Bone Marrow Protein Oxidation in Response to Ionizing Radiation in C57BL/6J Mice
Abstract
:1. Introduction
2. Experimental
2.1. Chemicals
2.2. Animals
2.2. Irradiation
2.4. Protein Oxidation Detection by OxyBlot
2.5. 2-D Gel Electrophoresis
2.6. Peptide Mass Fingerprinting for Protein Identification
3. Results and Discussion
3.1. Radiation-Induced Protein Carbonylation in Bone Marrow Tissue
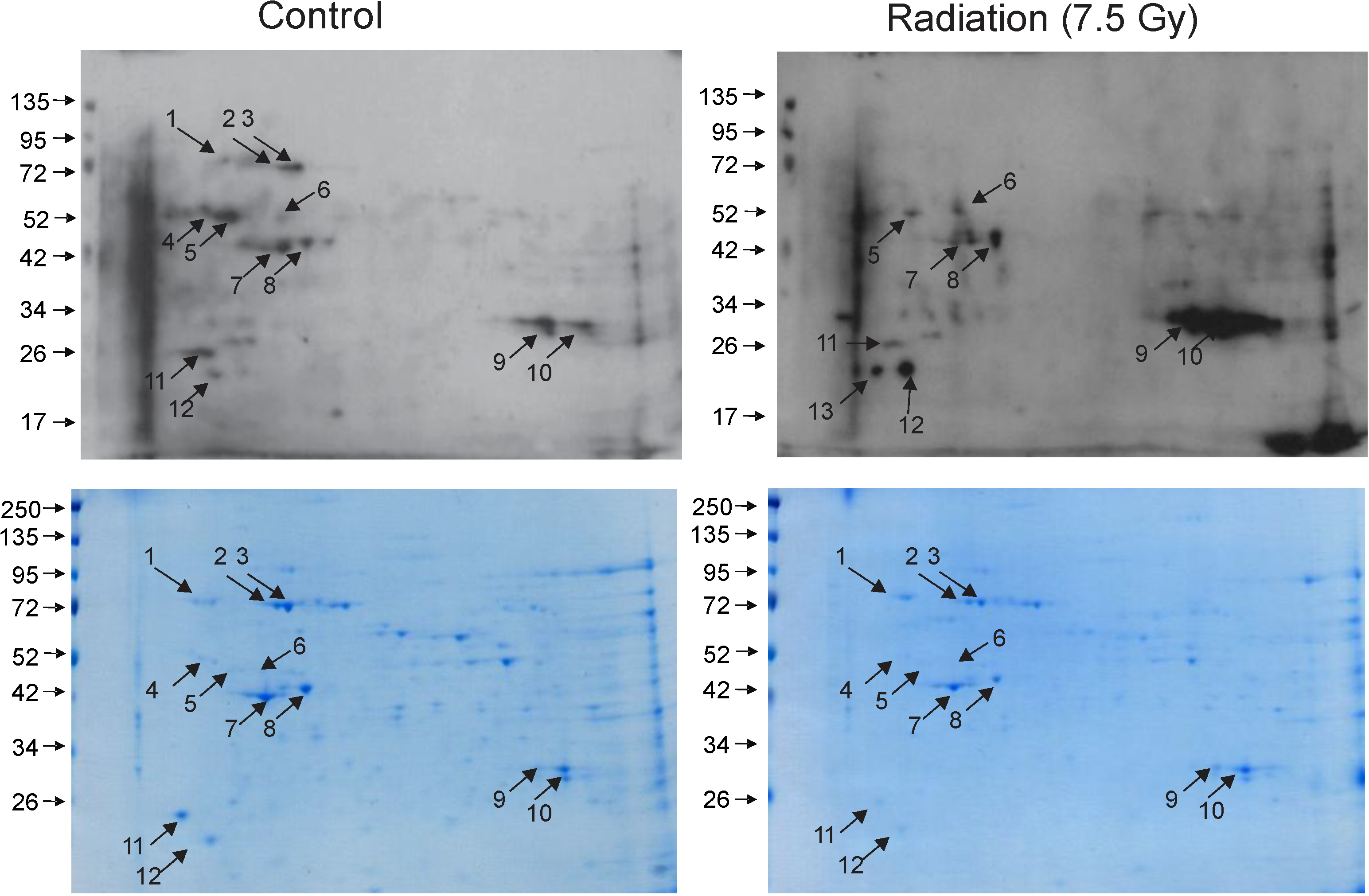
3.2. Identification of Carbonylated Proteins from Control and Irradiated Bone Marrow Tissue
| Spot No. | Protein ID | # Peptides | % Coverage | Control | Radiation |
|---|---|---|---|---|---|
| 1 | 78 kDa glucose-regulated protein (GRP78) | 22 | 37.5 | + | − |
| 2 | Heat shock cognate 71 kDa protein (HSC71) | 19 | 38.9 | ++ | − |
| 3 | Heat shock cognate 71 kDa protein (HSC71) | 29 | 52.5 | ++ | − |
| 6 | Chitinase-like protein 3 (CHI3L1 or YKL-40) | 19 | 53.8 | + | ++ |
| 7 | Actin, cytoplasmic | 15 | 49.6 | + | + |
| 8 | Actin, cytoplasmic | 10 | 29.3 | + | + |
| 10 | Carbonic anhydrase 2 (CAII) | 12 | 67.7 | + | +++ |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Meder, J.; Michalowski, A. Changes in cellularity and/or weight of mouse hemopoietic Tissues as a measure of acute radiation effects. Arch. Immunol. Ther. Exp. (Warsz) 1980, 28, 9–18. [Google Scholar]
- Mauch, P.; Constine, L.; Greenberger, J.; Knospe, W.; Sullivan, J.; Liesveld, J.L.; Deeg, H.J. Hematopoietic stem cell compartment: Acute and late effects of radiation therapy and chemotherapy. Int. J. Radiat. Oncol. Biol. Phys. 1995, 31, 1319–1339. [Google Scholar] [CrossRef]
- Davis, T.A.; Landauer, M.R.; Mog, S.R.; Barshishat-Kupper, M.; Zins, S.R.; Amare, M.F.; Day, R.M. Timing of captopril administration determines radiation protection or radiation sensitization in a murine model of total body irradiation. Exp. Hematol. 2010, 38, 270–281. [Google Scholar] [CrossRef]
- Passegue, E.; Wagers, A.J.; Giuriato, S.; Anderson, W.C.; Weissman, I.L. Global analysis of proliferation and cell cycle gene expression in the regulation of hematopoietic stem and progenitor cell fates. J. Exp. Med. 2005, 202, 1599–1611. [Google Scholar] [CrossRef]
- Cheng, T.; Rodrigues, N.; Shen, H.; Yang, Y.; Dombkowski, D.; Sykes, M.; Scadden, D.T. Hematopoietic stem cell quiescence maintained by p21cip1/waf1. Science 2000, 287, 1804–1808. [Google Scholar]
- Davis, T.A.; Mungunsukh, O.; Zins, S.; Day, R.M.; Landauer, M.R. Genistein induces radioprotection by hematopoietic stem cell quiescence. Int. J. Radiat. Biol. 2008, 84, 713–726. [Google Scholar] [CrossRef]
- Wang, Y.; Schulte, B.A.; LaRue, A.C.; Ogawa, M.; Zhou, D. Total body irradiation selectively induces murine hematopoietic stem cell senescence. Blood 2006, 107, 358–366. [Google Scholar] [CrossRef]
- Hirabayashi, Y. Radiation-induced, cell cycle-related gene expression in aging hematopoietic stem cells: Enigma of their recovery. Ann. N. Y. Acad. Sci. 2014, 1310, 69–73. [Google Scholar]
- Hirabayashi, Y.; Inoue, T. Prediction of epigenetic and stochastic gene expression profiles of late effects after radiation exposure. In Toxicology and Epigenetics; Sahu, S.C., Ed.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2012; pp. 475–510. [Google Scholar]
- Zhang, J.; Yang, Y.; Wang, Y.; Wang, Z.; Yin, M.; Shen, X. Identification of Hub genes related to the recovery phase of irradiation injury by microarray and integrated gene network analysis. PLoS One 2011, 6, e24680. [Google Scholar]
- Sanzari, J.K.; Wambi, C.; Lewis-Wambi, J.S.; Kennedy, A.R. Antioxidant dietary supplementation in mice exposed to proton radiation attenuates expression of programmed cell death-associated genes. Radiat. Res. 2011, 175, 650–656. [Google Scholar] [CrossRef]
- Chen, C.; Lorimore, S.A.; Evans, C.A.; Whetton, A.D.; Wright, E.G. A proteomic analysis of murine bone marrow and its response to ionizing radiation. Proteomics 2005, 5, 4254–4263. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, L.; Pazhanisamy, S.K.; Li, H.; Meng, A.; Zhou, D. Total body irradiation causes residual bone marrow injury by induction of persistent oxidative stress in murine hematopoietic stem cells. Free Radic. Biol. Med. 2010, 48, 348–356. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, Y.A.; Meng, A.; Yan, H.; Wang, X.; Niu, J.; Li, J.; Wang, H. Inhibiting TGFbeta1 has a protective effect on mouse bone marrow suppression following ionizing radiation sxposure in vitro. J. Radiat. Res. 2013, 54, 630–636. [Google Scholar] [CrossRef]
- Daly, M.J.; Gaidamakova, E.K.; Matrosova, V.Y.; Vasilenko, A.; Zhai, M.; Leapman, R.D.; Lai, B.; Ravel, B.; Li, S.M.; Kemner, K.M.; et al. Protein oxidation implicated as the primary determinant of bacterial radioresistance. PLoS Biol. 2007, 5, e92. [Google Scholar] [CrossRef]
- Du, J.; Gebicki, J.M. Proteins are major initial cell rargets of hydroxyl free radicals. Int. J. Biochem. Cell Biol. 2004, 36, 2334–2343. [Google Scholar] [CrossRef]
- Panganiban, R.A.; Mungunsukh, O.; Day, R.M. X-irradiation induces ER stress, apoptosis, and senescence in pulmonary artery endothelial cells. Int. J. Radiat. Biol. 2012, 89, 656–667. [Google Scholar] [CrossRef]
- McPherson, C.W. Reduction of pseudomonas aeruginosa and coliform bacteria in mouse drinking water following treatment with hydrochloric acid or chlorine. Lab. Anim. Care 1963, 13, 737–744. [Google Scholar]
- Steiner, S.R.; Philbert, M.A. Proteomic identification of carbonylated proteins in 1,3-dinitrobenzene neurotoxicity. Neurotoxicology 2011, 32, 362–373. [Google Scholar] [CrossRef]
- Image J Software. Available online: http://rsbweb.nih.gov/ij/ (accessed on 18 August 2012).
- Wong, C.M.; Cheema, A.K.; Zhang, L.; Suzuki, Y.J. Protein carbonylation as a novel mechanism in redox signaling. Circ. Res. 2008, 102, 310–318. [Google Scholar] [CrossRef]
- Muth, E.; Driscoll, W.J.; Smalstig, A.; Goping, G.; Mueller, G.P. Proteomic analysis of rat atrial secretory granules: A platform for testable hypotheses. Biochim. Biophys. Acta 1999, 71, 2871–2882. [Google Scholar]
- Proteinprospector Ms-Fit Search Engine. Available online: http://prospector.ucsf.edu/prospector/mshome.htm (accessed on 6 October 2013).
- Clauser, K.R.; Baker, P.; Burlingame, A.L. Role of accurate mass measurement (+/− 10 ppm) in protein identification strategies employing MS or MS/MS and database searching. Anal. Chem. 1999, 71, 2871–2882. [Google Scholar] [CrossRef]
- Day, R.; Barshishat-Kupper, M.; Mog, S.R.; McCart, E.; Prasanna, P.; Davis, T.; Landauer, M. Genistein protects against biomarkers of delayed lung sequelae in nice surviving high dose total body irradiation. J. Radiat. Res. 2008, 49, 361–372. [Google Scholar] [CrossRef]
- Farber, J.M.; Levine, R.L. Sequence of a peptide susceptible to mixed-function oxidation. Probable cation binding site in glutamine synthetase. J. Biol. Chem. 1986, 261, 4574–4578. [Google Scholar]
- Stadtman, E.R.; Levine, R.L. Protein Oxidation. Ann. N. Y. Acad. Sci. 2000, 899, 191–208. [Google Scholar] [CrossRef]
- Madian, A.G.; Myracle, A.D.; Diaz-Maldonado, N.; Rochelle, N.S.; Janle, E.M.; Regnier, F.E. Differential carbonylation of proteins as a function of in vivo oxidative stress. J. Proteome Res. 2011, 10, 3959–3972. [Google Scholar] [CrossRef]
- England, K.; Cotter, T. Identification of carbonylated proteins by MALDI-TOF mass spectroscopy reveals susceptibility of ER. Biochem. Biophys. Res. Commun. 2004, 320, 123–130. [Google Scholar] [CrossRef]
- Mori, H.; Oikawa, M.; Tamagami, T.; Kumaki, H.; Nakaune, R.; Amano, J.; Akinaga, Y.; Fukui, K.; Abe, K.; Urano, S. Oxidized proteins in astrocytes generated in a hyperbaric atmosphere induce neuronal apoptosis. J. Alzheimers Dis. 2007, 11, 165–174. [Google Scholar]
- Linares, M.; Marín-Garcíía, P.; Méndez, D.; Puyet, A.; Diez, A.; Bautista, J.M. Proteomic approaches to identifying carbonylated proteins in brain tissue. J. Proteome Res. 2011, 10, 1719–1727. [Google Scholar] [CrossRef]
- Sánchez, O.; Rodriguez-Sureda, V.; Dománguez, C.; Ferníndez-Figueras, T.; Vilches, A.; Llurba, E.; Alijotas-Reig, J. Study of biomaterial-induced macrophage activation, cell-mediated immune response and molecular oxidative damage in patients with dermal bioimplants. Immunobiology 2012, 217, 44–53. [Google Scholar] [CrossRef]
- Görgens, S.W.; Eckardt, K.; Elsen, M.; Tennagels, N.; Eckel, J. Chitinase-3-like protein 1 protects skeletal muscle from TNFalpha-induced inflammation and insulin resistance. Biochem. J. 2014, 459, 479–488. [Google Scholar] [CrossRef]
- He, C.H.; Lee, C.G.; Dela Cruz, C.S.; Lee, C.M.; Zhou, Y.; Ahangari, F.; Ma, B.; Herzog, E.L.; Rosenberg, S.A.; Li, Y.; et al. Chitinase 3-like 1 regulates cellular and tissue responses via IL-13 receptor alpha2. Cell Rep. 2013, 4, 830–841. [Google Scholar] [CrossRef]
- Lee, C.G.; Da Silva, C.A.; Dela Cruz, C.S.; Ahangari, F.; Bing, M.; Kang, M.-J.; He, C.-H.; Takyar, S.; Elias, J.A. Role of Chitin and chitinase/chitinase-like proteins in inflammation, tissue remodeling, and injury. Ann. Rev. Physiol. 2011, 73, 479–501. [Google Scholar] [CrossRef]
- Fermoselle, C.; Rabinovich, R.; Ausin, P.; Puig-Vilanova, E.; Coronell, C.; Sanchez, F.; Roca, J.; Gea, J.; Barreiro, E. Does oxidative stress modulate limb muscle atrophy in severe COPD patients? Eur. Respir. J. 2012, 40, 851–862. [Google Scholar] [CrossRef]
- Barreiro, E.; Hussain, S.N. Protein carbonylation in skeletal muscles: Impact on function. Antioxid. Redox Signal. 2010, 12, 417–429. [Google Scholar] [CrossRef]
- Suyama, K.; Watanabe, M.; Sakabe, K.; Otomo, A.; Okada, Y.; Terayama, H.; Imai, T.; Mochida, J. Grp78 suppresses lipid peroxidation and promotes cellular antioxidant levels in glial cells following hydrogen peroxide exposure. PLoS One 2014, 9, e86951. [Google Scholar]
- Chao, Y.M.; Lai, M.D.; Chan, J.Y. Redox-sensitive endoplasmic reticulum stress and autophagy at rostral ventrolateral medulla contribute to hypertension in spontaneously hypertensive rats. Hypertension 2013, 61, 1270–1280. [Google Scholar] [CrossRef]
- Mozzini, C.; Fratta Pasini, A.; Garbin, U.; Stranieri, C.; Pasini, A.; Vallerio, P.; Cominacini, L. Increased endoplasmic reticulum stress and Nrf2 repression in peripheral blood mononuclear cells of patients with stable coronary artery disease. Free Radic. Biol. Med. 2014, 68, 178–185. [Google Scholar] [CrossRef]
- Banerjee, A.; Abdelmegeed, M.A.; Jang, S.; Song, B.J. Zidovudine (AZT) and hepatic lipid accumulation: Implication of inflammation, oxidative and endoplasmic reticulum stress mediators. PLoS One 2013, 8, e76850. [Google Scholar]
- Ding, Y.; Li, X.R.; Yang, K.Y.; Huang, L.H.; Hu, G.; Gao, K. Proteomics analysis of gastric epithelial Ags cells infected with Epstein-Barr virus. Asian Pac. J. Cancer Prev. 2013, 14, 367–372. [Google Scholar] [CrossRef]
- Castegna, A.; Aksenov, M.; Aksenova, M.; Thongboonkerd, V.; Klein, J.B.; Pierce, W.M.; Booze, R.; Markesbery, W.R.; Butterfield, D.A. Proteomic identification of sxidatively modified proteins in Alzheimer’s Disease brain. Part I: Creatine kinase Bb, glutamine synthase, and ubiquitin carboxy-terminal hydrolase L-1. Free Radic. Biol. Med. 2002, 33, 562–571. [Google Scholar] [CrossRef]
- Fuentes-Almagro, C.A.; Prieto-Alamo, M.J.; Pueyo, C.; Jurado, J. Identification of proteins containing redox-sensitive thiols after Prdx1, Prdx3 and Gclc silencing and/or glucose oxidase treatment in Hepa 1–6 Cells. J. Proteomics 2012, 77, 262–279. [Google Scholar]
- Nabeshi, H.; Yoshikawa, T.; Kamada, H.; Shibata, H.; Sugita, T.; Abe, Y.; Nagano, K.; Nomura, T.; Minowa, K.; Tsunoda, S.; Tsutsumi, Y. Arsenic trioxide induces down-regulation of Gp46 via protein oxidation: Proteomics analysis of oxidative modified proteins in As2o3-treated HTLV-1-infected cells. Pharmazie 2010, 65, 702–707. [Google Scholar]
- Wong, C.M.; Marcocci, L.; Liu, L.; Suzuki, Y.J. Cell Signaling by Protein Carbonylation and Decarbonylation. Antioxid. Redox Signal. 2010, 12, 393–404. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kim, Y.-C.; Barshishat-Kupper, M.; McCart, E.A.; Mueller, G.P.; Day, R.M. Bone Marrow Protein Oxidation in Response to Ionizing Radiation in C57BL/6J Mice. Proteomes 2014, 2, 291-302. https://doi.org/10.3390/proteomes2030291
Kim Y-C, Barshishat-Kupper M, McCart EA, Mueller GP, Day RM. Bone Marrow Protein Oxidation in Response to Ionizing Radiation in C57BL/6J Mice. Proteomes. 2014; 2(3):291-302. https://doi.org/10.3390/proteomes2030291
Chicago/Turabian StyleKim, Yong-Chul, Michal Barshishat-Kupper, Elizabeth A. McCart, Gregory P. Mueller, and Regina M. Day. 2014. "Bone Marrow Protein Oxidation in Response to Ionizing Radiation in C57BL/6J Mice" Proteomes 2, no. 3: 291-302. https://doi.org/10.3390/proteomes2030291





