Certain Malvaceae Plants Have a Unique Accumulation of myo-Inositol 1,2,4,5,6-Pentakisphosphate
Abstract
:1. Introduction
2. Results and Discussion
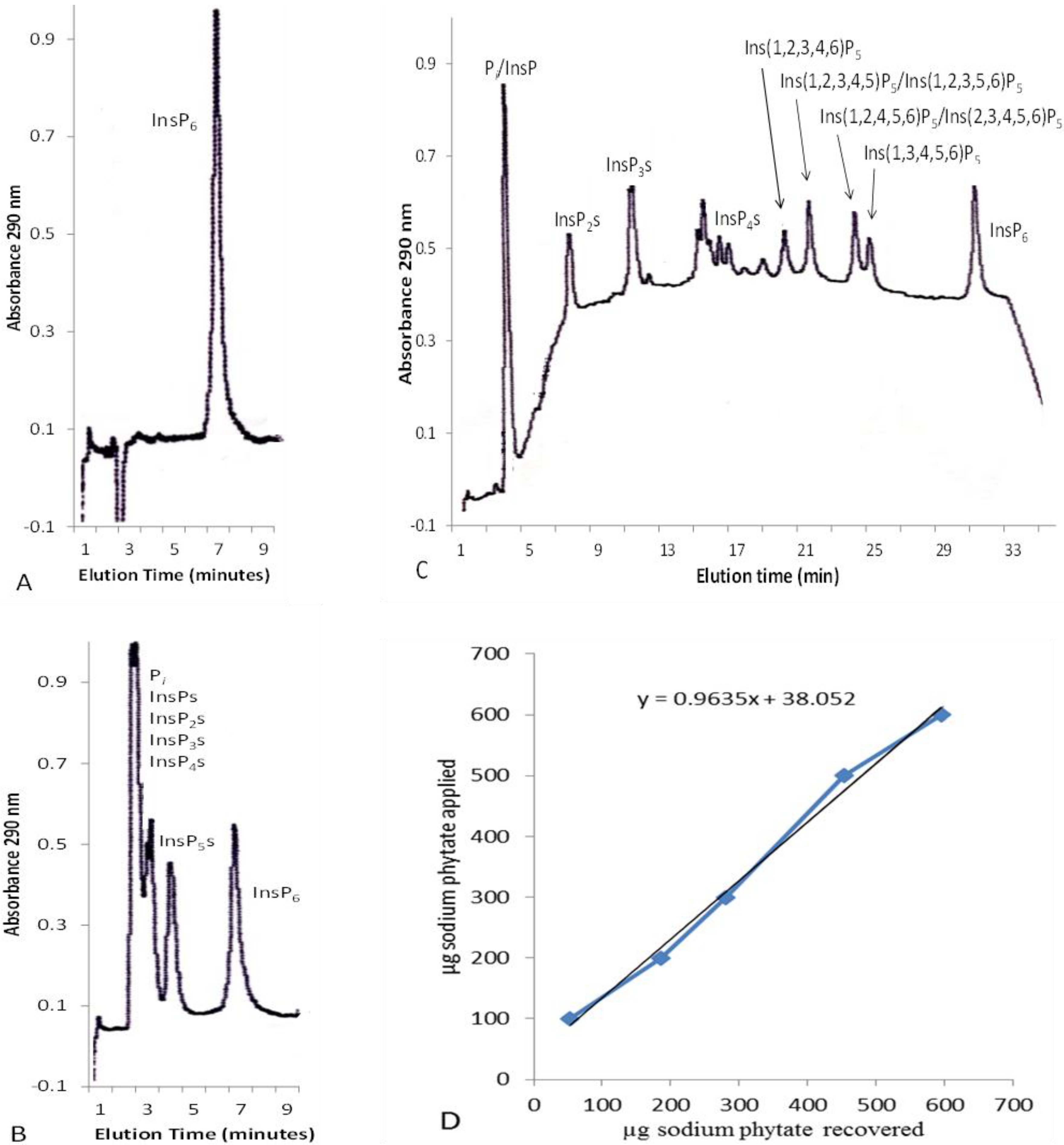
2.1. Concentration of Inositol Phosphates in Leaf Extracts
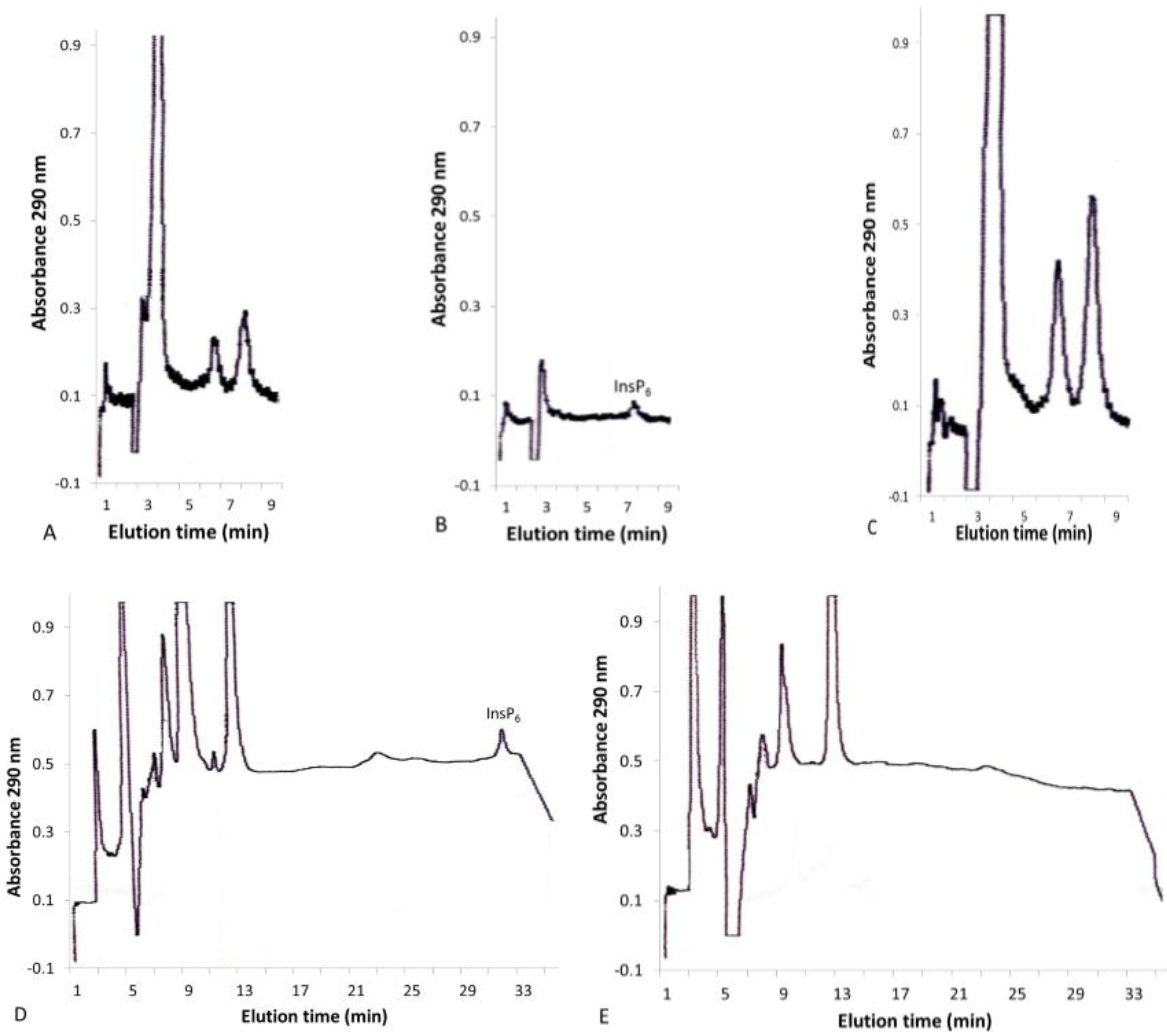
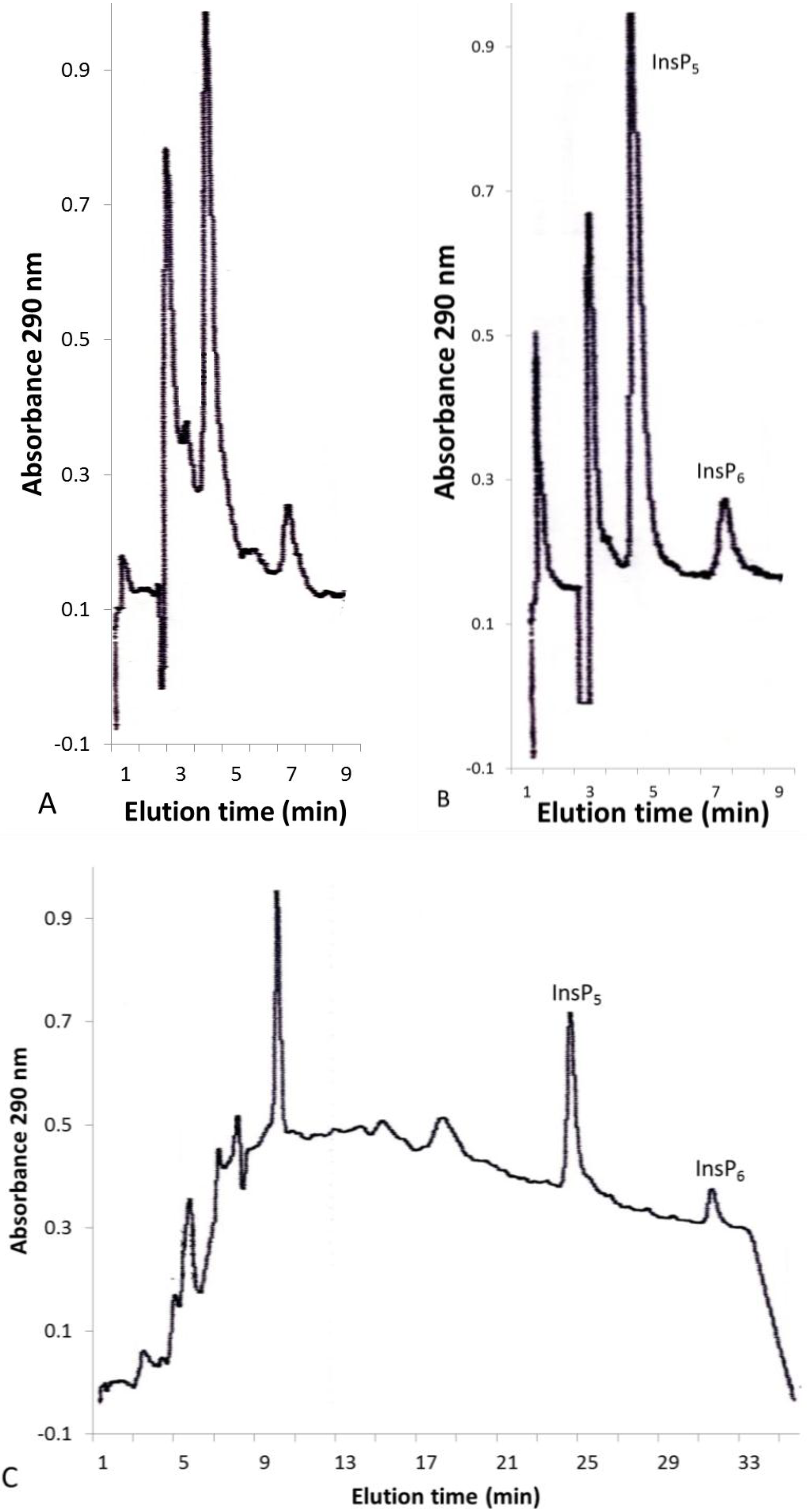
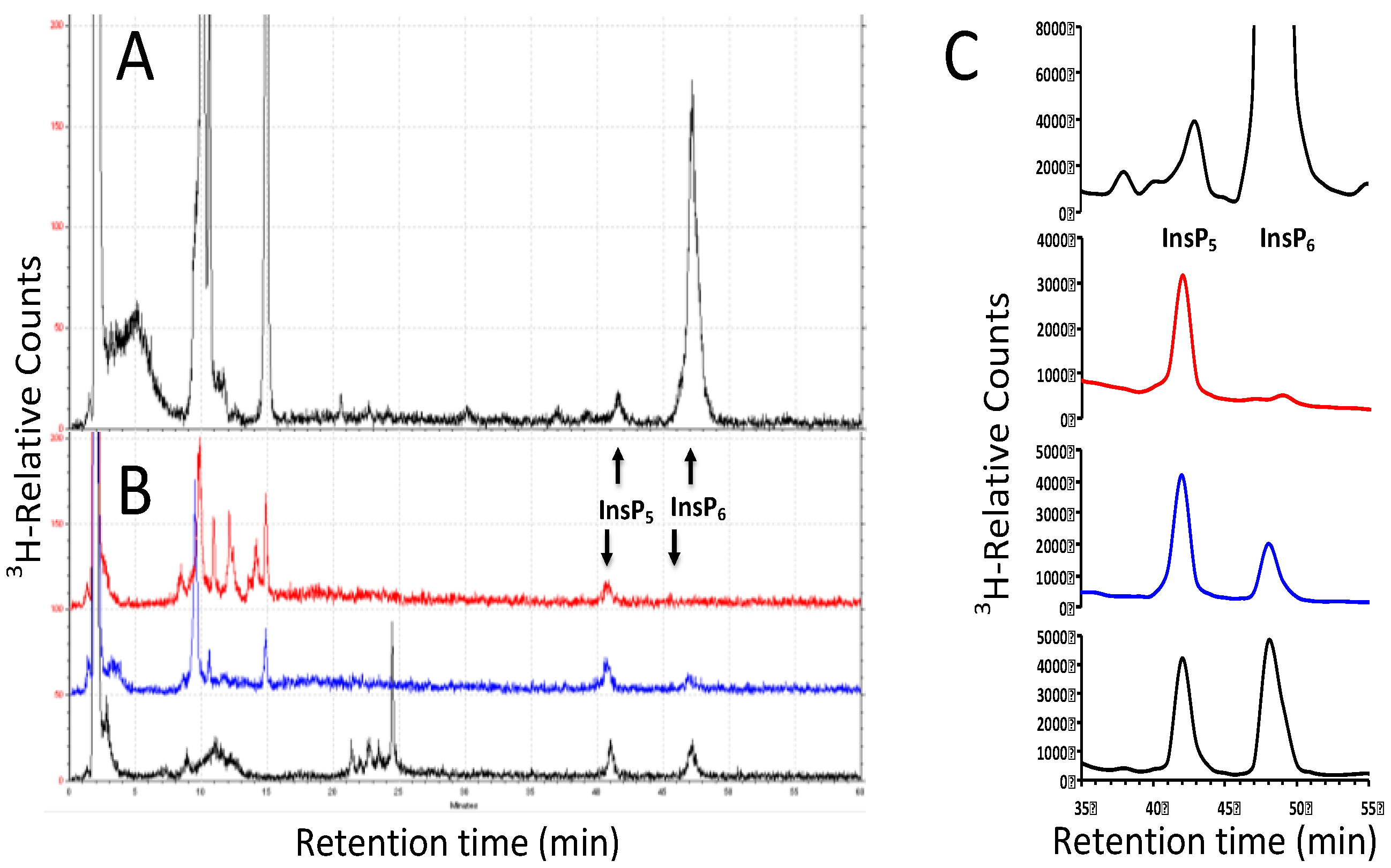
2.2. Solid-Phase Extraction of Inositol Phosphate Concentrates
| Plant | InsP5 Added | InsP6 Added | InsP5 Determined | InsP6 Determined | InsP5 Recovered | InsP6 Recovered |
|---|---|---|---|---|---|---|
| Tobacco | 0 µg | 0 µg | 0 µg | 37 µg | - | - |
| Tobacco | 50 µg | 0 µg | 49 µg | 42 µg | 49 µg | - |
| Cotton | 0 µg | 0 µg | 40 µg | 66 µg | - | - |
| Cotton | 0 µg | 132 µg | 48 µg | 196 µg | - | 130 µg |
2.3. InsP6 and InsP5 Levels in Various Plants
| Plant | Tissue | InsP6 (µM) | Ins(1,2,4,5,6)P5 (µM) |
|---|---|---|---|
| Zea mays | leaves | 101–198 (3) | nd |
| Oryza sativa | leaves | 9–49 (6) | nd |
| Arabidopsis thaliana | leaves | 29–89 (9) | 2–22 (5) |
| Arabidopsis thaliana | seedlings | 47–110 (4) | nd |
| Nicotiana tabacum | leaves | 11–24 (3) | nd |
| Solanum lycopersicum | leaves | 39–65 (3) | nd |
| Trifolium repens | leaves | 21 (1) | 6 (1) |
| Acer rubrum | leaves | 35 (1) | 2 (1) |
| Ginkgo biloba | leaves | 61 (1) | nd |
| Camelina sativa | leaves | 70 (1) | 6 (1) |
| Glycine max | leaves | nd (1) | nd |
| Pueraria labata | leaves | nd (1) | nd |
| Plant | Tissue | InsP6 (µM) | Ins(1,2,4,5,6)P5 (µM) |
|---|---|---|---|
| Gossypium hirsutum | leaves | 11–169 (6) | 5–510 (6) |
| Gossypium hirsutum | seedlings | 77–140 (3) | 86–118 (3) |
| Gossypium hirsutum | 14–17 dpa fiber | 43 (1) | nd |
| Gossypium hirsutum | 16 dpa seeds | 105 (1) | nd |
| Gossypium barbadense | leaves | 60–76 (2) | 329–507 (2) |
| Gossypium raimondii | leaves | 71–86 (2) | 578–589 (2) |
| Abelmoschus. esculentus | leaves | 90–336 (4) | 3 (1) |
| Abelmoschus. esculentus | pod (husk) | 100 (1) | nd |
| Abelmoschus manihot | leaves | 250–822 (4) | 2–7 (2) |
| Alcea rosea | leaves | 50–52(2) | 10(2) |
| Althea officinalis | leaves | 95–217 (2) | 38–74 (2) |
| Hibiscus syriacus | leaves | 27–34 (3) | 10–19 (3) |
| Hibiscus moscheutos | leaves | 39–73 (2) | 219–491 (2) |
| Hibiscus rosa-sinensis | leaves | 31 (1) | 392 (1) |
| Hibiscus cannabinus | leaves | 172–491 (5) | 19–57 (4) |
| Hibiscus coccineus | leaves | 97–140 (2) | 536–647 (2) |
| Hibiscus sabdariffa | leaves | 153 (1) | 114 (1) |
| Hibiscus mutabilis | leaves | 49 (1) | 46 (1) |
| InsP6 | InsP5 | InsP5:InsP6 | ||
|---|---|---|---|---|
| % of Total InsP | % of Total InsP | Ratio | ||
| Arabidopsis | seedlings 1 | 27.8 | 2.70 | 0.097 |
| G. hirsutum | seedling shoot | 1.18 | 4.35 | 3.7 |
| G. hirsutum | seedling root | 3.78 | 7.20 | 1.9 |
| G. hirsutum | young leaf | 9.47 | 7.07 | 0.75 |
| Stage | Location | InsP6 (µM) | InsP5 (µM) |
|---|---|---|---|
| lowering | bottom near trunk | 11 | 5 |
| flowering | middle near trunk | 20 | 14 |
| flowering | middle near bolls | 33 | 126 |
| flowering | top | 169 | 492 |
| post-anthesis | top | 22 | 8 |
3. Experimental Section
3.1. Plant Material
3.2. Inositol Phosphate Standards
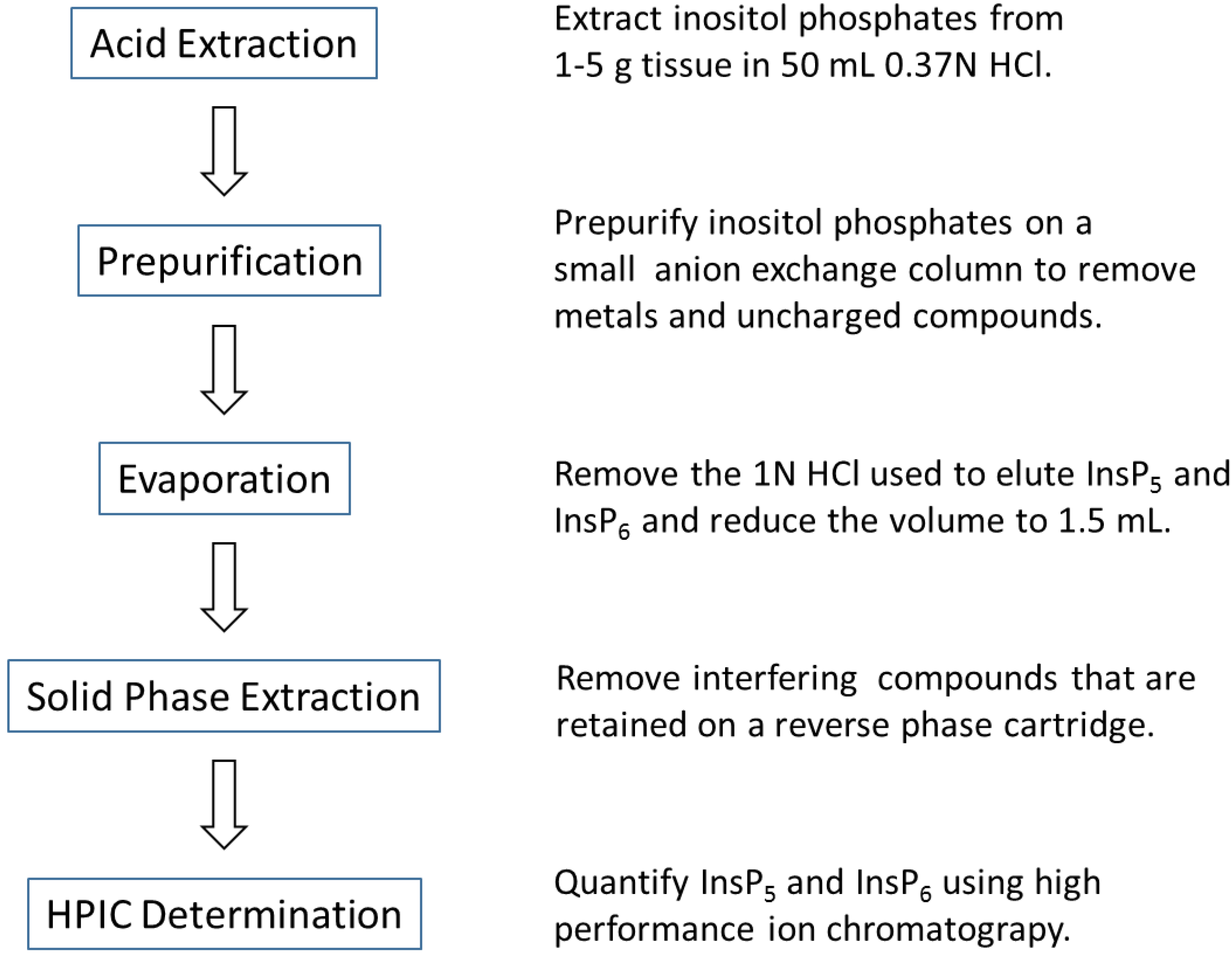
3.3. HCl Extraction and Concentration of Inositol Phosphates
3.4. Solid-Phase Extraction
3.5. HPIC
3.6. Recovery Experiments
3.7. Radiolabeling Plant Tissue with [3H]Inositol
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Streb, H.; Irvine, R.F.; Berridge, M.J.; Schulz, I. Release of Ca2+ from a nonmitochondrial intracellular store in pancreatic acinar cells by inositol-1,4,5-trisphosphate. Nature 1983, 306, 67–69. [Google Scholar] [CrossRef] [PubMed]
- Stevenson-Paulik, J.; Phillippy, B.Q. Inositol polyphosphates and kinases. In Lipid Signaling in Plants; Book Series: Plant Cell Monographs; Minnik, T., Ed.; Springer-Verlag Berlin: Berlin, Germany, 2010; Volume 16, pp. 161–174. [Google Scholar]
- Irvine, R.F. Inositide evolution—Towards turtle domination? J. Physiol. 2005, 566, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Irvine, R.F.; Schell, M.J. Back in the water: The return of the inositol phosphates. Nat. Rev. Mol. Cell Biol. 2001, 2, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Brehm, M.A.; Schenk, T.M.H.; Zhou, X.; Fanick, W.; Lin, H.; Windhorst, S.; Nalaskowski, M.M.; Kobras, M.; Shears, S.B.; Mayr, G.W. Intracellular localization of human Ins(1,3,4,5,6)P-5 2-kinase. Biochem. J. 2007, 408, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Monserrate, J.P.; York, J.D. Inositol phosphate synthesis and the nuclear processes they affect. Curr. Opin. Cell Biol. 2010, 22, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Lee, D.H.; Cho, H.K.; Kim, S.H.; Auh, J.H.; Pai, H.S. InsP6-sensitive variants of the Gle1 mRNA export factor rescue growth and fertility defects of the ipk1 low-phytic-acid mutation in Arabidopsis. Plant Cell 2015, 27, 417–431. [Google Scholar] [CrossRef] [PubMed]
- Graf, E.; Empson, K.L.; Eaton, J.W. Phytic acid—A natural antioxidant. J. Biol. Chem. 1987, 262, 11647–11650. [Google Scholar] [PubMed]
- Doria, E.; Galleschi, L.; Calucci, L.; Pinzino, C.; Pilu, R.; Cassani, E.; Nielsen, E. Phytic acid prevents oxidative stress in seeds: Evidence from a maize (Zea mays L.) low phytic acid mutant. J. Exp. Bot. 2009, 60, 967–978. [Google Scholar] [CrossRef] [PubMed]
- Kuo, H.F.; Chang, T.Y.; Chiang, S.F.; Wang, W.D.; Charng, Y.Y.; Chiou, T.J. Arabidopsis inositol pentakisphosphate 2-kinse, AtIPK1, is required for growth and modulates phosphate homeostasis at the transcriptional level. Plant J. 2014, 80, 503–515. [Google Scholar]
- Stevenson-Paulik, J.; Bastidas, R.J.; Chiou, S.T.; Frye, R.A.; York, J.D. Generation of phytate-free seeds in Arabidopsis through disruption of inositol polyphosphate kinases. Proc. Natl. Acad. Sci. USA 2005, 102, 12612–12617. [Google Scholar] [CrossRef] [PubMed]
- Lemtiri-Chlieh, F.; MacRobbie, E.A.C.; Webb, A.A.R.; Manison, N.F.; Brownlee, C.; Skepper, J.N.; Chen, J.; Prestwich, G.D.; Brearley, C.A. Inositol hexakisphosphate mobilizes an endomembrane store of calcium in guard cells. Proc. Natl. Acad. Sci. USA 2003, 100, 10091–10095. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Calderon-Villalobos, L.I.A.; Sharon, M.; Zheng, C.X.; Robinson, C.V.; Estelle, M.; Zheng, N. Mechanism of auxin perception by the TIRI1 ubiquitin ligase. Nature 2007, 446, 640–645. [Google Scholar] [CrossRef] [PubMed]
- Murphy, A.M.; Otto, B.; Brearley, C.A.; Carr, J.P.; Hanke, D.E. A role for inositol hexakisphosphate in the maintenance of basal resistance to plant pathogens. Plant J. 2008, 56, 638–652. [Google Scholar] [CrossRef] [PubMed]
- Sheard, L.B.; Tan, X.; Mao, H.; Withers, J.; Ben-Nissan, G.; Hinds, T.R.; Kobayashi, Y.; Hsu, F.F.; Sharon, M.; Browse, J.; et al. Jasmonate perception by inositol-phosphate-potentiated COI1-JAZ co-receptor. Nature 2010, 468, 400–405. [Google Scholar] [CrossRef] [PubMed]
- Mosblech, A.; Thurow, C.; Gatz, C.; Feussner, I.; Heilmann, I. Jasmonic acid perception by COI1 involves inositol polyphosphates in Arabidopsis thaliana. Plant J. 2011, 65, 949–957. [Google Scholar] [CrossRef] [PubMed]
- Phillippy, B.Q. Inositol phosphates in foods. Adv. Food Nutr. Res. 2003, 45, 1–60. [Google Scholar] [PubMed]
- Bentsink, L.; Yuan, K.; Korneef, D.; Vreugdenhil, D. The genetics of phytate and phosphate accumulation in seeds and leaves of Arabidopsis thaliana, using natural variation. Theor. Appl. Genet. 2003, 106, 1234–1243. [Google Scholar] [PubMed]
- Zhang, W.; Gruszewski, H.A.; Chevone, B.I.; Nessler, C.L. An Arabidopsis purple acid phosphatase with phytase activity increases foliar ascorbate. Plant Physiol. 2008, 146, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Jamar, D.C.L.; Lou, P.; Wang, Y.; Wu, J.; Wang, X.; Bonnema, G.; Koornneef, M.; Vreugdenhil, D. Quantitative trait loci analysis of phytate and phosphate concentrations in seeds and leaves of Brassica rapa. Plant Cell Environ. 2008, 31, 887–900. [Google Scholar] [CrossRef] [PubMed]
- Nagy, R.; Grob, H.; Weder, B.; Green, P.; Klein, M.; Frelet-Barrand, A.; Schjoerring, J.K.; Brearley, C.; Martinoia, E. The Arabidopsis ATP-binding cassette protein AtMRP5/AtABCC5 is a high affinity inositol hexakisphosphate transporter involved in guard cell signaling and phytate storage. J. Biol. Chem. 2009, 284, 33614–33622. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, S.I.; Tai, T.H. Identification of genes necessary for wild-type levels of seed phytic acid in Arabidopsis thaliana using a reverse genetics approach. Mol. Genet. Genomics 2011, 286, 119–133. [Google Scholar] [CrossRef] [PubMed]
- Harland, B.F.; Morris, E.R. Phytate: A good or a bad food component? Nutr. Res. 1995, 15, 733–754. [Google Scholar] [CrossRef]
- Alkarawi, H.H.; Zotz, G. Phytic acid in green leaves. Plant Biol. 2014, 16, 697–701. [Google Scholar] [CrossRef] [PubMed]
- Alkarawi, H.H.; Zotz, G. Phytic acid in green leaves of herbaceous plants—Temporal variation in situ and response to different nitrogen/phosphorus fertilizing regimes. AoB Plants 2014, 6. [Google Scholar] [CrossRef] [PubMed]
- Stevenson-Paulik, J.; Chiou, S.T.; Frederick, J.P.; dela Cruz, J.; Seeds, A.M.; Otto, J.C.; York, J.D. Inositol phosphate metabolomics: Merging genetic perturbation with modernized radiolabeling methods. Methods 2006, 39, 112–121. [Google Scholar] [CrossRef] [PubMed]
- Gillaspy, G.E. The cellular language of myo-inositol signaling. New Phytol. 2011, 192, 823–839. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Villalta, P.W.; Sturla, S.J. Simultaneous determination of inositol and inositol phosphates in complex biological matrices: Quantitative ion-exchange chromatography/tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2009, 23, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Phillippy, B.Q.; Bland, J.M.; Evens, T.J. Ion chromatography of phytate in roots and tubers. J. Agric. Food Chem. 2003, 51, 350–353. [Google Scholar] [CrossRef] [PubMed]
- Phillippy, B.Q.; Wyatt, C.J. Degradation of phytate in foods by phytases in fruit and vegetable extracts. J. Food Sci. 2001, 66, 535–539. [Google Scholar] [CrossRef]
- Desai, M.; Rangarajan, P.; Donahue, J.A.; Williams, S.P.; Land, E.S.; Mandal, M.K.; Phillippy, B.Q.; Perera, I.Y.; Raboy, V.; Gillaspy, G.E. Two inositol hexakisphosphate kinases drive inositol pyrophosphate synthesis in plants. Plant J. 2014, 80, 642–653. [Google Scholar] [CrossRef] [PubMed]
- Phillippy, B.Q.; Bland, J.M. Gradient ion chromatography of inositol phosphates. Anal. Biochem. 1988, 175, 162–166. [Google Scholar] [CrossRef]
- Hanke, D.E.; Parmar, P.N.; Caddick, S.E.K.; Green, P.; Brearley, C.A. Synthesis of inositol phosphate ligands of plant hormone-receptor complexes: Pathways of inositol hexakisphosphate turnover. Biochem. J. 2012, 444, 601–609. [Google Scholar] [CrossRef] [PubMed]
- Brearley, C.A.; Hanke, D.E. Inositol phosphates in the duckweed Spirodela polyrhiza L. Biochem. J. 1996, 314, 227–233. [Google Scholar] [PubMed]
- Phillippy, B.Q.; Ullah, A.H.J.; Ehrlich, K.E. Purification and some properties of inositol 1,3,4,5,6-pentakisphosphate 2-kinase in soybean seeds. J. Biol. Chem. 1994, 269, 28393–28399. [Google Scholar] [PubMed]
- Raboy, V. myo-Inositol-1,2,3,4,5,6-hexakisphosphate. Phytochemistry 2003, 64, 1033–1043. [Google Scholar] [CrossRef]
- Müller, D.; Leyser, O. Auxin, cytokinin and the control of shoot branching. Ann. Bot. (Lond.) 2011, 107, 1203–1212. [Google Scholar] [CrossRef] [PubMed]
- Rameau, C.; Bertheloot, J.; Leduc, N.; Andrieu, B.; Foucher, F.; Sakr, S. Multiple pathways regulate shoot branching. Front. Plant Sci. 2015, 5, 1–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cuḗllar Pḗrez, A.; Goossens, A. Jasmonate signalling: A copycat of auxin signalling? Plant Cell Environ. 2013, 36, 2071–2084. [Google Scholar]
- Loss, O.; Wu, C.T.; Riccio, A.; Saiardi, A. Modulation of inositol polyphosphate levels regulates neuronal differentiation. Mol. Biol. Cell 2013, 24, 2981–2989. [Google Scholar] [CrossRef] [PubMed]
- Guse, A.H.; Greiner, E.; Emrich, F.; Brand, K. Mass changes of inositol 1,3,4,5,6-pentakisphosphate and inositol hexakisphosphate during cell cycle progression in rat thymocytes. J. Biol. Chem. 1993, 268, 7129–7133. [Google Scholar] [PubMed]
- Mountford, J.C.; Bunce, C.M.; French, P.J.; Michell, R.H.; Brown, G. Intracellular concentrations of inositol, glycerophosphoinositol and inositol pentakisphosphate increase during hematopoetic cell differentiation. Biochim. Biophys. Acta 1994, 1222, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Wundenberg, T.; Grabinski, N.; Lin, H.Y.; Mayr, G.W. Discovery of InsP(6)-kinases as InsP(6)-dephosphorylating enzymes provides a new mechanism of cytosolic InsP(6) degradation driven by the cellular ATP/ADP ratio. Biochem. J. 2014, 462, 173–184. [Google Scholar] [CrossRef] [PubMed]
- Heinonen, J.K.; Lahti, R.J. A new and convenient colorimetric determination of inorganic ortho-phosphate and its application to the assay of inorganic pyrophosphatase. Anal. Biochem. 1981, 113, 313–317. [Google Scholar] [PubMed]
- Phillippy, B.Q.; Johnston, M.R. Determination of phytic acid in foods by ion chromatography with post-column derivitization. J. Food Sci. 1985, 50, 541–542. [Google Scholar] [CrossRef]
- Azevedo, C.; Saiardi, A. Extraction and analysis of soluble inositol phosphates from yeast. Nat. Protoc. 2006, 1, 2416–2422. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Phillippy, B.Q.; Perera, I.Y.; Donahue, J.L.; Gillaspy, G.E. Certain Malvaceae Plants Have a Unique Accumulation of myo-Inositol 1,2,4,5,6-Pentakisphosphate. Plants 2015, 4, 267-283. https://doi.org/10.3390/plants4020267
Phillippy BQ, Perera IY, Donahue JL, Gillaspy GE. Certain Malvaceae Plants Have a Unique Accumulation of myo-Inositol 1,2,4,5,6-Pentakisphosphate. Plants. 2015; 4(2):267-283. https://doi.org/10.3390/plants4020267
Chicago/Turabian StylePhillippy, Brian Q., Imara Y. Perera, Janet L. Donahue, and Glenda E. Gillaspy. 2015. "Certain Malvaceae Plants Have a Unique Accumulation of myo-Inositol 1,2,4,5,6-Pentakisphosphate" Plants 4, no. 2: 267-283. https://doi.org/10.3390/plants4020267





