeation of bacterial components. |
| 51Cr-EDTA | 51Cr-EDTA crosses the intestinal barrier via the paracellular route and has similar physiological properties to oliogosaccharides. | Whole intestine | Urine | Invasive and complex detection method. Not readily available. Radioactivity. Impractical in clinical setting. Does not show permeation of bacterial components. |
| PEG4000/400 | Polyethylene glycol, an inert molecule of different sizes. | Whole intestine | Urine | Time-consuming. The exact route of PEG is not well defined [70], thus implications in interpreting results. Does not show permeation of bacterial components. |
| Gadolinium-based MRI contrast agent [71] | Gadolinium (500–1000 Da) | Whole intestine | 24-h urine collection | Lack of evidence in human studies. More expensive and may have higher toxicity than conventional sugars. Partial hepatobiliary elimination. Contraindicated in renal impairment. |
| Ussing chambers | Ion transport across the intestinal epithelium tissue sample is measured using a short circuit current. | Site-specific | Biopsy | Invasive and complex detection method. Ex-vivo. Lack of correlation between Ussing chamber and other permeability assays. |
| Imaging | ||||
| Confocal laser endomicroscopy | Intravenously-administered fluorescent contrast is seen to leak through the small intestinal mucosa under real time endoscopy. | Terminal ileum, colon, duodenum | Endoscopy | Invasive. Time-consuming (average of 46.5 minutes [60]). Validated measurement scores include the Watson grade (semi-quantitative [60]) and confocal leak score (quantitative) [5]. Requires special training of the endoscopist. Does not show permeation of bacterial components. |
| Biomarkers of Intestinal Permeability | ||||
| Claudin-3 [27] | Epithelial tight junction protein | NA | Urine | Limited data and lack of randomised trials. |
| Bacteria-Related Markers | ||||
| Lipopolysaccharide (LPS) assay | Show endotoxemia from bacterial translocation due to barrier function failure. | Colon | Blood (portal venous) | Technical limitation in detecting low levels of LPS in the peripheral blood. Requires careful standardization of the measurement. Evidence of use in Inflammatory Bowel Disease (IBD). |
| Circulating endotoxin core antibodies | An indirect measure of translocation of bacterial products by quantifying immunoglobulins (IgG, IgM and IgA) against the inner core of endotoxin for acute phase of intestinal barrier damage and function [72]. | Colon | Blood | Only study done on post-operative patients, not patients with chronic gastrointestinal disease. Evidence for use in IBD. |
| Plasma d-lactate | d-lactate is produced by the gut bacteria and translocated across the intestinal mucosa with barrier dysfunction. | Colon | Blood | False positive test with bacterial over growth. Limited use in critically ill patients (e.g., ischemic colonic injury, acute necrotizing pancreatitis). |
| Faecal butyrate concentrations | Butyrate is a barrier enhancing substance, modifying claudin-1 and -2 to preserve intestinal barrier function and preventing bacterial translocation. | Colon | Faeces | Poorly established. The test relies on the principle that butyrate as a single major component of the barrier function rather than a complex and interactive entity. |
| Bacteria-derived haemolysin | Toxin that impair the intestinal barrier. | Colon | Poorly established. Results are attributed to only haemolysin-producing bacteria. | |
| Assessment of fatty liver disease | Inflammation and fatty liver disease result from translocation of bacteria and its products into the portal system. | Whole intestine | Imaging | Poor specificity. |
| Reference | Sample | Sample Size | Technique | Findings |
|---|---|---|---|---|
| Gassler et al., 2001 [9] | Surgical specimen | 10 ulcerative colitis(UC) 10 Crohn’s disease(CD) 10 sporadic colon cancer | Reverse transcription Quantitative PCR and sequencing reaction Immunofluorescence staining and immunoblotting Immunohistochemistry Western blot and densitometric analysis | In actively inflamed Inflammatory Bowel Disease(IBD) tissue: desmosome protein expressions (desmoplakin-1, desmoglein-2 and desmocllin-2) decreased with severity of inflammation in IBD tissue (p < 0.05); Adherens junction(AJ) proteins such as E-cadherin and α-catenin were highly reduced; APC, p 120, plakophilin-2, β-catenin and plakoglobin were decreased and correlated with degree of inflammation in UC; plakophilin-2 and plakoglobin, but not β-catenin or APC proteins were reduced in actively inflamed CD; Tight junction(TJ) strands were discontinuous with reduced ZO-1 and occludin expression. In inactive IBD tissue: AJ-associated proteins were affected, but not desmosomes and TJs. Therefore, these alterations are not a primary occurrence in IBD. |
| Kucharzik et al., 2001 [30] | Colonic biopsy | 11 active UC 9 active CD 29 control (normal colorectal mucosa or surgical resection of colon cancer) | Immunofluorescence Immunohistochemistry Western blotting | Global downregulation of occludin in IBD compared to controls. In epithelial cells adjacent to transmigrating polynorphonuclear leukocytes(PMNs), expressions of other TJ and AJ proteins were also downregulated (i.e.,zonulin-1 (ZO-1), claudin-1, junction adhesion molecule(JAM), beta-catenin, and E-cadherin). |
| Blair et al., 2006 [39] | Biopsy | 5 UC 15 CD 6 control (adenocarcinoma) | Quantitative immunofluorescence microscopy | Epithelial MLCK expression mildly upregulated in inactive IBD and further upregulated in active disease (increase in Myosine Light Chain Kinase(MLCK) expression correlate with histological disease activity). MLCK phosphorylation is also significantly increased in active, but not inactive IBD. |
| Zeissig et al., 2007 [24] | Sigmoid colon biopsy | 23 active CD 22 control 15 inactive CD 15 UC | Ussing chamber Freeze fracture electron microscopy Western blot Immunohistochemistry | Occludin (p < 0.05), claudin-5 (p < 0.05) and -8 (p < 0.001) were downregulated and re-distributed in active CD compared to controls but not in inactive state. Claudin-2 was strongly upregulated and inducible by Tumor Necrosis Factor- α (TNF-α). Other claudins were unchanged (-1, -4. -7) or not detectable in sigmoid colon (claudin-11, -12, -14, -15, and -16). There were reduced and discontinuous TJ strands. Focal epithelial lesions (e.g., microerosions) did not contribute to barrier dysfunction in CD. However, epithelial apoptosis was increased in active but not inactive CD. |
| Vetrano et al., 2008 [75] | Tissue specimen | 11 control 13 CD 15 UC | Western blot Immunofluorescence staining for anti-JAM-A, E-cadherin and ZO-1 and confocal fluorescence microscopy | Loss of JAM-A expression in actively inflamed IBD (p < 0.01) but not in uninvolved mucosa of IBD. Western blot showed significantly lower JAM-A levels in inflamed mucosa of IBD (p < 0.05) compared to the controls. |
| Oshima et al., 2008 [25] | Rectum biopsy | 5 active UC 5 control | Antibody staining (for claudin-1, 2, 3, 4, and 7) Immunofluorescence microscopy Western blot Real-time PCR | Expression of claudin-4 and -7 were decreased; claudin-2 was elevated and claudin-1 and -3 remained unchanged, compared to the control patients. |
| Thuijls et al., 2010 [27] | Colonic biopsy (only from IBD group) Urine samples | 10 healthy 10 IBD remission (5 CD, 5 UC) 10 active IBD (4 CD, 6UC) | Immunostaining of claudin-3 Western blot for urinary claudin-3 | Less staining of claudin-3 was observed in tissue samples of active IBD compared to controls and IBD patients in clinical remission. This correlated with urinary claudin-3 levels (p < 0.001). |
| Poritz et al., 2011 [29] | Mucosa sample | UC CD Control | Western blot | Decrease in occludin and an increase in claudin-1, thus significant increase in claudin-1: occludin (C:O) ratio in diseased UC colon compared to non-diseased UC colon (p < 0.001) and normal colon tissue (p < 0.01). In CD, C:O ratio elevated in all CD tissue, irrespective of disease status. |
| Vetrano et al., 2011 [73] | Colon biopsy | 16 healthy 12 active CD 13 active UC | Immunohistochemistry Flow cytometry RT-PCR | EPCR (endothelial cell PC receptor) and PC (protein C) expression in inflamed tissue samples from UC and CD was significantly lower compared to healthy individuals (p < 0.001). EPCR, PC and PAP-1 (Protease-activated receptor-1) were expressed by epithelial cells of both healthy and IBD but the expression was decreased in IBD epithelial cells by 47% and 30%. Downregulation of mRNA for EPCR, PC as well as PAR-1 in active IBD. |
| Das et al., 2012 [28] | Colonic biopsy | 11 active CD 10 active UC 10 untreated colonic tuberculosis 6 IBS as control | Immunohistochemistry Transmission electron microscopy | Claudin-2 upregulated in all disease groups (p = 0.002). Claudin-2 was expressed the full length of ICJ in IBD group while it was localised to the upper one-third in cTB and control groups. Claudin-4 expression was lower in disease compared to controls groups (p < 0.01). ZO-1 expression was reduced and focal in all disease group while it was diffused in control. Occludin expression were not significantly deviated in disease groups versus the control. Pentalaminar structure of TJ destroyed in IBD patients. |
| Petit et al., 2012 [23] | Colon samples from IBD patients | 24 IBD patients Control (colonic diverticulitis) | Immunohistochemistry Electron microscopy Immunoblotting | PrPc was concentrated at cell-cell junction and largely co-localised with beta-catenin in controls. This was disorganised in the junctions of IBD mucosa, accompanied by an increase in intracellular signal. However, the mRNA and protein level of PrPc was not significantly deviated compared to the controls. |
| Goswami et al., 2014 [10] | Duodenal biopsy | 24 Celiac disease 28 active CD 15 functional dyspepsia as controls | Light microscopy Immunohistochemistry Western blot Transmission electron microscopy | Overexpression of claudin-2 (p = 0.001 at villi and p = 0.007 at crypts) that did not reverse with six months of treatment. Occludin was significantly overexpressed (p < 0.001) compared to controls that did not decrease with treatment. ZO-1 was reduced in mucosal crypts (p = 0.004) that did not alter with treatment, however, western blotting did not find consistent results. No change in JAM-1 protein. Altered ultrastructure of TJs such as pentalaminarstructure and TJ dilatation. |
| Rodriguez-Feo et al., 2015 [74] | Tissue biopsy | 15 inflamed CD 6 non-inflamed CD (control) | Immunohistochemistry, confocal microscopy, real-time PCR, Western blotting | IBD patient samples showed significant reduction of RTN-4B/NOGO-B expression in inflamed mucosa compared to non-inflamed mucosa which show patchy staining pattern mostly at surface epithelium. |
| Gu et al., 2017 [76] | Colon biopsy | 40 IBD in remission (assessed at 6, 12, 24 months after baseline colonoscopy) | Quantitative real-time PCR Gene array | Baseline expression of platelet endothelial cell adhesion molecule (PECAM-1) (2.4 fold elevation, p = 0.02), ICAM-3 (1.9fold elevation, p = 0.03) and VCAM-1 (1.4fold elevation, p = 0.02) were significantly higher in patients who flared than those who did not. Elevation in PECAM-1 and ICAM-3 were significant as early as six months. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.Y.; Wasinger, V.C.; Yau, Y.Y.; Chuang, E.; Yajnik, V.; Leong, R.W. Molecular Pathophysiology of Epithelial Barrier Dysfunction in Inflammatory Bowel Diseases. Proteomes 2018, 6, 17. https://doi.org/10.3390/proteomes6020017
Lee JY, Wasinger VC, Yau YY, Chuang E, Yajnik V, Leong RW. Molecular Pathophysiology of Epithelial Barrier Dysfunction in Inflammatory Bowel Diseases. Proteomes. 2018; 6(2):17. https://doi.org/10.3390/proteomes6020017
Chicago/Turabian StyleLee, Jessica Y., Valerie C. Wasinger, Yunki Y. Yau, Emil Chuang, Vijay Yajnik, and Rupert WL. Leong. 2018. "Molecular Pathophysiology of Epithelial Barrier Dysfunction in Inflammatory Bowel Diseases" Proteomes 6, no. 2: 17. https://doi.org/10.3390/proteomes6020017
APA StyleLee, J. Y., Wasinger, V. C., Yau, Y. Y., Chuang, E., Yajnik, V., & Leong, R. W. (2018). Molecular Pathophysiology of Epithelial Barrier Dysfunction in Inflammatory Bowel Diseases. Proteomes, 6(2), 17. https://doi.org/10.3390/proteomes6020017





